Abstract
The ability to discriminate between Alzheimer’s disease (AD), subcortical vascular disease, and other cognitive disorders is crucial for diagnostic purposes and clinical trial outcomes. Patients with primarily subcortical vascular disease are unlikely to benefit from treatments targeting the AD pathogenic mechanisms and vice versa. The Gothenburg mild cognitive impairment (MCI) and dementia studies are prospective, observational, single-center cohort studies suitable for both cross-sectional and longitudinal analysis that outline the cognitive profiles and biomarker characteristics of patients with AD, subcortical vascular disease, and other cognitive disorders. The studies, the first of which started in 1987, comprise inpatients with manifest dementia and patients seeking care for cognitive disorders at an outpatient memory clinic. This article gives an overview of the major published papers (neuropsychological, imaging/physiology, and neurochemical) of the studies including the ongoing Gothenburg MCI study. The main findings suggest that subcortical vascular disease with or without dementia exhibit a characteristic neuropsychological pattern of mental slowness and executive dysfunction and neurochemical deviations typical of white matter changes and disturbed blood–brain barrier function. Our findings may contribute to better healthcare for this underrecognized group of patients. The Gothenburg MCI study has also published papers on multimodal prediction of dementia, and cognitive reserve.
Keywords
Introduction
Several attempts have been made, largely based on results from animal models and familial Alzheimer’s disease (AD) cases, to counteract the effects of amyloid-
Cerebrospinal fluid (CSF) markers—a key focus in the Gothenburg studies—are assumed to reflect pathogenic events as they occur in the brain directly in living patients. Incipient and manifest AD are characterized by increased CSF levels of phosphorylated tau (P-tau) linked to formation of neurofibrillary tangles, and ‘total’ tau (T-tau) mirroring axonal degeneration together with decreased levels of A
Subcortical vascular disease in white and gray matter is a common disorder, especially among the elderly, but often overlooked in clinical practice. In one prevalence study, it was found to constitute as much as 50% of cases with vascular dementia. 10 In its pronounced pure form, subcortical vascular disease gives rise to a characteristic phenotype with predominant speed/attention and executive impairments called subcortical (ischemic) vascular dementia (SVD). 11 Not only angiopathic lesions of the nonamyloid hypertensive type (arteriolosclerosis) but also cerebral amyloid angiopathy may play a role in the pathogenesis of SVD. Both types of lesions may lead to parenchymal changes involving white matter microinfarcts, lacunar infarcts, rarefaction, and microbleeds. 12 Hitherto there are only few CSF studies on patients with SVD. 13
The neuropathology underlying cognitive impairment in later life is often a combination of AD and microvascular brain damage, which may overlap and synergize to amplify the risk of clinical symptoms. 14 The clinical counterpart of the combination of AD and subcortical vascular disease, mixed-type dementia (MixD), is less studied and its prevalence among patients with dementia is unclear. A significant span in reported prevalence rates (2% to 60%) for the combination of AD and vascular dementia have been reported. 15
Dementia means cognitive dysfunction pronounced enough to lead to impaired activities of daily living, whereas cognitive impairment refers to the whole range of severities from the very mild to the most pronounced manifestations. Diagnostic criteria for AD and cerebrovascular disease associated with cognitive impairment have focused on the most severe form of cognitive impairment, i.e., dementia. However, the focus has shifted to identify the early characteristics of pathologic changes taking place in patients with MCI that will progress to dementia. They are better candidates for clinical trials since they are in the early phases of disease with lower burden of pathology.
In addition, most data suggest that pathologic brain changes start to accumulate many years, perhaps even decades, before they result in readily identifiable clinical symptoms. 14 Here, refined biomarker tools, representing cognitive, neuroimaging, and neurochemical alterations, may prove particularly useful to outline the underlying disease process.
The Gothenburg MCI and dementia studies attempt to increase the knowledge about subcortical vascular disease with and without AD and its relationship to pure AD at early and manifest stages. The design of the Gothenburg MCI study is presented in a sister publication (Wallin et al., p. 114, this issue). Here, we summarize the results of the ongoing Gothenburg MCI study and its predecessors.
The Gothenburg mild cognitive impairment and dementia studies
Publications from the Gothenburg MCI and dementia studies and the Gothenburg part of the LeukoAraiosis and DISability (LADIS) study are included in the current overview. The dementia studies consist of the prospective dementia study (P study) and the revised prospective dementia (P-rev) study; both predecessors to the Gothenburg MCI study.
Neuropsychological findings.
