Abstract
After a period of global cerebral ischemia, CO2 reactivity and the hemodynamic–metabolic activation to functional stimulation are transiently suppressed. This raises the question of whether the impaired functional coupling reflects disturbances of functional integrity of the brain or an impaired cerebrovascular reactivity. We, therefore, compared the recovery of CO2 reactivity with that of somatosensory evoked potentials, functional flow activation and neurologic deficits in a rodent model of cardiac arrest-induced cerebral ischemia, followed by up to 7 days of reperfusion. Cardiac arrest of 10 minutes' duration was produced in 24 animals by electrical fibrillation of the heart. Five animals were sham-operated controls. Resuscitation was performed by external cardiac massage, using standard resuscitation procedures. Functional activation was carried out under chloralose anesthesia by electrical stimulation of forepaws. CO2 reactivity was tested by ventilation of animals with 6% CO2. During functional and hypercapnic stimulation CBF was measured in the somatosensory cortex using laser–Doppler flowmetry, and at the end of the experiment by 14C-iodoantipyrine autoradiography. Neurologic deficits were scored by evaluating consciousness and various sensory and motor functions. In control animals 6% CO2 increased CBF measured by laser–Doppler flowmetry by 28.8% ± 8.7%. Forepaw stimulation generated somatosensory evoked potentials with an amplitude of 750 ± 217 μV and increased CBF measured by laser–Doppler flowmetry by 86.0% ± 18.1%. After return of spontaneous circulation, CO2 reactivity was transiently reduced to about 30% of control at 1 hour of reperfusion (P < 0.05) but returned to near control at 5 hours. Somatosensory evoked potential amplitudes were reduced to 15% of control at 45 minutes of reperfusion and returned to only 50% to 60% at 3 and 7 days after return of spontaneous circulation (P < 0.05). Functional activation of blood flow was completely suppressed during the first hour after return of spontaneous circulation but also recovered to 50% to 60% of control at 3 days after return of spontaneous circulation (P < 0.05). Linear regression analysis revealed a significant correlation between recovery of functional activation of blood flow and both recovery of the amplitude of somatosensory evoked potentials (P = 0.03) and the neurologic deficit score (P = 0.02), but not between neurologic deficit score and recovery of CO2 reactivity or somatosensory evoked potential amplitudes. These data demonstrate that the suppression of functional activation of blood flow after 10 minutes cardiac arrest is not related to impairment of coupling mechanisms but reflects ongoing disturbances of the functional integrity of the brain. Assessment of functional flow coupling is a reliable way to study postischemic recovery of the brain.
Keywords
In the intact brain neuronal activation is closely coupled to an increase in blood flow and metabolic activity (Sokoloff, 1977). Regional measurements of cerebral blood flow or of glucose utilization are, therefore, widely used to detect functionally active brain regions. The most obvious application of this approach is the localization of normal brain functions, a method referred to as functional neuroanatomy (Fox et al., 1986). But there is also a clinical interest in identifying and localizing disturbances of brain function at an early stage of disease, as in Alzheimer's disease or other neurodegenerative disorders (Kessler et al., 1991). Conversely, functional activation of cerebral blood flow or metabolism is monitored to study functional recovery, particularly after transient episodes of ischemia (Dietrich et al., 1986; Ueki et al., 1988).
Obviously, the validity of this approach depends on the preservation of the coupling mechanisms that mediate the signals arising from the activated neuronal network to the regulatory elements of brain metabolism and blood flow. There are indications that under pathophysiologic conditions these mechanisms may be disturbed, leading to a dissociation between neuronal activity and the associated hemodynamic or metabolic response. After prolonged cerebral ischemia, CO2 reactivity of cerebral blood flow—which is thought to reflect the coupling between metabolic activity and cerebral vascular tone—is suppressed for extended periods despite progressive recovery of spontaneous electrocortical activity (Schmidt-Kastner et al., 1986). Cortical potentials evoked by somatosensory stimulation may also begin to recover in the absence of any regional changes of glucose metabolism or blood flow (Ueki et al., 1988). On the other hand, topical application of acidic mock cerebrospinal fluid produces vasodilation of pial arteries (Hossmann et al., 1976), and potassium-induced spreading depression increases blood flow and metabolism under the same experimental conditions (Kocher, 1990).
These dissociations could be explained in various ways. Either spontaneous electrocortical activity or evoked cortical potentials are not equivalent with the kind of neuronal activity that is coupled to the metabolic–hemodynamic response of the tissue, or the functional metabolic coupling is mediated by signal transduction systems that may be selectively impaired by the pathologic process. This raises the questions of whether CO2 reactivity provides any meaningful information about the intactness of the coupling mechanisms and whether recordings of metabolic or blood flow changes are able to monitor functional activity under pathophysiologic conditions.
To address these questions we evaluated the temporal relationship between the suppression and recovery of CO2 reactivity, functionally evoked blood flow response, evoked cortical potentials and neurologic performance after a period of global cerebral ischemia. For the production of global brain ischemia we used a rodent model of reversible cardiac arrest. This model was chosen because cardiocirculatory arrest is the most frequent clinical cause of this type of ischemia, and because the results obtained can be expected to bear direct clinical relevance.
MATERIALS AND METHODS
Experiments were carried out in accordance with the German legislation on animal care, and approved by the local authorities. Twenty-nine adult male Sprague-Dawley rats weighing 330 to 450 g were used. Five animals were sham-operated, nonischemic controls, the other 24 animals were submitted to 10 minutes of cardiac arrest followed by 6 hours, 1 day, 3 days, or 7 days reperfusion, respectively (n = 6 for each survival time).
Cardiac arrest and resuscitation
Animals were anesthetized with 1.5% halothane in a mixture of 70% nitrous oxide and 30% oxygen. A femoral or tail artery and a femoral vein were cannulated with PE-50 catheters for blood sampling, blood pressure recording, drug administration, and continuous saline infusion (2 mL·kg−1·h−1). Animals were intubated orotracheally, paralyzed with pancuronium bromide (0.2 mg·kg−1·h−1), and artificially ventilated (Harvard Rodent Ventilator, Harvard Apparatus, South Natick, MA, U.S.A.). Blood gases were repeatedly measured and maintained within physiologic limits by appropriate adjustments of the ventilator. Body temperature was kept constant at 37°C using a feedback-controlled heating pad.
