Abstract
To investigate putative interacting or distinct pathways for hippocampal complex substructure (HCS) atrophy and cognitive affection in early-stage Alzheimer's disease (AD) and cerebrovascular disease (CVD), we recruited healthy controls, patients with mild cognitive impairment (MCI) and poststroke patients. HCSs were segmented, and quantitative white-matter hyperintensity (WMH) load and cerebrospinal fluid (CSF) amyloid-β concentrations were determined. The WMH load was higher poststroke. All examined HCSs were smaller in amyloid-positive MCI than in controls, and the subicular regions were smaller poststroke. Memory was reduced in amyloid-positive MCI, and psychomotor speed and executive function were reduced in poststroke and amyloid-positive MCI. Size of several HCS correlated with WMH load poststroke and with CSF amyloid-β concentrations in MCI. In poststroke and amyloid-positive MCI, neuropsychological function correlated with WMH load and hippocampal volume. There are similar patterns of HCS atrophy in CVD and early-stage AD, but different HCS associations with WMH and CSF biomarkers. WMHs add to hippocampal atrophy and the archetypal AD deficit delayed recall. In line with mounting evidence of a mechanistic link between primary AD pathology and CVD, these additive effects suggest interacting pathologic processes.
INTRODUCTION
Traditionally, cerebrovascular disease (CVD) has been an exclusion criterion for Alzheimer's disease (AD), but today there is strong evidence for a connection between AD and CVD, 1 and there are indications of a direct mechanistic and reciprocal link between cerebral vascular dysfunction and AD-linked neurodegeneration. Cerebral microvasculature in AD is consistently shown distorted with endothelial atrophy and pericyte degeneration, 2 and similar capillary affection is shown in most conditions predisposing to stroke. 3 The presence of large-vessel cerebral atherosclerosis is associated with amyloid plaques postmortem, 4 and the neurovascular unit and blood–brain barrier are disrupted in both CVD and AD, leading in turn to neurochemical imbalance. 5
White-matter hyperintensities (WMHs) are characterized by demyelination and axonal rarefaction, and are used as surrogate markers of small vessel CVD. 6 Though associated with lower levels of cerebrospinal fluid (CSF) amyloid β 1–42 (Aβ42) (and other amyloid precursor protein metabolites), 7 hypoxia is known to upregulate β-secretase activity and is associated with amyloid deposition and neuritic plaque formation. 8 Hippocampal atrophy in CVD is well described in patients with and without evidence of amyloid plaques, 9 and up to 50% of poststroke patients will develop dementia. 10
The hippocampus consists of several histologically heterogeneous subregions: the cornu ammonis (CA) comprising sectors 1 to 4 and the dentate gyrus (DG). AD typically involves atrophy of the hippocampal complex (the hippocampus and interconnected subhippocampal entorhinal (ERC) and perirhinal (PRC) cortices), and recent reports suggest selective and consecutive vulnerability of pertinent hippocampal complex substructures (HCSs) and cerebral cognitive networks. 11 Substantial atrophy of ERC layer II is seen in very mild AD, 12 and ensuing degeneration of the perforant pathway (originating in this layer and a major hippocampal input for memory processing) is important in hippocampal neocortical deafferentation. 13 ERC atrophy is also a predictor of deterioration from mild cognitive impairment (MCI, a heterogeneous clinical condition associated with an increased risk of dementia 14 ) to Alzheimer's dementia, 15 but whether neurons within the HCS are similarly affected in CVD and early-stage AD remains an open question.
Early-stage neurofibrillary pathology is seen in the medial temporal lobe, first affecting the PRC, then the ERC, 16 before moving on to the hippocampus (first appearing in CA1). Both CSF total microtubule-associated protein tau (T-tau) and phosphorylated microtubule-associated protein tau (P-tau) are established biomarkers of AD neurofibrillary pathology, but T-tau is an unspecific marker of neurite damage (and increased in both AD and poststroke), whereas increased P-tau is considered AD specific. 17
To reveal putative interacting or distinct pathways for HCS atrophy and cognitive affection in predementia AD and CVD, we included healthy controls, poststroke cases, and patients with MCI (stratified according to CSF Aβ42 levels into amyloid-positive MCI and all-cause MCI) to answer the following research questions (RQ):
MATERIALS AND METHODS
Eligibility Criteria, Measures of Cognitive Impairment, and Ethical Conduct
Patients with MCI were recruited from a university-hospital based memory clinic. Spouses of participating patients were potentially eligible as controls provided clinically established normality with regard to memory, emotionality, and tempo. The inclusion procedure adheres to the Petersen criteria and is extensively documented elsewhere. 18
Stroke patients were recruited from a university-hospital based stroke unit. Inclusion criteria for these patients were cortical and lacunar ischemic infarctions, between 40 and 79 years of age, and mini-mental state examination
19
(MMSE) score ≥ 23, no severe problems of language, and visual/auditory neglect. Exclusion criteria were a history of somatic or psychiatric disorder including depression. Only patients who agreed to undergo CSF sampling were included. Thirty-seven poststroke subjects fulfilled these criteria, but ten were excluded due to pathologic CSF Aβ42 (see
To obtain CSF from controls, a secondary control group was recruited before lumbar puncture in conjunction with orthopedic surgery. Thirty-four of these subjects assented and successfully underwent magnetic resonance imaging (MRI) (on a different scanner). Normality with regard to memory, emotionality, and tempo (as with the primary control group) was established in a subsequent clinical interview. Three subjects were excluded due to pathologic CSF Aβ42, and five subjects were excluded due to cognitive complaints, leaving twenty-six subjects for the secondary control group.
The patient populations and the primary control group were directly comparable, but CSF was not available for the primary control group (Supplementary Tables 1 and 2).
All subjects gave their written consent, and the Regional Committee for Medical and Health Research Ethic South-East evaluated (based on the Norwegian Health and Research Act and the Helsinki Declaration of 1964; revised 2013) and approved the study. All further study conduct was based on and in line with these guidelines.
