Abstract
Neural stem cells (NSCs) show therapeutic potential for ischemia in young-adult animals. However, the effect of aging on NSC therapy is largely unknown. In this work, NSCs were transplanted into aged (24-month-old) and young-adult (3-month-old) rats at 1 day after stroke. Infarct volume and neurobehavioral outcomes were examined. The number of differentiated NSCs was compared in aged and young-adult ischemic rats and angiogenesis and neurogenesis were also determined. We found that aged rats developed larger infarcts than young-adult rats after ischemia (
Introduction
Ischemic stroke is the third common cause of death and the leading cause of disability in industrialized nations. Until now, the treatment of ischemic stroke remains a daunting task due to the lack of effective therapeutic strategies. Over the past decade, numerous neuroprotective drugs, which are effective for treating acute stroke in experimental animal stroke models, generally failed in clinical trials. 1 One important reason for the failure is that these studies normally use young-adult animals. 2 However, stroke is truly an age-related cerebrovascular disorder. Statistics from American Heart Association revealed that older adults in the United States had higher stroke prevalence including silent cerebral infarction. 3 Nearly 75% of stroke patients are older than 65 years. Reports show that each successive 10 years after age 55, stroke occurrence is more than doubled in both men and women. 4 More importantly, the efficacy of biologic reagents including cells are significantly varied between young-adult and aged in both human and experimental animals. Therefore, using aged animals to evaluate those potentially effective reagents are essential for stroke research and clinical translation.
Neural stem/progenitor cells (NSCs) transplantation is a potential strategy for protecting and restoring brain functions after stroke. Studies showed that NSCs from rodents, 5 non-human primates, 6 and humans 7 could survive and differentiate into functional neurons, attenuate infarction and improve neurobehavioral recovery after stroke. Despite these exciting initial successes, most of these experiments are performed in young-adult animals. Whether these treatments would benefit aged subjects remains unknown.
Aging is a critical factor in the occurrence and development of cerebrovascular diseases especially in ischemic stroke. A series of cellular and molecular changes alter the structure and function of the adult brain during the aging process. Studies showed that aging process is associated with structural and cellular functional changes in brain, 8 heart, 9 and cardiovascular system, 10 and metabolic alteration in brain; 11 aging also affects kidney structural and morphologic changes.12,13
Aging could also significantly change the microenvironment of the brain. For example, neuronal functions could be impaired by aging due to increased oxidative damage, reduced metabolic activity, and protein and lipid by-products accumulation. However, growth factors known to influence neural proliferation and survival, including vascular endothelial growth factor (VEGF), brain-derived neurotrophic factor and insulin-like growth factor-1, decline with age. 14 Since aging brain possesses a different environment from the young-adult brain, the receptivity to NSC transplants in aging rat brain needs to be explored.
In this study, we investigated whether the transplantation of NSCs in aged rats could have similar salutary effects as it does in young-adult rats after transient middle cerebral artery occlusion (tMCAO). In addition, the mechanisms by which NSCs facilitate their protective function in aged rats after ischemia were also explored.
Materials and Methods
Cell Culture, Characterization, Proliferation, and Differentiation Animal work in this study was performed in accordance with ARRIVE guidelines and animal protocol was approved by the Institutional Animal Care and Use Committee of University of North Texas Health Science Center at Fort Worth, Texas, USA (Permission number: 2011/12–21-A05) and Shanghai Jiao Tong University, Shanghai, China (Permission number: Bioethics 2012022). Neural stem cells were isolated from the telencephalon of the E14 green fluorescent protein (GFP) transgenic mice (Animal Research Center of Nanjing University, Nanjing, China) as previously described. 15 After 2 weeks expansion of monolayer cultures on a laminin-coated dish in the DMEM/F12 (1:1) medium (Gibco, Carlsbad, CA, USA) supplemented with B27 supplement (Gibco), basic fibroblast growth factor (20 ng/ml; Gibco), and epidermal growth factor (20 ng/ml; Gibco). The primary passage of NSCs was not pure, and contains mixed cells. After passage 4, cultured NSCs start to show signs of reduced viability and differentiation capacity. Therefore, we chose NSCs from passages 2 and 4 in our study. These cells strongly maintained their proliferation and differentiation ability.
To characterize cells, cultured cells were grown on ploy-L-ornithine hydrobromide (Sigma, St Louis, MO, USA) and laminin (Sigma)-coated glass coverslips in a 24-well plate (BD Falcon, San Jose, CA, USA). Cells were then immunostained with mouse anti Nestin (Millipore, Billerica, MA, USA), mouse anti Tuj-1 (Millipore), rabbit anti-glial fibrillary acidic protein+ (GFAP+) (Millipore), and mouse anti-Galactocerebroside (Millipore). To induce differentiation, neurospheres were plated on ploy-L-ornithine hydrobromide and laminin-coated glass coverslips in a 24-well plate, cultured in the DMEM/F12 (1:1) medium supplemented with B27 supplement and 1% fetal bovine serum (Gibco). The medium was changed every 3 days and after 7 days, cells were immunostained with mouse anti-Nestin, mouse anti-Tuj-1, rabbit anti-GFAP, and mouse anti-Galactocerebroside. To assess the proliferation rate of NSCs, NSCs were cultured in proliferative medium containing 20 ng/ml basic fibroblast growth factor, 20 ng/ml epidermal growth factor, and 10 μmol/L 5-Bromo-2′-deoxyuridine (BrdU) (Sigma) for 24 hours. Then, NSCs were washed with phosphate-buffered saline (PBS) for three times and fixed with 4% paraformaldehyde. After incubation in 2 mol/L HCl at 37°C for 30 minutes, and rinsed with 0.1 mol/L boric acid (pH 8.5) at room temperature for lOminutes, cells were blocked with 10% bovine serum albumin and stained with mouse anti-BrdU (1:100; Santa Cruz Technology, Santa Cruz, CA, USA). To test whether GFP signal was diluted in the proliferated NSCs, BrdU staining was also performed in GFP+ NSCs in the same way.
The total number of cells was counted using DAPI staining and the number of Tuj-1, GFAP, and BrdU immunoreactive cells was also counted in a random field on each coverslip using a fluorescent microscope under × 20 objective lens (Leica, Solms, Germany). Three coverslips were evaluated in each immunostaining group and six fields were randomly chosen on each coverslip. The percentage of Tuj-1, GFAP, and BrdU-positive cells was calculated.