Cardiac arrest was induced by electrical fibrillation (alternating current, 12 V, 50 Hz) using a pediatric pacing catheter advanced into the esophagus, and confirmed by arterial blood pressure recording (Kombidyn Monitoring Set, Braun AG, Melsungen, Germany). For the duration of cardiac arrest, artificial ventilation, the heating system, and saline infusion were transiently switched off. Ten minutes after the beginning of ventricular fibrillation, cardiopulmonary resuscitation was started using standard resuscitation procedures (Böttiger et al., 1997). Closed-chest cardiac massage was performed by applying sternal compression at a rate of about 200/min. At the same time, artificial ventilation was resumed with 100% oxygen at a frequency 50% above control conditions, and bolus injections of epinephrine (0.02 mg/kg body weight) and sodium bicarbonate (NaHCO3, 0.5 mEq/kg body weight) were given intravenously. Direct current countershocks (5 J, DEFIPORT SCP912, Hellige, Freiburg, Germany) were applied 120 seconds after the onset of cardiopulmonary resuscitation and repeated after 30 to 60 seconds, if necessary. After return of spontaneous circulation (ROSC), as defined by spontaneous cardiac action with mean arterial pressure of more than 50 mm Hg, the heating pad was again switched on, and saline infusion was resumed. Arterial blood samples were taken 5, 30, and 60 minutes after resuscitation, and blood gases (Model 288, Ciba-Corning, Fernwald, Germany) were adjusted as fast as possible by appropriate adjustments of the ventilator speed and inspired oxygen concentration, and controlled application of NaHCO3.
Animals selected for 1, 3, and 7 days reperfusion were weaned from the respirator 60 minutes after ROSC. The wounds were infiltrated with lidocaine and properly closed. Then the animals were extubated and kept for another hour in an oxygen-enriched atmosphere before return to their cages. To prevent dehydration, 20 mL Ringer's lactate solution per day was given subcutaneously until the rats started to drink spontaneously.
Recording of cerebral blood flow
Animals were fixed in a stereotactic head holder, the skin over the apex of the skull was removed, and the bone over the right somatosensory cortex was thinned with a liquid-cooled dental drill for improving translucency. A laser–Doppler flow probe (0.8 mm, PeriMaster 4001, PeriMed, Järfälla, Sweden) was placed over this region and fine-adjusted for maximal response during somatosensory stimulation (Lindauer et al., 1993; Schmitz et al., 1996). The output of the flowmeter was continuously recorded on a polygraph (DASH 8, Astromed, Rodgau, Germany) and processed using a personal computer and the DasyLab data acquisition software (DataLog, Mönchengladbach, Germany).
Electrical forepaw stimulation was carried out as recently described (Schmitz et al., 1996). Before functional activation, halothane/N2O was replaced by alpha-chloralose anesthesia (60 to 80 mg/kg intravenously) and ventilation was carried out with a 7:3 mixture of N2/O2. For electrical stimulation of the right somatosensory cortex, small needle electrodes were inserted subcutaneously into the contralateral forepaw, and rectangular pulses of 0.3-millisecond duration were applied for 1 minute at a frequency of 3 Hz. The current of stimulation pulses (0.5 to 1.0 mA) was adjusted to evoke maximum amplitude of somatosensory evoked cortical potentials (SEP). Specificity of the response was ascertained by alternate stimulation of the two forepaws.
In the 6-hour survival group, functional activation was carried out at 45 minutes of reperfusion and then always 30 minutes after testing CO2 reactivity (Fig. 1). Animals investigated at 1 to 7 days after cardiac arrest were reanesthetized and instrumented as described above. These and control animals were submitted to functional stimulation after stabilization of all physiologic variables, again 30 minutes after testing CO2 reactivity.
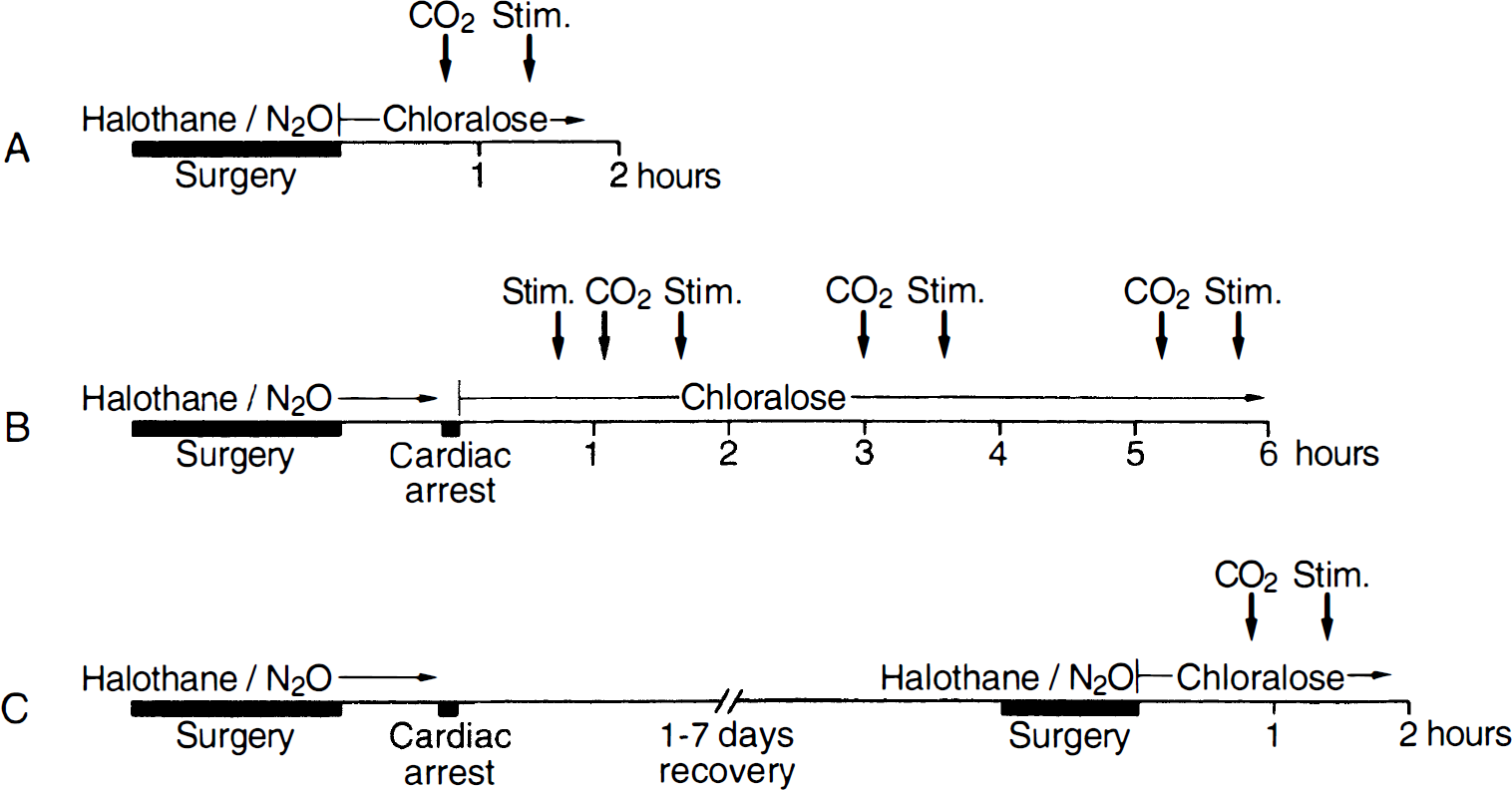
Experimental protocol.