Magnetic Resonance Imaging
For the primary control group, the MCI and stroke groups, MRI scans were obtained from a Siemens Espree 1.5 T system (Siemens Medical Solutions, Erlangen, Germany), one MPRAGE 3D sequence was acquired (repetition time/echo time/inversion time (TI)/flip angle (FA) = 2,400/3.65/1,000/8°, matrix = 240 × 192), 160 sagittal slices, thickness = 1.2 mm, in-plane resolution of 1 mm × 1.2 mm. The protocol also included 2D axial FLAIR (fluid-attenuated inversion recovery) images with the following parameters: repetition time/echo time/inversion time (TI) = 13,420/121/2,500, 36 slices, spaced at 3.0 mm and 3.9 mm thick.
For the secondary control group, MRI scans were acquired on a Philips Achieva 3 T system (Philips Medical Systems, Best, The Netherlands). At 3 T, a single 3D turbo field echo sequence was acquired for morphometric analysis with the following sequence parameters: repetition time/echo time/inversion time (TI)/flip angle (FA) = 4.5 ms/2.2 ms/853 ms/8°, matrix = 256 × 213, 170 slices, thickness = 1.2 mm, in-plane resolution of 1 mm × 1.2 mm.
Magnetic resonance imaging was performed consecutively after inclusion in the control and MCI groups, and 3 months after stroke. Stroke location, type, and size were determined (Supplementary Table 3).
Population characteristics
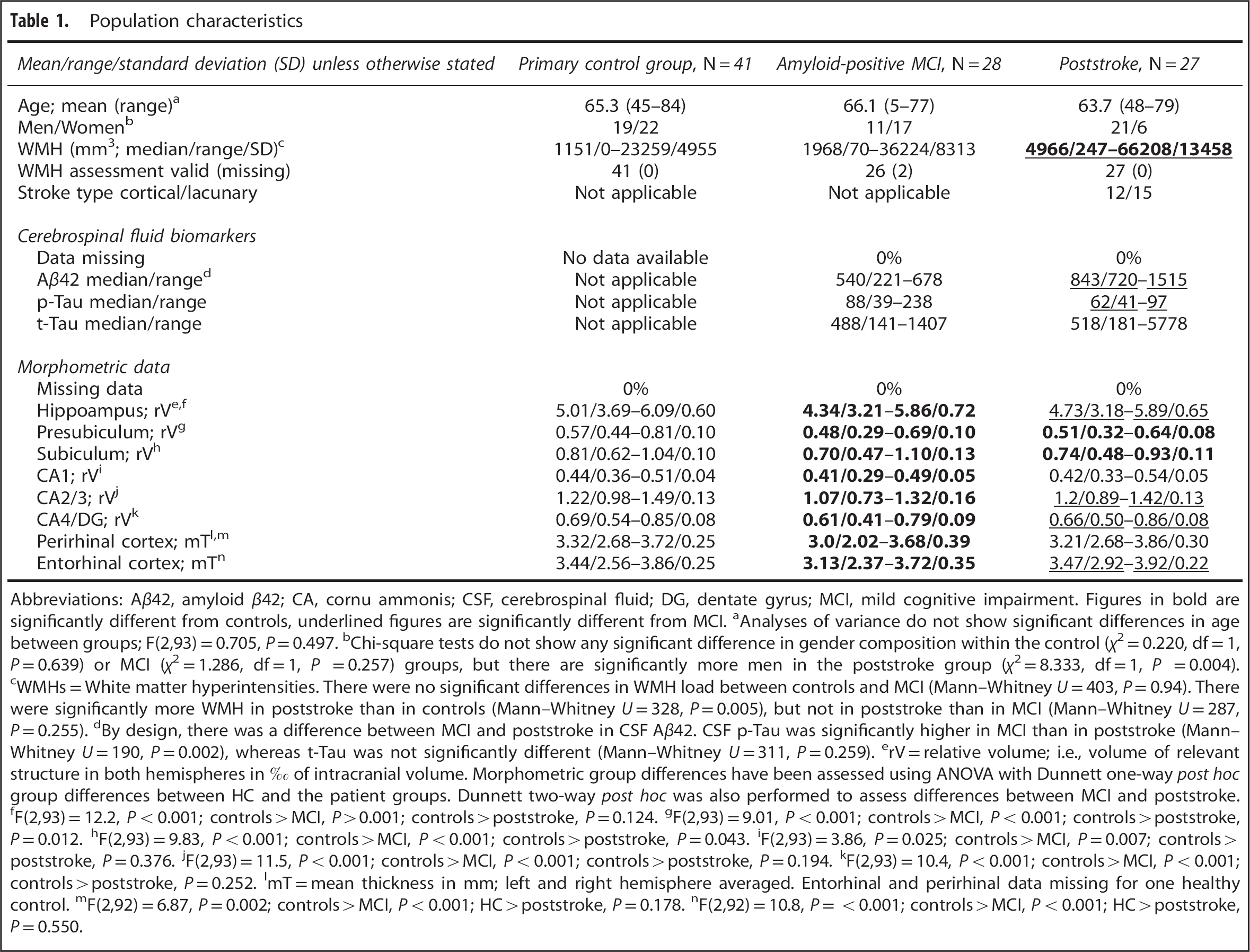
Abbreviations: Aβ42, amyloid β42; CA, cornu ammonis; CSF, cerebrospinal fluid; DG, dentate gyrus; MCI, mild cognitive impairment. Figures in bold are significantly different from controls, underlined figures are significantly different from MCI.
Analyses of variance do not show significant differences in age between groups; F(2,93) = 0.705,
Chi-square tests do not show any significant difference in gender composition within the control (χ2 = 0.220, df = 1,
WMHs = White matter hyperintensities. There were no significant differences in WMH load between controls and MCI (Mann–Whitney
By design, there was a difference between MCI and poststroke in CSF Aβ42. CSF p-Tau was significantly higher in MCI than in poststroke (Mann–Whitney
rV = relative volume; i.e., volume of relevant structure in both hemispheres in %o of intracranial volume. Morphometric group differences have been assessed using ANOVA with Dunnett one-way
F(2,93) = 12.2,
F(2,93) = 9.01,
F(2,93) = 9.83,
F(2,93) = 3.86,
F(2,93) = 11.5,
F(2,93) = 10.4,
mT = mean thickness in mm; left and right hemisphere averaged. Entorhinal and perirhinal data missing for one healthy control.
