Background and Purpose
Immunophilin ligands, such as cyclosporin A and FK506, have neuro-protective effects in experimental stroke models, although the precise mechanism is unclear. Cyclophilin C-associated protein (CyCAP) is a natural cellular ligand for the immunophilin, cyclophilin C, and has a protective effect against endotoxins by down-modulating the proinflammatory response. The present study examined the pattern of expression of CyCAP and cyclophilin C mRNA within the ischemic hemisphere using Northern blotting and in situ hybridization at several time points (at 2 hours, 24 hours, 3 days, and 7 days) after focal cerebral ischemia.
Methods
Permanent middle cerebral artery occlusion model (Tamura model) was employed in halothane anesthetized adult Sprague Dawley rats (300–330 g, n= 30). Rats were decapitated at 2 hours, 24 hours, 3 days, and 7 days after ischemia. Sections of 20 μm thickness were cut on a cryostat and were stained with hematoxylin and eosin, and examined by light microscopy. For in situ hybridization, these sections were applied by Digoxigenin -labeled antisense cRNA for CyCAP and cyclophilin C probes. For Northern blot analysis, total RNA was isolated from dissected region such as ipsilateral remote cortex, ischemic core cortex, and contralateral cortex. The [α-32P] UTP- labeled cRNA probes for CyCAP and cyclophilin C were used.
Results
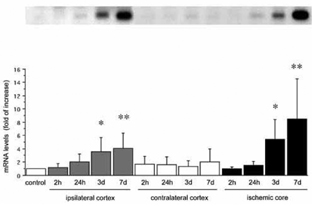
Both CyCAP and cyclophilin C mRNAs were ubiquitously distributed in the neurons of the normal brain. Expression increased in neurons of the peri-infarct zone up to 7 days after MCA occlusion. The neuronal distribution was confirmed by counter-immunostaining of NeuN. Both mRNAs were predominantly expressed in microglia of the ischemic core at 7 days, confirmed by immunostaining with the microglial marker, ED1. The quantification of CyCAP and cyclophilin C mRNAs at 7 days by Northern blot analysis showed the 8.5 fold increase (P<0.005, n=6) and 6.8 fold increase (P<0.005, n=6), respectively, in ischemic core compared with control.
Conclusions
The observed changes in CyCAP and cyclophilin C mRNA expression in response to permanent focal cerebral ischemia strongly suggest that these molecules participate in intrinsic tissue protection by providing neuroprotection as a mimic of cyclosporin A; by recruiting microglia/macrophages to remove potentially deleterious debris, promote tissue repair by secreting growth factors, and facilitate the return to tissue homeostasis; and by suppressing pro-inflammatory cytokines in response to ischemia. Preservation of these effects could represent a novel pharmacological approach to counter the inflammatory reaction triggered by cerebral ischemia. In addition, overexpression of CyCAP in the penumbra suggests a role in cell survival, an intriguing possibility that deserves further investigation (Figure 1).