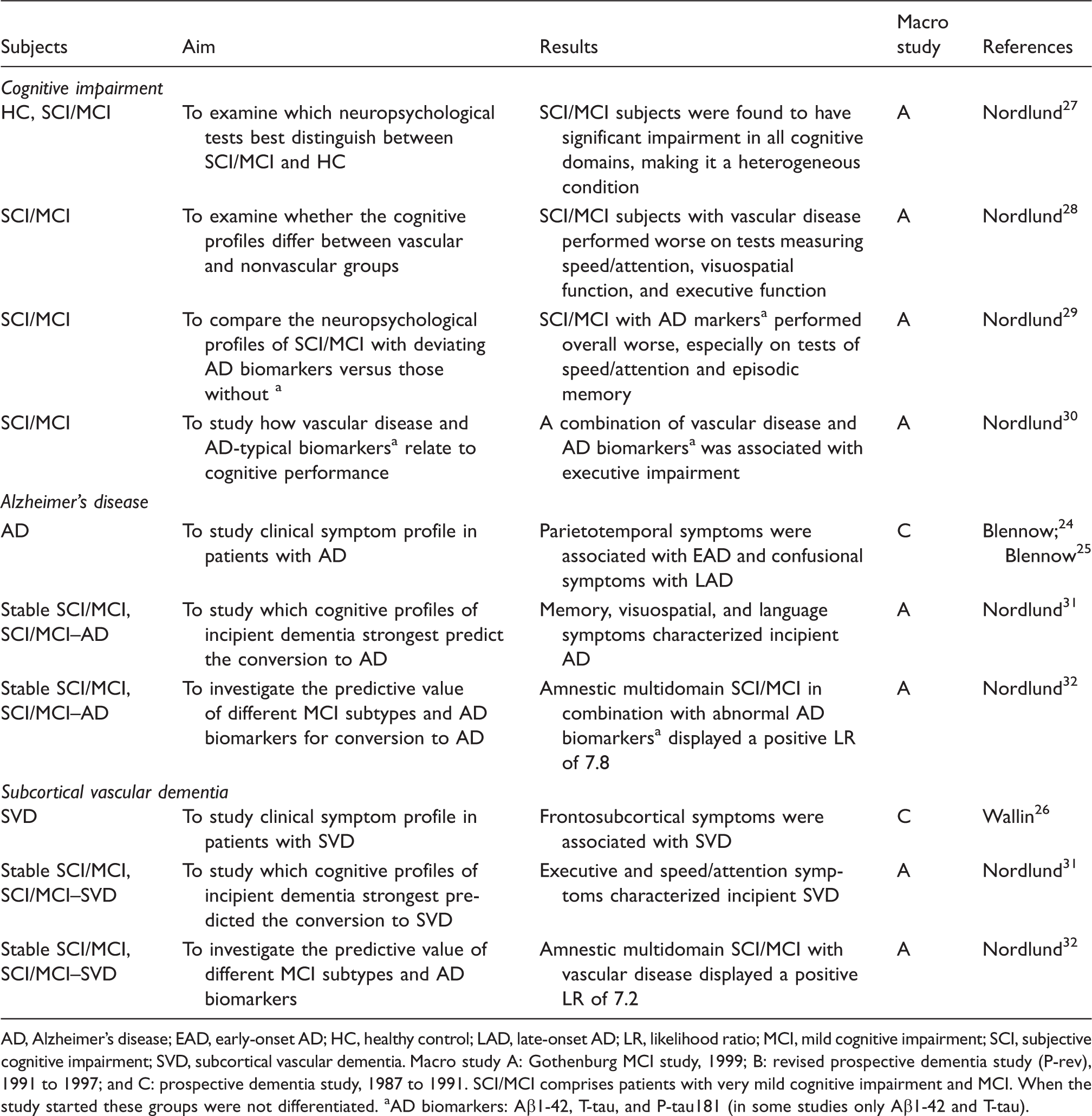
AD, Alzheimer’s disease; EAD, early-onset AD; HC, healthy control; LAD, late-onset AD; LR, likelihood ratio; MCI, mild cognitive impairment; SCI, subjective cognitive impairment; SVD, subcortical vascular dementia.
Macro study A: Gothenburg MCI study, 1999; B: revised prospective dementia study (P-rev), 1991 to 1997; and C: prospective dementia study, 1987 to 1991.
SCI/MCI comprises patients with very mild cognitive impairment and MCI. When the study started these groups were not differentiated.
AD biomarkers: Aβ1-42, T-tau, and P-tau181 (in some studies only Aβ1-42 and T-tau).
Brain imaging and physiologic findings.
