Abstract
Symptoms of acute mountain sickness (AMS) may appear above 2,500 m altitude, if the time allowed for acclimatization is insufficient. As the mechanisms underlying brain adaptation to the hypobaric hypoxic environment are not fully understood, a prospective study was performed investigating neurophysiological changes by means of near infrared spectroscopy, electroencephalograpy (EEG), and transcranial doppler sonography at 100, 3,440 and 5,050 m above sea level in the Khumbu Himal, Nepal. Fourteen of the 26 mountaineers reaching 5,050 m altitude developed symptoms of AMS between 3,440 and 5,050 m altitude (Lake-Louise Score ≥ 3). Their EEG frontal beta activity and occipital alpha activity increased between 100 and 3,440 m altitude, i.e., before symptoms appeared. Cerebral blood flow velocity (CBFV) in the anterior and middle cerebral arteries (MCAs) increased in all mountaineers between 100 and 3,440 m altitude. During further ascent to 5,050 m altitude, mountaineers with AMS developed a further increase in CBFV in the MCA, whereas in all mountaineers CBFV decreased continuously with increasing altitude in the posterior cerebral arteries. These results indicate that hypobaric hypoxia causes different regional changes in CBFV despite similar electrophysiological changes.
Keywords
INTRODUCTION
Nowadays an ever increasing number of people travel to high-altitude destinations for recreation or work. The incidence of acute mountain sickness (AMS) during an ascent on foot to altitudes between 2,800 and 5,336 m varies between 43% and 69% in different population studies. 1 The incidence of possibly life-threatening forms of AMS-like high-altitude cerebral edema (HACE) and high-altitude pulmonary edema is much lower than for AMS: estimates range between 0.1% and 4.0%. 2 Altitude-related illnesses have contributed to the death of 17% of British climbers attempting peaks > 7,000 m. 3
The main risk factors for developing high-altitude illness include the rate of ascent, individual susceptibility, and the altitude reached (sleeping altitude). 2 Other risk factors are permanent residence < 900 m. 1 Physical fitness is not a contributing factor, and people aged > 50 years may be even less susceptible. 2 The reasons for the considerable variation in individual susceptibility are unclear, but most likely diverse interactions between genetic and environmental factors have a major role. 1 The Lake-Louise Consensus Group defined AMS as the presence of headache in an unacclimatized person who has recently arrived at an altitude above 2,500 m in addition to one or more of the following: gastrointestinal symptoms (anorexia, nausea, or vomiting), insomnia, dizziness, and lassitude or fatigue. 1 AMS appears 4 to 36 hours after ascent to high altitudes. 1
It is not fully understood which factors are important for a good or poor adaptation to high altitude and how the brain reacts to these different states. Furthermore, it remains unclear which factors allow AMS to evolve directly into HACE. In this prospective study, we investigated early physiological and pathophysiological changes that occur at high altitudes. They include mechanisms of adaptation to high altitude such as brain activity measured by electroencephalography (EEG) and cerebral blood flow velocity (CBFV) measured by transcranial doppler sonography (TCD). Blood pressure, peripheral O2 saturation (SpO2), endtidal CO2 (EtCO2), and brain oxygen saturation, using near infrared regional saturation (NIRS), were measured as cofactors during a joint trekking trip to Everest Base Camp in the Khumbu Himal, Nepal. We hypothesized that the CBF increases because of the known decrease of peripheral and central (brain) oxygenation as compensatory responses to restore the blood and oxygen supply to the brain. Beginning brain hypoxia may lead to an increase of neuronal cell activity, resulting in an increased EEG background rhythm.
MATERIALS AND METHODS
Thirty-two healthy volunteers (12 females, 20 males; 43.5 ± 2.2 years; range 19 to 66 years; all hobby mountaineers) were examined during a joint, 22-day-long, high-altitude trekking tour in Sagarmatha National Park, Nepal. All participants were neurologically normal and had no history of neurological or psychiatric disorders, head trauma, or drug abuse. The most recent exposure to high altitude > 2,500 m had ended at least 6 months prior to the study.
