Background
Spinal motor neurons are one of the most vulnerable cells to hypoxia/ischemia, and are also selectively damaged in amyotrophic lateral sclerosis (ALS), a fatal neurodegenerative disease. Recently, mice with deletion of the hypoxia-response element (HRE) in the vascular endothelial growth factor (VEGF) promoter gene have been generated. The mice showed late-onset progressive and selective motor neuron degeneration reminiscent of human ALS, and therefore, the vulnerability of motor neurons against hypoxia has been argued as the possible mechanism of the disease.
Methods
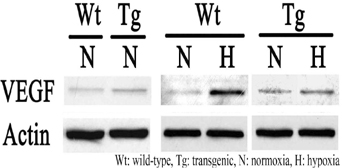
Localization and hypoxic induction of VEGF was examined in the spinal cord of transgenic mice carrying a mutation (G93A) in the superoxide dismutase 1 gene, an animal model of ALS. Animals were exposed to hypoxia (7% oxygen) for 2 hours, and sacrificed 6 hours after hypoxia. Immunohistochemical, immunofluorescent, and immunoblotting analyses were performed in transgenic (Tg) mice and wild-type (Wt) littermates (n=6, respectively) under normoxia and hypoxia.
Results
Immunohistochemical and immunofluorescent study demonstrated that VEGF is mainly expressed in motor neurons before and after hypoxia. In immunoblotting, baseline expression of VEGF was higher in Tg mice than in Wt littermates. However, VEGF was hardly induced after hypoxia in Tg mice, whereas Wt mice showed approximately a 9-fold increase. Impaired VEGF induction was obvious in Tg mice at 12 weeks of age, when they were still presymptomatic. In contrast, baseline and hypoxic expression of brain derived neurotrophic factor and glial cell line-derived neurotrophic factor did not differ between Tg and Wt mice.
Conclusion
The present study demonstrated that hypoxic induction of VEGF in Tg mice is selectively impaired from an early stage, suggesting profound involvement in the pathogenesis of motor neuron degeneration in hypoxia, as well as ALS (See Figure 1).