Introduction
Traumatic and ischemic brain injury (TBI) is accompanied by excessive glutamate efflux which results in increased opening of and ion flux through NMDA receptors (NMDAR). Because this may cause excitotoxicity, the activational status of NMDAR and its change over time have important clinical implications. We have labeled the compound CNS-5161 with tritium and carbon-11 1 and studied its sensitivity to changes in NMDAR activation by binding, autoradiography and microPET. Our hypothesis was that NMDAR activation by glutamate, NMDA or brain injury would increase [11C]CNS-5161 affinity, an event followed by NMDAR hypofunction and metabolic depression 2 .
Methods
[3 H]CNS-5161 (0.5 mCi/Kg, N=3) was injected in the tail vein of rats 5 min after NMDA (40 mg/Kg SC) or saline and brains removed 20 or 60 min later for autoradiography. Serial microPET experiments in rats (n=6) were performed using [11C]CNS-5161 (0.5–1.9 mCi/animal; specific activity 2.2–6.3 Ci/μmole) at 3 times: either prior to, minutes or hours after surgical brain injury consisting of microdialysis probe insertion (stereotaxic surgery) targeting the right striatum. ROI analysis was performed using PMOD software to co-register images and apply an atlas template.
Results
[3 H]CNS-5161 binding affinity to brain membranes was increased in presence of glutamate and glycine (Kd=2. 3+/−0.08 Vs. 5.6+/−0.4 nM in the absence of glutamate). In intact controls, the frontal cortex/cerebellum ratio was 1.2+/−0.15, blocking with MK801 gave 0.9+/−0.1 and NMDA pretreatment to open channels gave 1.7+/−0.2. [3 H]CNS-5161 data provided evidence that injury-induced glutamate release increases radiotracer affinity for the open channel and suggested that the changing slope of the washout would appropriately reflect radiotracer affinity. Consistently, there was a significant increase in [11C]CNS-5161 affinity immediately following the trauma (inversely proportional to a 72 and 95% decrease in the rate of dissociation in the left and right cortex, respectively) that receded by 2 hours after the surgery. There was no significant change in [11C]CNS-5161 affinity in the cerebellum at any point, consistent with in vitro data (13.6 and 6.7% change in the left and right washout, p<0.05).
Discussion
These results suggest CNS-5161 is a radiotracer with in vivo sensitivity to the activated state of NMDAR, whose affinity depends on glutamate-induced activation of the ion channel complex. Injured animals demonstrated more sustained uptake in cortex, hippocampus and striatum but not in the cerebellum, which was undistinguishable from controls. Future PET studies using [11C]CNS-5161 in stroke or TBI victims may help determine the functional status of the receptors in individual subjects and the potential efficacy of NMDAR inhibition or stimulation (See Figure 1).
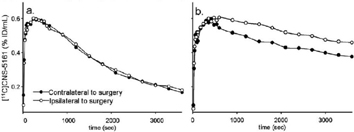
Time activity of [11C]CNS-5161 in the frontal/parietal cortex before (a) and 7 min after (b) stereotaxic surgery using a paired bolus paradigm.