Transient Middle Cerebral Artery Occlusion and NSCs Transplantation
In all, 3-month-old (young-adult;
Twenty-four hours after tMCAO, rats were randomly divided into four groups for NSCs or vehicle injection: (1) young-adult rats treated with PBS (vehicle,
Immunohistochemistry
Histologic cryosections (20 μm in thickness) from anterior commissure to hippocampus were collected for immunohistochemical analysis. Serial frozen sections, 20 μm in thickness and 200 μm in interval from the frontal cortex, were stained with 0.1% cresyl violet (Sinopharm Chemical Reagent Co., Shanghai, China). Infarct volume was determined by subtracting the area of cresyl violet staining in the ipsilateral hemisphere from that of the contralateral hemisphere using the NIH image J software, then multiplied by the section interval thickness.
Immunohistochemistry was performed as previously descri bed. 17 After blocking with 10% BSA (Sigma), brain sections were incubated with GFP (1:100; Cell Signaling, Beverly, MA, USA), neuronal marker Tuj-1 (1:100; Millipore), neuroblast marker DCX (1:100; Santa Cruz Technology), astrocyte marker GFAP (1:100; Millipore) at 4°C overnight. Some brain sections were double immunostained using endothelial cell (EC) marker CD 31 (1:200; R&D Systems, Tustin, CA, USA) and VEGF (1:100; Santa Cruz Technology); GFAP and VEGF. Secondary antibodies were appropriate Alexa Fluor 488, 594 or 647-labeled IgG (Invitrogen, Carlsbad, CA, USA).
Double Immunostaining
To determine the proliferation of ECs and neuroblasts, brain sections were incubated in 2 mol/L HCl at 37°C for 30 minutes, and rinsed with 0.1 mol/L boric acid (pH 8.5) at room temperature for lOminutes. After blocking with 10% bovine serum albumin, the sections were stained with CD-31 or DCX and BrdU (1:100; Santa Cruz Technology). Fluorescence signals were detected using a TCS SP5 Confocal Scanning System mounted on a TCS SP5 200 inverted microscope (Leica) equipped with LASOS laser (Lasertechnik GmbH, Jena, Germany). Images were acquired using the LAS AF Software (Leica).
Apoptosis of neurons was evaluated by terminal-deoxynucleotidyl transferase mediated nickend labeling (TUNEL) and NeuN (1:100; Millipore) double immunostaining according to the manufacturer's protocol (Roche Diagnostics, Basel, Switzerland). Every five section (200 μm apart) containing the grafted region was chosen for TUNEL staining. The sections were then stained with anti-NeuN. Slices were covered with anti-fade mounting medium (Vector Labs, Burlingame, CA, USA) and visualized under a fluorescent microscope at × 20 objective lens (Leica). The apoptotic cells were calculated from six areas per section, five sections (200 μm apart) per animal by two observers blinded to the experimental group.
Behavioral Tests
Neurobehavioral tests were performed before tMCAO and 1, 3, 7, and 14 days after tMCAO by an investigator who was blinded to the experimental groups. Rats were trained for 3 days before tMCAO with three consecutive trials to generate stable baseline values.
Rotarod test is an index of fore and hind limb motor coordination and balance. 18 The duration that rats remained on the accelerating rotating rod was measured. The velocity was slowly increased from 20 to 40 r.p.m. in 5 minutes. Each animal was given three trials, and the time that the animals spent on the rungs or gripped the device and spun around for two consecutive revolutions was recorded. Data were analyzed as the average duration (three trials) on the rotarod.
Modified neurologic severity scores of the animals were graded on a scale of 0 to 14, which is a composite of motor, reflex, and balance tests. 19 The higher the score, the more severe injury.
The elevated body swing test was used to test asymmetric motor behavior. 20 In the test, rats were held by the base of the tail and raised 10 cm above the testing surface. The initial direction of swing, defined as the turning of the upper body by >10 degrees to either side, was recorded in 20 trials in each rat, performed over 5 minutes. The number of turns in each (left or right) direction was recorded for each rat.
Quantification of Survived Implanted NSCs and Microvessel Counting
All the GFP-positive cells were counted on nine serial coronal sections per brain (anterior-posterior: −0.8 mm to +0.8 mm, 200 μm apart). Blood vessel counting through CD31 staining was a simple and reproducible way to morphologically identify the number of blood vessels. Numerous studies of manual and computer-assisted methods confirm minimal interobserver variability in the blood vessel counting. 21 Two brain coronal sections from the CD31 and BrdU double stained brain, 1 mm frontier and 1 mm posterior from the ischemic core, were chosen. Three areas of blood vessels, just in the peri-focal striatum region, were chosen at low power objective lens (× 20) and blood vessels were counted on these pictures. Two investigators blinded to the experimental group assessed blood vessel counts separately. Only vessels with a clearly defined lumen or a well-defined linear vessels shape were taken into account. Single EC was ignored. The number of blood vessels was calculated as the mean of the blood vessel counts obtained from the six pictures. Newly formed neuroblasts were quantified in the similar ways.
Western Blot Analysis
Tissue samples were collected from the striatum of the ipsilateral hemisphere, and quantified with BCA protein assay (Pierce, Rockford, IL, USA). To analyze protein levels, equal amounts of total proteins were subjected to 10% (W/V) SDS-PAGE and transferred onto nitrocellulose membranes (Whatman, Piscataway, NJ, USA). Membranes were then blocked with 5% skim milk for 1 hour at room temperature and probed with anti-VEGF (1:500; Santa Cruz Technology) and anti-jS actin (1:2,000; Santa Cruz Technology) antibodies at 4°C overnight. Subsequently, membranes were incubated with horseradish peroxidase-conjugated goat anti rabbit or rabbit anti-goat IgG for 1 hour at room temperature after three times TBST washing, and then reacted with an enhanced ECL substrate (Pierce). The result of chemiluminescence was recorded with an imaging system and semiquantified using the Quantity One software (Bio-Rad, Hercules, CA, USA).
