Abstract
In this study, we sought to identify a disease-related spatial covariance pattern of spontaneous neural activity in Parkinson's disease using resting-state functional magnetic resonance imaging (MRI). Time-series data were acquired in 58 patients with early to moderate stage Parkinson's disease and 54 healthy controls, and analyzed by Scaled Subprofile Model Principal Component Analysis toolbox. A split-sample analysis was also performed in a derivation sample of 28 patients and 28 control subjects and validated in a prospective testing sample of 30 patients and 26 control subjects. The topographic pattern of neural activity in Parkinson's disease was characterized by decreased activity in the striatum, supplementary motor area, middle frontal gyrus, and occipital cortex, and increased activity in the thalamus, cerebellum, precuneus, superior parietal lobule, and temporal cortex. Pattern expression was elevated in the patients compared with the controls, with a high accuracy (90%) to discriminate the patients from the controls. The split-sample analysis produced a similar pattern but with a lower accuracy for group discrimination in both the derivation (80%) and the validation (73%) samples. Our results showed that resting-state functional MRI can be potentially useful for identification of Parkinson's disease–related spatial covariance patterns, and for differentiation of Parkinson's disease patients from healthy controls at an individual level.
Keywords
INTRODUCTION
Parkinson's disease (PD) is a common neurodegenerative disorder. The diagnosis of PD is based mainly on clinical assessments but the diagnostic accuracy is low during early stages of PD. 1 There is an increasing interest in finding viable biomarkers that can be reliably used in identifying at risk individuals, aiding early differential diagnosis, measuring disease progression, and monitoring treatment effects in patients with PD.
Previous neuroimaging studies with 18 F-fluorodeoxyglucose (FDG) positron emission tomography (PET) and multivariate network analysis have shown that PD is associated with a specific disease-related spatial covariance pattern (PDRP) of regional cerebral glucose metabolism.2,3 The expression of PDRP network can be quantified prospectively with FDG PET in individual subjects to distinguish PD patients from healthy controls,4–6 correlate with independent measures of disease severity, 4 evaluate modulations by novel experimental therapeutics,5,6 and discriminate PD patients from patients with multiple system atrophy or progressive supranuclear palsy.7,8 PDRP can also be reliably assessed by imaging regional cerebral blood flow with other radiotracer techniques (PET and SPECT)2,9 and perfusion magnetic resonance imaging (MRI).10,11
Functional MRI (fMRI) has been used to show changes in neural activity12–14 and connectivity in the resting state in PD patients.15–18 Regional differences in low-frequency fMRI signal in the resting state were also reported to be reliable for discriminating PD patients from healthy controls. 13 These findings show that resting-state fMRI (rsfMRI) can be used to reveal the distribution of PD-related neural activity and assess its correlations with clinical symptoms. However, the majority of rsfMRI studies was based chiefly on the group-level statistics and is less helpful in prospective clinical applications with individual subjects. In this study, we sought to identify an analogous spatial covariance pattern of spontaneous neural activity in PD using rsfMRI, and examine if this method can be potentially used to discriminate PD patients from healthy controls at an individual level.
SUBJECTS AND METHODS
Subjects
We investigated 58 patients with idiopathic PD (age 61.4 ± 9.0 years, range 45 to 78 years; 25 women, 33 men). The diagnosis of PD was based on medical history, physical and neurologic examinations, response to levodopa, and laboratory tests and MRI scans to exclude other diseases. All patients presented motor symptoms of rigidity and bradykinesia at early to moderate stages but were chosen to have at most a mild tremor to avoid the disturbance of fMRI signal. The Unified Parkinson's Disease Rating Scale (UPDRS) 19 motor score at off state was 24.8 ± 8.2. The Hoehn and Yahr disability scale 20 was 2.0 ± 0.6. All patients had participated in a recent study. 21 The control group included 54 age- and gender-matched healthy subjects (age 61.1 ± 9.4 years, range 45 to 78 years; 26 women, 28 men). Mini-Mental State Examination was ≥ 26 in all subjects, with no differences between the patients and the controls. The primary analysis of network identification was performed in these PD patients and healthy subjects defined as cohort A. For the secondary analysis of network validation, subjects in cohort A were randomly divided into a derivation sample (cohort B) of 28 PD patients (age 59.9 ± 9.1 years, Hoehn and Yahr 1.9 ± 0.6, and motor UPDRS 24.2 ± 7.9) and 28 normal controls (age 63.7 ± 9.6 years), and a prospective validation sample (cohort C) of 30 PD patients (age 62.7 ± 8.9 years, Hoehn and Yahr 2.0 ± 0.5, and motor UPDRS 25.3 ± 8.6) and 26 normal controls (age 58.3 ± 8.4 years). Subject groups in these two cohorts were matched for age and clinical symptoms. The experiments were performed according to the Declaration of Helsinki and were approved by the Institutional Review Board of Xuanwu Hospital in Beijing, China. All subjects gave their written informed consent for the study.