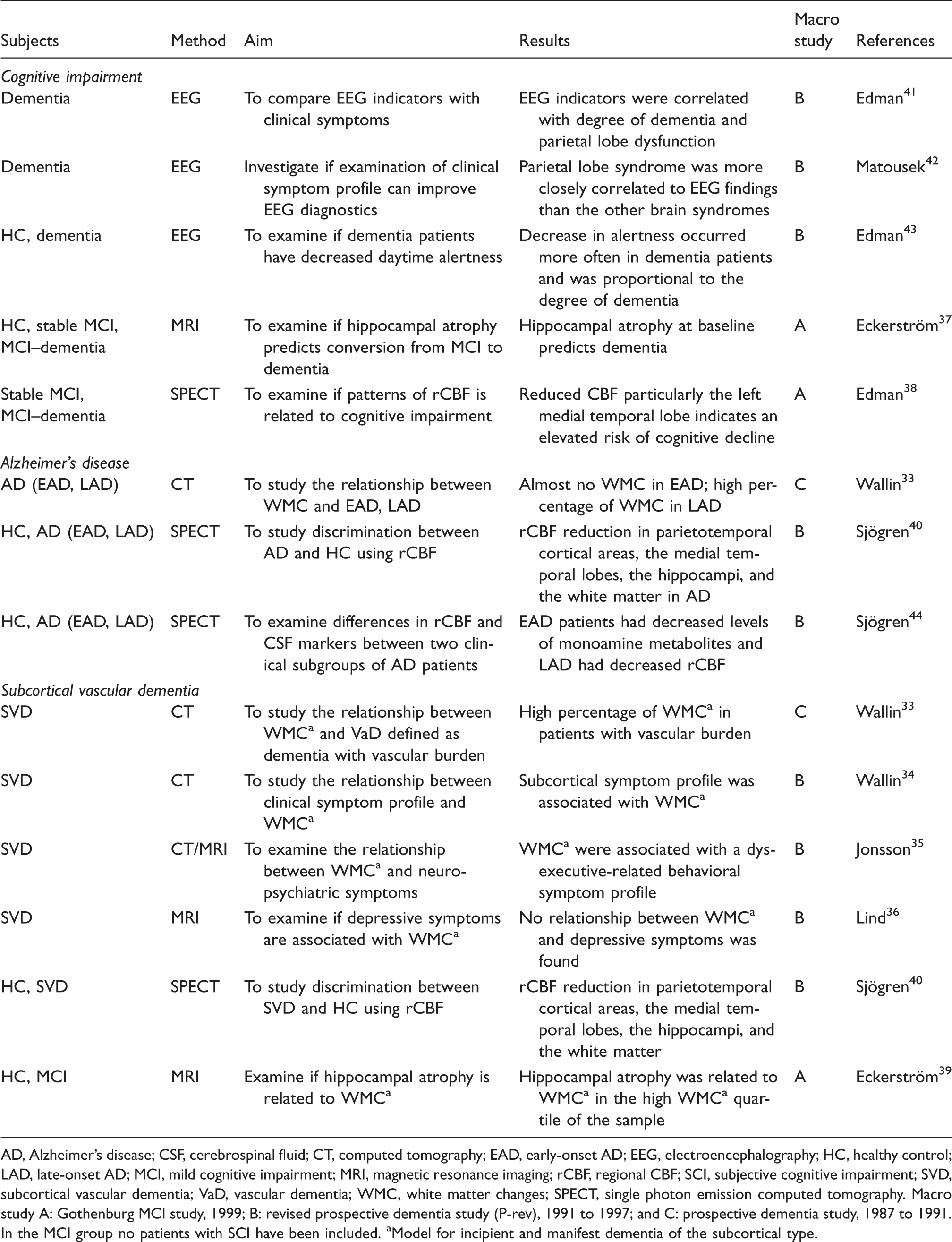
AD, Alzheimer’s disease; CSF, cerebrospinal fluid; CT, computed tomography; EAD, early-onset AD; EEG, electroencephalography; HC, healthy control; LAD, late-onset AD; MCI, mild cognitive impairment; MRI, magnetic resonance imaging; rCBF, regional CBF; SCI, subjective cognitive impairment; SVD, subcortical vascular dementia; VaD, vascular dementia; WMC, white matter changes; SPECT, single photon emission computed tomography.
Macro study A: Gothenburg MCI study, 1999; B: revised prospective dementia study (P-rev), 1991 to 1997; and C: prospective dementia study, 1987 to 1991.
In the MCI group no patients with SCI have been included.
Model for incipient and manifest dementia of the subcortical type.
Biochemical findings.

AAT, α-1-antitrypsin; Aβ, amyloid-β; AChE, acetylcholinesterase; AD, Alzheimer’s disease;
Macro study A: Gothenburg MCI study, 1999; B: revised prospective dementia study (P-rev), 1991 to 1997; C: prospective dementia study, 1987 to 1991; L: LeukoAraiosis and DISability (LADIS study, 2001).
AD biomarkers: Aβ1-42, T-tau, and P-tau181 (in some studies only Aβ1-42 and T-tau).
No obvious separation between SCI and MCI has been performed. Therefore, in the MCI group SCI patients may have been included.
The Gothenburg MCI study is an ongoing longitudinal, clinical–observational, single-center study using a predefined case report form. It is designed to obtain clinical, neuropsychological, neurochemical, genetic, and imaging/physiologic knowledge about cognitive impairment in patients seeking care or referred to an outpatient memory clinic. In particular, the study deals with AD, SVD, and associated disorders at their early and manifest stages. The nosological approach is in agreement with the basic ideas of the P and P-rev studies. However, the Gothenburg MCI study specifically focuses on the earlier manifestations of the diseases, which is at variance with the P and P-rev studies. The recruitment of patients started in December 1999 and the examinations performed are part of the clinical practice. Details about the design of the study, diagnostic procedures, methodology and baseline, and follow-up characteristics after 2 and 6 years have been described in a sister publication (Wallin et al., p. 114, this issue). The diagnostic procedures were strictly standardized in accordance with established criteria.4,16– 21 The syndromal and etiological entities were systematically diagnosed by experienced clinicians masked to psychometrics, CSF, and quantitative imaging data.
The examinations included methods from various modalities. The cognitive modality consisted of neuropsychological testing comprising
The LADIS study is a European multicenter collaboration that started in 2001 with the aim of assessing the role of white matter changes (WMC) in predicting disability in subjects aged 65 to 84. 23 One of the centers, the Gothenburg center, also collected CSF of the patients included in the study. The majority of the included patients were also examined under the umbrella of the Gothenburg MCI study; the others as regular patients at the memory clinic. As the general idea of the LADIS study is in agreement with that of the clinical Gothenburg studies, the LADIS CSF papers have been included in this overview.
