Abstract
We investigated the contribution of blood flow elevation in the cerebrovasculature to physical training-induced brain-derived neurotrophic factor (BDNF) levels elevation in the brain. Brain-derived neurotrophic factor protein levels were measured in the motor cortex 24 h after the last session of a forced treadmill walking (30 minutes a day, 18 m/minute for 7 consecutive days). Unilateral common carotid artery occlusion and modulation of exercise intensity (0 versus — 10% inclination of the treadmill) were used as strategies to reduce the (normal) elevation of flow in the cerebrovasculature occurring during exercise. Administration of N-nitro-L-arginine methyl ester (L-NAME, 60 mg/kg before each exercise sessions) and genetic hypertension (spontaneously hypertensive rats) were used as approaches to reduce stimulation of nitric oxide production in response to shear stress elevation. Vascular occlusion totally and partially abolished the effect of physical training on BDNF levels in the hemisphere ipsilateral and contralateral to occlusion, respectively. BDNF levels were higher after high than low exercise intensity. In addition, both genetic hypertension and L-NAME treatment blunted the effects of physical training on BDNF. From these results, we propose that elevation of brain BDNF levels elicited by physical training involves changes in cerebral hemodynamics.
INTRODUCTION
In the last 20 years, the promotion of the regular practice of physical activity has become an important public health message. This can be attributed primarily to the cardiovascular benefits of physical training that are, at least in part, due to elevation of flow in the vasculature and subsequent modification of endothelial phenotype through activation of shear stress-dependent mechanisms. 1 Among the panoply of endothelium-derived factors that are secreted in response to shear stress elevation, nitric oxide (NO) is probably the most important and best characterized mediator. More recent evidence suggest that physical training has positive effects on brain function and relate brain benefits to an augmentation of brain-derived neurotrophic factor (BDNF) levels in the brain. 2 Surprisingly, the physiologic mechanisms that drive the elevation of BDNF in the brain remain speculative whereas their elucidation represents a rational basis for the design of physical training modalities aimed at improving brain health.
As neuronal activity is well known to regulate bdnf transcription and maturation of BDNF messenger RNA into mature BDNF, 3 high BDNF levels observed in the brain after training have been implicitly related to neuronal hyperactivity that occurs during each exercise boot in motor-related and -unrelated brain areas.4,5 An additional mechanism that has been proposed is the involvement of mediators originating from active skeleton muscles and able to induce bdnf gene in the brain such as insulin-like growth factor 1 or irisin.6,7 Surprisingly, whereas blood flow (ml/minute) and blood flow velocity (cm/second) were reported to increase in the common and internal carotid arteries, vertebral arteries, and major cerebral arteries in proportion to exercise intensity8,9 and although a reciprocal interaction exist between NO and BDNF synthesis, 10 the contribution of cerebral hemodynamics to the effects of physical training on brain BDNF levels has never been explored.
The present study explores the hypothesis that flow elevation in the cerebrovasculature during exercise is involved in physical training-induced BDNF levels increase in the rat brain. For this purpose, we investigated to what extent strategies aimed at reducing the (normal) flow increases in the cerebrovasculature (occlusion of a common carotid artery, modulation of exercise intensity) or at decreasing endothelial production of NO in response to shear stress (genetic hypertension and NO synthase inhibition) did blunt the effect of physical training on brain BDNF. Physical training consisted in a daily walking activity on a motorized treadmill for 7 consecutive days. Changes in treadmill inclination (horizontal versus —10% decline) were used to modulate exercise intensity. Levels of mature BDNF and synaptophysin (SYN) as a marker of synaptogenesis and synaptic activity11,12 were measured in the cortex 24 h after the last session of exercise using western blot analysis.
MATERIALS AND METHODS
Ethical Approval
All experiments were carried out (license 21CAE102, French Department of Agriculture) in accordance with the European Committee's Council Directive and performed to comply with the ARRIVE (Animal Research: Reporting In Vivo Experiments) guidelines. A total of 80 rats were used in the completion of this study. Young adult male (13 weeks old) were used so that excluding age-associated endothelial dysfunction and interaction between female hormones and BDNF as confounding factors. Among these rats, there were 56 Wistar rats, 12 Wistar-Kyoto (WKY) rats, 12 spontaneously hypertensive rats (SHR). Wistar-Kyoto and SHR were purchased from Charles River (L'Arbresle, France) and other rats from Janvier (Le Genest Saint Isle, France). Animals were maintained in a temperature-controlled room, on a 12/12 hours light/dark cycle with food and water ad libitum. They were housed five per cage to maintain social interaction. To reduce the animal's stress level, the same operator performed all steps of experiments. Efforts were made to reduce the number of rats.