Resting-state Functional Magnetic Resonance Imaging Protocols and Processing
Functional MRI acquisition was performed on a 3 T Magnetom Trio system (Siemens, Erlangen, Germany). Parkinson's disease patients were scanned after their medication had been withdrawn for at least 12 hours. High-resolution anatomic images were acquired with a three dimensional magnetization prepared rapid acquisition gradient echo sequence (repetition time (TR)/echo time (TE) = 2,530/3.39 ms, 128 axial slices, 1.33 mm thickness, and field of view = 256 mm). Functional MRI data were collected with a gradient-echo echo-planar sequence (TR/TE = 2,000/30 ms, flip angle = 90°, 33 axial slices, 3.5 mm thickness, no gap, field of view = 220 mm, and matrix size = 64 × 64). During rsfMRI, subjects were instructed to keep their eyes closed, to remain motionless, and to not to think of anything in particular. The rsfMRI scanning session lasted for 8 minutes.
Functional MRI data were preprocessed using Statistical Parametric Mapping (SPM8, http://www.fil.ion.ucl.ac.uk/spm) and Data Processing Assistant for Resting-State fMRI (DPARSF) V2.0 advanced edition (http://www.restfmri.net). Time-series data were first slice-time corrected and aligned to the first image for motion correction. Each subject's head motion parameters were examined. No subject had more than 1.5 mm maximum translation in
To identify a specific pattern of neural activity, we analyzed rsfMRI data with the amplitude of low-frequency fluctuation (ALFF) approach, which measures the regional spontaneous activity at rest. 23 Amplitude of low-frequency fluctuation was calculated using the same procedures reported previously, 23 with Resting-State fMRI Data Analysis Toolkit (REST, http://www.restfmri.net). The time courses of all voxels were first converted to the frequency domain using a fast Fourier transform, and the power spectrum was obtained. The square root of the power spectrum was calculated at each frequency and then averaged across 0.01 to 0.08 Hz for each voxel. This averaged square root was taken as an index of ALFF. The ALFF of each voxel was divided by the global mean ALFF value within the brain tissue mask to standardize data across subjects.
Network Analysis
Scaled subprofile model (SSM), which is one of the multivariate spatial covariance techniques based on principal component analysis (PCA), was applied to assess subject-by-voxel effects on ALFF maps in both PD patients and control subjects with Scaled Subprofile Model Principal Component Analysis (SSMPCA) toolbox (http://www.feinsteinneuroscience.org). In contrast to PET or SPECT images, log-transformation was not used for fMRI data because of its additive variability. A PD-related covariance pattern was identified from a linear combination of principal components whose expression in individual subjects maximized the separation between the two groups. This was conducted by logistic regression analysis in the set of contiguous principal components that accounted for ~50% of subject × voxel variance in the SSMPCA operation. The sign of the resultant pattern was defined such that PD patients had elevated mean expression compared with controls. The reliability of this pattern was assessed on a voxel basis by a bootstrapping estimation algorithm.