Statistical Analysis
The values were given as mean ± standard deviation. Parametric data in different groups were analyzed by one-way analysis of variance, followed by Tukey
RESULTS
Characterization, Differentiation, Proliferation of NSCs, and Timeline of Experiments
Neural stem cells were generated from the telencephalon of El4 mice and characterized by immunocytochemistry. Results showed that cells were nestin+, while Tuj-1−, GFAP− and galactocerebroside− (Gale−, Figure 1Aa–d). When cells were induced to differentiate, 9.4 ± 4.3% of cells were Tuj-1+ and 90.3 ± 4.2% of cells were GFAP+, while nestin− andGalc− (Figures 1Ae-h and B). In addition, the proliferation rate was evaluated by BrdU incorporation (Figure 1 Ai and j). The proliferation rate of NSCs was 14.6 ± 5.8% (Figure 1B), and the proliferated NSCs were still GFP+ (Supplementary Figure 1). Neural stem cells were then transplanted to rats' brain at 1 day after stroke (Figure 1C).
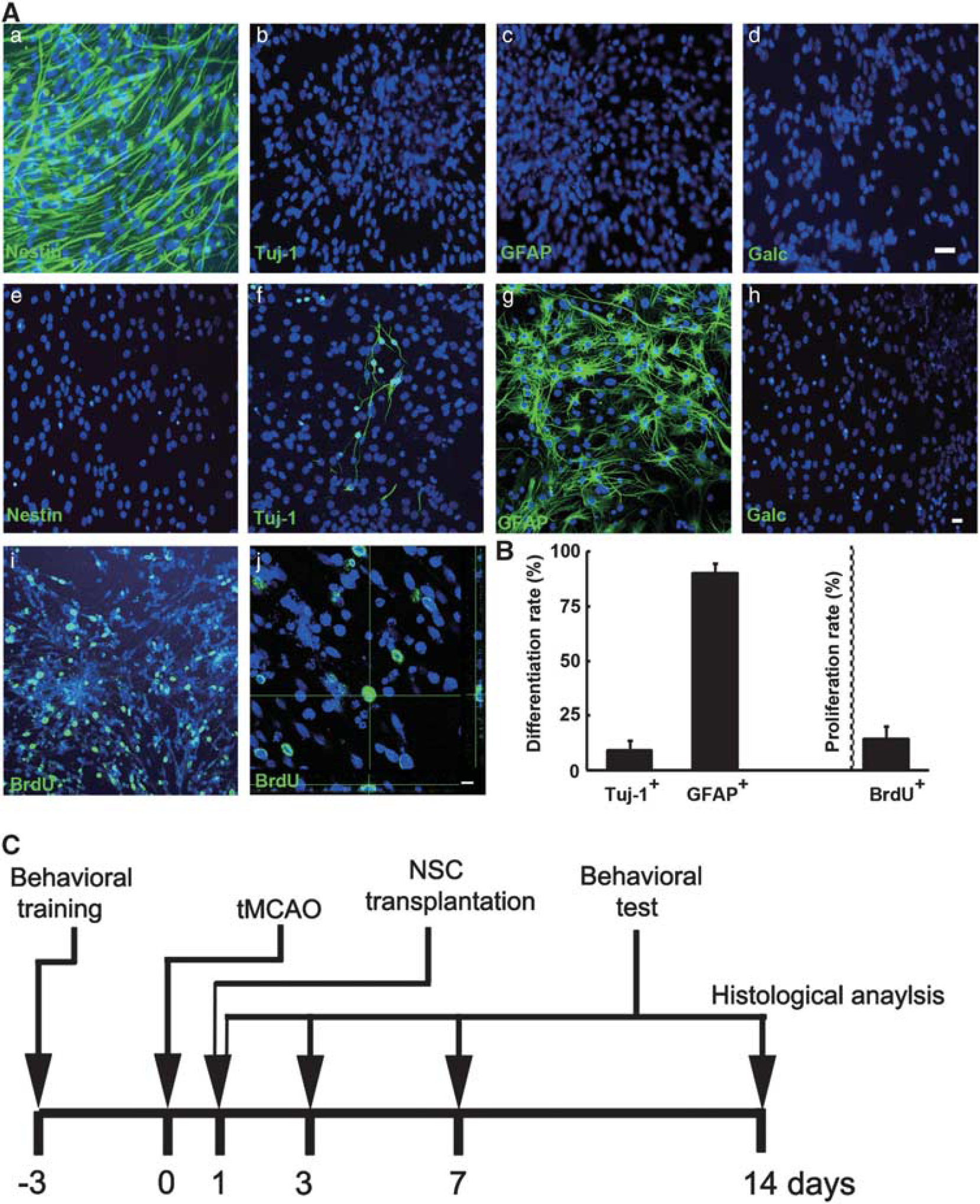
Neural stem cells (NSCs) characterization, differentiation, proliferation, and timeline of experiments. (
NSCs Treatment Attenuated Brain Infarct Volume and Improved Neurobehavioral Outcomes in Aged Rats after Transient Middle Cerebral Artery Occlusion
Mean arterial blood pressure, pH, pCO2, pO2, and glucose concentration were measured before tMCAO, cerebral blood flow was recorded before the surgery, after filament insertion, and after suture withdraw. The physiologic parameters were similar among them (Table 1).
