Introduction
In recent intravenous thrombolysis trials 1 , some benefit was noted for stroke patients treated between 3–6 h, though caution was advised for its administration beyond the recommended 3 h therapeutic time window 2 . It has been speculated that diffusion-weighted (DWI) and perfusion-weighted (PWI) MRI, may provide an objective guideline for identifying patients most likely to benefit. Multiparametric algorithms combining these modalities have been shown to accurately predict tissue infarction 3 . However, duration of ischemia has not been explicitly incorporated in such algorithms. This study investigates the spatio-temporal dynamics of infarction risk as a function of occlusion duration using a rat embolic stroke model.
Methods
Unilateral stroke was induced by embolic occlusion of the middle cerebral artery (n=8). DWI and PWI experiments were performed at 44±10, 76±5, 139±6, 259±6, 318±5 min post-occlusion. Apparent diffusion coefficient (ADC), CBF, CBV and mean transit time (MTT) maps were calculated, normalized with respect to contralateral values and used along with occlusion duration as covariates in predictive algorithms which outputs the probability of infarction on a voxel-wise basis 3 . Lesions were defined as tissue with ADC < 2 SD from contralateral values at the final MRI. Predicted lesion volumes consisted of tissue exhibiting > 50% risk.
Results
Figure 1 demonstrates the spatiotemporal evolution of infarction risk as a function of occlusion duration calculated using MR data acquired at the initial time point for one rat. The predicted risk of infarction was found to significantly increases with occlusion time for all animals (79±5%, 80±5%, 81±5%, 84±5%, 85±5%) (p<.05). Also evident is the spatial heterogeneity of infarction risk within the lesion. Predicted lesion volumes were significantly correlated to measured hemispheric lesion volume assuming different occlusion times (p<.05).
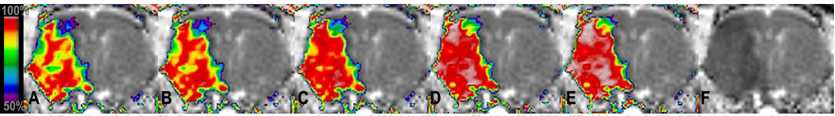
predicted risk of infraction assuming occlusion of (A) 52, (B) 85, (C) 150, (D) 273, (E) 329 min & (F) ADC values at 329 min.
Discussion
This study shows infarction risk increases with occlusion time, consistent with clinical and experimental experience. Importantly, these time-dependence findings were determined algorithmically without prior assumptions. This supports the concept that infarction likelihood depends on both depth and duration of ischemic injury. In addition, the changing spatial heterogeneity of infarction risk exhibited over time may reflect the transition of potentially salvageable to irreversibly damaged tissue. Further studies with histological validation are needed to fully explore this aspect. In conclusion, this study demonstrates the potential of statistical algorithms for improved characterization of ischemic injury as well as for identifying salvageable tissue after stroke.