24
Brain regions deemed reliable from this
Statistical Analysis
PDRP scores in all subjects were
RESULTS
Parkinson's Disease–Related Covariance Pattern
SSMPCA analysis produced a spatial covariance pattern of neural activity in PD (PDRP–ALFF) characterized by decreased activity in the bilateral caudate nucleus and anterior putamen, bilateral middle frontal gyrus, rostral supplementary motor area (pre-SMA), right lingual and middle occipital gyri, left precuneus and inferior temporal gyrus, and right supramarginal and posterior cingulate gyri, and increased activity in the thalamus, bilateral cerebellum, right medial frontal gyrus/rectus, bilateral precuneus, left superior parietal lobule, and bilateral temporal cortices including posterior insula (Figure 1A and Table 1). This pattern was defined from a set of principal components accounting for a total subject × voxel variance of 34.2% and found to be reliable (
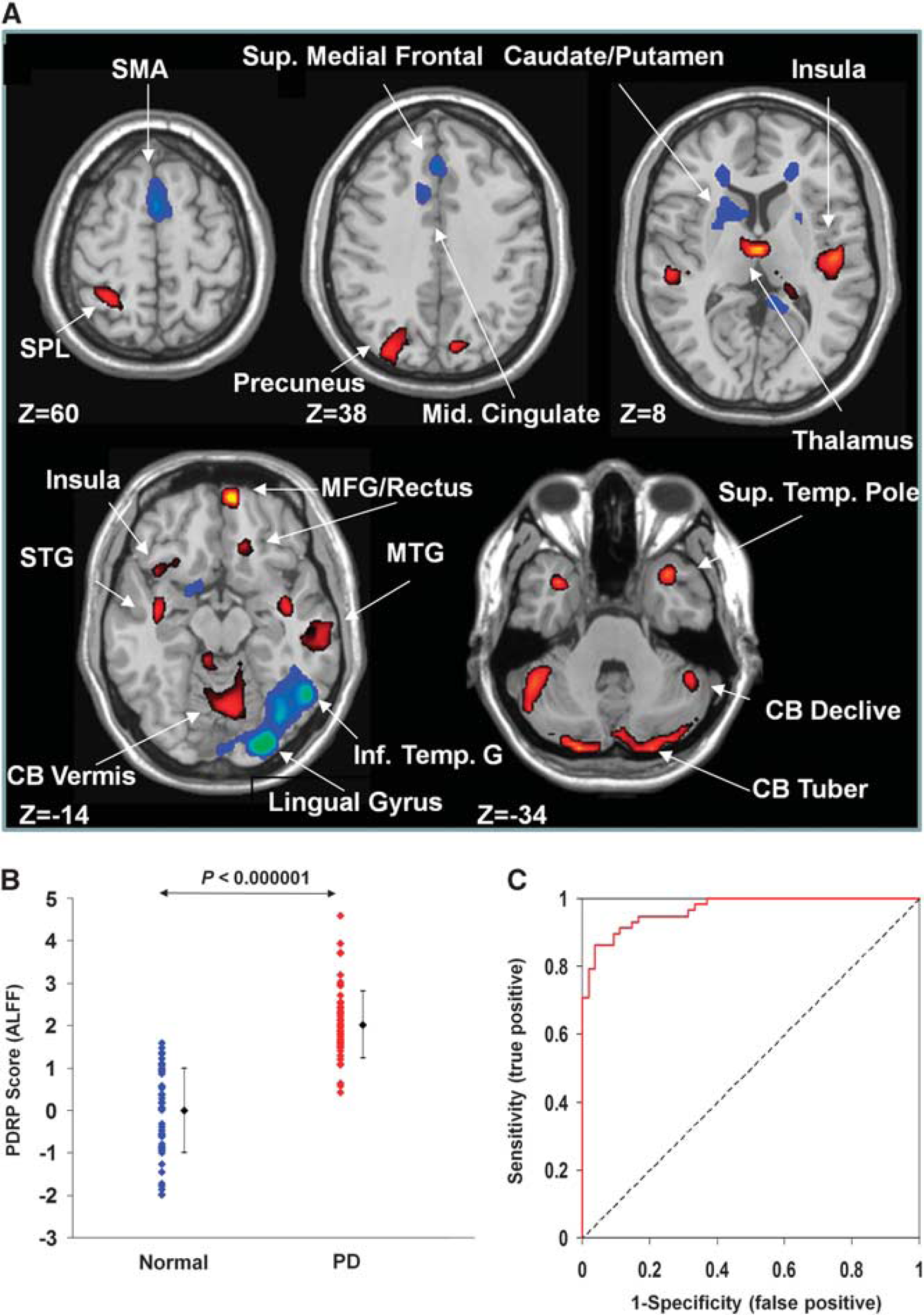
Parkinson's disease–related spatial covariance pattern–amplitude of low-frequency fluctuation (PDRP–ALFF) identified with resting-state functional magnetic resonance imaging (fMRI). (
Anatomic regions of PDRP–ALFF derived from resting-state fMRI
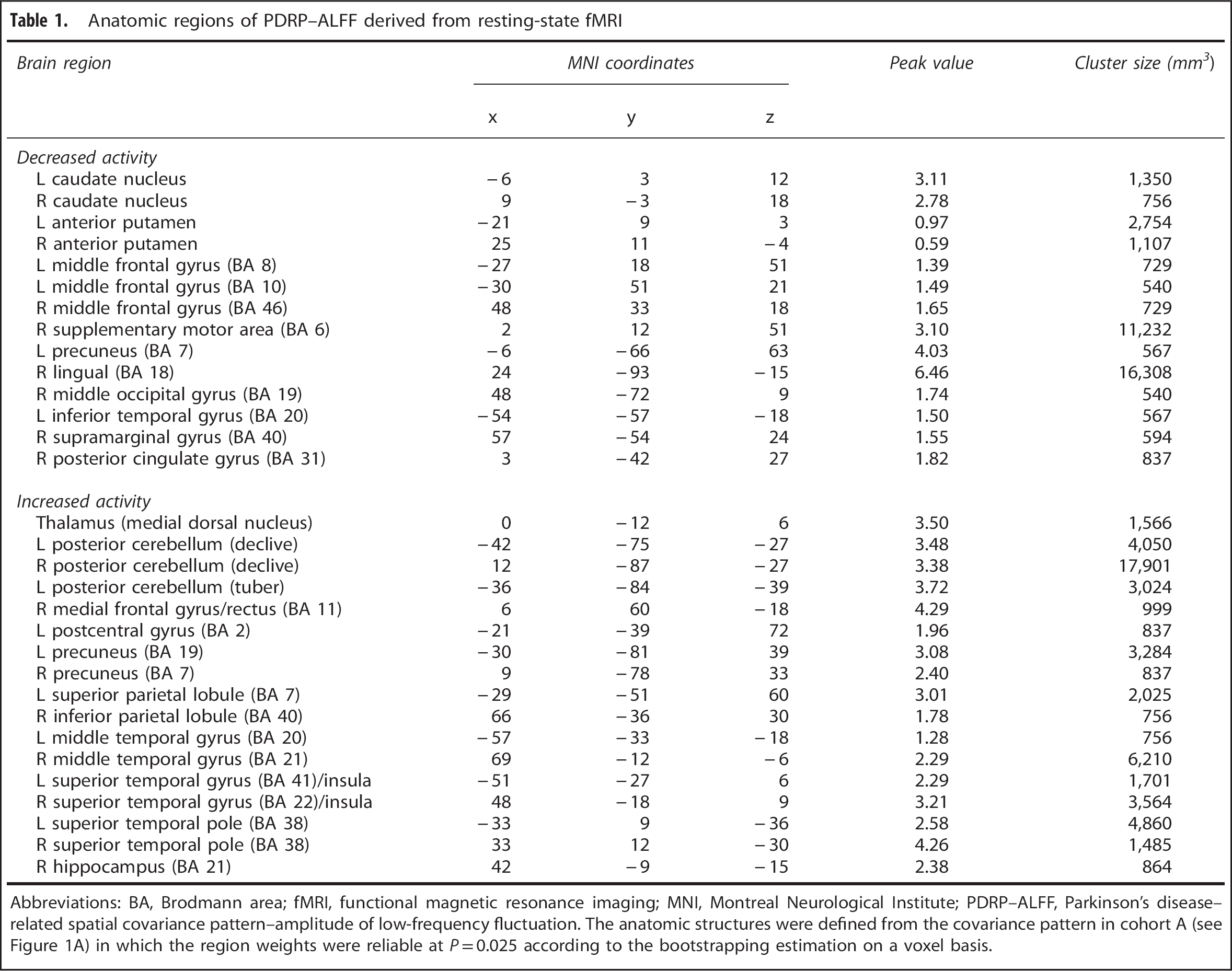
Abbreviations: BA, Brodmann area; fMRI, functional magnetic resonance imaging; MNI, Montreal Neurological Institute; PDRP–ALFF, Parkinson's disease–related spatial covariance pattern–amplitude of low-frequency fluctuation. The anatomic structures were defined from the covariance pattern in cohort A (see Figure 1A) in which the region weights were reliable at
Subject scores of PDRP–ALFF networks and ROC parameters in patients with PD and normal controls
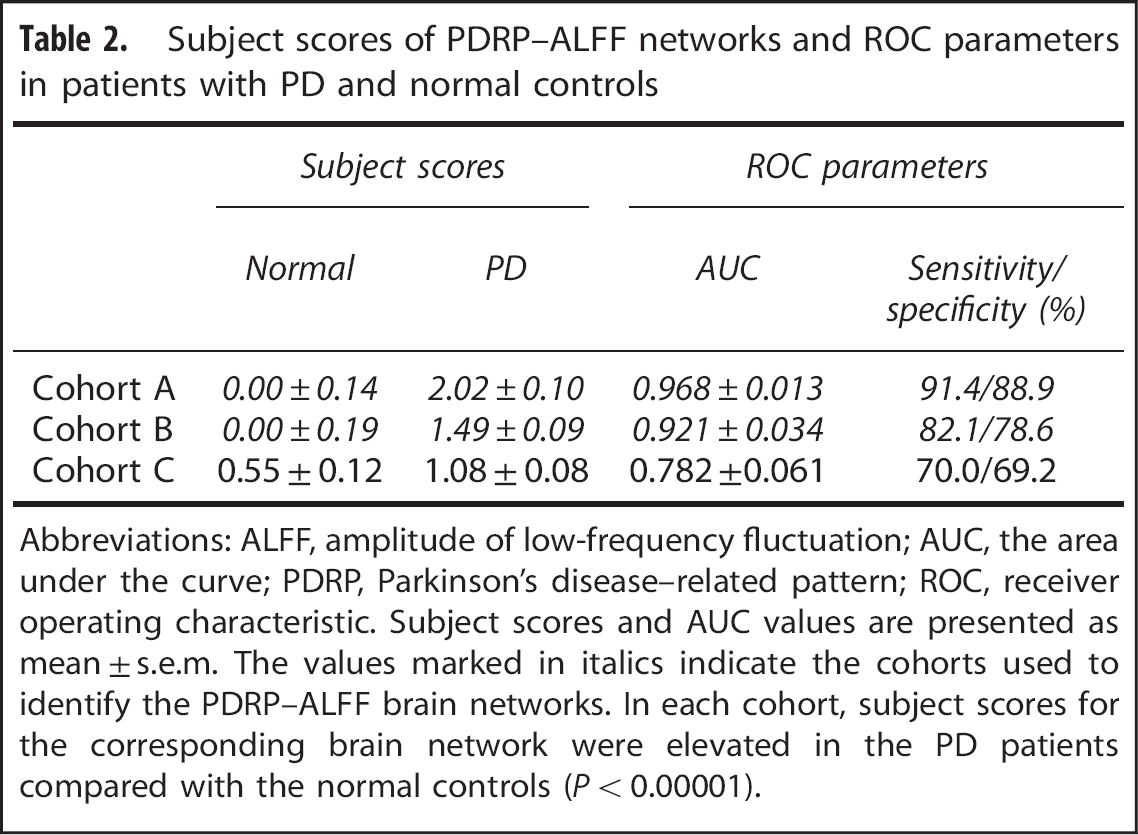
