Introduction
Consolidated ischaemic brain lesions are invariably associated with lesions of the blood brain barrier (BBB) and increased permeability of tracers of almost all molecular sizes. In contrast to necrosis, excitotoxic brain lesions are often thought not to involve, at least, early changes in the cerebral vasculature.
The object of this preliminary investigation was to study the kinetics of evolution of the brain lesions and the BBB properties following the injection of an excitotoxic agent (NMDA) into the striatum.
Methods
Under sevoflurane anaesthesia, female Sprague dawley rats were placed in a stereotaxic frame and a cannula was inserted into the ventral striatum (coordinates 0 mm posterior, 3 mm lateral, 6 mm ventral to the bregma). NMDA (75 nmol) was injected in a 3 μl volume of PBS. In control animals, only PBS was administered into the striatum. Euthanasia was performed 3, 6, 24 and 48 hours after the administration of NMDA. Two hours before euthanasia, each animal received an intravenous injection of 1 ml of a 2% solution of Evans blue which binds to albumin (EBA, 65 KDa). The brains were cut in 20 μm sections using a cryostat, and 2 adjacent sections in every 40 were collected, one for quantification of the lesion and the other for evaluation of Evans blue extravasation, an indicator of changes in BBB permeability.
Results
The administration of NMDA into the striatum induced well demarcated areas not stained with thionin, as early as 3 hours following the injection. The volume of this lesion displayed a significant and progressive enlargement during, at least the first 24 hours (figure 1a). The administration of the vehicle did not induce any discernable lesion at all the time points examined. The extravasation of Evans blue was visible even 3 h following the administration of NMDA and also displayed a progressive evolution with a temporal profile not dissimilar to that of the excitotoxic lesion (figure 1b)
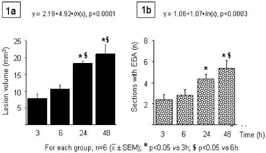
Conclusions
The findings indicate that in rat, NMDA-induced extravasation of Evans blue evolves in parallel to the volume of neuronal loss. Many points remain to be resolved: the molecular size of non-permeable tracers; the degree of plasma binding; the ultrastructural basis of the changes in BBB permeability. Nonetheless, the use of this model of brain lesion should take into account these alterations in the cerebral vasculature.