Physical Training
To familiarize the rats with the operator, they were gently handled twice a day for 1 week. During the following week, all the rats were familiarized with the treadmill apparatus (model Exer-3/6, Linton Instrumentation, Norfolk, UK). A 2-minute long walking session (horizontal position of the treadmill) was given twice a day, using mild intensity (0.4 mA) of foot shocks as negative reinforcement. Then, rats were randomly separated in sedentary and trained rats. We have been concerned with using exercise ‘dosage’ in rats mimicking recommendations of physical activity to improve human health. Therefore, physical training consisted in a daily walking of 30-minute duration at a rapid speed (18 m/minute). The physical training duration was however restricted to 7 days that is short enough to not increase capillary density in the brain. Treadmill activity (without electrified grid) was induced in the morning between 0900 hours and 1200 hours. Corresponding sedentary rats were kept in their housing cage that was placed at the proximity of the treadmill apparatus. We previously showed that such a modality of physical training (treadmill activity with a horizontal position, 30 minutes/day, 18 m/minute, for 7 consecutive days) induced a mild increase in citrate synthase activity in skeleton muscles, did not change blood pressure in WKY rats and SHR, 13 and resulted in a twofold increase in mature BDNF levels in the cortex when induced in control Wistar rats. 14
Strategies Used to Reduce the Increase in Blood Flow Within the Cerebrovasculature During Exercise
Common Carotid Artery Occlusion. After anesthesia (chloral hydrate, 400 mg/kg, intraperitoneal) and a midline ventral incision of the neck, the left common carotid artery was definitively occluded and the incision was sutured. Once awake, rats returned to their housing cage. Sham-operated controls received the same surgical procedure without vessel ligation. Training was started 3 days after the vascular occlusion (VO) or at equivalent time in sham rats. Importantly, even though occlusion of a common carotid artery leads to an immediate decrease in cerebral blood flow in the hemisphere ipsilateral to occlusion, cerebral blood flow recovers preoccluded values as soon as 24 hours after occlusion induction.15,16 Consistently, long-term unilateral occlusion does not induce neuronal suffering. 17
Changes in Exercise Intensity. Flow changes in the cerebrovasculature are dependent on exercise intensity, 18 the ability to increase cardiac output, 19 and the arterial partial pressure in carbon dioxide. 20 Thus, blood flow in the carotid and vertebral circulations increases in proportion to exercise intensity from rest to moderate-intensity exercise. 9 In the present study, modulation of exercise intensity was achieved by changing the slope of the treadmill (0 versus —10% decline at a same speed). Indeed, whole body oxygen consumption as well as hindlimb muscle blood flow and cardiac output have been shown to be lower for downhill than level locomotion at the same speed. 21
Strategies Used to Explore Shear Stress-Dependent Mechanisms in the Effect of Physical Training on Brain-Derived Neurotrophic Factor
A well-documented response of endothelium to shear stress elevation is an overproduction of NO by the endothelium. Moreover, endothelial NO production is reduced in cerebral microvessels from SHR compared with WKY rats
22
and SHR exhibit a decrease in flow-mediated dilation,23,24 a process that largely involves endothelium-derived NO. Thus, to assess the role of shear stress in the effects of physical training, we investigated whether genetic hypertension and treatment with a NO synthase inhibitor can reduce the effect of physical training on BDNF. N-nitro-
Western Blot Analysis
Brain BDNF and SYN levels were measured 24 hours after the last treadmill session or at equivalent time in sedentary rats. After chloral anesthesia (400 mg/kg, intraperitoneal) and transcardial perfusion with saline to eliminate blood from the cerebral vasculature, the right and left hemispheres were separated. After dissection of the motor areas, samples were processed as described previously in details. 14 Blots were probed with anti-BDNF (rabbit monoclonal, Epitomics 3160-1, Euromedex, Souffelweyersheim, France) or anti-SYN antibodies (rabbit polyclonal, Thermo Scientific RB-1461-P1, Interchim, Montluçon, France) and anti-β-actin antibodies (mouse monoclonal, A5441, Sigma-Aldrich). β-actin was used as an internal control. BDNF and SYN levels were analyzed after normalization to β-actin levels. Multiple groups of rats were compared two by two to investigate (i) the proper effects of VO, hypertension, and L-NAME treatment, (ii) the impact of exercise intensity, (iii) the interaction between VO, hypertension, or L-NAME treatment and training effects. Accordingly, BDNF and SYN levels were measured in 14 groups of rats: sedentary rats with VO (sed VO, n = 6), sed sham (n = 6), trained VO (training was induced in rats with previous VO, n = 6), trained sham (n = 6), sed WKY (n = 6), trained WKY (n = 6), sed SHR (n = 6), trained SHR (n = 6), trained 0% (n = 6), trained −10% (n = 6), sed L-NAME (sedentary rats received L-NAME 60 mg/kg, n = 4), trained L-NAME (n = 6), sed saline (n = 4), trained saline (n = 6). For all groups of trained rats, there was no inclination of the treadmill except in group trained −10% for which the treadmill was −10% decline. Two groups of rats were analyzed on the same gel and gels were run in triplicate. Brain-derived neurotrophic factor and SYN were calculated from a representative gel. The number of rats in each group is appropriate to deal with inter-individual variability and appropriate to a statistical analysis.
Statistical Analysis
Results were expressed by means ± s.d. Statistical analysis between two groups was performed using Wilcoxon test as a non-parametric tests for non-paired observations. Differences were considered significant when P < 0.05.