Abbreviations: ALFF, amplitude of low-frequency fluctuation; AUC, the area under the curve; PDRP, Parkinson's disease–related pattern; ROC, receiver operating characteristic. Subject scores and AUC values are presented as mean ± s.e.m. The values marked in italics indicate the cohorts used to identify the PDRP–ALFF brain networks. In each cohort, subject scores for the corresponding brain network were elevated in the PD patients compared with the normal controls (
Reproducibility in the Split Sample
A PD-related spatial covariance pattern of neural activity was produced in cohort B and characterized by decreased activity in the pre-SMA, precuneus, bilateral caudate nucleus, and right lingual and inferior temporal gyri, and increased activity bilaterally in the middle temporal gyrus and cerebellum (Figure 2B). This pattern was defined from a set of principal components accounting for a total subject × voxel variance of 35.3% and found to be reliable (
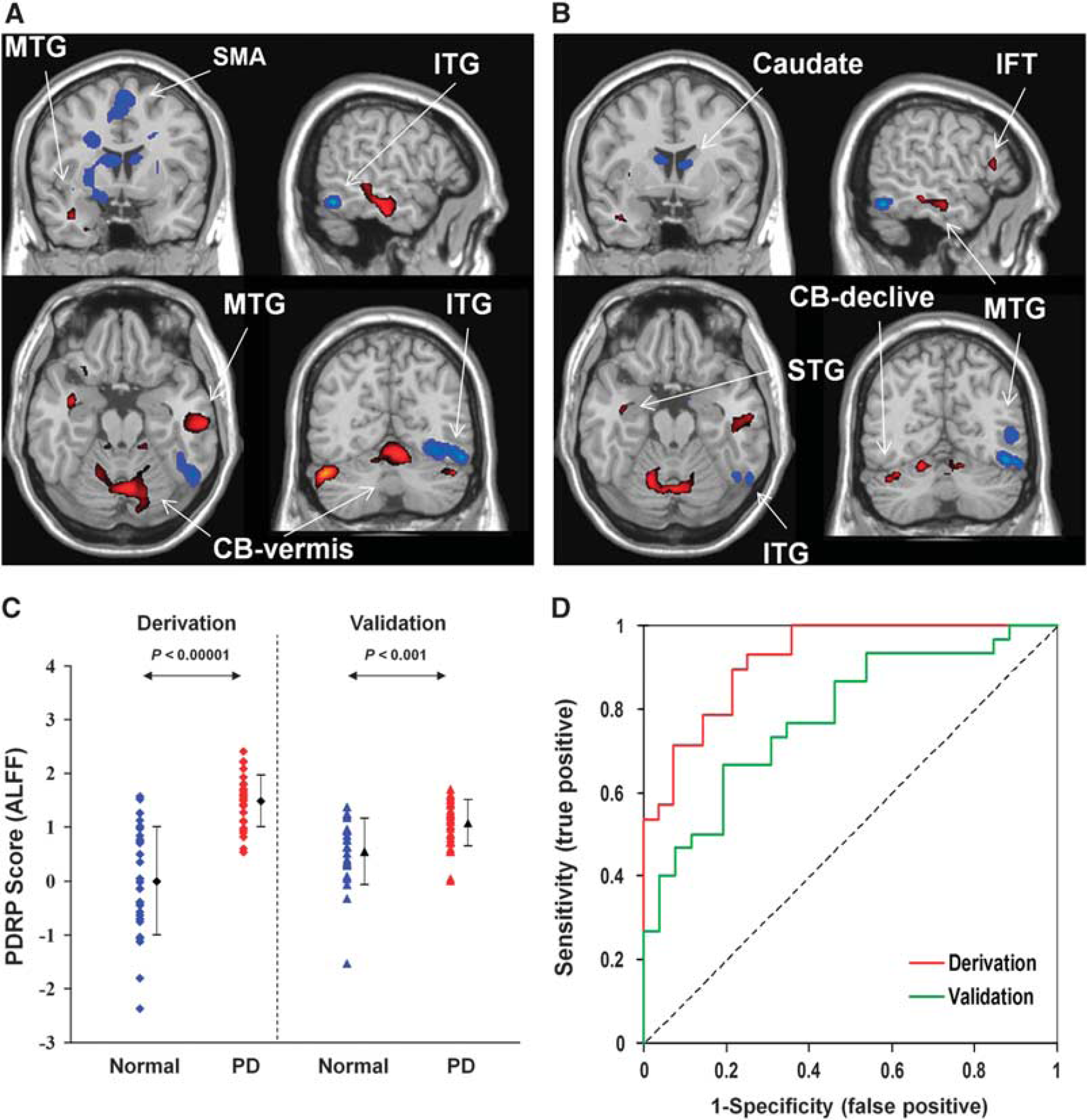
The comparison of Parkinson's disease–related spatial covariance pattern–amplitude of low-frequency fluctuation (PDRP–ALFF) brain networks and group discrimination in patients with PD and healthy controls in the split-sample analysis. (
DISCUSSION
This is the first study that used multivariate SSMPCA methodology to investigate disease-related brain networks in rsfMRI time series. The PD-related spatial covariance pattern we have identified is, to a large part, topographically comparable to the previous results with regional homogeneity, 12 or cross-validation analysis of ALFF in PD patients.13,14 Although the physiology of fMRI signal is not fully understood, these related studies reveal local and system-level abnormalities in a large number of widely distributed regions and relay stations within the basal ganglia–thalamo–cortical and cerebello–thalamo–cortical circuits.
