Tumor necrosis factor-α (TNFα), a proinflammatory cytokine can play a prominent role in CNS injury putatively in part through cerebrovascular dysfunction. The aim of our study was to demonstrate how cerebral blood flow and microvascular morphology are affected by circulating TNF-α.
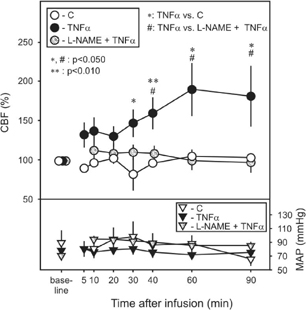
Fifty male Wistar rats were anesthetized with chloral hydrate (400 mg/kg, i.p.). Five animals were administered by 2.5 μg/kg TNFα (in 1 ml saline) infused unilaterally to the left common carotid artery. Control animals received 1 ml saline (n=5). Another group of rats received l-nitroarginine methyl ester (20 mg/kg/0.5 ml/2 min, L-NAME), a nitric oxide synthase (NOS) inhibitor into the right femoral vein prior to TNFα or saline infusion (n=5/group). Mean arterial pressure (MAP) was monitored through a tail artery cannula, while cerebrocortical blood flow (CBF) was recorded by a laser-Doppler probe positioned above the frontoparietal cortex. In a next set of experiments, rats were infused with TNFα or saline, and were sacrificed for electron microscopic analysis at various survival times (45 min, 4 h and 8 h post-infusion, n=5/group). While MAP values did not differ between groups, TNFα significantly increased cortical CBF with the progress of time to 190 % of baseline at 60 min after TNFα infusion. The administration of L-NAME prevented TNFα-induced CBF elevation. Damage to the blood-brain barrier (BBB) was observed in the form of perivascular edema. Approximately 50% of cerebrocortical capillaries demonstrated severe swelling of astrocytic endfeet at 45 min, while about 65 % of the vessels were damaged at 8 h post-infusion. The average lumen diameter of investigated capillaries decreased gradually from 4.5 μm to 3.6 μm by 8 h after TNFα infusion. Our results demonstrate that circulating TNFα is a vasoactive compound in the CNS. It affects cerebral blood vessels both at functional and morphological levels. Since NOS inhibition could prevent CBF increase, TNFα action is suggested to be mediated by NO from an unclarified source. Further, our data support the notion, that circulating TNFα can impose morphological damage at the BBB and can lead to CNS complications (See Figure 1).
Footnotes
Acknowledgements
The project was supported by the Hungarian Scientific Research Fund (OTKA F042803, T32566 and T046531), and the Bolyai János Research Scholarship of the Hungarian Academy of Sciences to E.F.