RESULTS
Carotid Occlusion Alleviates the Effect of Physical Training on Brain-Derived Neurotrophic Factor
When induced in rats with normal carotid circulation (sham rats), horizontal physical training induced an increase in BDNF levels that reached +150% (Figure 1A). These effects of training were abolished when training was induced in rats with previous unilateral common carotid artery occlusion. Thus, training became unable to increase BDNF levels in the hemisphere ipsilateral to the occlusion; (Figure 1B) while the increase in BDNF levels was restricted to + 36% in the hemisphere contralateral to the occlusion (Figure 1C). Notably, unilateral common carotid artery occlusion had no impact on brain BDNF levels in sedentary rats regardless the considered hemisphere (Figures 1D and E).
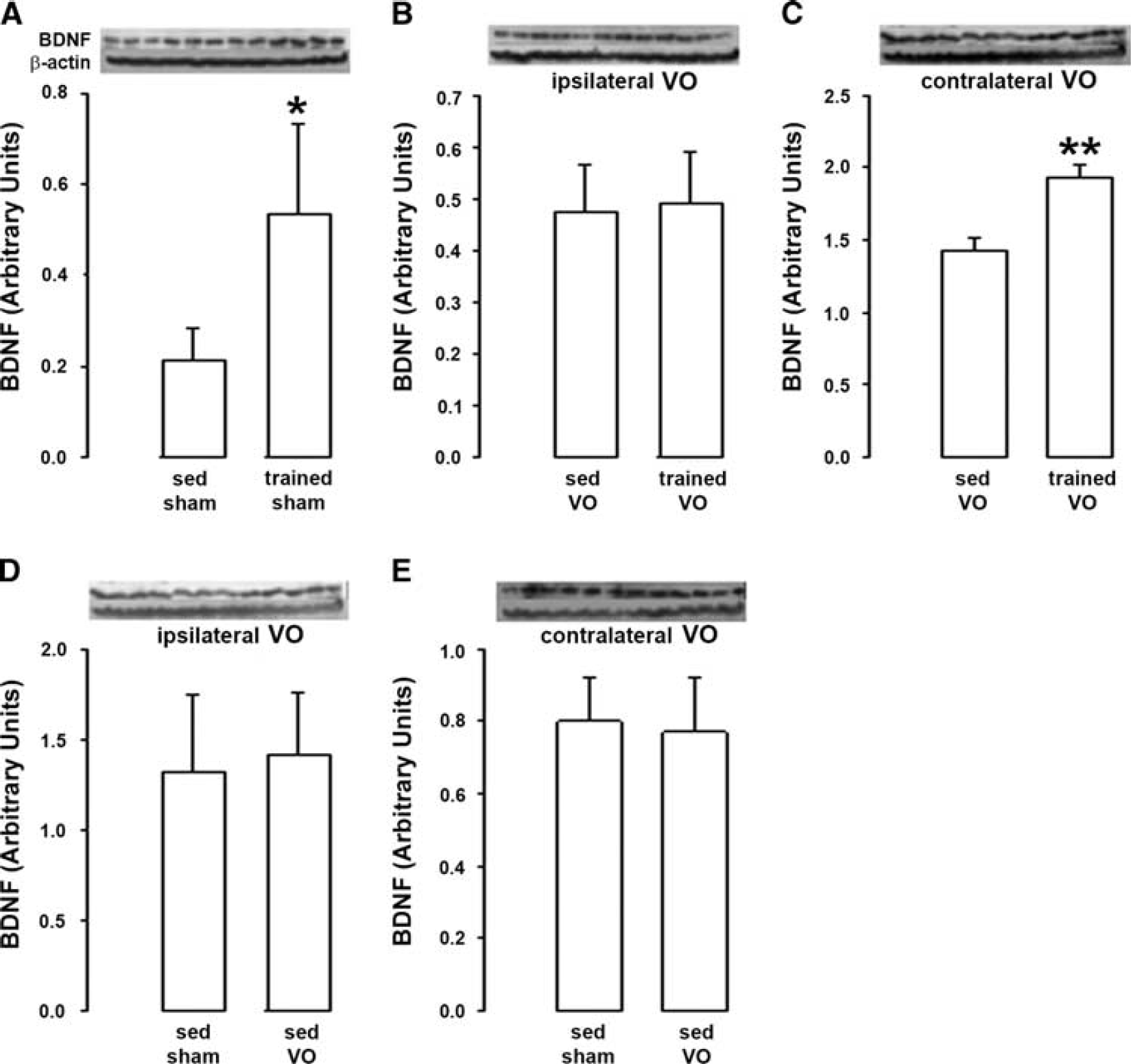
Effect of physical training on brain-derived neurotrophic factor (BDNF) after unilateral common carotid artery occlusion. (
Brain Brain-Derived Neurotrophic Factor Levels in Trained Rats are Dependent on Physical Training Intensity
Physical training with low exercise intensity (− 10% inclination of the treadmill) significantly increased BDNF levels as compared with sedentary rats (+ 30%, Figure 2A), but in a lesser extent than physical training with high exercise intensity (no inclination of the treadmill, + 150%, see Figure 1A). Thus, BDNF levels in trained rats were significantly lower (− 50%) for downhill than horizontal training (Figure 2B).