The PDRP derived from FDG PET is characterized by increased metabolic activity in the posterior putamen, globus pallidus, thalamus, sensorimotor cortex, pons, and cerebellum, along with decreased metabolic activity in the premotor, SMA, and parietooccipital association cortices.25,26 Compared to the PDRP from FDG PET, the PDRP derived from rsfMRI not only shows some overlap, i.e., decreased activity in the SMA and occipital cortex, and increased activity in the thalamus and cerebellum but also exhibits important differences. For example, neural activity is decreased in the caudate nucleus and anterior putamen, and increased in the parietal cortex but does not change in the globus pallidus, sensorimotor cortex, and premotor areas in the PDRP from rsfMRI (Figure 1A). These differences should be due mostly to methodological factors. 18 F-fluorodeoxyglucose PET data measure synaptic neural activity via regional cerebral glucose metabolism, whereas ALFF may reflect the variability of local spontaneous neural activity in fMRI signal. The relationship between fMRI signal and FDG uptake is likely to be very complex and may be better understood by multimodal neuroimaging, neurophysiologic, and pathologic studies in animal models.
Several rsfMRI studies have investigated PD-related spontaneous neural activity or network connectivity.12–18,27 These studies have provided useful information on understanding the neural correlates of clinical symptoms in PD, like tremor, 15 apathy, depression and motor dysfunction, 14 and cognitive impairment, 27 indicating that rsfMRI is useful in the investigation of neurophysiologic mechanisms underlying PD. Our findings further show that rsfMRI may be used to identify PDRP for possible assessment of network abnormality in individual subjects even though our study sample consisted mostly of patients at early to moderate stages.
Most regions in our PDRP–ALFF are involved in the corticobasal ganglia–thalamo–cortical loops, like the SMA, thalamus, caudate nucleus, putamen, temporal cortex, and limbic lobe, which include several parallel circuits such as sensorimotor, associative, and limbic circuits. The sensorimotor circuit projects somatotopically from the primary motor cortex, premotor area, and SMA to the putamen, then through the thalamus projects back to these cortical motor areas. 28 The dysfunction of the sensorimotor circuit has been reported to cause motor difficulties in PD, like akinesia and bradykinesia. 29 The associative circuit projects from associative cortical areas, like the prefrontal cortex, and the temporal regions to the caudate nucleus and the putamen, 30 whereas the limbic circuit projects from the limbic cortices, the amygdala and the hippocampus to the ventral striatum. 31 It has been shown that some nonmotor signs in PD, like cognitive decline, could be linked to the reduced 18 F-fluorodopa uptake in the associative circuit, 32 as well as in the limbic circuit. 33
The PDRP–ALFF reported in this study also contains areas outside the corticobasal ganglia–thalamo–cortical loops, e.g., the cerebellum. The cerebellum is known to influence motor and cognitive operations via the cerebello–thalamo–cortical circuit. 34 Increasing evidence suggests that this circuit is an important mechanism underlying pathophysiology of the PD resting tremor.15,35 However, as all patients in this study have mild tremor at most, the results reported here were not likely to be influenced by the factors related to resting tremor. The increased activity in the cerebellum is possibly a compensation for basal ganglia dysfunction. 36
The PDRP–ALFF expression from rsfMRI showed a high accuracy of ~90% to discriminate PD patients from healthy controls in the large sample of cohort A (91% sensitivity and 89% specificity). Despite methodological differences between studies this finding was in line with several previous reports,13,18 suggesting that rsfMRI could be potentially useful for differentiating PD patients from healthy controls at an individual level. Although the subject scores of the PDRP–ALFF did not change with regard to gender and age in the normal controls, they were higher in men and correlated positively with age in the patients. The latter confirmed with our patient sample in which men were slightly older and had marginally more severe motor symptoms than women.