F(2,92) = 6.87,
F(2,92) = 10.8,
Population neuropsychological characteristics
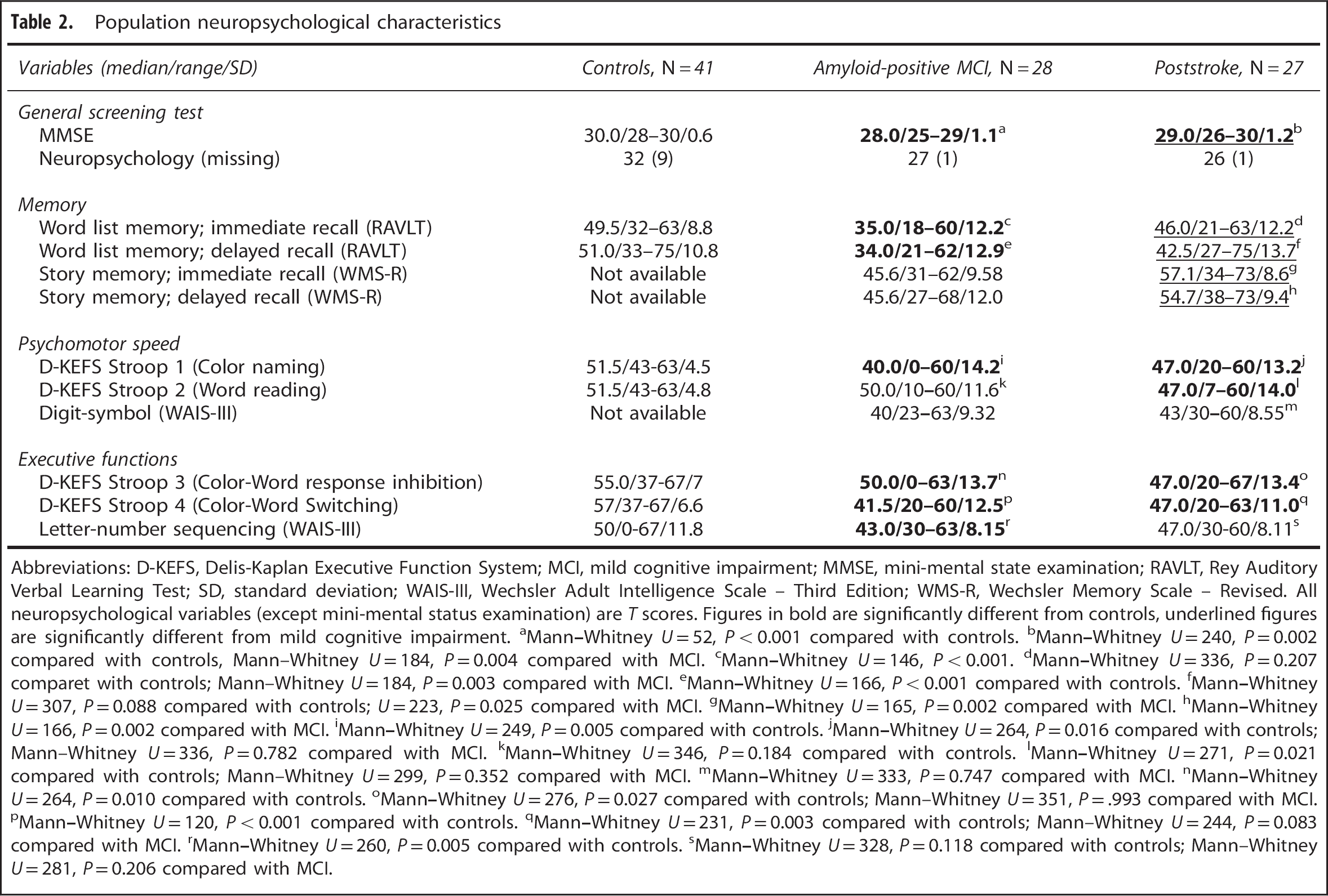
Abbreviations: D-KEFS, Delis-Kaplan Executive Function System; MCI, mild cognitive impairment; MMSE, mini-mental state examination; RAVLT, Rey Auditory Verbal Learning Test; SD, standard deviation; WAIS-III, Wechsler Adult Intelligence Scale – Third Edition; WMS-R, Wechsler Memory Scale – Revised. All neuropsychological variables (except mini-mental status examination) are
Mann–Whitney
Mann–Whitney
Mann–Whitney
Mann–Whitney
Mann–Whitney
Mann–Whitney
Mann–Whitney
Mann–Whitney
Mann–Whitney
Mann–Whitney
Mann–Whitney
Mann–Whitney
Mann–Whitney
Mann–Whitney
Mann–Whitney
Mann–Whitney
Mann–Whitney
Mann–Whitney
Mann–Whitney
Within group Spearman correlations between WMH load and hippocampal complex structure sizes
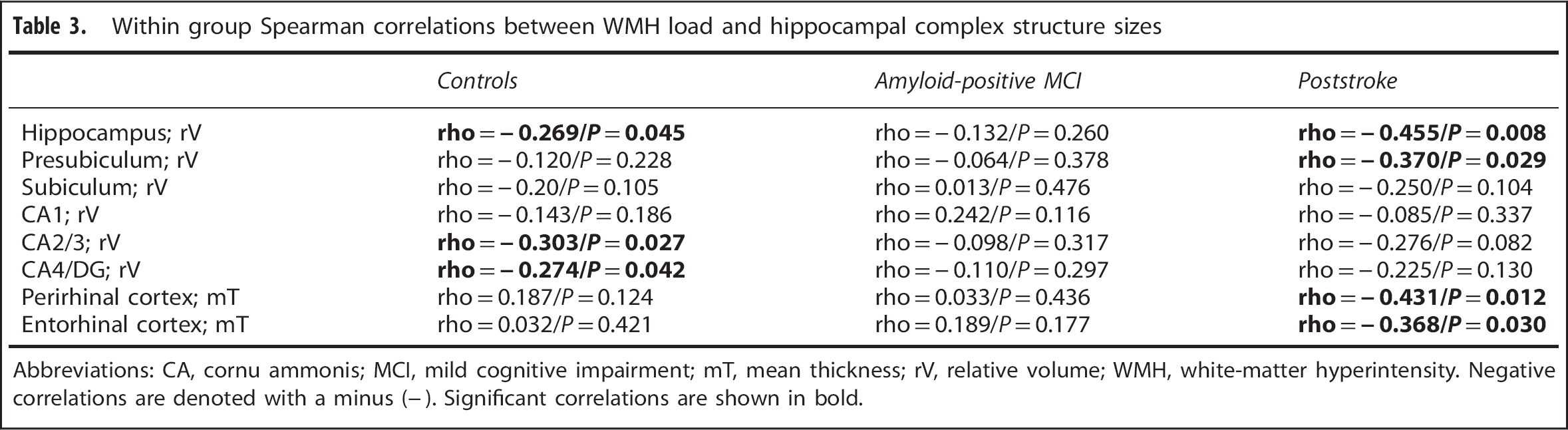
Abbreviations: CA, cornu ammonis; MCI, mild cognitive impairment; mT, mean thickness; rV, relative volume; WMH, white-matter hyperintensity. Negative correlations are denoted with a minus (−). Significant correlations are shown in bold.
