Introduction
Poly-ADP-ribosylation is an important post-translational modification of proteins involved in DNA repair, maintenance of genomic integrity and transcriptional regulation. The addition of poly-ADP-ribose polymers (PADPR) is an energy requiring process utilizing NAD; and is mediated through poly-ADP-ribose polymerases (PARP). Although PARP plays an important role in cell homeostasis, over-activation under conditions of extensive DNA damage can lead to energy failure, and cell death via the release and nuclear translocation of mitochondrial apoptosis-inducing factor (AIF). We have previously demonstrated the presence of PARP and PADPR in the mitochondria. However, the specific mitochondrial targets of poly-ADP-ribosylation remain undefined.
Hypothesis
Target proteins of poly-ADP-ribosylation in the mitochondria include those involved in energetics and apoptosis.
Methods
We first verified the presence of PARP and PADPR in isolated rat brain mitochondria and in mitochondria of injured brain after experimental controlled cortical impact (CCI) by immuo-electronmicroscopy. To identify potential substrates of poly-ADP-ribosylation, immunoprecipitation using an antibody against PADPR was performed in isolated brain mitochondria after nitrosative stress with peroxynitrite and in mitochondrial-enriched fractions of injured brain after CCI, followed by small format 2-D gel electrophoresis. Peptides of interests were analyzed by matrix assisted laser desorption/ionization mass spectroscopy (MALDI-MS). Identified proteins of interest were then confirmed by standard Western Blot.
Results
Immuno-electronmicroscopy demonstrated the presence of PARP-1 and PADPR both in isolated mitochondria ex vivo and in mitochondria of injured brain in vivo. 2-D gel electrophoresis detected multiple poly-ADP-ribosylated mitochondrial proteins. Several potentially important proteins were identified as PARP substrates, including (see figure 1): the β subunit of F1F0 ATPase (complex V), voltage-dependent anion channel-1 (a key component of the mitochondrial permeability transition pore), cytochrome oxidase subunit Va, glyceraldehyde-3-phophate dehydrogenase and mitofilin (mitochondrial inner membrane protein).
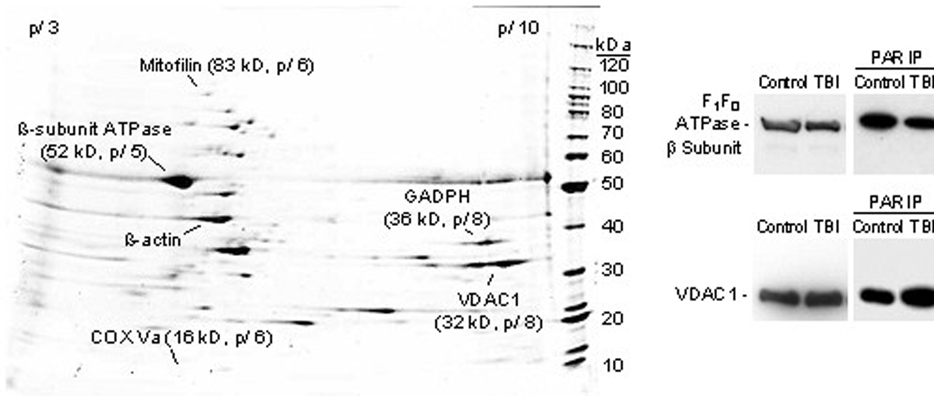
Conclusions
These data show that poly-ADP-ribosylation is a prevalent post-translational modification of mitochondrial proteins. The finding that components of the electron transport chain, glycolysis and mitochondria membrane permeability transition pore are substrates of poly-ADP-ribosylation points to additional mechanisms by which PARP may regulate mitochondrial energetics and contribute to the release of apoptogenic factors after brain injury.
Footnotes
Acknowledgements
Support: NS38620/NS30318/HD40686