We assessed the reproducibility of PDRP–ALFF in the split-sample analysis, which gave rise to a topographic pattern very similar to that from the entire cohort. Pattern expression was abnormally elevated (
Research over the last decade has shown that FDG PET and fMRI provide unique and complementary information in measuring changes in local neural activity. Despite currently inferior signal-to-noise ratio compared with FDG PET, fMRI modality does offer several unique advantages. In addition to higher spatial and temporal resolutions, MRI scanners are more widely available and do not involve ionizing radiation. This allows a single subject to be scanned repeatedly, and more subjects to be scanned to increase statistical power. Compared to the conventional task-based fMRI methods, rsfMRI is easy to perform and not too demanding on the participants, and can circumvent task-related confounds, increase signal-to-noise ratio, and expand patient populations. It is now possible to use rsfMRI to evaluate neuronal activity instead of FDG PET as shown by many studies cited in this paper and elsewhere (see the reviews by Prodoehl
It is unclear whether our findings can be generalized to all PD patients because of some limitations in this study. First, our patients had mild tremor at most to facilitate rsfMRI. The FDG PET study has shown that tremor-dominant PD patients have a distinct spatial covariance pattern of regional cerebral glucose metabolism. 35 Further rsfMRI studies are necessary to explore tremor-related spatial covariance pattern of brain activity. Second, our patients exhibited typical motor symptoms of rigidity and bradykinesia mostly at early to moderate stages. This narrow range of disease severity resulted in part from the recruitment of a large PD cohort with mild tremor. Extensive work in parkinsonian patients at early stage, and with atypical symptoms is still needed before rsfMRI can be used as a biomarker to identify PD patients at an individual level. It is also necessary to examine the relationship between the PDRP–ALFF scores and clinical symptoms by studying patients with a wide spread of UPDRS motor ratings or Hoehn and Yahr stages. Third, nonmotor symptoms like cognitive, psychiatric, and emotional impairments were not examined in our patients. It is well recognized that nonmotor problems are present in most PD patients and their neuroimaging correlates can be assessed by rsfMRI. 14 Thus, the influences of these nonmotor symptoms on PDRP and corresponding cognitive-related patterns need to be investigated in the future.
An inherent limitation in using rsfMRI on PD is that tremor can disturb fMRI signal. Thus, up to now, most fMRI studies have excluded patients with obvious tremor. Some improvement in data acquisition and analysis can be helpful in overcoming this problem. For example, simultaneous electromyography monitoring during scanning is helpful to regress out the influence of tremor on fMRI signal. Motion correction during data analysis also has benefits in reducing movement artifacts. With continued improvement in imaging and data analysis techniques, the application of rsfMRI to tremor-dominant PD is increasingly becoming feasible.
In summary, we have shown that rsfMRI can be used for identification of disease-related covariance patterns, and has the potential for differentiation of PD patients from healthy controls on a single-case basis. It is critically important to further develop and validate this technique, and optimize imaging protocols ranging from innovations in coil design and acquisition sequences/parameters to advances in signal-processing strategies and data analytical methodologies. Further studies are needed to test the replicability of PDRP–ALFF prospectively in new patient samples, validate its test–retest stability and reproducibility with different MRI scanners, and assess its clinical correlation and modulation by antiparkinsonian therapies. Such comprehensive technical improvement in the rsfMRI method and more rigorous validation in independent patient populations can be helpful for establishing a simple and noninvasive imaging biomarker for PD and related disorders.
Footnotes
TW was involved in conception, organization, and execution of the research; writing of the first draft, review, and critique of the manuscript. YM participated in conception of the research, analysis of the data, review, and critique/revision of the manuscript. ZZ was involved in analysis of the data. SP was responsible for network analysis of the data and preparation of the results. XW was involved in execution of the research. DE gave valuable suggestions on the design of data analysis strategy and was responsible for the revision of the manuscript. PC was involved in conception of the research.
The authors declare no conflict of interest.