Magnetic Resonance Imaging Segmentations and Analyses
Cortical reconstruction and volumetric segmentation were performed with the FreeSurfer image analysis suite version 5.3.0 (http://surfer.nmr.mgh.harvard.edu/). This includes segmentation of the subcortical white-matter and deep gray-matter volumetric structures
21
and parcellation of the cortical surface. This labels cortical sulci and gyri, and thickness values are calculated in the regions of interest. Further, an automated procedure for subsegmentation of the hippocampus, as implemented in FreeSurfer, was employed.
22
The thickness values of the ERC and the PRC were calculated using methods based on ultra-high resolution
The WMH load was estimated by a recently developed in-house object-based supervised machine learning algorithm for automated segmentation and quantification of WMHs based on FLAIR image intensity and masks of tissue types. 24 This provides an automated quantitative assessment of WMH load (Figure 1). All segmentations were manually inspected for accuracy; areas radiologically considered to represent acute stroke were manually masked and excluded from the analyses.
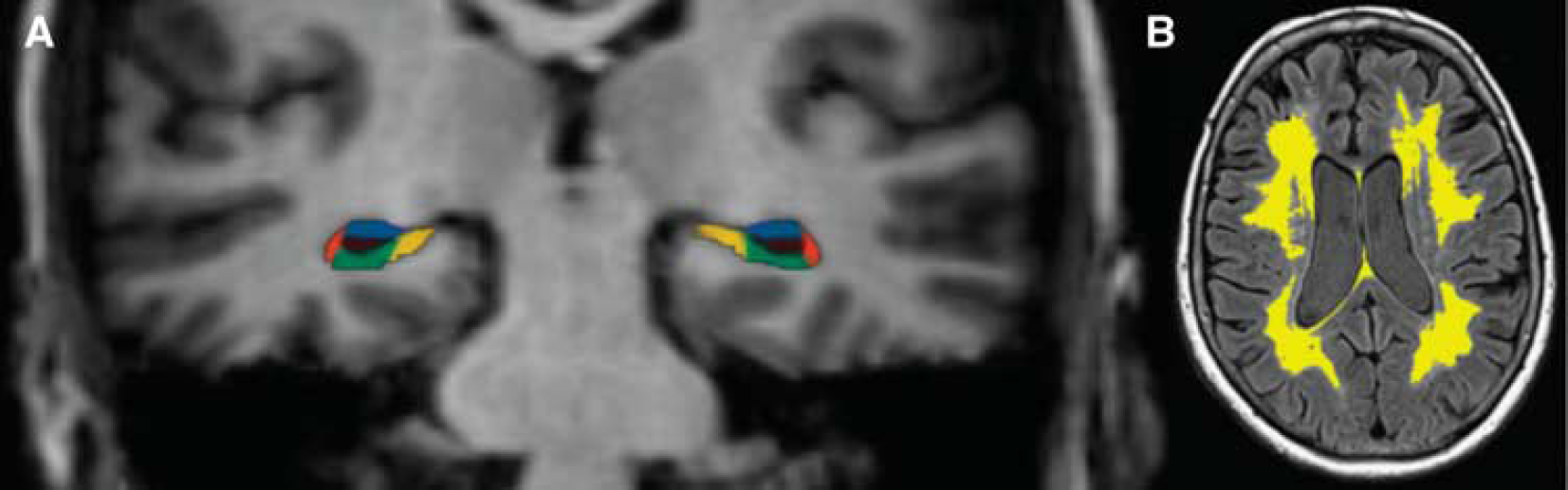
(
Cerebrospinal Fluid Biomarkers
Cerebrospinal fluid samples were collected from all patients by lumbar puncture through the L3/L4 or L4/L5 intervertebral space. The lumbar puncture was performed between 0900 and 1200 hours, and consecutively after inclusion in the MCI group, 7 to 10 days after stroke or on the day of inclusion for the secondary control group. Cerebrospinal fluid Aβ42, T-tau, and P-tau were routinely examined. The CSF T-tau level was considered as abnormal if T-tau ≥ 300 ng/L for patients under 50 years, > 450 ng/L for patients from 50 to 69 years, and ≥ 500 ng/L for patients from 70 years and above. Cerebrospinal fluid P-tau was considered as pathologic if ≥ 80 ng/L, and CSF Aβ42 was considered as pathologic if < 700 ng/L.