Multimodal prediction.
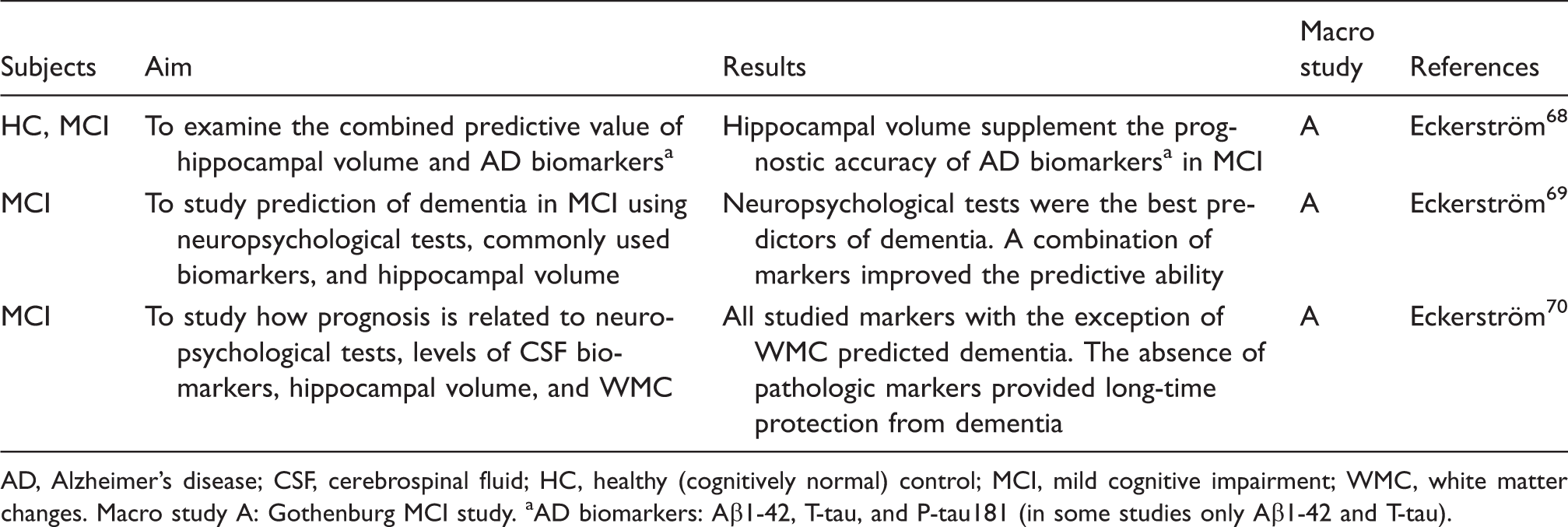
AD, Alzheimer’s disease; CSF, cerebrospinal fluid; HC, healthy (cognitively normal) control; MCI, mild cognitive impairment; WMC, white matter changes.
Macro study A: Gothenburg MCI study.
AD biomarkers: Aβ1-42, T-tau, and P-tau181 (in some studies only Aβ1-42 and T-tau).
The course of the disease and cognitive reserve.
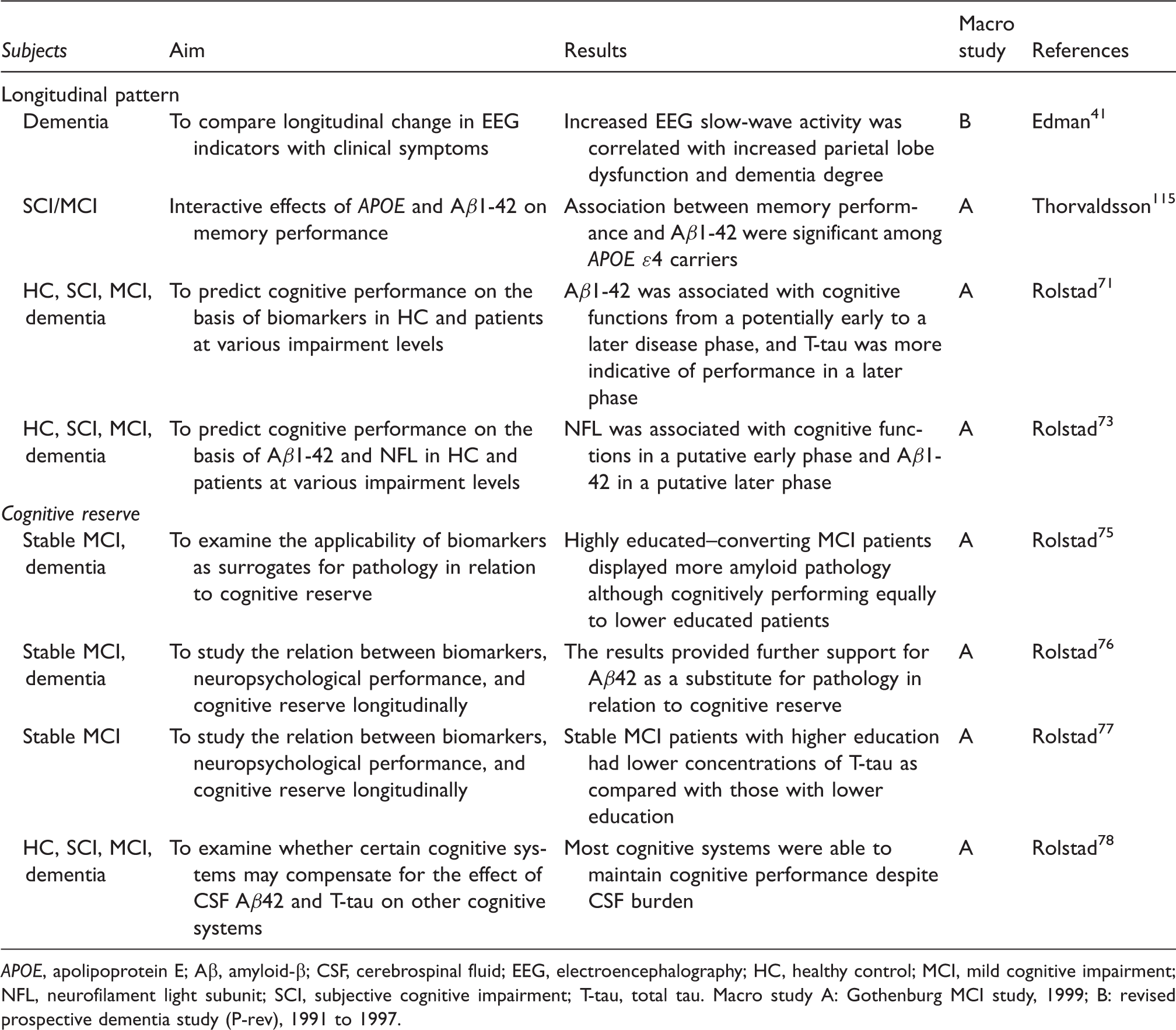
Macro study A: Gothenburg MCI study, 1999; B: revised prospective dementia study (P-rev), 1991 to 1997.
The term MCI has not been consistently used. In some earlier papers, patients with subjective cognitive impairment (SCI) have been included in the MCI group whereas in others they have been explicitly excluded. When it is obvious that the MCI group also includes patients with SCI, it has been renamed SCI/MCI. If it is not clearly stated whether MCI includes SCI a # sign has been added in the table column.
Results
In the following section, results from different outcome modalities in the Gothenburg MCI and dementia studies are presented.
Neuropsychology
In the P study, it was found that parietotemporal symptoms, i.e., impairments in interpreting sensory information and performing practical tasks, were typical of manifest early-onset AD (onset before 65 years of age), 24 which corresponds to AD without cerebrovascular disease. In contrast, late-onset AD (onset after 65 years of age) was characterized by memory impairment together with confusional symptoms, but absence of or less severe parietotemporal symptoms. 25 In another of the P studies frontosubcortical symptoms with mental slowness and loss of initiative were found to be characteristic of manifest SVD. 26
The first study that reported results from the Gothenburg MCI study was a cross-sectional study on patients with SCI/MCI. Etiological factors such as the influence of vascular disease were not taken into account. It was found that SCI/MCI was a highly heterogeneous diagnostic entity with significant impairments in all cognitive domains (Table 1). 27
As for vascular and other markers of potential interest in SCI/MCI, when grouped according to the presence of vascular disease, the cognitive profiles of the groups were found to differ, with SCI/MCI with vascular disease performing generally more poorly. 28 The cognitive profile of MCI subjects with AD-typical neurochemical biomarkers differed from that of those without. 29 Neither vascular disease nor altered levels of neurochemical biomarkers were alone associated with a specific cognitive profile in SCI/MCI, whereas a combination of them was associated with executive impairment. 30
In a recent follow-up study with comprehensive neuropsychological examination memory, visuospatial, and language symptoms characterized incipient AD, whereas speed/attention deficits and executive dysfunction preceded manifest SVD. 31 In a longitudinal study on MCI subtypes and neurochemical biomarkers, the converting SCI/MCI patients had a very high degree of impairment in all cognitive domains, memory impairment not being more predictive than any other cognitive domain for conversion; however, in combination with neurochemical biomarkers it could predict incipient AD and with vascular burden incipient SVD. 32
Brain Imaging and Physiology
An early report from the P study found that WMC are highly prevalent in patients with vascular burden. 33 Later in the P-rev study, WMC were found to be associated with a subcortical symptom profile, 34 and a dysexecutive behavioral symptom profile with apathy as the most prominent feature 35 rather than classical depressive symptoms. 36