The response of blood flow to hypercapnia was tested by adding 6% CO2 for 3 minutes to the inspiration gas in nonischemic controls, and at 1, 3, and 6 hours, and 1, 3, and 7 days of reperfusion. Arterial blood samples were taken for measurement of arterial Pa
At the end of each experiment, quantitative measurement of regional blood flow was carried out using the 14C-iodoantipyrine autoradiographic technique (Sakurada et al., 1978). The tracer (40 μCi/mL; Amersham, Braunschweig, Germany) was infused intravenously for a period of 60 seconds, and arterial blood samples were collected during infusion at 6- to 8-second intervals on preweighed filter paper. At the end of the infusion period, brains were frozen in situ, removed from the skull at −20°C, and cut with a cryostat microtome. Coronal 20-μm cryostat sections were sampled in steps of 200 μm from bregma to 2 mm rostral to bregma, because this part includes the somatosensory cortex activated by electrical forepaw stimulation (Schmitz et al., 1996). Quantitative autoradiography was performed using 14C-standards, a charge-coupled device camera connected to a Macintosh computer (Apple, Cupertino, CA, U.S.A.), and the image processing software IMAGE (National Institutes of Health, Bethesda, MD, USA). 14C-radioactivity of blood samples was measured in a liquid scintillation counter (Wallac 1410, Pharmacia, Freiburg, Germany), and blood flow was calculated as described by Sakurada et al. (1978). Blood flow in somatosensory cortex was expressed as the average of values determined in 8 to 10 autoradiograms obtained from consecutive cryostat sections.
Recording of somatosensory evoked potentials
Primary cortical potentials evoked by somatosensory stimulation of the contralateral forepaw (SEP) were recorded with a small calomel electrode (Busch et al., 1995) placed over the contralateral somatosensory cortex at 0.5 mm behind the laser–Doppler probe and fine-adjusted for maximum amplitude. The indifferent calomel electrode was positioned on the nasal bone. Somatosensory evoked potentials were averaged using the DasyLab data acquisition software. For the measurement of peak-to-peak amplitude, averages of 100 consecutive SEP were taken.
Neurologic deficit score
In control animals and in animals allowed to survive for 1, 3, and 7 days after cardiac arrest, neurologic deficit scores (NDS) were determined according to Katz et al. (1995) and Neumar et al. (1995). Scoring included five parameters: general behavior (consciousness and respiration), cranial nerve function (corneal reflex, whisker movement, hearing, vision, food sniffing), motor function (leg and tail movement), sensory function (leg and tail movement on pinching), and coordination (travelling ledge, righting reflex, placing test, stop at table edge) with NDS of 0% reflecting normal brain function and NDS of 100% brain death. Because of the experimental design it was not possible to determine NDS in the 6-hour survival group. Therefore, 6 animals from the 1- to 7-day survival groups were randomly assigned for NDS evaluation after 6 hours.
Data analysis
The changes in laser–Doppler flow during hypercapnia or functional activation are expressed in percent of the baseline value. For this purpose data points were sampled for 60 seconds before and during hypercapnia or functional activation. Regional blood flow of somatosensory cortex measured by quantitative autoradiography is given in absolute numbers. Measurements (means ± SD) were tested for normal distribution using the Kolmogorov-Smirnov test, and differences were evaluated for statistical significance using one-way analysis of variance or Kruskal-Wallis-analysis of variance on ranks followed by Student-Newman-Keuls test. Correlations were tested by linear regression analysis (STATISTICA for Windows, StatSoft Inc., Tulsa, OK, U.S.A.). A probability value of P < 0.05 was considered significant.
RESULTS
Cardiac arrest and resuscitation
Before cardiac arrest, physiologic variables were in the normal range (Table 1), and there were no differences between groups of animals selected for different survival times. Ventricular fibrillation caused instantaneous cessation of cardiac function and a drop of blood pressure to baseline (Fig. 2). Cardiopulmonary resuscitation succeeded in ROSC in all animals within 4 minutes (duration of cardiopulmonary resuscitation, 181 ± 44 seconds). There were no differences between the various survival groups as regards to the amount of epinephrine (0.022 ± 0.006 mg/kg), sodium bicarbonate (1.6 ± 0.3 mEq/kg), or direct current countershocks (2.4 ± 1.5) required for successful resuscitation.
