Introduction
Vascular endothelial growth factor (VEGF) mediator family and their receptors are involved in co-ordinated regulation of angiogenesis and neurogenesis after stroke 1 . We have recently shown that placenta-derived growth factor (PlGF), a ligand for both VEGFR1 and neuropilin-1 (NP-1), plays a permissive role for VEGF- and astrocyte-induced angiogenic responses of human brain endothelial cells (BEC) 2 . The role of PlGF in hypoxia-induced angiogenesis remains unclear.
Methods
The role of PlGF in hypoxia-induced brain angiogenesis was investigated using cultured BEC and astrocytes (AST) from PlGF wild-type (PlGF+/+) and knockout mice (PlGF−/−). Cells were subjected to either 3-h or 6-h hypoxia alone or followed by a 16-h reoxygenation. Capillary-like tube (CLT) formation by BEC grown in MatrigelTM (an in vitro measure of angiogenesis) was determined as described 2 . The expression of PlGF and VEGF/PlGF receptor mRNA was determined by quantitative PCR.
Results
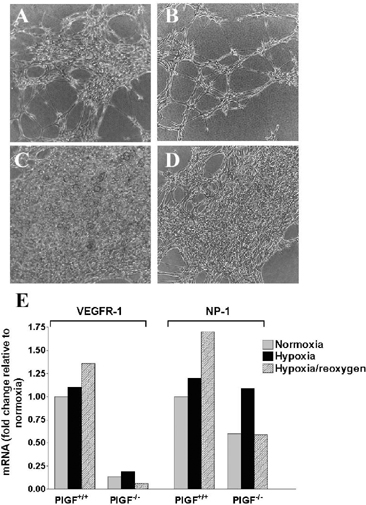
PlGF mRNA increased >2 fold in both BEC and AST from PLGF+/+ animals subjected to hypoxia, but returned to control levels after 16 h of reoxygenation. PlGF+/+ BEC exposed to 3 (Fig 1A) or 6 (Fig 1B) h hypoxia followed by 16 h reoxygenation formed increasing number of CLTs, whereas PlGF−/− BEC failed to respond to hypoxia by angiogenic conversion (Fig. 1C–D). VEGFR1 (Fig. 1E) and neuropilin-1 (NP-1, Fig. 1E) mRNA expression was significantly lower (∼10 and 2-fold, respectively) in PlGF−/− compared to PlGF+/+ BEC. A significant up-regulation of both VEGFR1 and NP-1 mRNAs in PLGF+/+ BEC was noted during reperfusion, whereas NP-1 mRNA was transiently up-regulated during hypoxia in PLGF−/− BEC (Fig 1E). VEGFR-2 expression was not detected in BEC of either genotype.
Conclusions
The results suggest that PlGF may be important regulator of hypoxia-induced angiogenic transformation of BEC, since both BEC and AST up-regulate PlGF mRNA in response to hypoxia and PlGF−/− BEC fail to respond to hypoxia by forming CLTs. The lower basal and hypoxia/reoxygenation-induced VEGFR1 and NP-1 expression in PLGF−/− BEC likely contribute to poor angiogenic response of PlGF−/− BEC.