The route began in Lukla (2,160 m) and ended at the so-called Silver Pyramid (Italian–Nepali Research Center Ev-K2-CNR, Lobuche, 5,050 m), which is located near Everest Base Camp. One group reached 5,050 m via Gokyo Ri (5,340 m), the other via Island Peak (6,200 m). The rates of ascent were as follows: from 2,860 to 3,440 m in 2 days and from 3,440 to 5,340 m in 4 days or 6,200 m in 6 days and back to 5,050 m in 2 to 3 days. EEG and TCD were performed at baseline level (100 m), at 3,440 m and at the Silver Pyramid 5,050 m. SpO2, EtO2, and Lake-Louise AMS Score were evaluated twice daily by an expedition doctor. Severe AMS was treated immediately according to the guidelines of the Wilderness Medical Society. A mountain guide accompanied and observed those mountaineers who descended to a lower altitude because of AMS.
The Lake-Louise Score was used to ascertain the presence of symptoms of AMS. It contains a self-rating and a foreign-rating scale (recorded by expedition doctor), which ranges from 0 to 29. 1 Mountaineers with a score ≥ 3 were considered symptomatic if they had headache and one other AMS symptom.
Blood pressure was measured using the Riva–Rocci method, always by the same researcher and on the right arm, and after resting in a sitting position for at least 10 minutes. Systolic and diastolic blood pressure were recorded.
Oxygen saturation was measured using a finger clip on the right index finger, and EtCO2 (Oridion Oxymetry and Capnography, Needham, MA, USA) by nasal probe. Measurements were made after resting in a sitting position for at least 10 minutes.
An INVOS 5,100 cerebral oximeter (Somanetics, Troy, MI, USA) was used to measure changes in regional cerebral oxygenation (rSO2). Near infrared light (730 and 805 nm) is emitted through the forehead of the skull. Once the light passes through the cerebral cortex, the light that returns is detected at two distances from the light source (at 3 and 4 cm). The spectral absorption of blood in the brain can be determined on the basis of this principle and is defined as rSO2. Before measuring, the skin was cleaned with a prewrapped skin-prep pad. Then two sensors were applied on the right and left sides of fronto-temporal region in accordance with the manufacturer's recommendations. To minimize the influence of extracerebral light, the forehead was covered with a black kerchief during the recording procedure. The rSO2 data were recorded after a resting time of 3 minutes.
TCDs (DWL Elektronische Systeme, Sipplingen, Germany) were performed bilaterally of the middle cerebral artery (MCA) at 50-mm depth, bilaterally of the anterior cerebral artery (ACA) at 65-mm depth, and bilaterally of the posterior cerebral artery (PCA) at 65-mm depth using a 2-MHz sonde placed over the temporal scale. To avoid the well-known inter-rater variability in repeated TCD measurements, the same examiner performed all TCDs. To obtain the best possible results, the values were recorded after waiting for at least 1 minute for stable signals. All TCD measurements were made at 3,440 and 5,050 m the next day after arrival. As the study design focused on the assessment of relative changes in cerebral blood velocity, the baseline measurements at 100 m were set at 100%, and the relative changes were calculated.
EEGs (SIGMA Medizintechnik, Thum, Germany) were recorded for 10 minutes with 24 silver/silver chloride electrodes placed according to the 10 to 20 system using a sampling rate of 256 Hz; resistance was kept < 10 kOhm. Data analysis was performed using Fast-Fourier transformation of 15 × 4 seconds artefact-free EEGs. Constant vigilance during recording was ensured through irregular blinking. At 3,440 and 5,050 m, the EEG was performed the morning after arrival. FP2, FP1, F3, F4, and FZ were considered the frontal electrodes, and O1, O2, P3, P4, and PZ as the occipital electrodes. As the relative changes in the frontal background activity (beta band) and delta activity, as well as in occipital background activity (alpha band) and delta activity were of interest, the percentage changes of power activity in these bands were calculated for each altitude.
Although the investigators of NIRS, TCD, and EEG were not aware of the AMS Score, which was assessed by the expedition doctors independently, they were not blinded for clinical signs of AMS.