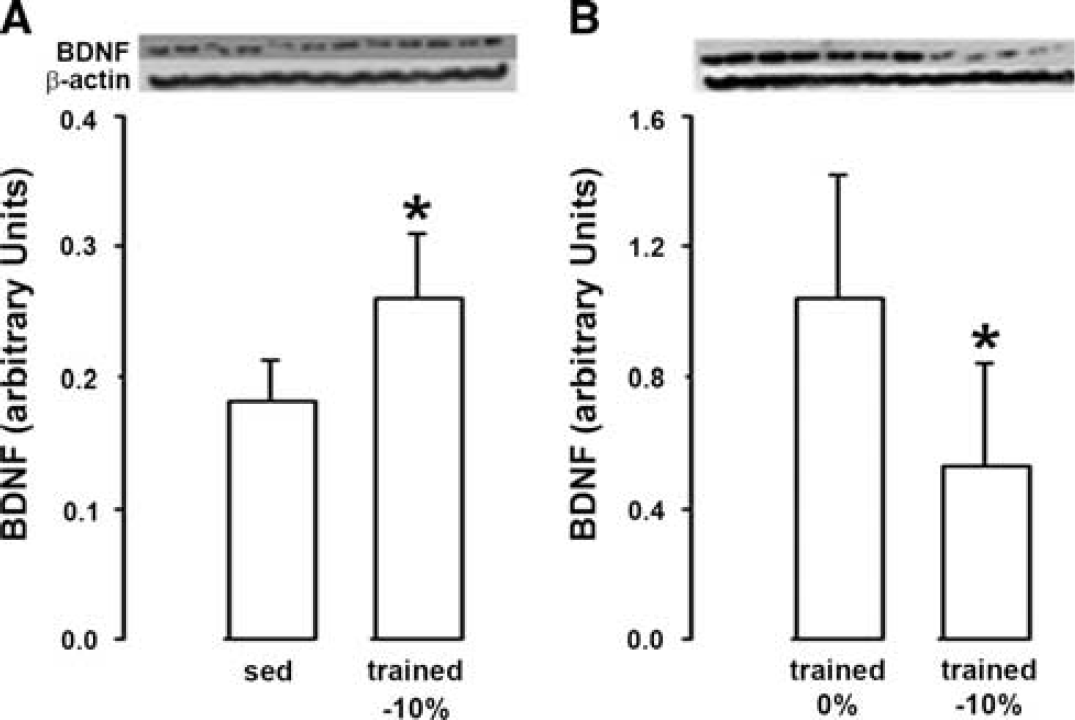
Impact of exercise intensity on brain-derived neurotrophic factor (BDNF). (
Genetic Hypertension Abolished the Effect of Physical Training on Brain-Derived Neurotrophic Factor
Although the increase in BDNF levels induced by training reached +114% in WKY rats (Figure 3A), training did not significantly change BDNF levels in SHR (Figure 3B). Moreover, in sedentary rats, BDNF levels were higher (+ 40%) in SHR than WKY (Figure 3C).
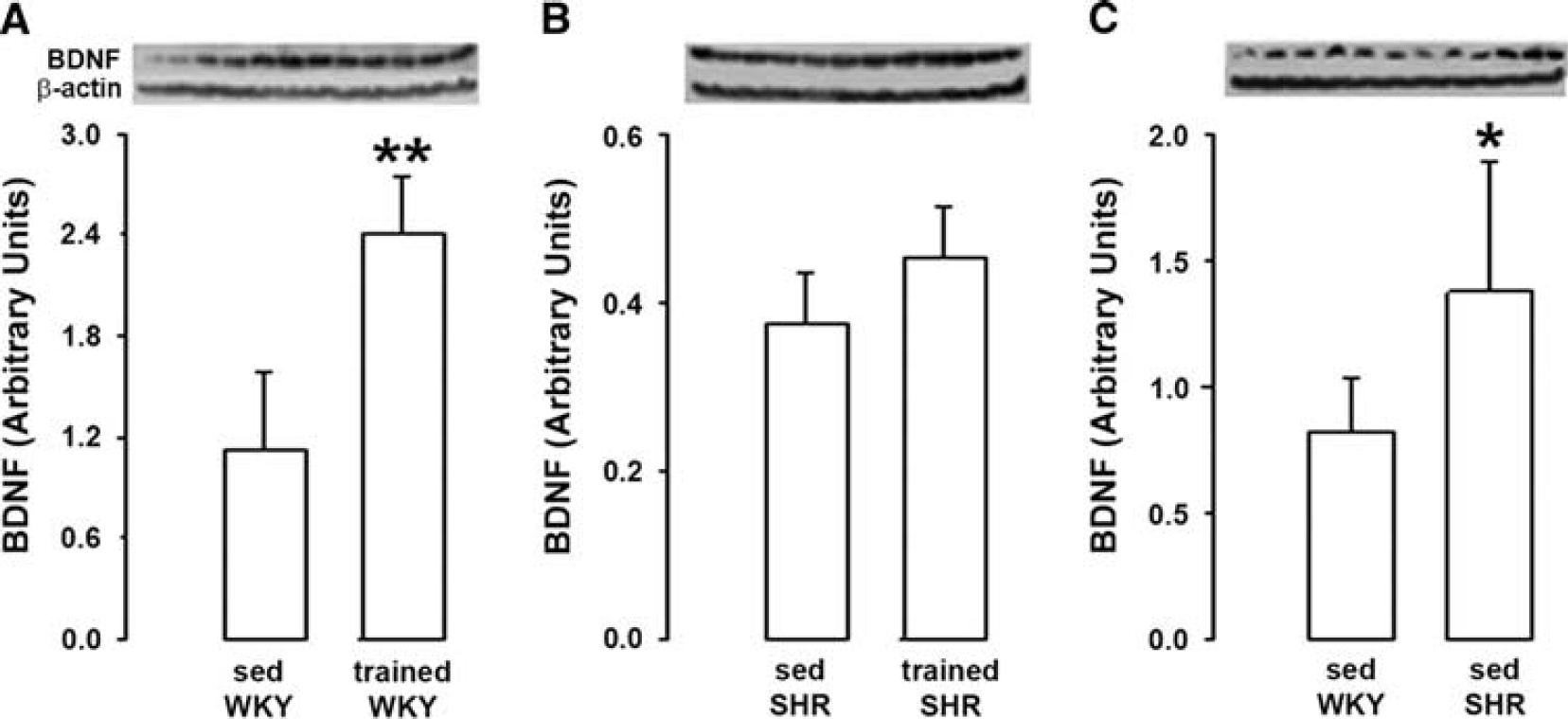
Effect of physical training on brain-derived neurotrophic factor (BDNF) in genetic hypertension. (
Physical Training did not Increase Brain-Derived Neurotrophic Factor Levels in N-Nitro-L-Arginine Methyl Ester-Treated Rats
When induced in saline-treated rats, training increased BDNF levels by a factor 2.4 (+ 140%, Figure 4A). By contrast, training did not modify BDNF levels when it was induced in L-NAME-treated rats (Figure 4B). Thus, BDNF levels were lower in trained L-NAME-treated rats than in trained saline-treated rats (− 44%, Figure 4C). Of note, no difference in BDNF levels was observed in sedentary rats between L-NAME- and saline-treated rats (Figure 4D).