Summary of physiologic parameters in rats
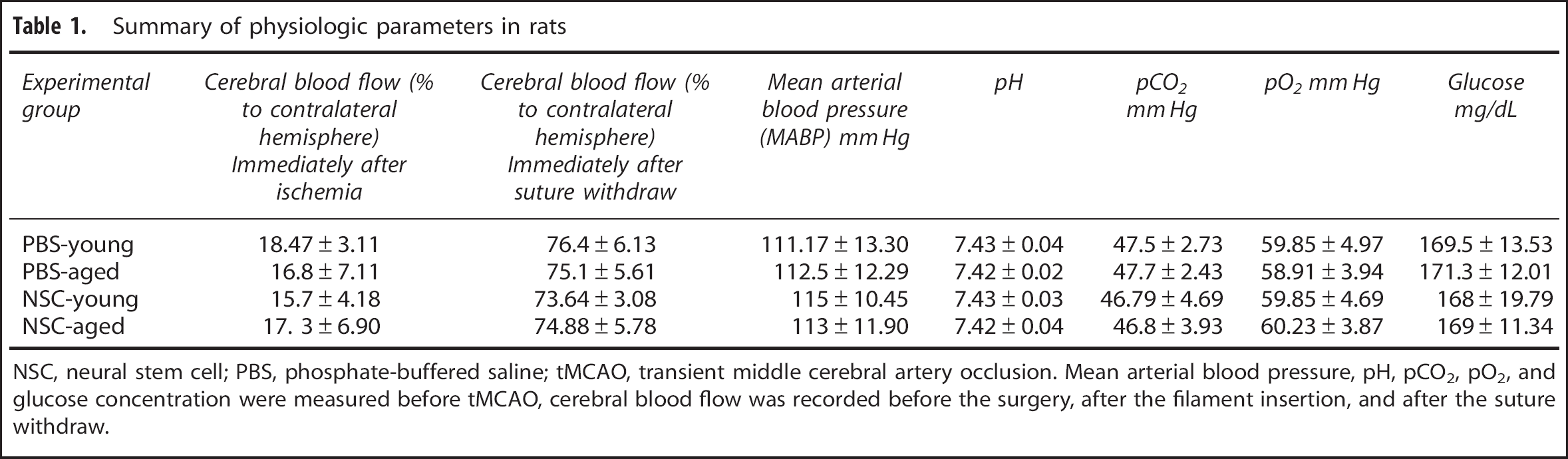
NSC, neural stem cell; PBS, phosphate-buffered saline; tMCAO, transient middle cerebral artery occlusion. Mean arterial blood pressure, pH, pCO2, pO2, and glucose concentration were measured before tMCAO, cerebral blood flow was recorded before the surgery, after the filament insertion, and after the suture withdraw.
To determine whether the benefits of NSC transplantation extend to aged rats, brain sections were stained with cresyl violet after 14 days of tMCAO. Aged rats developed larger infarcts in both cortex and striatum than young-adult rats (Figures 2A and 2B), NSCs treatment reduced infarct volume of striatum in both young-adult and aged rats, compared with the PBS group (Figures 2A and 2B,
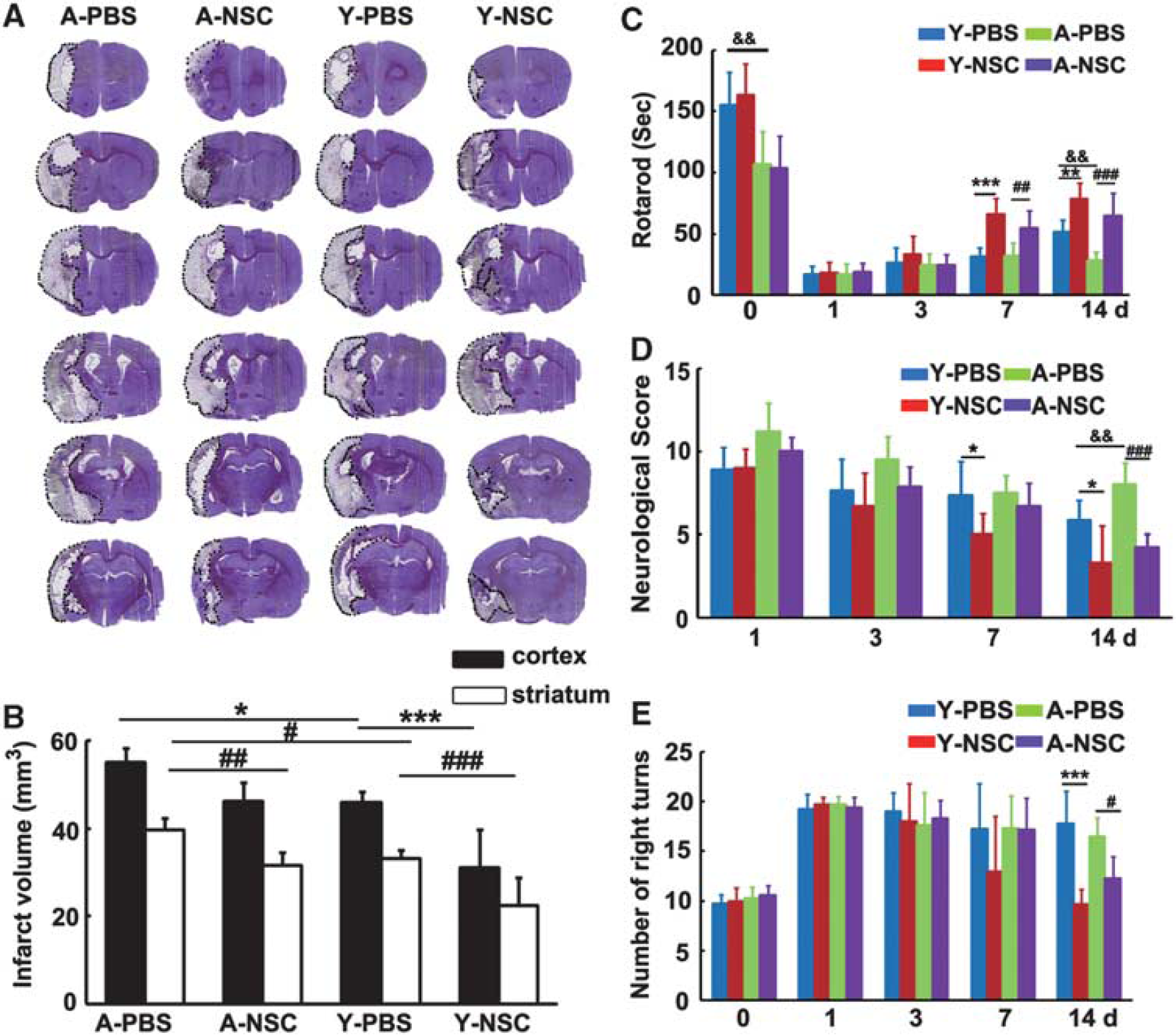
Neural stem cells (NSCs) transplantation reduced infarct volume and improved neurobehavioral recovery in aged ischemic rats. (
Survival and Differentiation Pattern of NSCs in Aged Rats after Transient Middle Cerebral Artery Occlusion