Neuropsychological Assessment
Neuropsychological examination was performed 3 months after stroke or within 3 months of MRI and CSF sampling in the MCI and control groups. In addition to the general screening test MMSE, tests of memory, processing speed, and executive functions have been administered. The memory tests included Logical Memory Subtest from the Wechsler Memory Scale – Revised (WMS-R) 25 and the Rey Auditory Verbal Learning Test (RAVLT). 26 The tests of processing speed and executive functions comprise the Digit Symbol subtest of the Wechsler Adult Intelligence Scale – Third Edition (WAIS-III), 27 Delis-Kaplan Executive Function System (D-KEFS) Color-Word Interference Test, 28 and The Letter-Number Sequencing task from WAIS-III.
Statistical Analyses
For the hippocampus and the hippocampal subregions, volumes from the left and right hemispheres were added, and relative volumes (per mille of total intracranial volume) were computed. For the ERC and the PRC, mean thickness of the left and right hemispheres was computed. Analyses of variance were used to assess differences in age between the groups. Chi-square tests were used to assess gender composition within the groups. The Mann–Whitney test was used to assess group differences in WMH load.
RQ 3 is performed with the secondary control group, the all-cause MCI group, and the poststroke group. Demographic data of the secondary control group and the all-cause MCI subgroup are presented in Supplementary Tables 1 and 2.
Due to skewed CSF biomarker data, one-way Spearman correlation was used to assess relation to HCS size (corrected for age and gender as in RQ2).
RESULTS
There were no significant age differences between groups, but there were significantly more men in the poststroke group (Table 1). The WMH load was modest in MCI and controls, and significantly more extensive in stroke. By design, CSF Aβ42 was lower in amyloid-positive MCI than in poststroke. Cerebrospinal fluid P-tau was significantly higher in amyloid-positive MCI than in poststroke, but there were no significant difference in CSF T-tau. On MMSE, controls performed best (median score 30), MCI worse (28), and poststroke in between (29). These differences were statistically significant (Table 2). Both immediate and delayed word list memory were significantly worse in MCI compared with controls and poststroke. Story memory data were not available for controls, but MCI performed worse compared with poststroke. Mild cognitive impairment performed worse than controls on one measure of psychomotor speed whereas poststroke performed worse on two measures. Mild cognitive impairment performed worse than controls on all three measures of executive function, whereas poststroke performed worse on two (Table 2).
All the investigated HCSs (relative volumes of the presubiculum, subiculum, CA1, CA2/3, and CA4/DG, and mean thickness of ERC and PRC) was significantly smaller in MCI than in controls (after correction for age and gender). The presubiculum and the subiculum were significantly smaller in the stroke group than in controls (Table 1).
In the stroke group, WMH load correlated with total hippocampal volume and volume of the presubiculum, PRC and ERC thickness. In MCI, WMH load did not correlate with size of any of the hippocampal complex structures, but in controls there were significant correlations with total hippocampal volume, CA2/3, and CA4/DG (Table 3).
This RQ was performed in the secondary control group, all-cause MCI group and poststroke groups (as accounted for under
Within group Spearman correlations between CSF biomarkers and hippocampal complex structure sizes
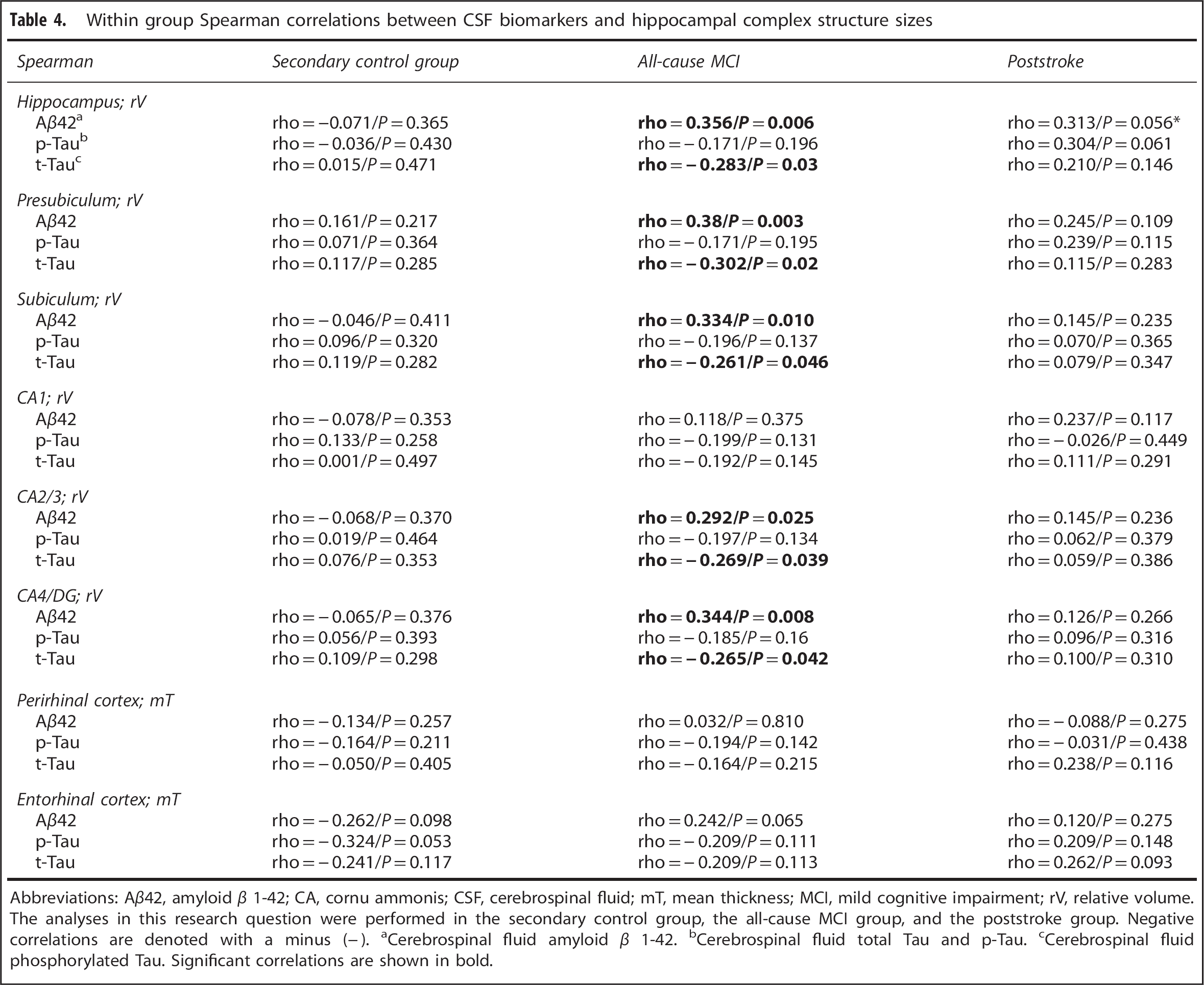
Abbreviations: Aβ42, amyloid β 1–42; CA, cornu ammonis; CSF, cerebrospinal fluid; mT, mean thickness; MCI, mild cognitive impairment; rV, relative volume. The analyses in this research question were performed in the secondary control group, the all-cause MCI group, and the poststroke group. Negative correlations are denoted with a minus (−).
