Abstract
13C Nuclear Magnetic Resonance (NMR) studies of rodent and human brain using [1-13C]/[1,6-13C2]glucose as labeled substrate have consistently found a lower enrichment (~25% to 30%) of glutamine-C4 compared with glutamate-C4 at isotopic steady state. The source of this isotope dilution has not been established experimentally but may potentially arise either from blood/brain exchange of glutamine or from metabolism of unlabeled substrates in astrocytes, where glutamine synthesis occurs. In this study, the contribution of the former was evaluated ex vivo using 1H-[13C]-NMR spectroscopy together with intravenous infusion of [U-13C5]glutamine for 3, 15, 30, and 60 minutes in mice. 13C labeling of brain glutamine was found to be saturated at plasma glutamine levels > 1.0 mmol/L. Fitting a blood–astrocyte–neuron metabolic model to the 13C enrichment time courses of glutamate and glutamine yielded the value of glutamine influx,
INTRODUCTION
Glucose, the primary source of energy in the mature brain, is mainly oxidized in neurons to support neuronal firing, and the release and recycling of neurotransmitters, glutamate, and GABA, through the neuron–astrocyte glutamate–glutamine and GABA–glutamine cycles. The metabolic pathways comprising these cycles have been revealed
A consistent finding in NMR studies of rodent and human brain metabolism using [1-13C] or [1,6-13C2]glucose as precursor is the lower fractional 13C enrichment of brain glutamate and glutamine at isotopic steady state compared with blood glucose,3–5 revealing the presence of constant ‘dilutional’ flows of unlabeled substrates into the respective tricarboxylic acid (TCA) cycles of neurons and astroglia. For glutamate, this dilution amounts to 25% to 30% in rodents5,6 (~14% in human brain
7
) of the theoretical maximum expected from the 13C-labeled glucose, and is thought to arise within neurons mainly from metabolism of blood-borne substrates such as lactate or ketone bodies and within the brain from glutamine produced in astrocytes through the glutamate–glutamine cycle.8–10 In addition to the glutamate-C4 dilution (relative to glucose-C1), 13C enrichment of glutamine-C4 is further ~20% to 30% less than that of glutamate-C4 at isotopic steady state.4,5,11–13 As the majority of glutamine-C4 labeling in these experiments arises from neuronal glutamate via the glutamate–glutamine cycle, the lower 13C enrichment of glutamine-C4 (i.e., Gln-C4fe/Glu-C4fe < 1) indicates non-neuronal inflow of unlabeled carbon8–10 atoms into glutamine. This inflow of unlabeled carbon could arise from blood-to-brain transport of unlabeled glutamine, or alternatively, astroglial oxidation of unlabeled substrates such as amino acids or short/medium-chain free fatty acids at the level of acetyl-CoA or
In this study, we infused [U-13C5]glutamine into mice and measured the 13C turnover of cortical glutamine and glutamate, while fitting a metabolic model modified to include glutamine transport between blood and brain. Our findings address quantitatively the contribution of glutamine influx from blood to the maintenance of brain glutamine levels and its role in the astroglial glutamine dilution.
MATERIALS AND METHODS
All the experimental procedures with animals were approved by the Institutional Animals Ethics Committee of Centre for Cellular and Molecular Biology (CCMB), Hyderabad, India and were conducted in accordance with the guidelines established by Committee for the Purpose of Control and Supervision on Experiments on Animals, Ministry of Environment and Forests, Government of India. ARRIVE guidelines were followed in the preparation of the manuscript. Male C57BL6 mice (2 months old) were maintained at 12 hour/12 hour light/dark cycle at the CCMB Animal House with temperature 22 ± 1°C and 60 ± 5% relative humidity, and received standard chow and water
Animal Preparation and Infusion of [U-13C5]Glutamine
Mice were anesthetized with urethane (1.5 g/kg intraperitoneally) and the lateral tail vein catheterized for infusion of 13C-labeled glutamine. The body temperature was maintained ~37°C with a heating pad warmed by a temperature-regulated recirculating water bath, and the respiration rate was monitored using a Biopac data acquisition system (Biopac Systems, Inc., Santa Barbara, CA, USA). Forty-five minutes after induction of anesthesia, [U-13C5]glutamine (250 mmol/L dissolved in deionized water, pH ~7.0) was administered intravenously for 3, 15, 30, or 60 minutes, using a bolus-variable rate infusion (1,220
Extraction of Metabolites from Brain Tissue
The brain was dissected in a cryo-chamber maintained at −20°C to isolate the cerebral cortex tissue. Metabolites were extracted from the frozen tissue as described previously.