The data of left and right values were pooled if the differences of the latter did not reach the level of significance. Only data for those mountaineers who reached 5,050 m altitude were used in further statistical analyses. As there were no differences between the two mountaineering groups (based on the ascent route via Gokyo Ri (5,340 m) or Island Peak (6,200 m)), the data were assigned to the AMS or non-AMS group regardless of the ascent route taken.
Data were analyzed using the SPSS software (release 11.0, SPSS, Chicago, IL, USA). The Friedmann analysis was performed for several connected samples, and the analysis was continued only if the
The study protocol was approved in advance by the Ethics Committee of the University of Vienna (Austria) according to the ethical standards of the Helsinki Declaration of 1975 (and as revised in 1983). We confirm that written, informed consent was obtained from each participant.
RESULTS
All participants reached Namche Bazaar at 3,440 m on day 2, and 26 of the 32 participants, the Silver Pyramid (5,050 m) between 12 and 13 days later. Because of technical difficulties, EtCO2, SpO2, and TCD data of the PCA were not obtained in one mountaineer at 5,050 m.
AMS Lake-Louise Score
Fourteen of 26 mountaineers who reached 5,050 m altitude developed symptoms of AMS between 3,440 and 5,050 m altitude (Lake-Louise Score ≥ 3). The mean AMS Score in this group was 4 ± 0 (min = 3; max = 7) and the mean duration of illness, 1 ± 0.4 day (min = 1; max = 5).