In one study from the Gothenburg MCI study using hippocampal volume as a biomarker it was found that MCI patients converting to dementia during the 2-year follow-up time had smaller baseline hippocampi compared with both stable MCI patients and controls, with the left hippocampus as the strongest predictor. 37 Similarly, the left medial temporal lobe was found to be the best discriminator in a subsequent study examining differences in regional cerebral blood flow between stable and progressive MCI patients. 38 The presence of WMC and hippocampal atrophy in patients with MCI, AD, and SVD has also been investigated. It was found that hippocampal atrophy was present both in AD and SVD patients and that there was an association between hippocampal atrophy and WMC in the 25% of patients showing the highest degree of WMC. 39 Interestingly, in an early study, SVD patients had reduced regional cerebral blood flow in the medial temporal lobe and increased gray matter atrophy compared with controls. 40
In addition, the P and P-rev studies used EEG to investigate neurophysiologic changes in subgroups of dementia patients. The first EEG study found that slow-wave abnormality correlated with dementia severity. 41 In an effort to increase EEG efficacy in dementia diagnostics, EEG changes were analyzed in dementia patients with different brain regional syndromes. It was found that EEG abnormalities could mainly be attributed to parietal lobe dysfunction suggesting that EEG may be most valuable in patients with early-onset AD. 42 The following EEG study applied an automatic method to analyze alertness in dementia patients. The results show that demented patients were less alert compared with controls and that the decrease in alertness was proportional to dementia severity. 43
Another approach was to use regional cerebral blood flow in combination with CSF monoamine metabolites to examine two clinical subgroups of AD patients. It was found that early-onset AD patients had lower levels of CSF monoamine metabolites and late-onset AD had reduced regional cerebral blood flow. 44
Biochemistry
In the P and P-rev studies, it was found that overt SVD exhibited increased levels of myelin lipid sulfatide 45 and neurofilament light (NFL) 46 in CSF. The findings of increased levels of CSF NFL have been corroborated in overt SVD patients47,48,49 and NFL has also been found to be increased in patients with MCI who later progress to SVD. 50 Furthermore, NFL has been shown to reflect degree of WMC,51,52 while sulfatide and the extracellular modulating matrix metalloproteinase 9 (MMP-9) have been shown to predict WMC progression as assessed with the modified Rotterdam Progression Scale 53 in nondisabled patients with WMC.51,54 Alterations in the blood–brain barrier (BBB) as indicated by an increased CSF/serum albumin ratio have consistently been found in patients with SVD34,47,49,55 though not always evaluated as a biomarker.49,50,55
Increased CSF levels of MMP-9 and tissue inhibitor of metalloproteinase-1 (TIMP-1) together with a higher concentration of myelin basic protein (MBP), NFL, and albumin ratio in this particular patient group suggest an inflammatory involvement in SVD.
49
When assessed with multivariate analysis MMP-9, TIMP-1, MBP, and NFL contributed to the separation of patients with SVD and MixD from patients with pure AD. Albumin ratio was not evaluated in the model.
49