Cerebrospinal fluid amyloid β 1–42.
Cerebrospinal fluid total Tau and p-Tau.
Cerebrospinal fluid phosphorylated Tau. Significant correlations are shown in bold.
In controls, there were neither significant correlations between WMH load and measures of neuropsychological function, nor hippocampal volume and measures of neuropsychological function. In MCI, there were correlations between WMH load and word list memory delayed recall, Stroop 1 and Letter-number sequencing. In poststroke, there were several near significant correlations between WMH load and memory. There were significant correlations between Stroops 1, 2, 4 and MMSE. In MCI, there were correlations between hippocampal volume and all tested neuropsychological variables of memory and executive function, but none of psychomotor speed. In CVD, there were correlations between hippocampal volume and word list memory immediate recall, story memory immediate and delayed recall as well as MMSE. Tables 5 and 6 summarize the results of RQ 4.
Within-group Spearman correlations between WMH load (corrected for age and gender) and neuropsychological function
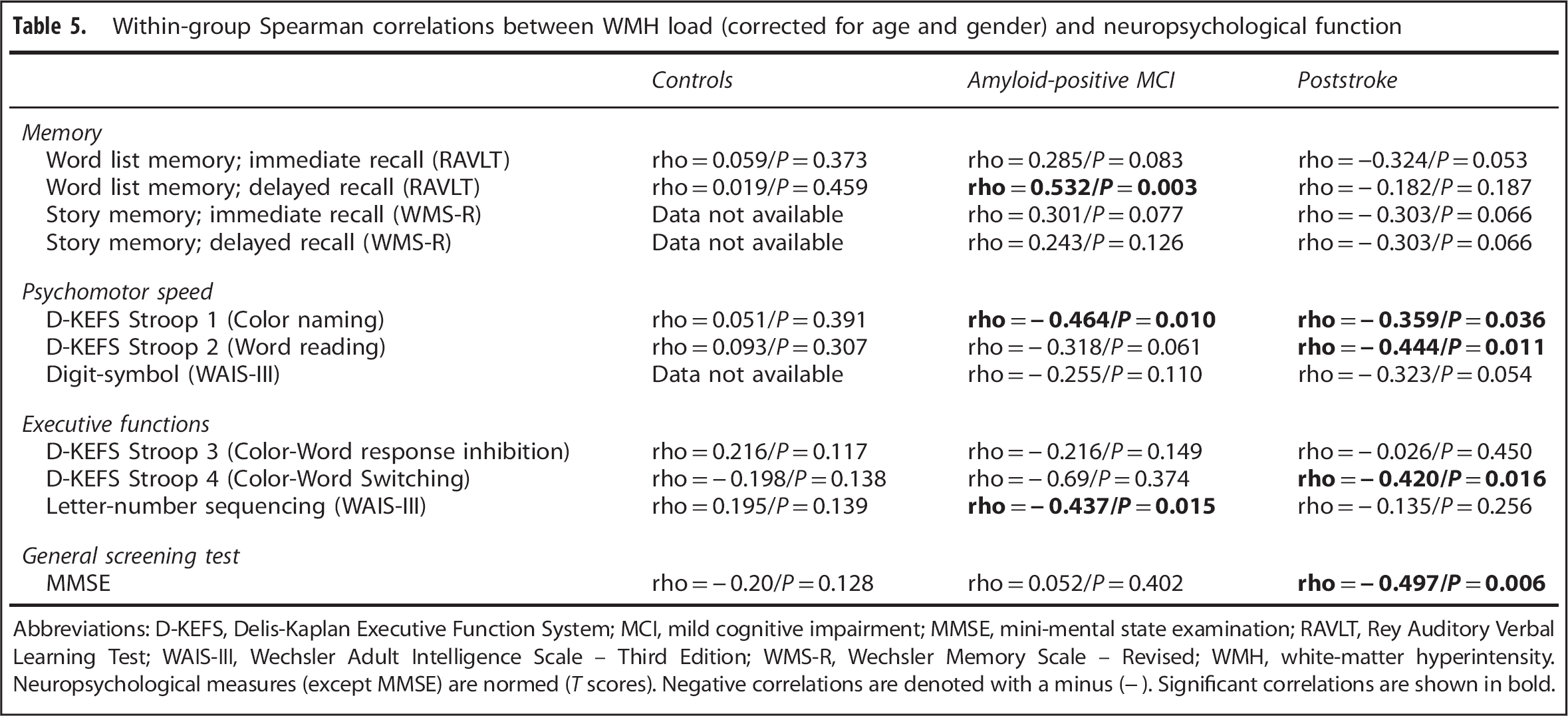
Abbreviations: D-KEFS, Delis-Kaplan Executive Function System; MCI, mild cognitive impairment; MMSE, mini-mental state examination; RAVLT, Rey Auditory Verbal Learning Test; WAIS-III, Wechsler Adult Intelligence Scale – Third Edition; WMS-R, Wechsler Memory Scale – Revised; WMH, white-matter hyperintensity. Neuropsychological measures (except MMSE) are normed (
Within-group Spearman correlations between relative hippocampal volume (corrected for age and gender) and
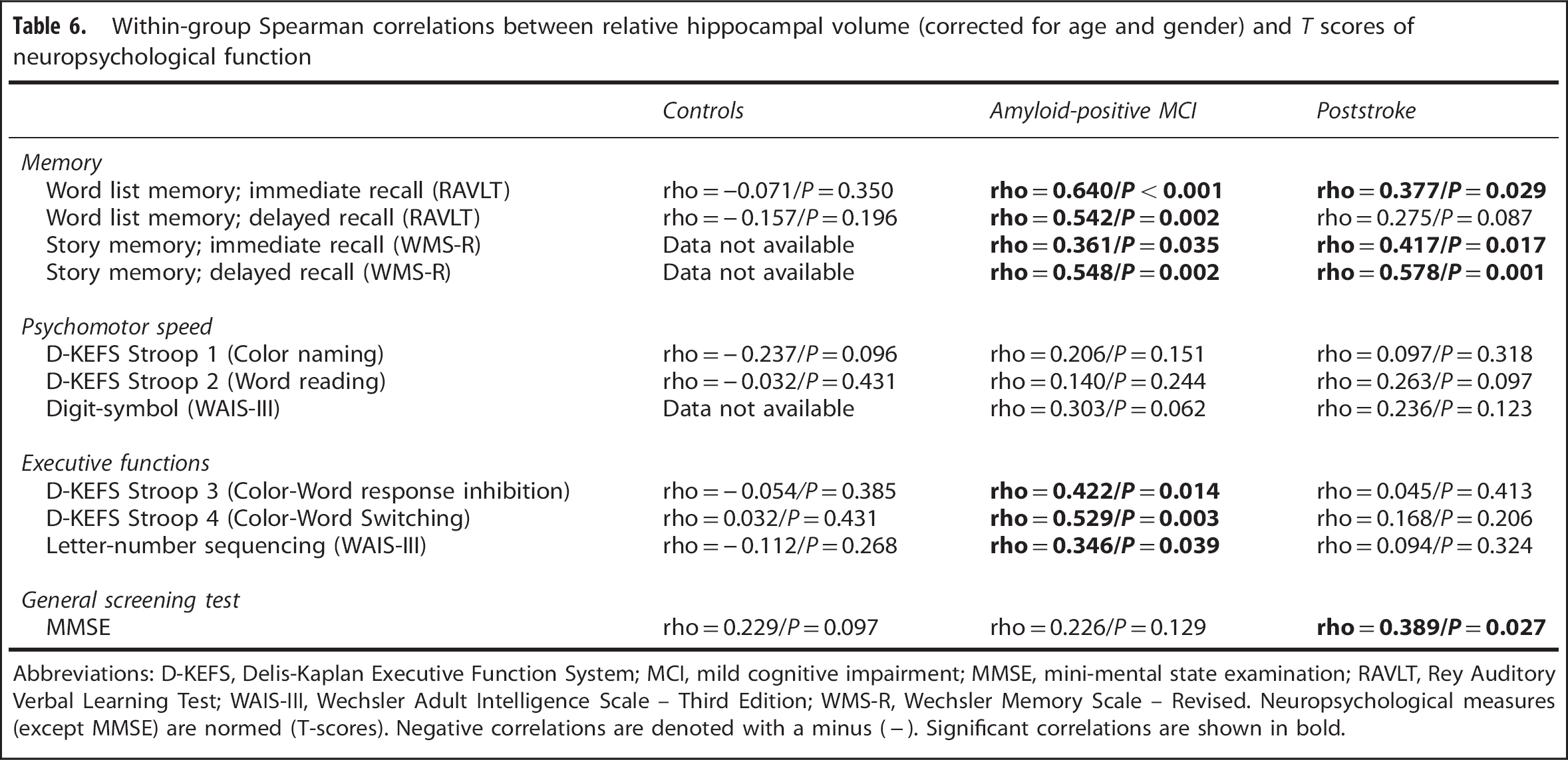
Abbreviations: D-KEFS, Delis-Kaplan Executive Function System; MCI, mild cognitive impairment; MMSE, mini-mental state examination; RAVLT, Rey Auditory Verbal Learning Test; WAIS-III, Wechsler Adult Intelligence Scale – Third Edition; WMS-R, Wechsler Memory Scale – Revised. Neuropsychological measures (except MMSE) are normed (T-scores). Negative correlations are denoted with a minus (−). Significant correlations are shown in bold.