Neural stem cells were transplanted into the striatum in ischemic rats. Photographs and statistical results were acquired from the boundary zone of ischemic core (Figure 3A, boxed areas). Thirteen days after transplantation, GFP staining revealed an extensive migration of the grafted cells toward to the peri-infarct regions in both the aged and young-adult ischemic rats. It was noted that cavity was formed after stroke and NSCs were accumulated in the boundary of the cavity (Figure 3A, asterisk). However, only a small amount of GFP+ cells with unclear morphology were accumulated in the injection area in sham rats (Figure 3B). We found the number of GFP+ cells, which migrated into the peri-infarct area, was similar between aged and young-adult ischemic rats (Figure 3C,
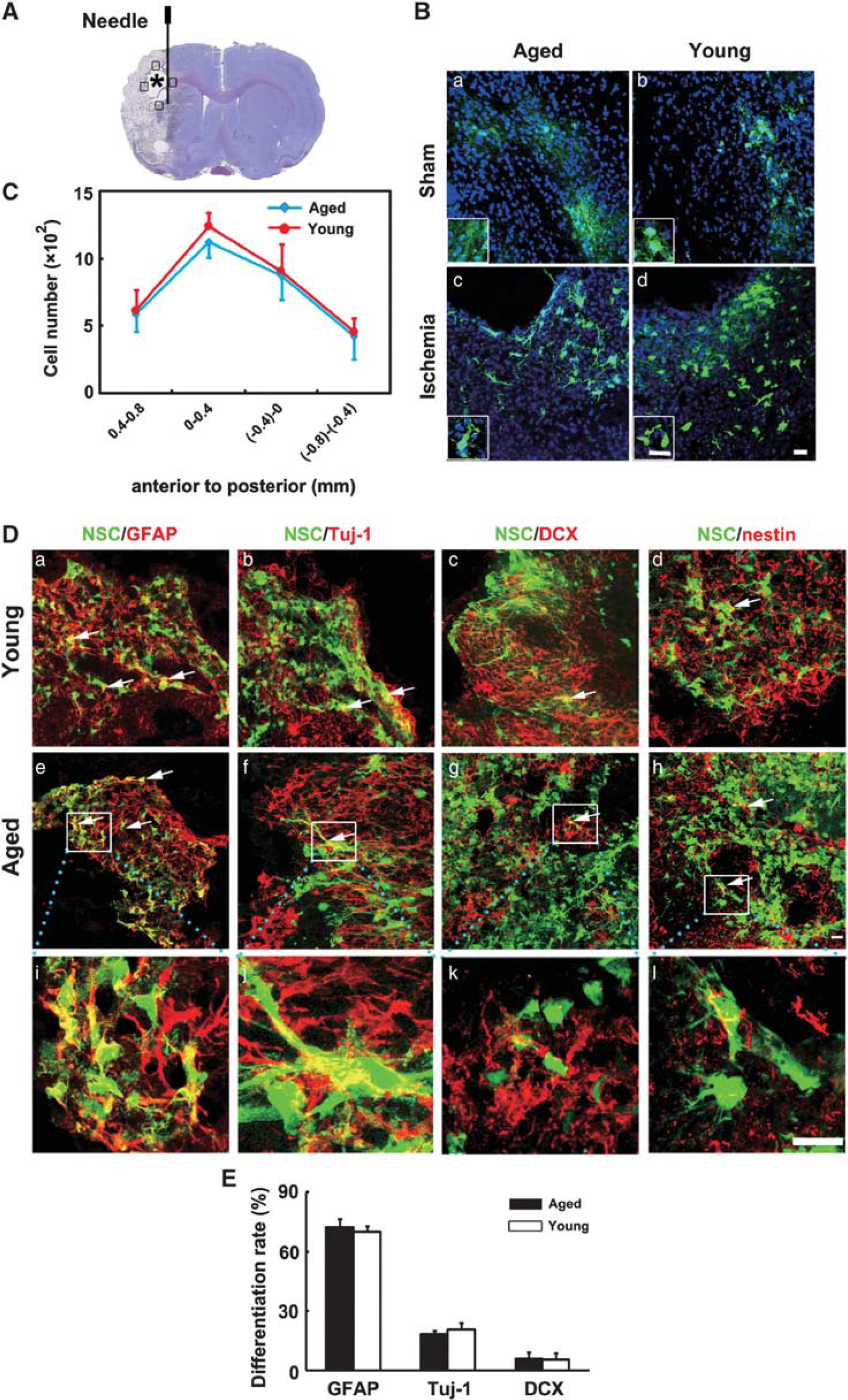
Survival and differentiation pattern of transplanted neural stem cells (NSCs) in aged rat brain after 14 days of transient middle cerebral artery occlusion (tMCAO). (
NSC Transplantation Reduced Apoptosis of Neurons in Aged Ischemic Rats
To test whether NSC could reduce neuron apoptosis in aged brain after ischemia, brain sections were double stained with TUNEL and NeuN. We found that after 14 days of tMCAO, the number of apoptotic neurons in aged rats was more than that in young-adult rats, while transplantation of NSCs greatly reduced the number of neuronal apoptosis in both aged and young-adult ischemic rats (Figures 4A–4C,