The alterations in albumin ratio have also been correlated to changes in TIMP-1 not only in patients with pure SVD and MixD but also in nondisabled patients with WMC.49,51 Furthermore, TIMP-1, plasminogen activator inhibitor-1,
One study found an increased cortisol awakening response in patients with MCI, as compared with controls. 65 In another study, a neuropsychological profile similar to that seen in prodromal AD was associated to the thyroid hormone triiodothyronine in patients with SCI/MCI. 66 Furthermore, SCI patients who subsequently progressed with MCI did not exhibit a deviating baseline AD biomarker profile, 67 which is not surprising as the majority of the MCI patients do not exhibit incipient AD.
Multimodal Prediction
To optimize the prediction of dementia, a number of studies have analyzed combinations of markers from different modalities. A study comparing the predictive ability of hippocampal volume to the CSF biomarkers A
The Course of the Disease and Cognitive Reserve
When simulating the course of an assumed disease using the associations between neurochemical biomarkers and neuropsychological deficits from cognitive health to manifest dementia it was found that A
When addressing the concept of CR, i.e., the capacity of cognitive processes to resist damage to the brain,
74
the conclusion was that MCI patients with longer education subsequently converting to dementia display more amyloid pathology as assessed by CSF determinations than less educated progressive MCI patients. Thus, high education could be used as a proxy for CR.
75
A longitudinal study provided further support for the applicability of A
Discussion
The current overview is a summary of the findings from the Gothenburg MCI and dementia studies, i.e., clinical–observational studies on AD and SVD among inpatiens and patients seeking care at a memory clinic. The methods used comprise psychometrics, biochemical methods mainly CSF determinations of substances reflecting disturbances of the brain, and imaging/physiologic methods. The majority of the listed papers are biochemical papers but there are also neuropsychological and imaging studies.
In particular the papers focus on subcortical vascular disease, which is a common cause of cognitive impairment and disability induced by cerebrovascular disease.12,13 Yet the impact of subcortical vascular disease is under debate and its manifestations are occasionally considered as epiphenomena. This opinion is not supported by the results from the Gothenburg MCI and dementia studies, which clearly indicate that SVD is an entity with features different from those of pure AD. There are also overlapping features between the disorders, which imply that they are both spectrum disorders.
Neuropsychology
The early studies showed that patients with manifest dementia and vascular burden showed high degrees of WMC. On the contrary, almost no patients with early-onset AD showed signs of WMC or significant vascular diseases. A high percentage of patients with late-onset AD also displayed WMC, which indicates presence of overlapping pathologies, i.e., late-onset AD appears to be equivalent to MixD as described in the Introduction section. Thus, the distribution of WMC in our early studies fits in the AD–SVD spectrum model. The symptom profiles also vary along the spectrum with mental slowness and executive dysfunction being characteristic of SVD at one end and impairments in interpreting sensory information and practical difficulties being characteristic of AD at the other; in the middle of the spectrum memory problems and concentration difficulties predominate.
