Introduction
Neurologic and neuropsychologic deficiencies are commonly found following cardiac arrest and cardiopulmonary resuscitation (CPR). Subtle changes in blood-brain barrier (bbb) permeability have been known to occur in experimental animal models 1 . It has previously been shown, using a cortical freezing injury model, that iron oxide nanoparticles can accumulate in damaged endothelial cells following i.v. injection, allowing mapping of the disrupted BBB-area by MR imaging 2 . We assessed whether these iron oxide contrast agents would allow MRI detection of endothelial permeability changes in the brain following cardiac arrest.
Materials and Methods
Cardiac arrest was induced in C57/BL6 mice by i. v. injection of KCl, with body cooling to 27 C and heating of the brain at 39 C. After 10 min of cardiac standstill, CPR was initiated and MION-46L nanoparticles were administered (1 mmole Fe/kg) immediately following CPR (n=3). As controls, two sham-operated and one normal animal received the same dose of MION-46L, and one cardiac arrest/CPR animal received no contrast agent. After 24 hrs following injection, the brains were removed for high-resolution MR imaging. Imaging was performed on a 9.4 Tesla instrument, using T2- and T2*-weighted imaging with a resolution of 70×66×63 μm. Histopathological correlation was performed using DAB-enhanced Prussian Blue staining for iron.
Results
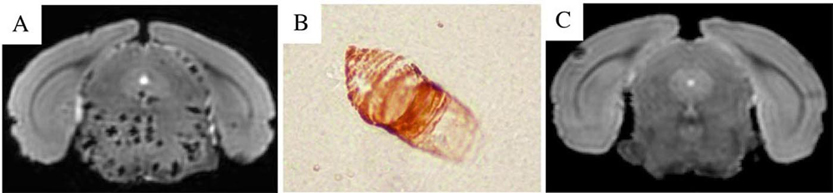
For all injured animals receiving MION-46L, numerous hypointense lesions were visible throughout the brain stem (Figure 1A), but not in other parts of the cerebrum or cerebellum. The lesions appeared substantially larger on the T2*-weighted images due to the iron-induced magnetic susceptibility effects. Histology revealed many dilated vessels in the brain stem containing endothelial cells that were uniformly filled with iron-positive magnetic nanoparticles (Figure 1B). No such lesions, either on MRI or histology, were observed in the four different controls (Figure 1C).
Discussion
We conclude that cardiac arrest followed by resuscitation can lead to a hitherto unreported damage of brain stem endothelial cells. We hypothesize that a transient high blood pressure (from a no-flow status) induces a loss of endothelial cell membrane integrity, resulting in leakage and uptake of the iron oxide nanoparticles. It is not known why the brain stem appears primarily affected, but it may be related to the predominant occurence of vasogenic edema in the brain stem of patients with essential hypertension, often referred to as hypertensive encephalopathy 3 .