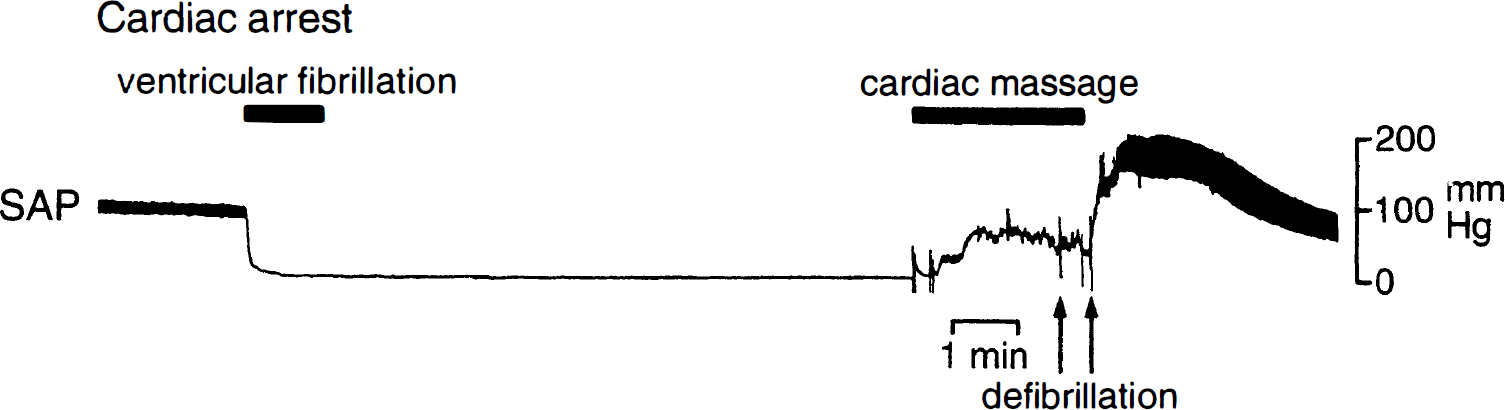
Recording of systemic arterial blood pressure (SAP) during and after 10 minutes of cardiac arrest. Note instantaneous fall of SAP after the onset of ventricular fibrillation and the prompt restoration of cardiac function after defibrillation.
Physiological variables before and after cardiac arrest
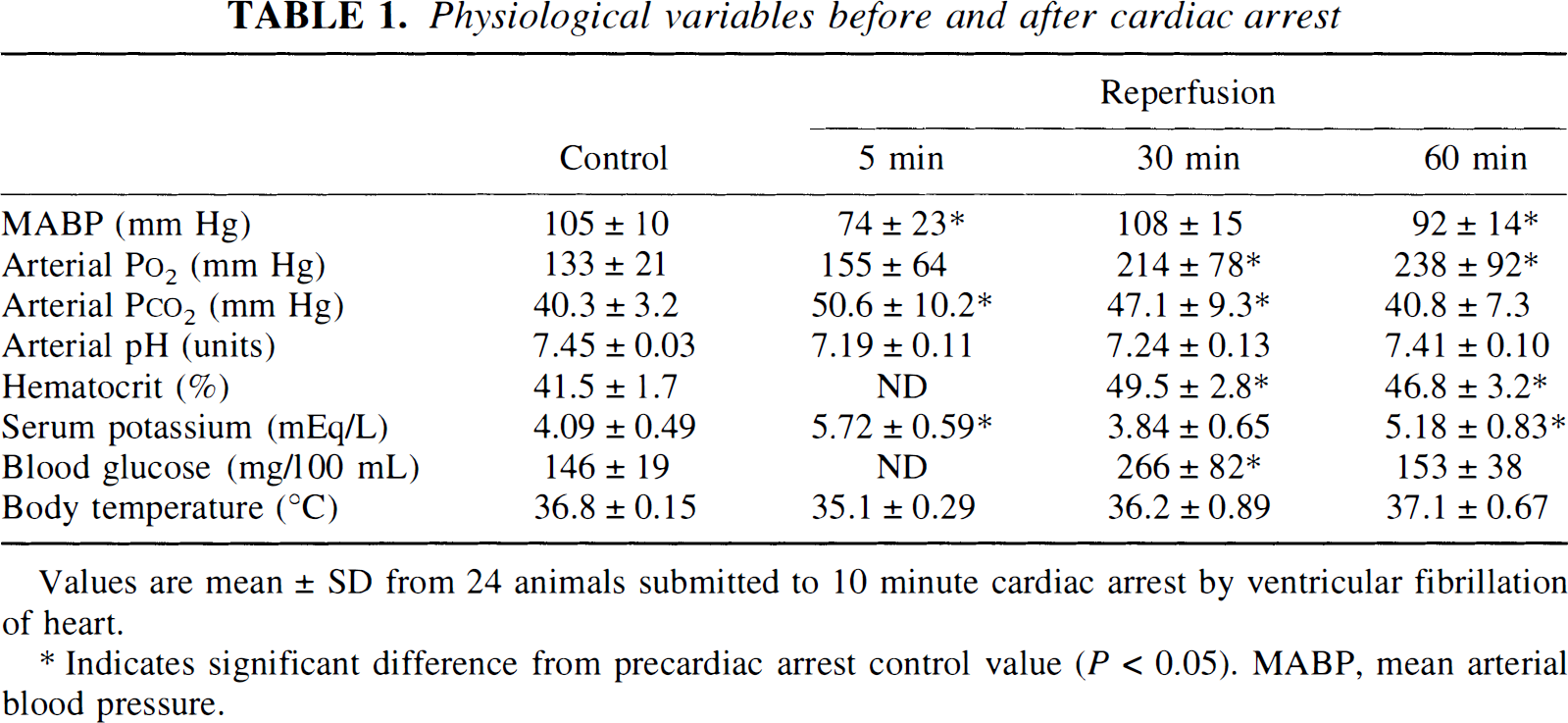
Values are mean ± SD from 24 animals submitted to 10 minute cardiac arrest by ventricular fibrillation of heart.
Indicates significant difference from precardiac arrest control value (P < 0.05). MABP, mean arterial blood pressure.
After return of spontaneous cardiac function, blood pressure rose sharply above control and peaked at 203 ± 33 mm Hg by 57 ± 18 seconds of reperfusion, followed by a return to or below baseline after 5 minutes (Fig. 2). Postcardiac arrest changes in blood variables, i.e., acidosis, hypercapnia, and hyperkalemia, were maximal at 5 minutes after ROSC, followed by gradual return to normal within 60 minutes (Table 1). Body temperature fell during cardiac arrest by 1.5° to 2°C but rapidly normalized after ROSC. Pulmonary function was not severely impaired after cardiac arrest, as evidenced by the rapid normalization of ventilation parameters required to maintain physiologic blood gas parameters.
Electrophysiologic and neurologic function
Under control conditions a high amplitude–low frequency EEG (mean amplitude: 611 ± 75 μV) with intermittent burst-suppression pattern was present that is typical for α-chloralose anesthesia (Ueki et al., 1988; Ueki et al., 1992) (Fig. 3). Electrical stimulation of the forepaw generated primary SEP with a peak-to-peak amplitude of 750 ± 217 μV.