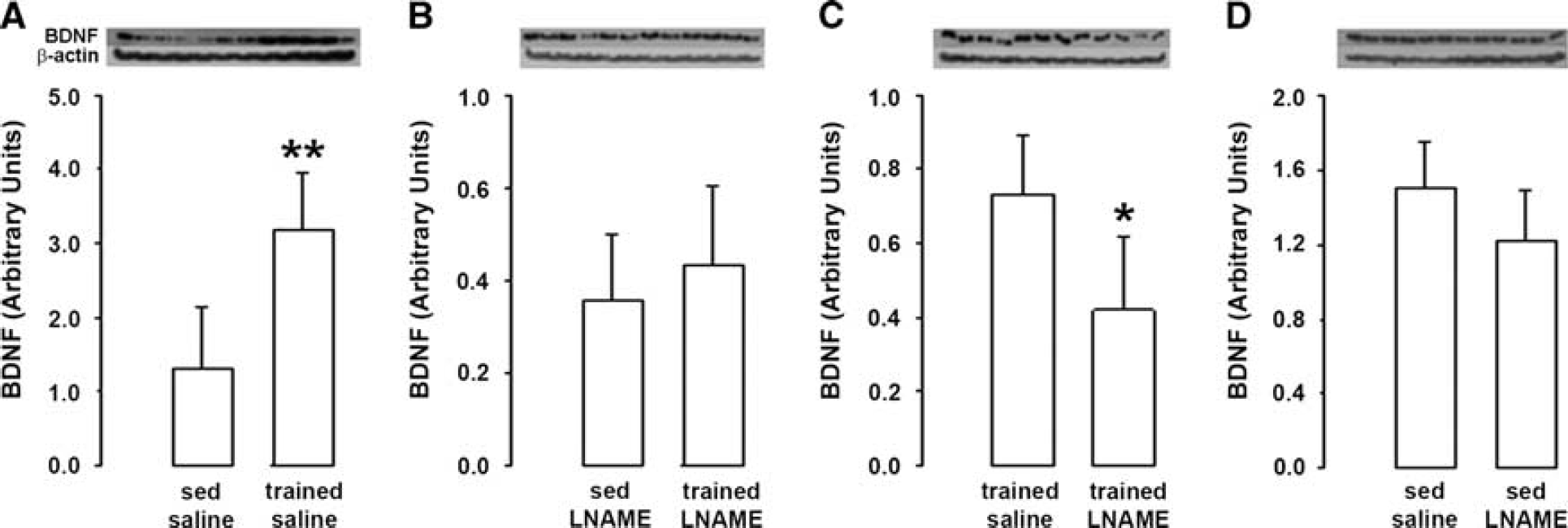
Effect of physical training on brain-derived neurotrophic factor (BDNF) after nitric oxide synthase inhibition. (
Brain-Derived Neurotrophic Factor Levels Match with Synaptophysin Levels
The results are summarized in Figure 5 and Table 1. When induced in rats with normal carotid circulation (sham rats), horizontal training increased SYN levels by + 160% (Figure 5A). By contrast, when induced in rats with unilateral common carotid artery occlusion (VO rats), training did not change SYN levels in the hemisphere ipisilateral to occlusion (Figure 5B) and restricted the increase to + 58% in the opposite hemisphere (Figure 5C). Moreover, physical training induced an elevation of SYN levels both in WKY rats (+ 116%, Figure 5D) and SHR (+ 60%, Figure 5E). By contrast, SYN levels were not changed by training when induced in L-NAME-treated rats (Figure 5F). Like horizontal training, downhill training increased SYN levels (Figure 5G) but to a lesser extent (+ 47 versus +160% as shown in Figure 5A). Finally, as shown in Figure 5H, SYN levels in sedentary rats were higher in SHR than in WKY rats (+131%).