DISCUSSION
To our knowledge, this is the first study to assess the relations between WMHs and HCSs in CVD, compared with relations in amyloid-positive MCI and controls. The main finding of this study is that the hippocampal complex is similarly affected poststroke and in early-stage AD, but there are different associations with WMH and CSF biomarkers. Also, we find a significantly higher WMH load poststroke compared with controls and amyloid-positive MCI. All examined HCSs were smaller in amyloid-positive MCI than in controls, and the presubiculum and the subiculum were smaller in poststroke than in controls. Neuropsychological measures of memory were reduced in amyloid-positive MCI as compared with controls, and measures of psychomotor speed and executive function were reduced in both poststroke and amyloid-positive MCI as compared with controls. Poststroke (but not MCI) WMH load correlated with total hippocampal and size of the presubiculum, PRC and ERC. In both poststroke and amyloid-positive MCI, there were several correlations between measures of neuropsychological function and both WMH load and hippocampal volume. Strikingly, in amyloid-positive MCI, there was a strong correlation between WMH load and the archetypal AD type impairment delayed recall.
The present results support previous reports that both CVD 9 and AD cause medial temporal lobe atrophy. Although the patterns of medial temporal lobe atrophy in MCI and CVD are similar, there are differential associations with WMHs and CSF biomarkers. In terms of medial temporal atrophy, CVD is important in poststroke patients but has a minor role in amyloid-positive MCI. Vice versa, CSF biomarker pathology is important in MCI, but not in stroke.