20
Briefly, weighed tissue was powdered with ice-cold 0.1 N HCl/Methanol, and [2-13C]glycine was added as an internal concentration reference. Tissue was homogenized in 60% ethanol and centrifuged at 14,000
Analysis of Plasma Glutamine
The plasma was mixed with D2O containing sodium formate (0.5 mmol/L) and passed through a centrifugal filter (10 kDa cutoff) to remove macromolecules. A 1H-[13C]-NMR spectrum of filtered plasma was acquired at 600 MHz NMR spectrometer to measure the concentration and 13C enrichment of glutamine (Bruker AVANCE II, Karlsruhe, Germany). 3 The concentration of glutamine was determined from the resonance intensity at 2.46 p.p.m. in the non-edited spectrum, relative to the formate resonance at 8.45 p.p.m. The percent enrichment of glutamine-C4 was quantified from the intensity ratio of the 2.46-p.p.m. resonances of the difference spectrum (13C only) and nonedited spectrum (12C + 13C), respectively.
Nuclear Magnetic Resonance Spectroscopy of Cortical Extracts
1H-[13C]-NMR spectroscopy of cortical tissue extracts was performed at 600 MHz NMR spectrometer using a triple resonance probe. All the pulses were replaced with adiabatic half or full passage as described earlier by de Graaf
Description of the Metabolic Model and Determination of Metabolic Rates
The time courses of 13C labeling of cortical amino acids were fitted with a metabolic model that incorporates glutamine influx and efflux between blood and brain (Figure 1; Supplementary Table S1). The metabolic model consists of three compartments (blood, astrocytes, and neurons) allowing for transport and metabolism of glutamine (13C-labeled) and glucose (unlabeled) in the two neural cell types. In this model, [U-13C5]glutamine enters the brain, mixes with the endogenous (unlabeled) glutamine, which can then provide the precursors for neurotransmitter glutamate synthesis in neurons through glutamate–glutamine cycling. Glutamate released during neuronal activity is taken