Neuropsychological findings also differentiated between AD and SVD at early stages. Executive and speed/attention symptoms were found to characterize incipient SVD, whereas incipient AD was characterized by memory, visuospatial, and language impairments although all of the above cognitive symptoms occurred in both disorders. Our findings are in agreement with those of a recent meta-analysis on patients with vascular cognitive impairment without dementia reporting that individuals with vascular MCI had significantly greater deficits in processing speed than those with nonvascular MCI who exhibited a greater relative deficit in delayed recall. 79 Thus, it is not unreasonable to assume that a large group of the vascular MCI patients in the meta-analysis belonged to the SVD group.
In the Gothenburg MCI study,31,32 all patients who converted to dementia had multiple-domain MCI at baseline, the majority had amnestic multiple-domain MCI. This supports earlier reports on impairment in several cognitive domains typically preceding dementia—regardless of specific diagnosis—whereas single-domain MCI seems to be a more benign condition.80,81,82 Our findings clearly contradict a number of even quite recent studies reporting that single-domain amnestic MCI is at the highest risk of developing AD.83,84 One conceivable reason for the differing results is that the test batteries used for the MCI subgrouping in the referred studies were limited, which prevents detection of impairments in domains not tested.
Brain Imaging and Physiology
The brain imaging research in the Gothenburg MCI study has mainly focused on the impact of WMC on cognition, clinical symptomatology and hippocampal volume, discrimination between specific dementia disorders, and prediction of dementia among patients with MCI.
White matter changes are commonly seen in elderly individuals and their importance for cognitive impairment is under debate. They are, however, viewed as the major hallmark in SVD related to the CSF neurochemical changes and causing the neuropsychological profile of the disease. In the clinical Gothenburg studies, WMC were associated with a subcortical symptom profile 34 and a dysexecutive behavioral profile, with apathy as the most pronounced feature. 35 White matter changes have been linked to depressive symptoms, 85 but although loss of motivation was linked to WMC there was no association between WMC and depressive mood indicating that WMC are directly linked to dysexecutive symptoms.35,36 Furthermore, WMC were found to be inversely associated with hippocampal volume in patients with high WMC load. 39 This finding suggests that the memory impairment often seen in patients with cerebrovascular disease 28 may be secondary to hippocampal atrophy caused by axonal disconnection of the hippocampi. Support for this interpretation is provided by a study using a mouse model for SVD where the induction of WMC was followed by hippocampal atrophy. 86
Taken together, the findings from the Gothenburg MCI study provide additional support that WMC are important not only in SVD but also in the development of cognitive impairment, regardless of underlying disease.
Hippocampal atrophy has been found to predict conversion from MCI to dementia, strengthening the view that the hippocampi play a vital role in the pathophysiologic process of cognitive decline. Interestingly, although hippocampal volume as a predictor of subsequent dementia performed in line with findings from similar studies,87,88 it was still less effective than both CSF and neuropsychological markers. A possible explanation for this could be the relatively young patients in the Gothenburg MCI study, who are up to 10 years younger than the patients in comparable studies.87,88 The younger patients may be less affected by age-related white matter atrophy and concomitant pathology, which could hamper the predictive ability of measures of atrophy. Another observation from the Gothenburg MCI study is that the left cerebral hemisphere may be affected before the right hemisphere in the development of cognitive impairment. Both reduced blood flow in the left medial temporal lobe and left hippocampal atrophy outperformed their right counterparts.37,38 Although there is some further support that the left hippocampus is more affected in AD, 89 further research is needed to establish a clear pattern.
Biochemistry
One of the first fluid biomarker findings that spoke in favor of the white matter involvement in SVD patients was the increased levels of NFL found in both overt SVD47,48,49 and also in its incipient form.
50
Sulfatide, another component of myelin, was increased in patients with white matter damage even before disease onset
52
and in the same study it was also found that increased levels of MMP-9 correlated with progression of WMC.
51
Our data indicates that damage to the subcortical white matter is a pathologic hallmark of SVD and that the pathology is possible to identify through biochemical markers. In addition to its extracellular remodeling function, MMP-9 is believed to be involved in the regulation of the BBB opening and its activity is counterbalanced by TIMP-1. Blood–brain barrier dysfunction is another hallmark of SVD that has been visualized in earlier studies through an elevated albumin ratio in patients with SVD and MixD, whereas BBB has been shown to be normal in patients with AD without vascular diseases.13,47,49,50,90 The albumin ratio has also been shown to correlate with the levels of TIMP-1 in both SVD patients and in patients with white matter damage,49,51 which give further support for the notion that BBB dysfunction play a role in SVD. In a pivotal study with carefully defined groups of patients using a multivariate analytic method, which has the advantage of pointing at the relative importance of a group of factors for a specific outcome, it was found that patients with SVD displayed elevated levels of MBP, NFL, MMP-9, and TIMP-1, whereas in AD the CSF levels of P-tau and T-tau were increased reflecting degeneration of cortical regions affected by AD pathology.
49
Since MBP and NFL are both major constituents of the subcortical axons and they have been found to correlate to white matter lesion load,
51
their increased levels likely reflect subcortical axonal/white matter damage, which could also include the MMP/TIMP system through possible myelin breakdown. Other studies have also found alterations in MMP-9 and other MMPs in patients with SVD.91,92 Furthermore, both TIMP-1 and plasminogen activator inhibitor-1, both involved in the regulatory system connected to MMP activity, have been shown to be increased in CSF in MCI patients that subsequently deteriorate to SVD.