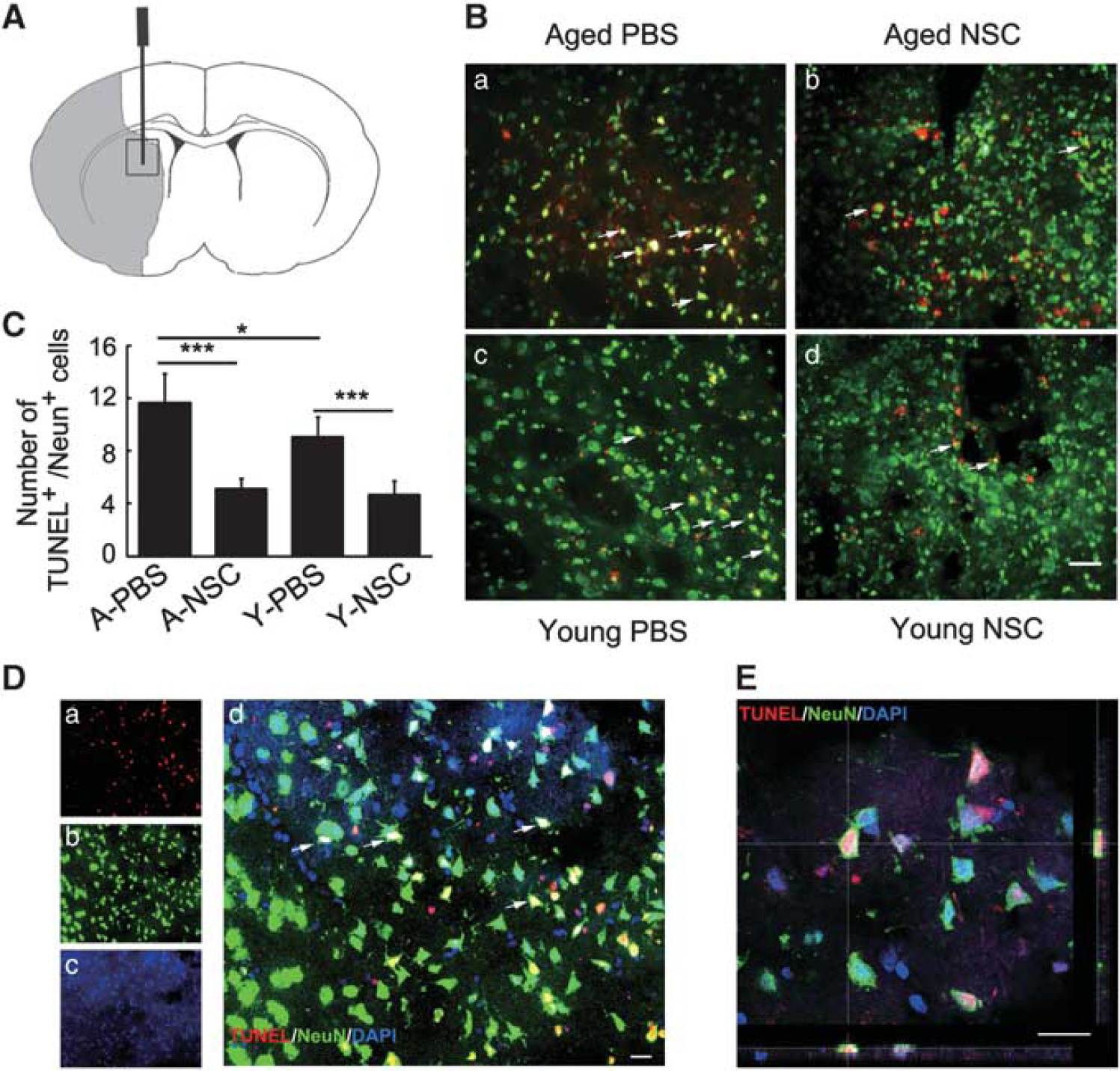
Neural stem cells (NSCs) transplantation attenuated neuronal apoptosis in aged and young-adult rat brain after 14 days of transient middle cerebral artery occlusion (tMCAO). (
NSC Transplantation Promoted Endogenous Angiogenesis and Neurogenesis in Aged Ischemic Rats
The number of newly formed microvessels and neuroblasts was analyzed in the ipsilateral striatum and subventricular zone after 14 days of tMCAO by CD31/BrdU (Figure 5A) and DCX/BrdU (Figure 5B) double immunostaining. Confocal images showed that CD-31/BrdU (Figure 5C) and DCX/BrdU (Figure 5D) were expressed in single cells. Statistical results showed that angiogenesis and neurogenesis were reduced in aged rats after 14 days of tMCAO, compared with the young-adult ischemic rats. However, angiogenesis and neurogenesis were significantly increased in both aged and young-adult rats after NSC transplantation compared with the control (Figures 5E and 5F,
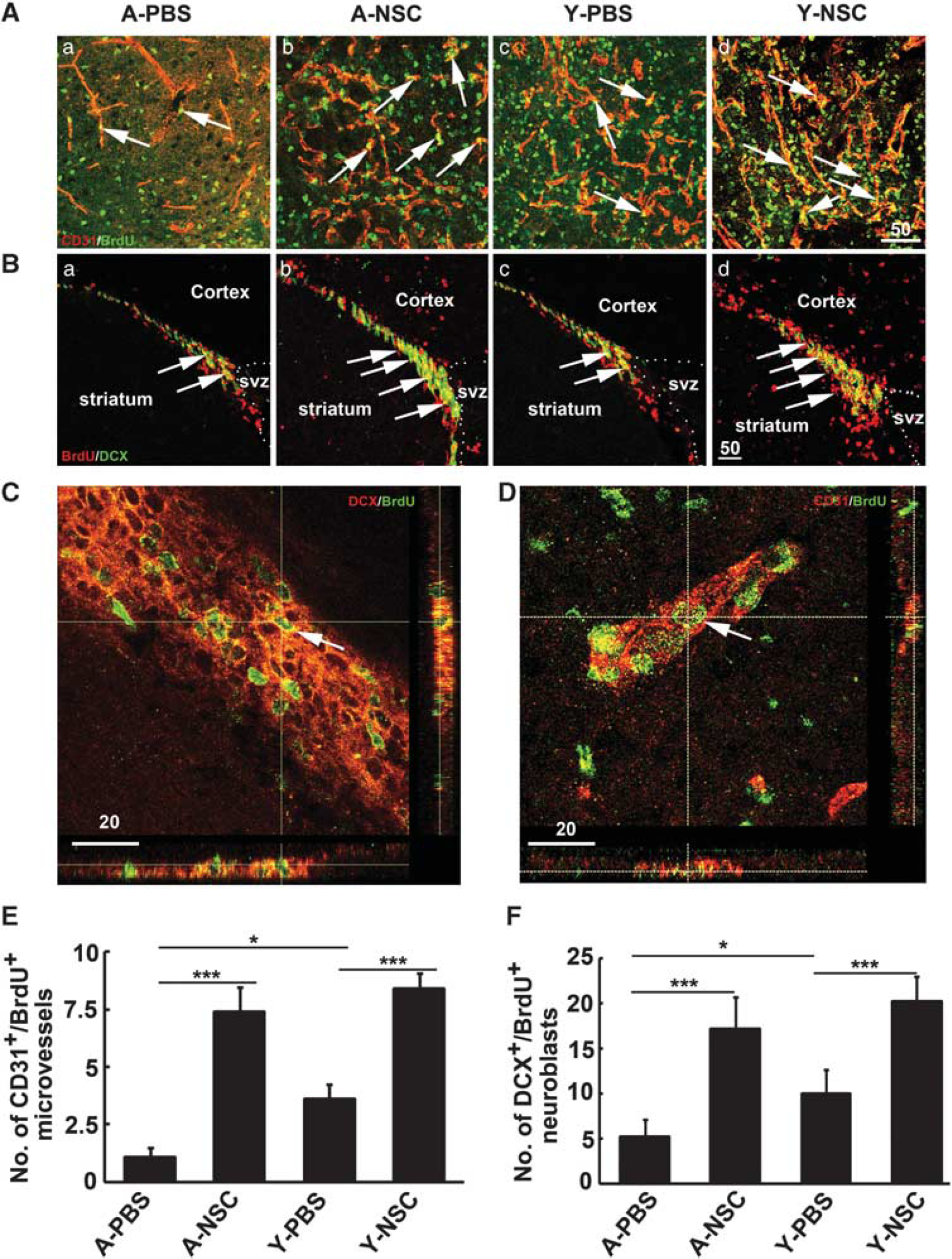
Neural stem cells (NSCs) transplantation increased angiogenesis and neurogenesis in aged ischemic rats. (
