Human albumin (ALB) therapy is highly neuroprotective in preclinical models of cerebral ischemia. Multiple mechanisms underlie ALB's efficacy: fatty-acid and transition-metal binding, antioxidant and oncotic actions, and salutary microcirculatory effects.
Design
In this dose-escalation trial, ALB (25% solution) was administered within 16 h of stroke onset to subjects with acute ischemic stroke and NIH Stroke Scale scores (NIHSSS) ≥ 6. Two cohorts were assessed: 1) those also receiving IV tPA; and 2) those not given tPA. Seventy subjects have been enrolled into 5 ALB dose-tiers: I [0.34 g/kg; n=9 (tPA) and 8 (non-tPA)]; II [0.68 g/kg; n=5 and 6], III [1.03 g/kg; n=6 and 6]; IV [1.38 g/kg; n= 9 and 9]; and V [1.71 g/kg; n=6 and 6]. Mean age (± SD) was 66 ± 15 years; 38 were male. ASPECTS CT-scan score was 8.1 ± 2.3. The 2-h ALB infusion was begun at 8.2 ± 3.7 h after stroke onset. Post-discharge assessments (modified Rankin (mRS), Barthel, NIHSSS) were conducted at 1 and 3 months.
Safety
Six of the 70 subjects died; all had severe strokes (NIHSSS 23-38). Pulmonary edema occurred in 3 subjects of tier IV (but in none of tier V, when prophylactic administration of furosemide was encouraged). One serious adverse event (SAE) (congestive failure with prolonged hospitalization) in tier III was possibly ALB-related. No tier IV or V subject had ALB-related SAE's.
Clinical course
Mean NIHSSS was 13. 7 ± 6.6 initially; 8.8 ± 8.6 at 72 h; and 5.4 ± 6.6 at 3 mo. In both cohorts, NIHSSS tended to improve more extensively over time at higher ALB dose-tiers, particularly in the tPA cohort (Figure 1). Overall, mean NIHSSS declined from 14.1 (initial) to 7.8 (3 mo) in tier I, from 16.1 (initial) to 3.4 (3 mo) in tier IV, and from 8.7 (initial) to 3.6 (3 mo) in tier V. Of note, the tier IV and tier V ALB doses (1.37 and 1.71 g/kg) exceeded the highly-protective per-kg dose in preclinical focal-ischemia studies (1.25 g/kg). In the tPA subgroup (n=35), tPA was given at 2.3 ± 0.9 h post-stroke onset, and i.v. ALB was started at 6.6 ± 3.2 h. By contrast, in the non-tPA subgroup, i.v. ALB was started 3 hours later (9.6 ± 3.5 h). In the tPA subgroup, significantly greater improvement in NIHSSS was observed at the higher ALB dose-tiers (IV + V) than at lower dose-tiers (Figure 1).
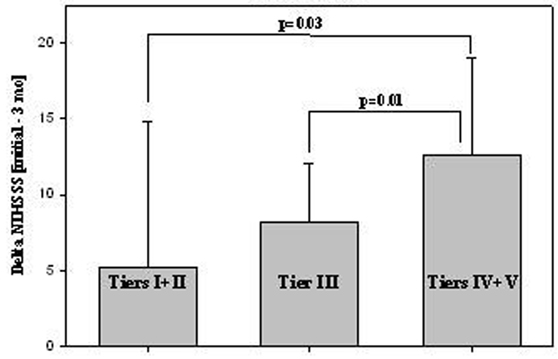
Conclusions
ALB therapy is well tolerated, and data suggest that higher-dose ALB may improve neurological outcome. We are planning to implement a randomized multicenter, placebo-controlled Phase III trial of this therapy – the ALIAS Trial.
Footnotes
Acknowledgements
Supported by NIH Grants NS40406 and NS48784.