Blood Pressure, Endtidal CO2, and Peripheral and Regional Cerebral Oxygenation
All participants did not exhibit a change in blood pressure during the ascent from 100 to 3,440 m and 5,050 m (mean 120/80 mm Hg (±2.7/±1.8) to 130/80 mm Hg (±3.3/±1.9) to 127.5/80 mm Hg (±2.8/±1.4); the blood pressure also did not differ between AMS and non-AMS mountaineers.
Endtidal CO2 levels decreased from 33 ± 0.5 to 31.5 ± 1.1 mm Hg at 3,440 m altitude (
Cerebral Blood Flow Velocity
The CBFV in the ACA of all mountaineers showed an increase in blood flow velocity between 100 and 3,440 m altitude, 100% ± 0: 119% ± 9;
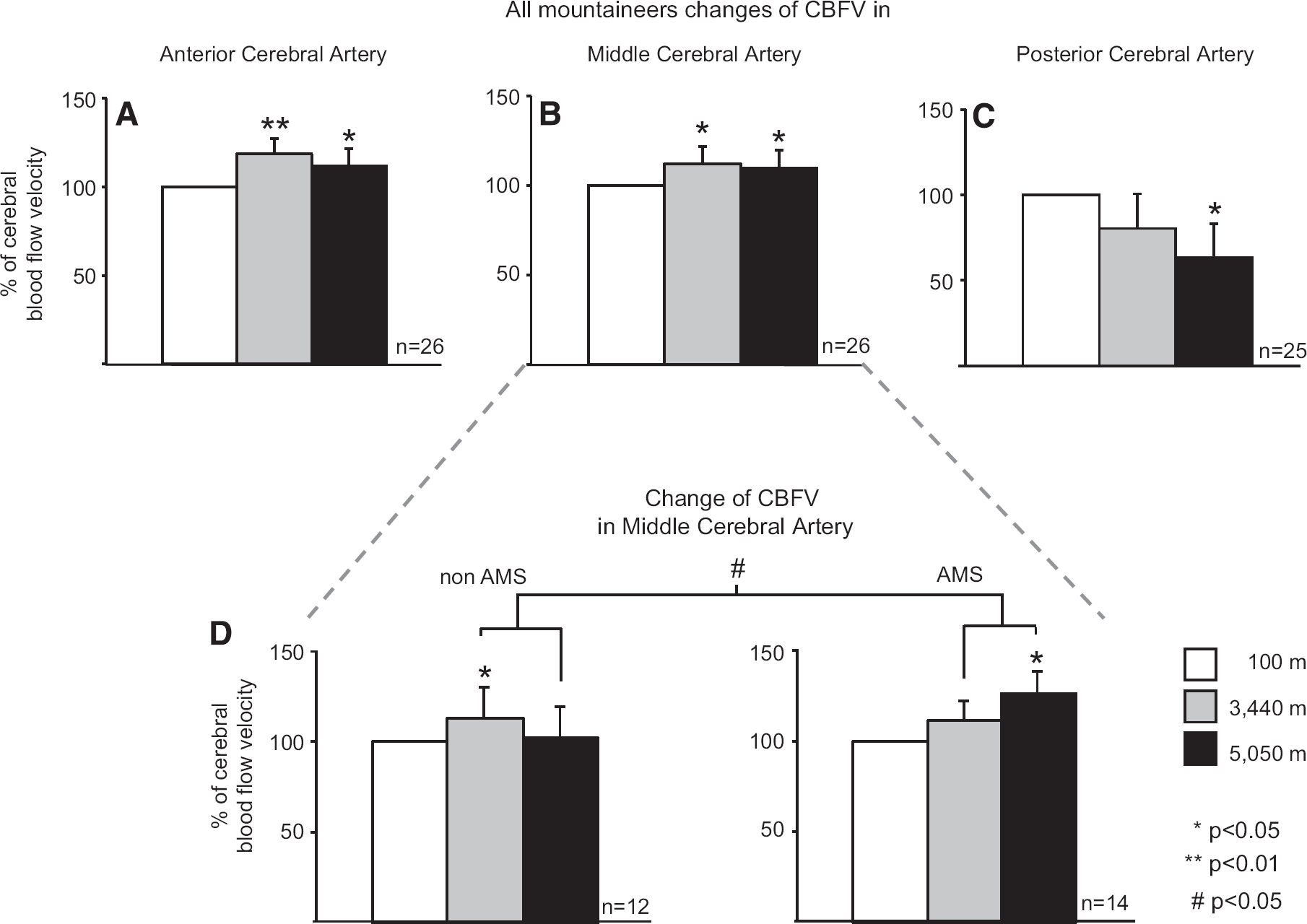
Changes in cerebral blood flow velocity (CBFV) for all mountaineers in the anterior cerebral artery (
CBFV in the MCA rose by 12% ± 10 from 100 to 3,440 m (
CBFV in the PCAs decreased continuously from 100 m over 3,440 m to 5,050 m; 100% ± 0: 80% ± 9: 64% ± 9,
The CBFV in the ACA and the PCA correlated with EtO2 levels, indicating that lower EtCO2 levels (better hyperventilation) correlated with higher CBFV values (for ACA:
Electroencephalography Activity
The background activity of the frontal lobe is the beta rhythm and that of the occipital lobe, the alpha rhythm. Whereas the frontal EEG beta activity increased in AMS mountaineers between 100 and 3,440 m altitude by 24% ± 14 (
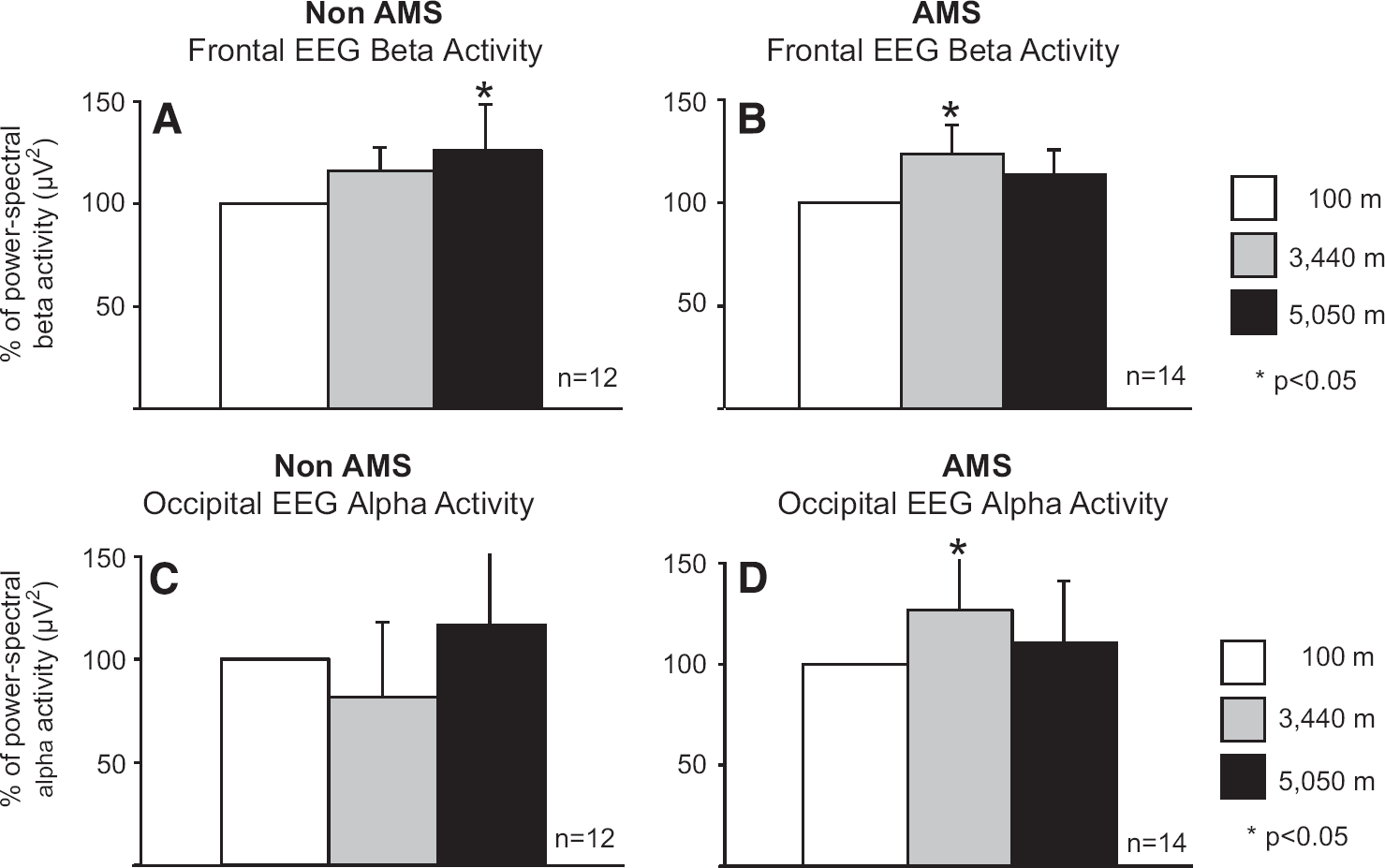
Changes in frontal beta activity in non-acute mountain sickness (non-AMS) (