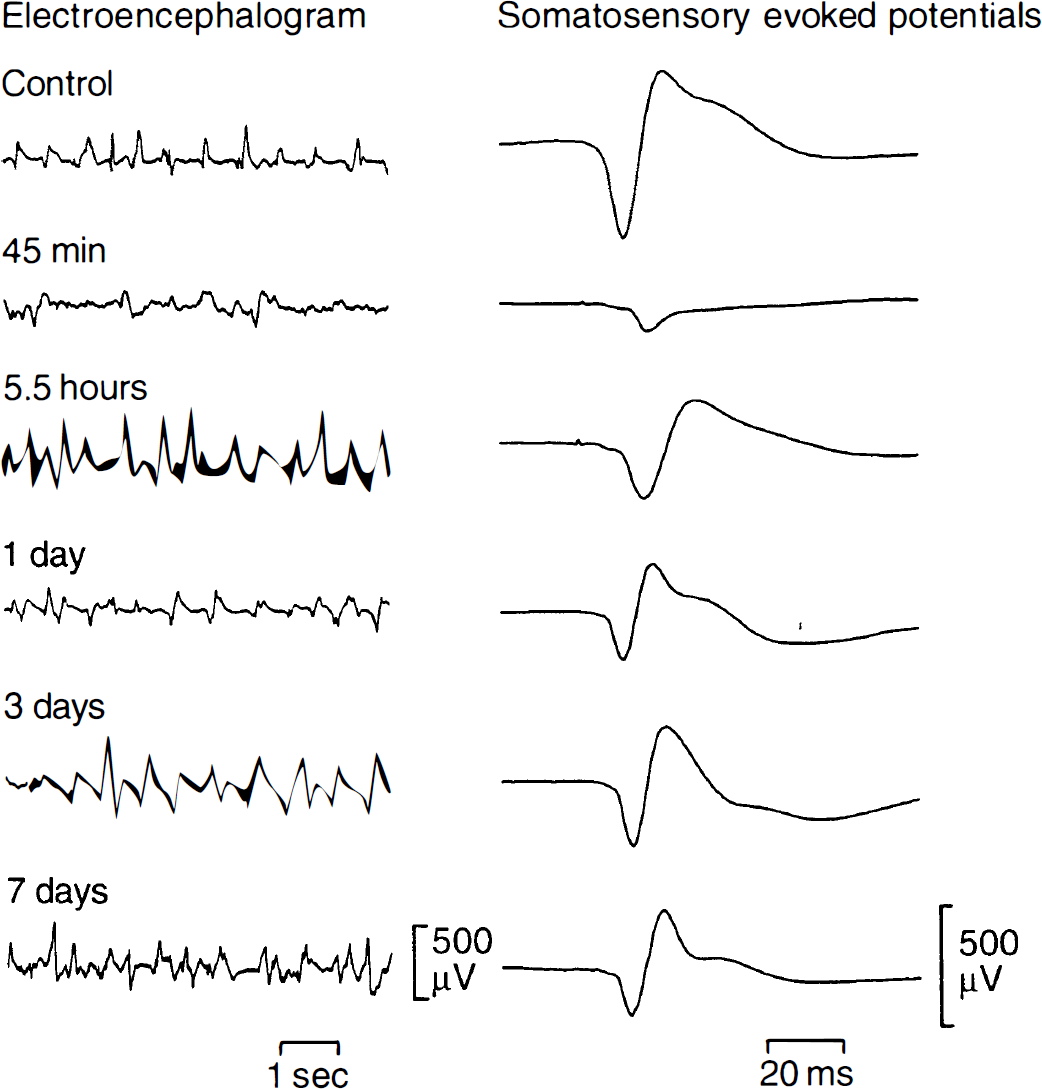
Recording of the electroencephalogram and of somatosensory evoked potentials before and at various recirculation times after 10 minutes of cardiac arrest. The spiky appearance of the electroencephalogram is characteristic for chloralose anesthesia. Note slow recovery of the amplitude of somatosensory evoked potentials as compared with the much faster restoration of the electroencephalogram.
Return of spontaneous circulation after 10 minutes of cardiac arrest was accompanied by the recovery of spontaneous EEG activity within 25 ± 1 minutes. Electroencephalographic amplitude returned to control within 6 hours of reperfusion, but then secondarily declined. The EEG median frequency was significantly slower during the first 3 days after resuscitation compared with control (Table 2).
Electrophysiological recordings
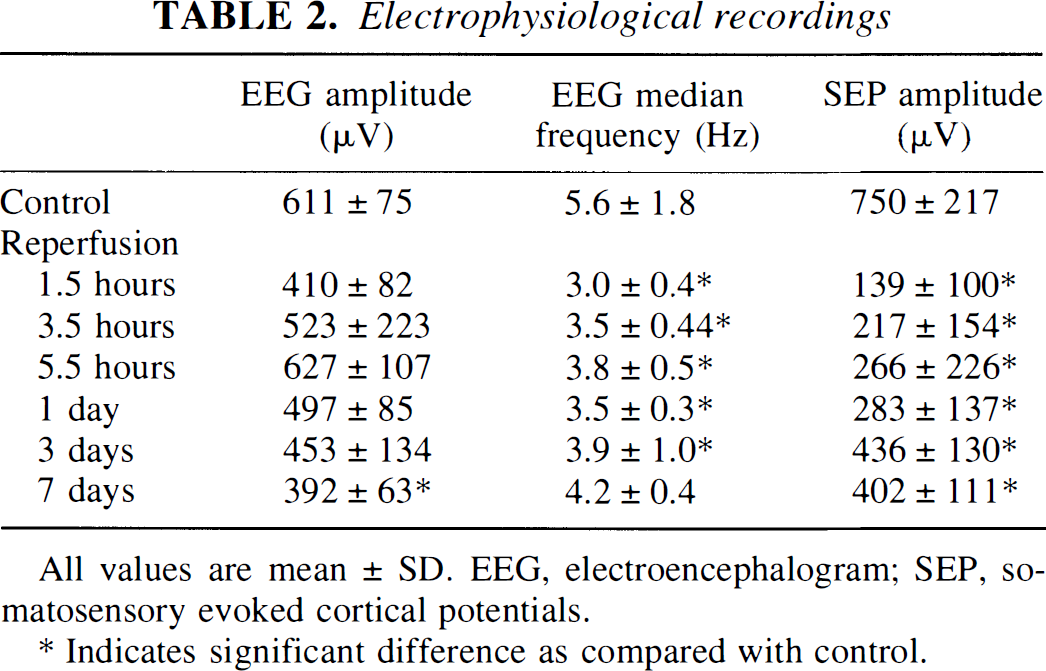
All values are mean ± SD. EEG, electroencephalogram; SEP, somatosensory evoked cortical potentials.
