Overproduction of the amyloid ß (Aß) peptide is a key factor in the pathogenesis of Alzheimer's disease (AD), but the mechanisms of its pathogenic effects have not been defined. Patients with AD have cerebrovascular alterations attributable to the deleterious effects of Aß on cerebral blood vessels. Furthermore, mice overexpressing the amyloid precursor protein (APP) have marked alterations in the cerebrovasodilation produced by neural activity (functional hyperemia). Experimental evidence suggests that these cerebrovascular alterations are mediated by reactive oxygen species (ROS). However, the source of the ROS mediating the dysfunction has not been defined. NADPH oxidase has recently emerged as a major source of ROS at the vascular level. In this study we tested the hypothesis that NADPH-derived ROS mediate the vascular dysfunction produced by Aß.
Methods
The increases in somatosensory cortex cerebral blood flow (CBF) induced by stimulation of the facial whiskers were monitored in urethane-chloralose anesthetized mice using laser-Doppler flowmetry. ROS production in the somatosensory cortex was monitored by the hydroethidine technique. The cerebrovascular effects of Aß were studied either by Aß1-40 superfusion on the exposed neocortex, or in mice overexpressing APP (Tg2576).
Results
The alterations in functional hyperemia induced by Aß (Figure 1A) were abrogated by the NADPH oxidase peptide inhibitor gp91ds-tat (ds; figure 1A), but not by its scrambled control (sds). CBF responses to adenosine were not affected by Aß (28±2%) or Aß+ds (30±3; n=5; p>0.05). The ds peptide blocked ROS production (n=5/group). Furthermore, the cerebrovascular dysfunction and ROS upregulation observed in wild-type mice (WT) did not occur in double-transgenic mice overexpressing the amyloid precursor protein but lacking the critical NADPH oxidase subunit gp91phox (Tg2576 gp910/0; Figure 1B).
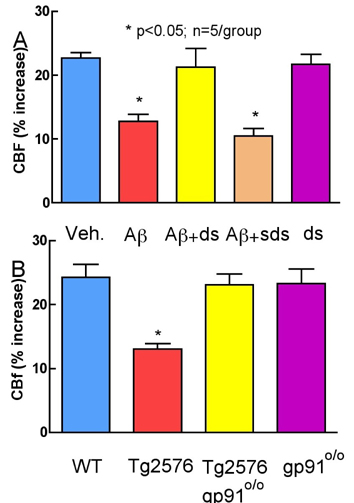
Conclusions
A gp91phox-containing NADPH oxidase is the critical link between Aß and cerebrovascular dysfunction, which may underlie the alteration in cerebral blood flow regulation observed in AD patients.