NSCs Transplantation Increased Vascular Endothelial Growth Factor Expression in Aged Ischemic Rats
Immunostaining results showed that NSC transplantation increased VEGF expression in both aged and young-adult rats (Figure 6A), which were confirmed by western blotting (Figures 6B and 6C,
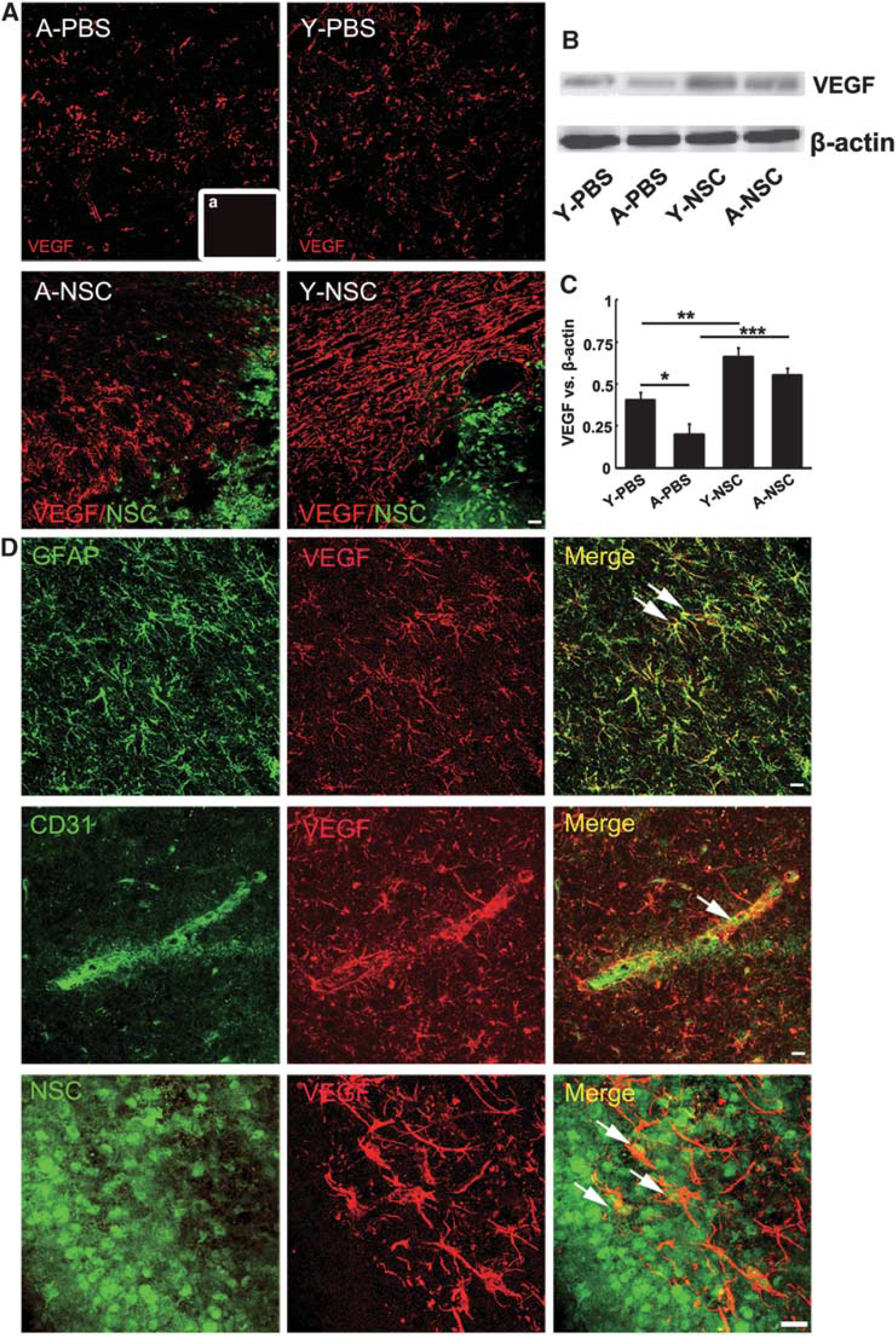
The expression of vascular endothelial growth factor (VEGF) was significantly increased in aged ischemic brain after neural stem cell (NSC) transplantation. (
Discussion
Previous studies showed that transplantation of NSCs modulated inflammatory response and microglia activation. The transplanted NSCs secreted neurotrophic factors, promoted endogenous angiogenesis and neurogenesis, enhanced plasticity, even differentiated into mature neurons with electrophysiological properties and received synaptic input from host neurons after cerebral ischemia. 22 These results suggest that NSC has great therapeutic potential in animal models. In contrast to these abundant studies in young-adult animals, little attention has been paid to aged rodents, which is of greater therapeutic significance since the occurrence of stroke is much higher in seniors.