Indicates significant difference as compared with control.
Somatosensory evoked potentials began to recover in less than 45 minutes, i.e., when the first postresuscitation stimulation was carried out. At this time the amplitude was only 15% ± 11% of control (P < 0.05) but it gradually improved and reached 58% of control after 3 days and 54% of control after 7 days of recirculation (Table 2; Figs. 3, 4). The reduced excitability of the cerebral cortex was also reflected by the stimulation current required for maximal response that had to be increased from 0.5 mA up to 0.7 to 1.0 mA during the first 24 hours after cardiac arrest.
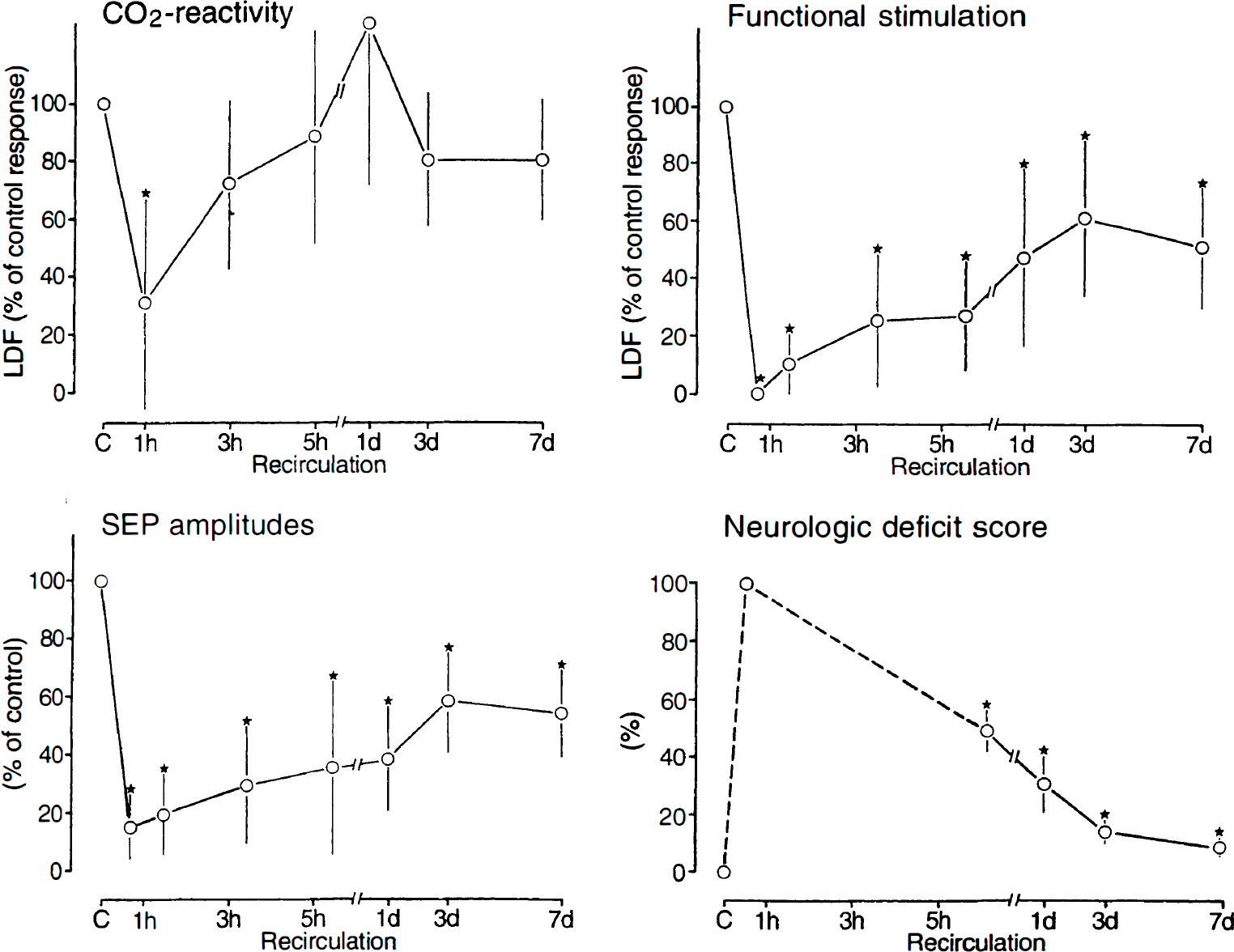
Comparison of the flow changes to hypercapnia (CO2 reactivity) and functional stimulation with the amplitude of somatosensory potentials (SEP) and the neurologic deficit score before and at various recirculation times after 10 minutes of cardiac arrest. Flow changes and the amplitude of SEP are expressed in percent of the preischemic control values. Note faster recovery of CO2 reactivity than both the flow response to functional stimulation and the recovery of SEP and neurologic performance.
The NDS determined at 6 hours and at 1, 3, and 7 days of reperfusion in 24 animals showed substantial improvement with ongoing recirculation (Fig. 4). All animals were conscious after 6 hours but exhibited coordination and sensory deficits, as well as bilateral spastic paralysis of the hind limbs. Coordination deficits and spastic paralysis disappeared after 3 days in all animals. After 7 days animals showed only slightly retarded reaction to visual stimulation and pinching of tail or hind-leg but no overt neurologic deficits.
CO2 reactivity of cerebral blood flow
Quantitative measurements of basal cerebral blood flow by 14C-iodoantipyrine autoradiography were carried out in nonischemic controls and at 6 hours to 7 days of survival after 10 minutes of cardiac arrest. In the sensorimotor cortex, regional cerebral blood flow was 67 ± 14 mL·100 g−1·min−1 (n = 4) before and 58 ± 9 mL·100 g−1·min−1 (n = 4) at 6 hours of recirculation after cardiac arrest. With ongoing survival times flow gradually declined to 53 ± 8 mL·100 g−1·min−1 after 1 day (n = 6; not significant[NS]), 48 ± 13 mL·100 g−1·min−1 after 3 days (n = 5; NS), and 47 ± 7 mL·100 g−1·min−1 after 7 days (n = 5; P < 0.05 versus control).