An increase of 27% ± 33 in occipital alpha activity in AMS mountaineers also occurred between 100 and 3,440 m (
Delta activity decreased in the frontal lobe by −34% ± 95 at 3,440 m and by −24% ± 188 at 5,050 m (not significant) as well as in the occipital lobe by −13% ± 15 at 3,440 m; a slight increase of +2% ± 13 was observed at 5,050 m (not significant).
DISCUSSION
Changes in Blood Pressure and Oxygenation
The blood pressure values did not change between the altitudes or between AMS and non-AMS mountaineers. These findings illustrate that the changes in CBFV did not contribute to a systemic increase in blood pressure. However, these results are limited, as we did not measure dynamic cerebral autoregulation using transfer-function gain, phase, and coherence between mean blood pressure and MCA velocity. 5 It is well known that air and O2 pressure decrease with rising altitude, resulting in hypobaric hypoxia. NIRS revealed that these effects lead to a decrease of peripheral O2 saturation as well as brain O2 saturation. 6 We could not demonstrate a correlation between the decrease in rSO2 and compensatory increase of frontal CBFV in our study. This may indicate that mechanisms other than those directly proceeding via hypoxia are involved in regulating the CBFV. 7 These results indicate that even an increase of CBF is not able to restore the oxygen level in the frontal cortex during an ascent to high altitude.