The hippocampal complex is vulnerable due to plasticity, neuronal projections, transmitter systems, and oxygen demand. There are high concentrations of NMDA receptors, the presubiculum has consistently been identified with strong acetylcholine staining, and several other diseases (e.g., temporal lobe epilepsy, posttraumatic stress disorder, depression, and schizophrenia) are associated with hippocampal degeneration. 29 Neurofibrillary pathology appears early in the hippocampal complex, 16 and soluble amyloid oligomers are neurotoxic. 17 As such, pre-plaque Aβ induced hippocampal complex degeneration is possible (also in the poststroke group). Aβ accumulates around and affects perivascular cells, and amyloid angiopathy is present in a majority of cases. Brain amyloid is in equilibrium with amyloid in CSF and plasma, and can be transported both ways across the blood–brain barrier. Damage to the neurovascular unit and the blood–brain barrier may disrupt this equilibrium. 5 Putatively (in CVD) this may cause or worsen pre-plaque amyloid dysmetabolism, or (in AD) amyloid dysmetabolism may cause or worsen impairment of cerebrovascular autoregulation; adding to the pathogenetic load. Further research must determine the exact underlying mechanisms and which factors predispose plaque deposition and which factors cause accelerated spreading of neurofibrillary pathology.
In line with the current findings, a recent neuropathological study did not find reduced neuronal volumes in the ERC (layer V) poststroke.
30
The current methodology has not allowed for testing the integrity of the perforant path
Although all segmentations were manually inspected and the image and segmentation quality and consistency were deemed good, the segmentation procedure was automated and not performed on ultra-high field strength, high-resolution MRI and segmented according to a harmonized protocol. When choosing hippocampal subregions of interest, we only included the larger structures with the best Dice overlap measures with manual delineation. 22 Nevertheless, when interpreting the results from the present study it should be noted that the subiculum is most likely overestimated on the expense of CA1, and that there does not exist a generally accepted protocol for subsegmenting the hippocampus.
In poststroke, the anterior part of the hippocampus is smaller than in controls and in particular the anterior subhippocampal areas (ERC and PRC) correlate (inversely) with WMH load. At the present stage, the hippocampal affection is more advanced than ERC and PRC, and there is a borderline significant correlation between delayed recall of story (context-rich) and WMH load, whereas not between WMH load and delayed word list recall (context free). This is in line with the supposition that anterior subhippocampal areas are important in context-free (or episodic) memory whereas the hippocampus is thought to be important in context-rich memory. 40 The observation that, in the poststroke group, delayed recall of story memory is strongly correlated with total hippocampal volume (with a smaller, only trend significant correlation for delayed word list memory) further underlines this (Table 6).
In MCI, the relatively moderate WMH load is most strongly (inversely) correlated with the early AD-specific context-free word list memory. Even though WMH does not seem to be a major factor in in the majority of MCI cases, its occurrence adds to the archetypal AD type impairment of context-free (episodic) memory. In line with this, a recent study 9 found that an amyloid-PET positive subgroup of patients with subcortical vascular dementia performed worse on neuropsychological tests and had more extensive hippocampal atrophy (but were also older, mean age 79.5 vs. 71.9 years) than the amyloid-PET negative subgroup.
Although we have included CSF Tau biomarker data also for the stroke group, lumbar puncture was performed at a time when CSF Tau (but not Aβ42) levels are expected to be increased due to acute stroke. 17 Also, it is not known whether very early atrophy presumptively due to neurofibrillary pathology would be reflected in CSF Tau levels.
A limitation that also needs to be addressed is the relatively moderate WMH load in the MCI group as compared with poststroke. It could be construed that with a more extensive WMH load, the effects on the hippocampal complex would be more pronounced. The relative sparseness of WMH in a memory-clinic recruited MCI population is nevertheless important to note, but even though on the same volume level as controls, WMH does affect neuropsychological function in MCI.
In conclusion, we show similar patterns of HCS atrophy in CVD and amyloid-positive MCI. Different associations with WMHs and CSF biomarkers underline the separate etiologies. The effects of WMHs on neuropsychological function are in line with mounting evidence for a mechanistic link between primary AD pathology and small vessel disease (e.g., putatively reciprocal effects of hypoperfusion and AD pathophysiology2,3 or effects of hypoxia on amyloid metabolism7,8), strengthening the connection between the two entities. The similarities in patterns of structural affection may, however, limit the applicability of structural scans for differential diagnosis of predementia cognitive affection.
Footnotes
PS contributed to the conception and design of the study, acquisition of data, analyzed the data, and contributed to the interpretation of the data. PS further drafted the article. RG contributed to the conception and design of the study, acquisition and interpretation of data (notably clinical and neuropsychological data), and revised the article. MR contributed to the acquisition and interpretation of data (notably MRI data), and revised the article. AB contributed to the conception and design of the study, acquisition and interpretation of data (notably MRI data), and revised the article. LG contributed to the conception and design of the study, interpretation of data, and revised the article. EH contributed to acquisition and interpretation of data (notably neuropsychological data), and revised the article. EA, KJ, BB, and IA contributed to the acquisition and interpretation of data (notably clinical data), and revised the article. PD-T contributed to the acquisition and interpretation of data, determined stroke location and size and revised the article. KV contributed to the acquisition and interpretation of data (notably MRI data), and revised the article. TF contributed to the conception and design of the study, acquisition and interpretation of data, and revised the article. All authors approved the final version.
MR reports grants from Iceland, Liechtenstein, and Norway through the EEA Financial Mechanism, during the conduct of the study. IA reports grants from South-Eastern Norway Regional Health Authority, grants from Research Council of Norway (NASATS-Nevronor), during the conduct of the study. TF reports grants from the Norwegian Research Council, during the conduct of the study. In addition, TF has a patent PCT/EP2009/001210 issued, and a patent PCT/US2011/062233 pending.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
