Introduction
Functional magnetic resonance imaging (fMRI) is a unique tool to study brain activity and plasticity changes. Most fMRI studies in rats have used α-chloralose to anesthetize the animals. Because of the organ toxicity and need for blood vessel catheterization, longitudinal studies cannot be performed with á-chloralose. Thus, we developed a totally noninvasive fMRI protocol, suitable to study recovery processes, using the α2-agonist medetomidine in combination with transcutaneous monitoring of blood gases, yielding robust patterns of functional brain activation at different time points, comparable with the results obtained under α-chloralose.
Methods
Wistar rats (n=4) were anesthetized at three different time points with halothane. After placement of a transcutaneous blood gas system (TCM4, Radiometer Copenhagen, Denmark) and forepaw stimulation electrodes, anesthesia was switched to a subcutaneous bolus (0.05 mg/kg) and continuous infusion (0.1 mg/kg/h) of medetomidine in the first two experiments, and intravenously applied α-chloralose (36 mg/kg/h) in the third experiment. In 3 additional rats, only one fMRI experiment was performed under medetomidine. After each fMRI session, medetomidine was antagonized with an intraperitoneal injection of atipamezole. BOLD fMRI experiments were performed on a 7 T animal scanner (Bruker BioSpin, Ettlingen, Germany). After pilot scans, multislice SE-EPI images were acquired using the following parameters: FOV: 2.56×2.56 cm2; 64×64 points; TR=3 s, TE = 30 ms. Both forepaws were stimulated alternately with rectangular pulses (2 mA, 3 Hz, 0.3 ms). 115 EPI images were acquired using a paradigm in which ON vs. OFF stimulation periods were switched in a 60 s cycle (15 OFF + 5 ON), repeated 5 times, and ending with 15 OFF images. Brain activation maps were constructed using a t-test (p<0.01) in the program STIMULATE (University of Minnesota, USA).
Results
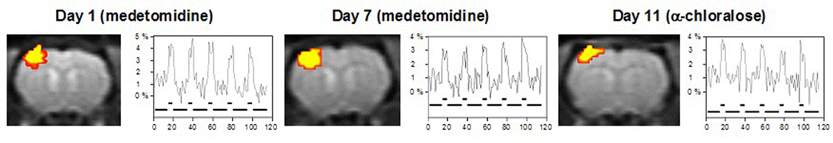
All animals tolerated the non-invasive fMRI experiments very well and fully recovered within 2 minutes after injection of atipamezol. Identical and highly reproducible BOLD activation areas were observed in the contralateral primary somatosensory cortex during forepaw stimulation using both medetomidine and α-chloralose (Fig. 1). The amplitudes of the BOLD signal were well comparable during stimulation under both anesthetics. Transcutaneous blood gas monitoring assured no significant changes in pCO2 levels during experiments.
Discussion
Using the α2-agonist medetomidine, we were able to perform repetitive and highly reproducible fMRI studies after forepaw stimulation in rats. Together with a continuous monitoring of transcutaneous pCO2 values, our protocol can be used for totally noninvasive fMRI studies, allowing to perform longitudinal studies in the same animal to study functional recovery processes upon therapeutical treatment.