Aging impairs the microenvironment of the brain. For example, aged mice suffered deficits in olfactory discrimination, most likely attributed to reduced neurogenesis. 23 In terms of cerebral ischemia, our previous study showed that ischemia-induced neurogenesis was preserved but reduced in the aged rat brain. 24 Angiogenesis was also reduced in the aged rodent brain after focal cerebral ischemia, compared with the young-adult animals. 25 The age-dependent decline of neurogenesis and angiogenesis was attributed to decreased growth factors. 14 In this study, we found that aged rats were more vulnerable than the young-adult rats to ischemic damage. Overall mortality in aged rats was significantly higher than that in the young-adult rats after tMCAO (6/20 in aged rats and 2/35 in young-adult rats). After cerebral ischemia, larger infarct volume, limited functional recovery, decreased angiogenesis and neurogenesis with decreased VEGF expression occurred in aged rats. 26 Interestingly, rotarod test, but not modified neurologic severity scores and elevated body swing test, showed that aging impaired motor function in normal aged rats, suggesting that different behavioral tests could target different aspects of functional outcomes. Additionally, rotarod and modified neurologic severity score tests showed worse in aged rats 14 days after tMCAO, which was associated with an age-dependent decrease in ischemia-induced angiogenesis, neurogenesis and VEGF expression. Based on these observations, it is reasonable to speculate that aging might affect the fate of implanted NSCs in the ischemic brain and its therapeutic efficiency after tMCAO.
We stereotactically injected NSCs into the brain at 1 day after tMCAO. Up to date, various stem cell delivery routes such as intravenous, intracerebral, intraartery, and intraventricular have been applied to explore their therapeutic benefits and the mechanism involved in stroke. 27 Intracerebral injection was chosen in our experimental design because the cells could be precisely transplanted into the striatum of ischemic brain without cell loss. Vascular injection could trap stem cells into systemic organs, with few stem cells could be homed to the ischemic brain hemisphere. 28 Previous studies showed that both early and delayed transplantation of NSCs reduced infarct volume, improved neurobehavioral outcomes, increased angiogenesis and neurogenesis. It is noted that cell survival was higher in early transplantation after stroke. 29 We transplanted NSC after 1 day of MCAO, which was practically difficulty for clinical application. Further work is needed to test whether transplantation of NSCs in later phases would also benefit for cerebral ischemia.
Our present study showed that GFP-labeled NSCs could survive in both aged and young-adult brain for at least 14 days after MCAO, and most of the transplanted NSCs were differentiated into GFAP+ astrocytes. However, when NSCs were injected into the sham surgery rats, unclear and atypical cell morphology could be observed in both aged and young-adult rats. No GFP signal could be found at 35 days after GFP+ NSC transplantation into sham rats (data not shown), indicating that normal rat brain could not provide suitable environment, which are critical for exogenous cell survival and differentiation. In addition, we showed that grafted NSCs' differentiation was similar between aged and young-adult rats after 14 days of tMCAO, suggesting that microenvironment in aged rat brain did not affect NSC differentiation.
Neuronal apoptosis occurs after brain ischemia is well documented. 30 Neural stem cells transplantation into young-adult ischemic rodents greatly reduced TUNEL+ apoptotic cells and inflammatory infiltration, further improved neurobehavioral outcomes. 31 We found that brain ischemia induced more neuronal apoptosis in aged rats compared with the young-adult rats, while NSC transplantation greatly attenuated neuronal apoptosis in both young-adult and aged rats. Furthermore, we also found that angiogenesis and neurogenesis were increased after NSC transplantation. Angiogenesis and neurogenesis are fundamental events during development and some disease processes. Cerebral ischemia induced angiogenesis and neurogenesis have been shown in a variety of rodent models and in humans. 32 Newly formed capillaries could supply oxygen and nutrition to the ischemic region, which is extremely important for the tissue repairing and remodeling. 33 Newly formed neuroblasts could migrate to the damaged area, where they become matured and functional cells to replace the dead neurons. 34 Thus, angiogenesis and neurogenesis are two important processes responsible for restoring brain functions after stroke.
It was reported that implantation of NSCs at 24 or 72 hours after stroke protected blood–brain barrier, reduced neuron apoptosis, facilitated neuroprotection, and attenuated infarct volume through multiple mechanisms. 35 Our present study further showed that transplanted NSCs reduced neuronal apoptosis, promoted focal angiogenesis and neurogenesis, increased VEGF expression in the lesion area, thus attenuated infarct volume at 14 days after stroke. This could be due to neuroprotection or neurorestoration. 36
Vascular endothelial growth factor is an important neurotrophic factor for angiogenesis and neurogenesis. Numerous studies show that brain injury upregulates VEGF, which is critical for angiogenesis and neurogenesis. 37 In addition, NSCs secreted VEGF promoted the density of capillary, enhanced axonal sprouting, and regulated microglia response. 38 We found that VEGF was mainly expressed in GFAP+ astrocytes, CD31+ ECs, and implanted NSCs, even in astrocytes derived from transplanted NSCs. In short, VEGF was greatly upregulated in both NSC-treated aged and young-adult ischemic rats. Upregulated VEGF subsequently increased focal angiogenesis and neurogenesis, and in the meantime reduced neuron apoptosis in aged and young-adult ischemic rats.
Astrocytes have a dual role after injury. Focal cerebral ischemia induces astrocytes swelling and leads to blood–brain barrier breakdown, reduces the uptake and release of glutamate. 39 Enhancing the survival of astrocytes restores blood–brain barrier and improves neuron survival in the acute phase of cerebral ischemia while glial scar formation during the subacute phase significantly inhibits functional recovery and process of regeneration. 40 In our study, we showed that limited transplanted NSCs were differentiated into neurons, and no direct evidence showed that dead neurons could be replaced by new neurons that were derived from transplanted NSCs. Neural stem cells differentiated into astrocytes and expressed VEGF (Supplementary Figure 2), which were defined as cytokine-activated astrocytes. These cytokine-stimulated astrocytes then promoted the recovery of central nervous system function. In addition, transplanted NSCs not only enhanced endogenous secretion of VEGF by astrocytes and ECs, but also protected endogenous neurons from apoptosis, consequently promoted angiogenesis and neurogenesis, thus reduced infarct volume and improved neurobehavioral recovery.
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