The flow changes during hypercapnia were assessed by laser–Doppler flowmetry. Ventilation with 6% CO2 increased arterial Pa
Physiologic variables before, during, and after hypercapnia
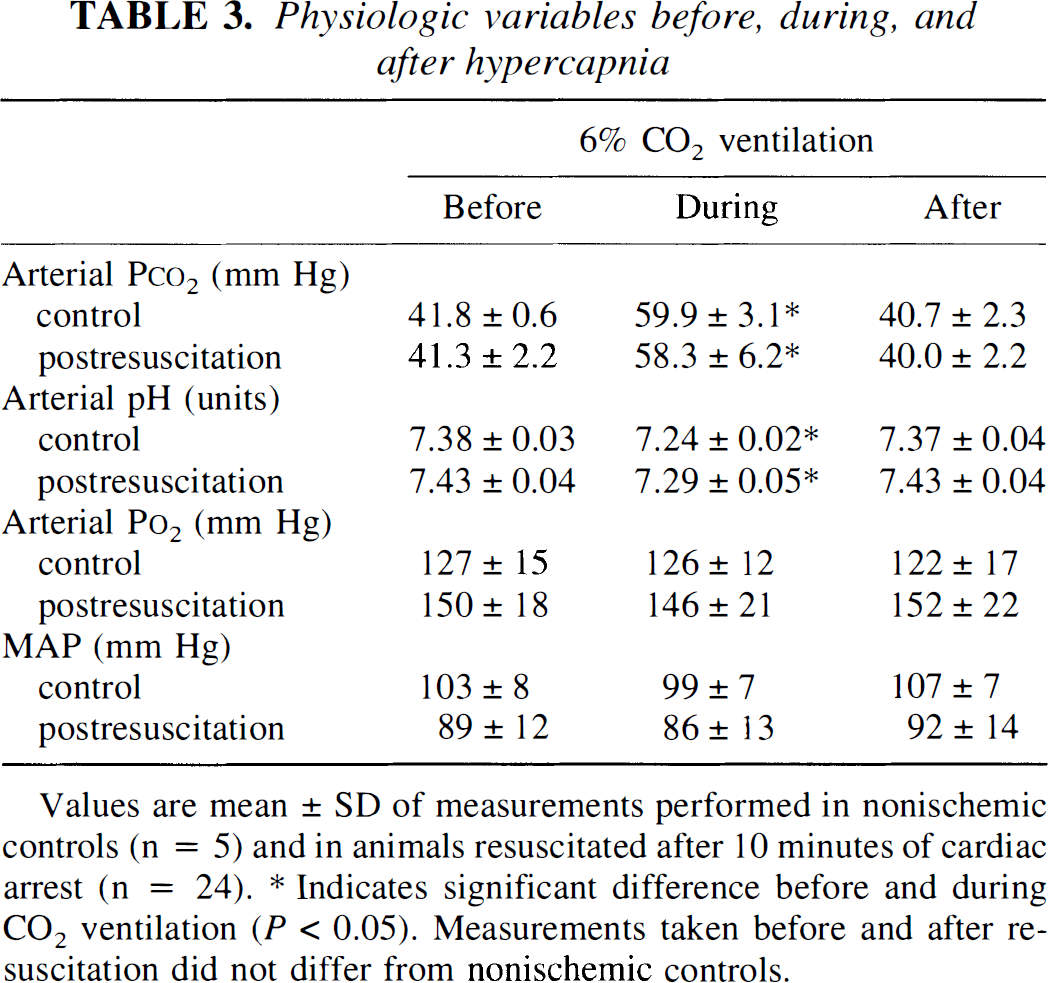
Values are mean ± SD of measurements performed in nonischemic controls (n = 5) and in animals resuscitated after 10 minutes of cardiac arrest (n = 24).
Indicates significant difference before and during CO2 ventilation (P < 0.05). Measurements taken before and after resuscitation did not differ from nonischemic controls.
Under control conditions hypercapnia led to an increase in laser–Doppler flow by 28.0% ± 8.7%. CO2 reactivity was reduced to 8.5% ± 10.1% (31% of control) at 1 hour of reperfusion (P < 0.05), but recovered to near normal at 5 hours of recirculation (Fig. 4).
Functional activation of cerebral blood flow
In the nonischemic control animals functional activation led to a sharp increase in laser–Doppler flow by 86.0% ± 18.1%. The specificity of this response was confirmed by the absence of any measurable change during stimulation of the ipsilateral forepaw. Somatosensory stimulation also failed to change arterial blood pressure. After 10 minutes of cardiac arrest followed by 45 minutes of recirculation, the blood flow response to forepaw stimulation was completely suppressed, although SEP had already begun to recover. With ongoing recirculation time, the blood flow response gradually recovered. In some but not all animals a beginning response could be observed after 1.5 hours of reperfusion, followed by a gradual improvement of 50% to 60% of control within 3 days (Fig. 4). However, even after 7 days of recirculation, the blood flow response was still significantly lower than in control animals.
Correlation analysis
Linear regression analysis revealed a significant correlation between the recovery of the peak-to-peak amplitude of SEP after cardiac arrest and the coupled blood flow response (r = 0.47; P < 0.05). There was no correlation between recovery of CO2 reactivity and the functional activation of blood flow (r = 0.21, NS) or SEP recovery (r = 0.28, NS).
The NDS correlated inversely with the recovery of blood flow response to forepaw stimulation (r = −0.48, P < 0.02) (Fig. 5). In contrast, there was no correlation between recovery of CO2 reactivity and NDS (r = −0.10, NS) or between NDS and the amplitude of SEP (r = −0.41, NS).