Changes of Cerebral Blood Flow Velocity
Another limitation of our study was that we did not investigate the CBFV in the basilary or vertebral circuits. Skow
Changes of Electroencephalography Activity
A decrease of overall occipital alpha activity was reported in a pressure chamber study simulating a fast ascent (150 m/min and rest of 25 minutes every 1,000 m) to 6,000 m altitude. However, this study did not report any clinical signs of AMS, 37 although they were probably present. A study investigating quantitative EEG changes revealed that hyperventilation caused an exponential increase in slow activity and a decrease in alpha power. 38 The rate of ascent was moderate in our study, allowing the brain time to adapt to hypobaric hypoxia. Delta activity did not increase in the frontal or occipital lobe, perhaps because none of the mountaineers studie, developed HACE.
It has been suggested that the changes in increase of occipital alpha activity may be induced by sympathetic hyperactivity. Such hyperactivity might be mediated by the release of norepinephrine in the brainstem, thus influencing the reticular activation system, 39 or induced via a central oxygen-chemosensitive network. 31
CONCLUSION
Two limitations of our study were the relatively small sample size and the sometimes difficult conditions in the field. The increase in CBFV in the anterior and MCA is a compensatory response to the hypobaric hypoxic environment, which restores blood and oxygen supply to the brain. The increase in CBFV in the MCA was delayed in mountaineers with AMS. Contrary to our hypothesis, CBFV decreased in the PCA with increasing altitude and hypoxia. This decrease was most likely because of differences in cerebrovascular reactivity to changes in CO2, resulting in a different balance of hypoxic increase and hypocapnic decrease of CBFV.
In summary, the results of MRI studies of simulated ascents to very high altitudes in pressure chambers suggest that mild cytotoxic edema might be more prevalent in persons with symptoms of AMS. 27 Such cytotoxic edema might occur if oxygen delivery is reduced because of the decreased perfusion in the posterior parts of the brain. This results in dysfunction of Na/K+ ATPase with consequently cytotoxic edema and AMS. If these effects are aggravated, AMS may evolve directly into HACE, which is characterized by dysfunction of the posterior parts of the brain. PRES may provide insights into interesting, analogous pathways in the pathophysiology of both conditions. It has been suggested that PRES develops because of the failure of autoregulation and hyperperfusion, but the theory of alternative endothelial dysfunction and hypoperfusion and vasoconstriction leading to altered integrity of the blood–brain barrier is favored. 21 Similar pathophysiological considerations might be valid for the development of HACE. Anatomical findings of the watershed area between the ACA and the PCA at the splenium might explain this predilection for microhemorrhages in HACE, in view of the continuous decrease of CBFV in the PCA.
These findings indicate that the physiological response of CBFV to hypobaric hypoxia differs in the anterior and posterior supratentorial parts of cerebral circulation despite their similar electrophysiological changes.
Footnotes
B Feddersen had the idea for the study, performed TCD and EEG studies in the field, analyzed the data, contributed to the manuscript, generated the figures, and was involved in the critical review of the article. P Neupane performed EEG studies in the field, analyzed the data, and was involved in the critical review of the article. F Thanbichler analyzed the data, contributed to the manuscript, and was involved in the critical review of the article. I Hadolt performed NIRS studies in the field, analyzed the data, and was involved in the critical review of the article. V Sattelmeyer performed NIRS studies in the field, analyzed the data, and was involved in the critical review of the article. T Pfefferkorn analyzed the data, contributed to the manuscript, generated the figures, and was involved in the critical review of the article. R Waanders had the idea for the study, was the scientific leader of the studies in the field, analyzed the data, contributed to the manuscript, and was involved in the critical review of the article. S Noachtar analyzed the data, contributed to the manuscript, and was involved in the critical review of the article. H Ausserer had the idea for the study, performed TCD studies in the field, analyzed the data, contributed to the manuscript, generated the figures, and was involved in the critical review of the article.
The authors declare no conflict of interest.
ACKNOWLEDGMENTS
The authors thank RONAST and Comitato Ev-K2-CNR Bergamo for providing free use of the Pyramid Laboratory, SIGMA-Medizintechnik, DWL Medizinische Systeme, Oridion, High Country Trekking, and all participants. The authors also thank Judy Benson for copyediting the manuscript.
