Purpose
NFκB is activated in brain cells after various insults, including cerebral ischemia. In cytoplasm, NFκB is tightly associated with inhibitory protein, IκBα. On activation by H2O2, IκBα is phosphorylated and degraded, exposing the nuclear localization signals on NFκB heterodimer. Cyclooxygenase (COX2) is known as an NFκB-inducible gene, expressed in leukocytes and brain. It is an important mediator of cell injury in inflammation. We investigated the effect of oxygen glucose deprivation (OGD) on IκBα phosphorylation, NFêB activation, COX2 expression and the relation of OGD to cell survival in SOD1 transgenic (Tg) mice.
Methods
Primary astrocytes from postnatal day 2–3 SOD1 Tg and wild type (Wt) mice were cultured for 14 days. Cells were subjected to OGD for 4 hrs and collected for the indicated time points. Small interfering RNAs (siRNA) of NFκB p50 were used to suppress the gene expression. NFκB p50, IκBα, phospho-IκBα and COX2 antibodies were used for immunoblotting. Cell death was measured by LDH assay.
Results
In Wt and SOD1 Tg astrocytes, IκBα level decreased rapidly after OGD, and recovered to basal level by 4 hr of reperfusion (Fig. 1). Phospho-IκBα was highly expressed after OGD and 2 h reperfusion in both cell types, which corresponded with the decreased level of IκBα at these time points. Cytosolic level of NFκB p50 in Wt gradually increased after OGD and decreased by 3.7-fold during reperfusion. NFκB p50 level increased by 1.9-fold after OGD in SOD1 Tg astrocytes. The reduced cytosolic NFκB p50 was consistent with for the increase in nuclear translocation. SOD1 transfected astrocytes exhibited enhanced elevation of NFêB at 2 hr of OGD. COX2 level was induced by 6-fold and 1.9-fold after OGD in Wt and SOD1 Tg astrocytes (Fig. 2). SOD1 Tg cells were3.3-fold more protected than Wt cells after OGD (p<0.05, n=6). However, SOD1 Tg cells transfected with siRNA showed 8-fold higher cell death rates and elevated COX2 expression. Immunostaining showed similar results.
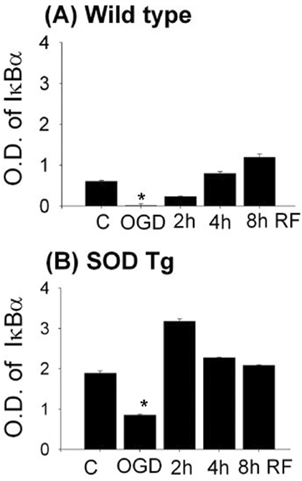
IκBα protein in Wt and SOD astrocytes after OGD and reperfusion.
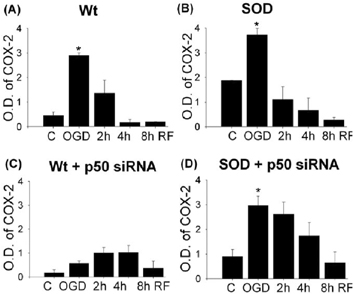
COX-2 levels in Wt (A), SOD(B) and SiRNA transfected astrocytes (C, D) after OGD reperfusion.
Conclusions
After OGD, IκBα was rapidly phosphorylated, leading to decreased IκBá and translocation of NFκB into the nucleus. NFκB had a protective role in cell survival after oxidative injury. However, blocking NFκB p50 expression by siRNA led to attenuation of COX2 expression in Wt, but enhanced COX2 expression and cell death in SOD1 Tg astrocytes. These results suggest that the transcriptional mechanism involved in COX2 gene expression may be different in SOD1 Tg astrocytes after OGD.
Footnotes
Acknowledgements
Supported by P50NS14543, PN5037520, RO1NS25372, RO1NS36147, RO1NS38653 and AHA Bugher Foundation.