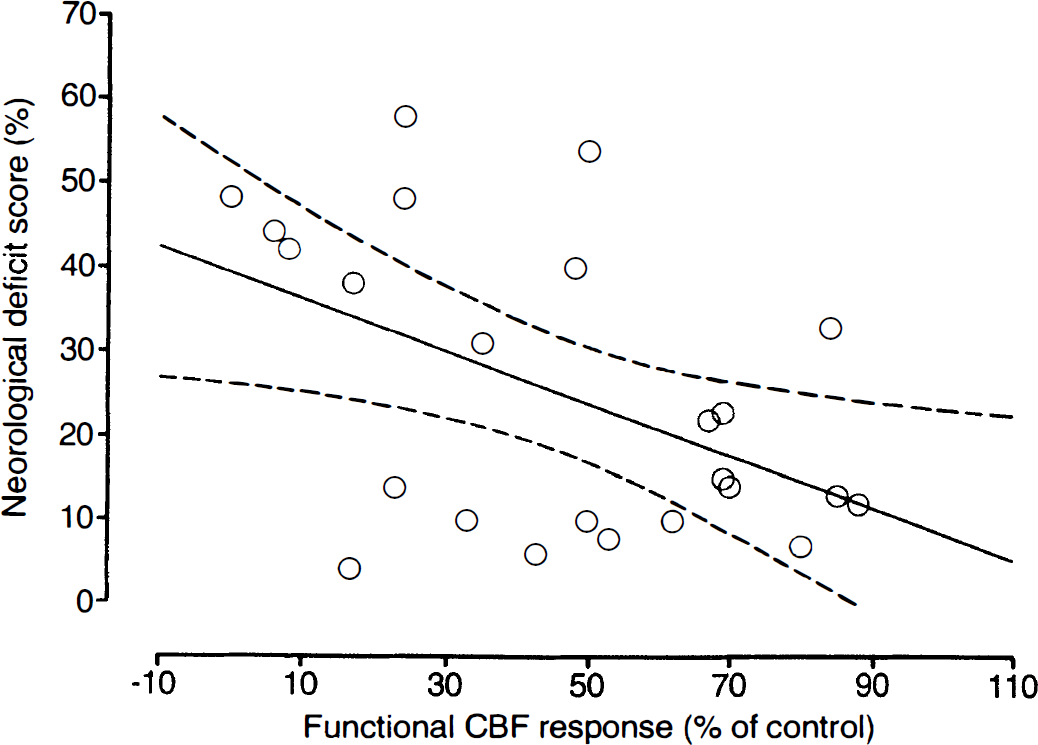
Scatter plot of the relationship between neurologic deficit score (NDS) and recovery of the flow activation induced by electrical forepaw stimulation in rats submitted to 10 minutes of cardiac arrest followed by 6 hours to 7 days of reperfusion. Each measurement refers to one animal investigated at the end of the reperfusion phase (total number of rats, n = 24).
DISCUSSION
The present study of the functional activation of cerebral blood flow after 10 minutes reversible cardiac arrest demonstrates that SEP and CO2 reactivity begin to recover earlier than the activation-induced blood flow response. However, with ongoing reperfusion the amplitude of SEP correlates directly and the NDS inversely with the increment of cerebral blood flow during functional activation. This finding differs from previous studies of 30 minutes of near complete forebrain ischemia of rat or of 60 minutes of complete brain ischemia of cat in which CO2 reactivity and the blood flow coupling to sensory stimulation were suppressed for extended periods, despite progressing recovery of electrophysiologic and neurologic recovery (Schmidt-Kastner et al., 1986; Ueki et al., 1988). In fact, CO2 reactivity, which is caused by a change of vascular tone analogous to that induced by metabolically produced carbon dioxide, was suppressed for several days after 60 minutes ischemia (Schmidt-Kastner et al., 1986) but recovered within less than 3 hours in the present model of 10 minutes of cardiac arrest.
This difference can be best explained by a more severe ischemic injury after the longer ischemia of the responsive elements that couple the hemodynamic reaction to the functional input. The much slower recovery of the blood flow response to functional activation in the present model is, therefore, not limited by a reduced cerebrovascular sensitivity, as after prolonged ischemia (Schmidt-Kastner et al., 1986). This explains that, contrary to this experimental situation, neurologic recovery is coupled to the recovery of the blood flow response.
In a previous study we demonstrated that in the chloralose-anesthetized rat brief hypercapnic periods markedly upregulate the local cerebral blood flow increase in response to electrical forepaw stimulation (Schmitz et al., 1996). Since hypercapnia is an unavoidable accompaniment of cardiac arrest, the possibility of a similar upregulation had to be considered in the present study. Obviously, the suppression of functional activation after cardiac arrest excludes such an upregulation during the early recirculation phase. However, to compensate for possible interactions after longer recirculation times, CO2 reactivity was always tested before functional stimulation to assure reproducible experimental conditions.
In contrast to the hemodynamic coupling no correlation was observed between the recovery of the amplitude of EEG or evoked potentials and the NDS. This indicates that the amplitude of the evoked cortical response is not a reliable measure of the metabolic workload imposed on the cerebral cortex during functional activation. We propose two explanations for this dissociation. First, the peak-to-peak amplitude of cortical evoked potentials does not discriminate between the afferent input and the efferent output of cortical activity. In fact, the form of the evoked potentials is greatly distorted during the early phase of postischemic recovery, indicating that the cerebral cortex responds not only quantitatively but also qualitatively in a different way to the afferent input as before ischemia.
It has been suggested that the metabolic workload during functional activation is mainly caused by the energy-dependent reuptake of neurotransmitters into astroglia (Pellerin and Magistretti, 1994). It is also known that after ischemia, presynaptic activity recovers faster than synaptic transmission (Hossmann, 1987). If the generation of presynaptic activity requires less energy than the generation of the efferent cortical output, the coupled blood flow response could, in fact, be smaller than the amplitude of the evoked potentials suggest.
The other possible explanation is a dissociation of the electrophysiologically detectable cortical response to the integrated activity of the activated neuronal network. This interpretation is in line with the well-known suppression of the metabolic–hemodynamic coupling by different anesthetics despite preservation of the generation of cortical evoked potentials (Ueki et al., 1992). The recovery of the blood flow response to functional stimulation could, therefore, be a more meaningful indicator of the recovery of integrated brain function than the reappearance of cortical evoked potentials.
With these considerations in mind, the hemodynamic coupling for the evaluation of functional recovery has to be interpreted with caution. If the coupling mechanisms per se are impaired, the absence of a blood flow response does not necessarily mean that the functional activity of the brain is lost. Recording of the CO2 reactivity is an easy means to exclude this possibility, and the current data clearly demonstrate that the suppression of the functional blood flow response after 10 minutes of cardiac arrest is not caused by impaired cerebrovascular reactivity. On the other hand, the activation-induced blood flow response is positive evidence for the functional recovery and, therefore, may provide more reliable information about the functional integrity of the neuropil than electrophysiologic recordings of spontaneous or evoked cortical activity, at least under the defined conditions of transient cardiac arrest. With the increasing availability of noninvasive methods for the detection of the blood flow response to functional activation, such as positron emission tomography (Fox et al., 1986), susceptibility (Kwong et al., 1992) and perfusion-weighted magnetic resonance imaging (Kerskens et al., 1996), or near-infrared spectroscopy (Villringer et al., 1993), this approach could therefore be of great interest for the early prediction of postcardiac arrest recovery of brain function.