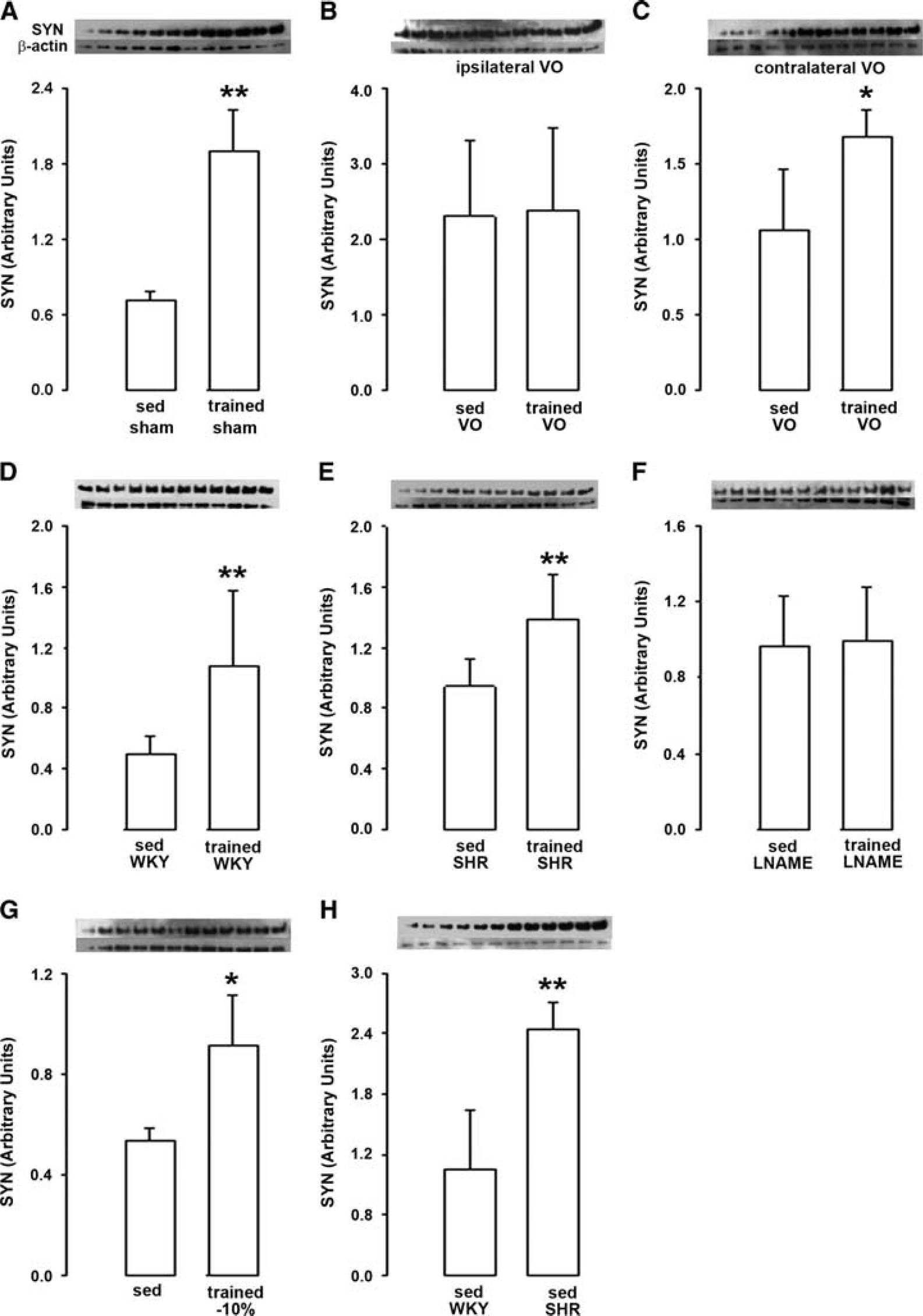
Synaptophysin (SYN) levels. (
Comparative changes in BDNF and SYN levels induced by physical training
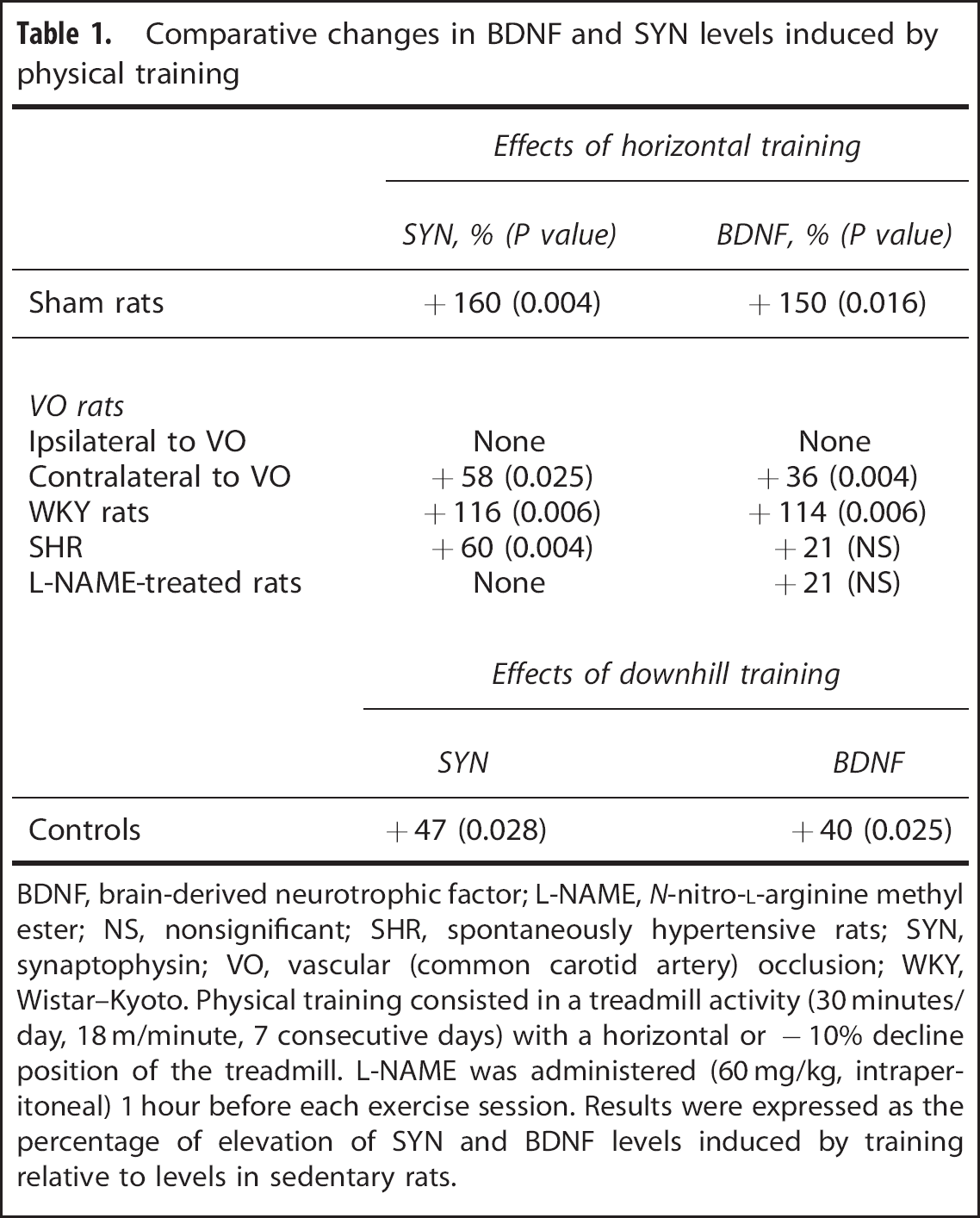
BDNF, brain-derived neurotrophic factor; L-NAME, N-nitro-L-arginine methyl ester; NS, nonsignificant; SHR, spontaneously hypertensive rats; SYN, synaptophysin; VO, vascular (common carotid artery) occlusion; WKY, Wistar–Kyoto. Physical training consisted in a treadmill activity (30 minutes/day, 18 m/minute, 7 consecutive days) with a horizontal or −10% decline position of the treadmill. L-NAME was administered (60 mg/kg, intraperitoneal) 1 hour before each exercise session. Results were expressed as the percentage of elevation of SYN and BDNF levels induced by training relative to levels in sedentary rats.
As shown in Table 1, changes in SYN levels induced by training matched with changes in BDNF levels in all animal groups, except in SHR in which training increased SYN levels (+ 60%) but not BDNF levels (Figure 3B).
DISCUSSION
The major findings of the present study are that (i) elevation of brain BDNF levels evoked by physical training was dependent on exercise intensity and totally abolished by previous occlusion of a common carotid artery, at least in the cortex ipsilateral to the occlusion, (ii) concurrent pharmacological inhibition of NO synthase and genetic hypertension prevented the effect of training on BDNF. Taken together, these findings connect changes in cerebral circulation to training-induced BDNF levels elevation in the brain.
