Purpose
Brain ischemia and reperfusion (I/R) cause phosphorylation of the alpha subunit of eukaryotic initiation factor 2 (eIF2 alpha), a reversible event associated with neuronal protein synthesis inhibition. However, irreversible translation arrest correlates with cell death. eIF2 alpha phosphorylation leads to formation of stress granules (SGs), cytoplasmic foci containing 48S preinitiation complexes and the mRNA binding protein TIA-1. SGs are sites of translationally inactive protein synthesis machinery, and their persistence may distinguish ischemic vulnerable brain regions. As cortical layers V and VI show ischemic vulnerability, we evaluated SG formation in layers II, V and VI neurons of motor cortex following global brain I/R.
Methods
SGs were evaluated by double-labeling immunofluorescence for two SG components: small ribosomal subunit protein S6 and TIA-1. 40X images were acquired in a Zeiss Apotome system. S6 was also evaluated by Western blot. Transient global brain ischemia was induced by cardiac arrest, and reperfusion was by critical care cardiopulmonary resuscitation with all animals maintained normothermic throughout. Experimental groups (n=3 per group) were: nonischemic controls (NIC), 10 min ischemia plus 10 min (10R), 90 min (90R) and 4 hr reperfusion (4hrR).
Results
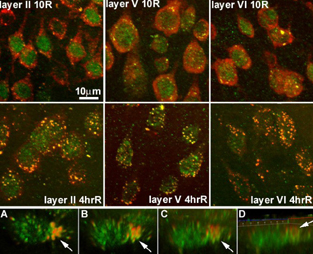
S6 staining (red) was cytoplasmic, and TIA-1 (green) was mainly nuclear. Colocalization of S6 and TIA-1 as yellow cytoplasmic foci defined SGs. Representative samples at 10R and 4hrR are shown in the Figure 1 for layers II, V and VI of motor cortex, as indicated. SGs were present at 10R in all three layers. SGs quantitatively increased in layers V and VI, but not II. By 4hrR, SGs coalesced into larger structures (Figure 1, middle panels). There was a noticeable loss of ribosomal protein S6 at 4hR that was most pronounced in layer 5, although there was cell to cell heterogeneity of S6 staining. By Western blot, S6 decreased 50% by 4hrR in unfractionated cortical homogenates. 3D reconstruction of a layer 5 pyramidal neuron showed SG localization mainly at the base of the apical dendrite, forming “trichromatic” columnar structures (rotation of 3D image shown in panels A-D, arrow points to SGs at base of apical dendrite. A is top view of neuron, D is side view).
Discussion
All three cortical layers showed evidence of SGs. By 4 hrR, SGs had become larger, and free S6 protein had decreased. Cells containing more S6 protein (a marker of the 40S ribosomal subunit) not confined to SGs will have a greater capacity for protein synthesis. That some layer 5 neurons showed large decreases in S6 protein may relate to the selective vulnerability of these neurons.
Footnotes
Acknowledgements
Work supported by NIH grant NS044100 (D.J.D.).