56
This would not only support an early inflammatory involvement in the disease process that is related to the BBB, but also suggests alterations in the coagulation system. It is therefore not unreasonable to assume that MMPs and TIMP-1 play a role in both BBB disruption and destruction of subcortical tissue in patients with SVD, as has been previously suggested.
93
A combined panel of markers could potentially be very important not only for early detection of patients with MCI that will progress to SVD, but also for early differential diagnostics. There are at present no such established markers for SVD. These markers could therefore in combination with markers that have repeatedly been shown to be altered in AD,49,57,58,59 i.e., T-tau, P-tau, and A
Multimodal Prediction
Mild cognitive impairment is a heterogeneous condition, and the prognosis differs within the group. To enable prediction of a specific etiological dementia diagnosis in patients with MCI the single-marker approach seems to be insufficient. It also seems that markers from more than one modality are needed to capture the specific features of the manifest disease. In our studies on patients included in the Gothenburg MCI study multivariate analyses revealed that multimodal combinations of variables outperformed the prognostic ability of single variables for dementia and AD. Absence of any pathologic markers greatly reduced the long-term risk of developing dementia. In the Gothenburg MCI study, the markers, alone and in combination, generally have higher predictive values compared with, for example, the ADNI study,87,95 or the DESCRIPA study. 88 One explanation is the study design; the Gothenburg MCI study is a single-center study whereas the ADNI and DESCRIPA are multicenter studies. Single-center studies can be expected to provide data with less variability related to local interpretation of inclusion/exclusion criteria, demographic differences, sampling techniques, scanner differences, etc., which may explain the high predictive values of the markers. The fact that combinations of markers provided less additional information than expected may be because of the already very high predictive value of the single markers. As mentioned previously, another factor to take into account is the relatively young age of the patients compared with, especially, the patients in the ADNI study.87,95 Although it is unclear how patient age affects the results, the main difference between the studies is that hippocampal volume is less predictive and CSF markers and neuro-psychological tests are more predictive in the younger Gothenburg MCI study patient material.
The Course of the Disease and Cognitive Reserve
In a series of studies, we have found that it is possible to study CR mechanisms at an early disease stage using CSF biomarkers. Amyloid-
Whereas quite a few studies have sought to disentangle the association between level of cognitive functioning and CSF biomarker burden, there has been a shortage of reports that have investigated the topic throughout the whole course of illness, from cognitive health to manifest dementia, using a comprehensive battery of cognitive tests. Our models of the disease course have given clues that may help us understand the disease processes. The sequence of neurochemical events seems to differ between those with and without vascular burden, and the intensity of the disease process seems most pronounced at the MCI stage. The latter result strengthens the view that the MCI stage is a feasible target for interventions.
Strengths and Weaknesses
Notably, early detection of cognitive impairment induced by subcortical vascular disease has not attracted a similar level of attention as AD. An important reason for this is certainly the predominant role of AD both in research and in the clinical implementation of knowledge in the field. In addition, subcortical vascular disease gives rise to cognitive symptoms that are difficult to detect or are said to be ‘clinically silent’. It is a strength of the Gothenburg MCI study that this disorder is paid serious attention. Other strengths are our fixed protocol, longitudinal design, inclusion of patients seeking help for cognitive disorders and a healthy control group, strict diagnostic criteria, and diagnostic assessment performed by a physician masked to the outcome variables. The study also has some limitations: few patients have converted from SCI/MCI to SVD, a significant number of patients drop out, overlap between the studies, and there may be potential cohort effects. Some research foci have not been pursued and no solid conclusions can thus be drawn from the preliminary results such as research using EEG and SPECT. Analyses of specific vascular factors and the impact of lifestyle have not yet been performed. We have not yet reported any specific results for MixD. There is also lack of studies on potential markers for congophilic versus arteriolosclerotic lesions.
Concluding Summary
The Gothenburg MCI and dementia studies are longitudinal, clinical–observational studies on the AD–SVD spectrum among patients seeking medical care at a memory clinic. Particular focus is on SVD, which is an underrecognized disorder in clinical practice and research. By using specific criteria for SVD and criteria in agreement with recently published clinical criteria for AD, not only AD but also SVD has been identified in the studies. Furthermore, it has revealed characteristic neuropsychological and neurochemical patterns for SVD, which are present also early in the course of the disease. Our nosological findings provide opportunities for trials in SVD and better definition of AD in AD trials.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Grants from Sahlgrenska University Hospital, Swedish Research Council, Swedish Brain Power, the Alzheimer’s Association, the Inga-Britt and Arne Lundberg Research Foundation, the Göteborg Medical Society, the Swedish Medical Society, the Swedish Dementia Foundation, Stiftelsen Gamla Tjänarinnor, Gun och Bertil Stohnes stiftelse, the Adlerbert Research Foundation, Swedish Alzheimer Foundation, Stiftelsen Psykiatriska forskningsfonden, Konung Gustaf V:s och Drottning Victorias Frimurarestiftelse, The Swedish Brain Foundation, The Sweden-America Foundation, and The Royal Swedish Academy of Sciences.
Footnotes
Acknowledgement
The authors thank Ewa Styrud, Christina Holmberg, Marie C Johansson, Ing-Marie Isgaard, and Eva Bringman for their technical support, and Niklas Klasson for comments.