As evoked in the introduction, elevation of BDNF levels in the brain after physical training may involve neuronal hyperactivity occurring during exercise. Therefore, to investigate the contribution of hemodynamics to the effects of training on BDNF, we were concerned with using strategies that would reduce exercise-induced elevation of blood flow in the cerebrovasculature but not reduce exercise-induced neuronal hyperactivity. These conditions were satisfied for model of unilateral carotid occlusion that alters neither basal neuronal activity 17 nor basal BDNF levels (the present results). They were also satisfied with downhill training. Indeed, although neuronal activation is dependent on exercise intensity, neuronal hyperactivity is expected to be higher during downhill (that predominantly involves eccentric contraction) than horizontal walking (that involves eccentric-concentric cycles) as suggested from studies that have compared the pattern of neuronal activation between concentric and eccentric contraction.30,31 Moreover, to further explore the contribution of cerebrovascular blood flow elevation to training effects, we investigated the impact of genetic hypertension and L-NAME treatment on training-induced BDNF levels elevation as pathologic and pharmacological approaches to induce alteration in shear stress signaling, respectively. In basal conditions (sedentary conditions), cortical BDNF levels were found higher in SHR than in WKY rats, suggesting an interaction between genetic hypertension and brain BDNF production. Consistently, as compared with WKY rats, SHR were previously reported to exhibit higher BDNF levels in the nucleus tractus solitarii 32 and lower levels in the hippocampus. 33 Such a structure-dependent effect of genetic hypertension is a priori not compatible with a negative interaction between hypertension and brain BDNF levels. In accordance with this hypothesis are our results showing no difference in cortical BDNF between saline- and L-NAME-treated rats while L-NAME was previously reported to induce hypertension when administered to rats at the dosage used in the present study.27,28 Further studies are needed to explore the mechanisms underlying higher cortical BDNF levels in sedentary SHR. Regardless of the mechanisms involved, these higher levels cannot however question the validity of the SHR model to investigate whether increased BDNF levels in the brain of trained rats relate to shear stress elevation in the cerebrovasculature and subsequent overproduction of NO during exercise. As shear stress in the cerebrovasculature is relatively constant in sedentary rats, higher BDNF levels in sedentary SHR and the lack of difference in BDNF levels between sedentary L-NAME- and saline-treated rats may suggest that BDNF levels at rest are independent on the basal production of NO by the vascular endothelium. This does not however exclude a control of brain BDNF levels by NO when NO is produced in excess.
The present study is the first to investigate the effect of physical training on BDNF levels in the presence of unilateral carotid occlusion. The results that reported a complete abolition on the effect of training on BDNF levels in the hemisphere ipsilateral to the occlusion and a partial abolition in the opposite hemisphere emphasize the contribution of cerebrovascular flow increase in training-induced BDNF levels elevation in the brain. This is further supported by our results showing that elevation of brain BDNF levels evoked by training was dependent on exercise intensity. Indeed, BDNF levels were lower after downhill than level locomotion activity. Only two previous studies from the same laboratory investigated the effect of downhill training on brain BDNF levels.34,35 The authors reported no difference in BDNF levels between horizontal and downhill modalities. However, discrepancies between these data and ours are may be because of difference in the modalities of training including the slope of the treadmill (− 29 versus −10% in our study), training duration (8 weeks versus 7 days in the present study), the regions examined (the hippocampus and the striatum versus the cortex in the present study), the procedure for BDNF levels measurements (ELISA tests that did not distinguish mature BDNF from its precursor proBDNF and only the mature form of BDNF using western blot analysis in the present study). Anyway, evidence that physical training-induced BDNF levels elevation in the brain is dependent on normal carotid circulation and exercise intensity emphasizes cerebral hemodynamics in the control of cerebral BDNF levels after physical training. This data question the traditional thinking that the unique role of elevation in cerebrovascular flow occurring during exercise serves to meet neuronal metabolic demand. It has to be approached to a previous study in which blood supply to hyperactive regions during exercise was reported to be larger than their metabolic oxygen demand. 36 We think that elevation of cerebrovascular flow during exercise beyond values required to satisfy metabolic needs may be a hitherto physiologic mechanism to induce brain plasticity through the control of brain BDNF levels. As an argument for this, changes in BDNF levels evoked by physical training parallel changes in SYN levels. Indeed, SYN is a biochemical marker of both synaptic density and activity, i.e., two processes underlying brain plasticity.
The emerging question is how changes in cerebral hemodynamics during exercise target brain BDNF levels. The present results support the involvement of endothelium-derived NO. First, we showed that physical training became unable to elevate brain BDNF levels in SHR, a model of endothelial dysfunction and altered production of NO by the endothelium in response to shear stress.22–24 Second, these effects of genetic hypertension were reproduced by administration of the NO synthase inhibitor L-NAME during physical training. Notably, L-NAME treatment was previously reported to reduce the elevation of brain BDNF levels evoked by free access to a running wheel as a model of physical training. 37 However, as a confounding factor L-NAME treatment was associated with a drastic decrease in the mean running distance. Importantly, in the present study, difference in brain BDNF levels between L-NAME- and saline-treated rats after training cannot be explained by a reduction in exercise duration/intensity, as all the rats were subjected to a same amount and intensity of physical training. Finally, a striking result of the present study was the differential response of SYN to physical training between SHR (+ 60%) versus L-NAME-treated rats (no change) while both genetic hypertension and L-NAME treatment annulled the effect of training on BDNF. A possible but speculative explanation may be a high TrkB receptor density in SHR cortex as compared with L-NAME-treated rats cortex.
In conclusion, our results provide converging evidence for a connection between elevation of BDNF levels after physical training and blood flow increases in the cerebrovasculature. This finding paves the way for further advancement in the mechanisms by which training improves brain health and provides a rational basis to optimize brain benefits of physical training. Our results also suggest that optimal modalities of training to improve brain function may differ between healthy beings and patients with endothelial dysfunction or asymptomatic carotid occlusion. Finally, the present study supports the emerging concept that cerebral endothelium is a key tissue linking cerebrovascular function and cognitive processes. 38
Footnotes
The authors declare no conflict of interest.