up by astrocytes and converted into glutamine, by the enzymatic action of glutamine synthetase, completing the cycle. Although glutamine is isotopically labeled with 13C in all five carbon atoms, only carbon 4 is depicted for glutamine and glutamate. The continuous metabolism of unlabeled glucose in the TCA cycles of neurons and astrocytes will introduce unlabeled carbons (dilution) in glutamine and glutamate at the level of acetyl-CoA produced from pyruvate. The mass balance and isotope rate equations describing the model appear in Supplementary Information (Supplementary Table S1).
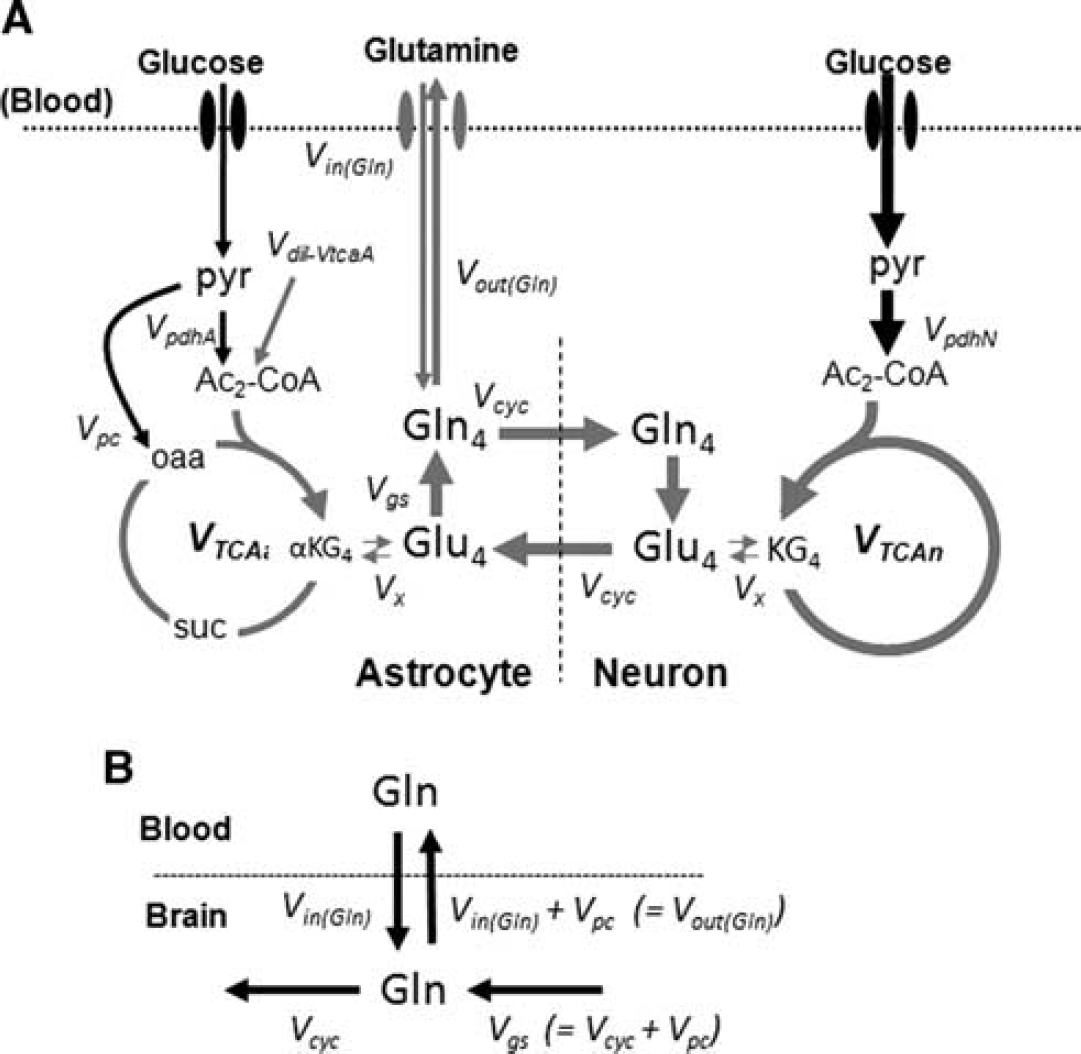
(A) Metabolic model of brain glucose and glutamine transport/metabolism. Glutamine is transported from blood-to-brain and brain-to-blood at rates
The driver input consisted of the time courses of blood plasma glutamine concentrations and 13C percentage enrichments. The target data to be fitted consisted of the time courses of cortical Glu-C4, C3 and Gln-C4 13C enrichments (Figure 1A) for the estimation of glutamine inflow
Statistics
Results consisting of concentrations and 13C enrichments are reported as group averages ± standard deviations (SDs). The uncertainties in the model parameters,
RESULTS
Concentration and 13C Enrichment of Plasma Glutamine
The baseline blood plasma glutamine concentration was 0.59 ± 0.08 mmol/L, as measured in a separate group of noninfused mice (
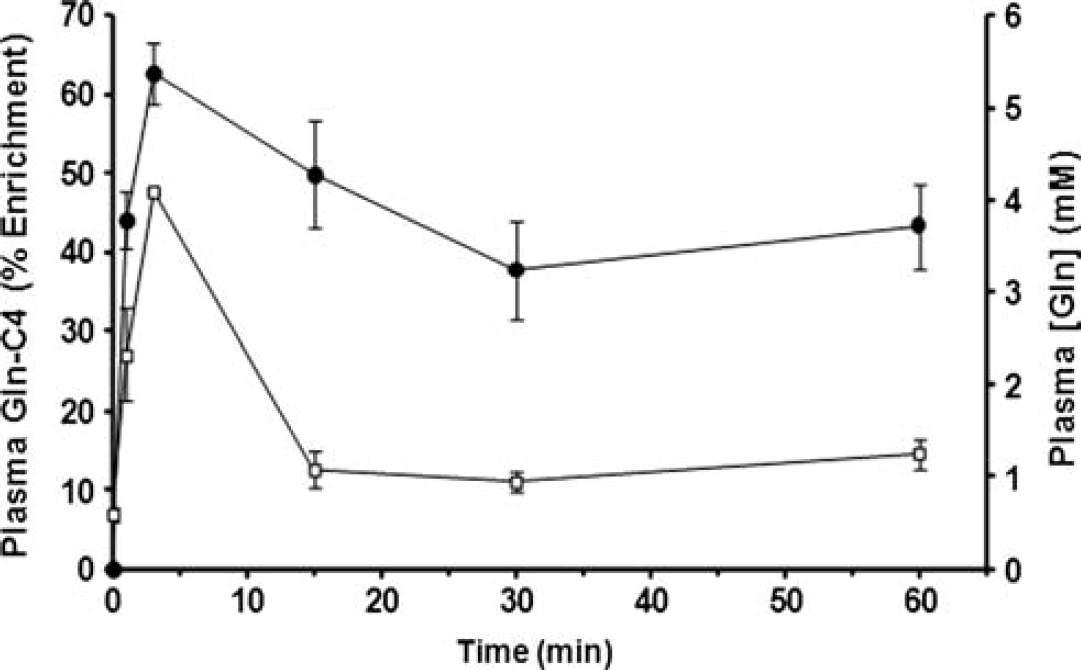
Plasma glutamine concentration and 13C percentage enrichment measured at different times during the intravenous infusion of [U-13C5]glutamine. Filled symbols, Gln-C4 enrichment (%), left vertical axis; open symbols, glutamine concentration in mmol/L, right vertical axis. These data were used as the plasma glutamine driver in the fitting of the time courses. Data are plotted as mean ± SD.
Concentrations (mmol/L) and 13C enrichments (%) of plasma and brain amino acids during intravenous [U-13C5]glutamine infusion
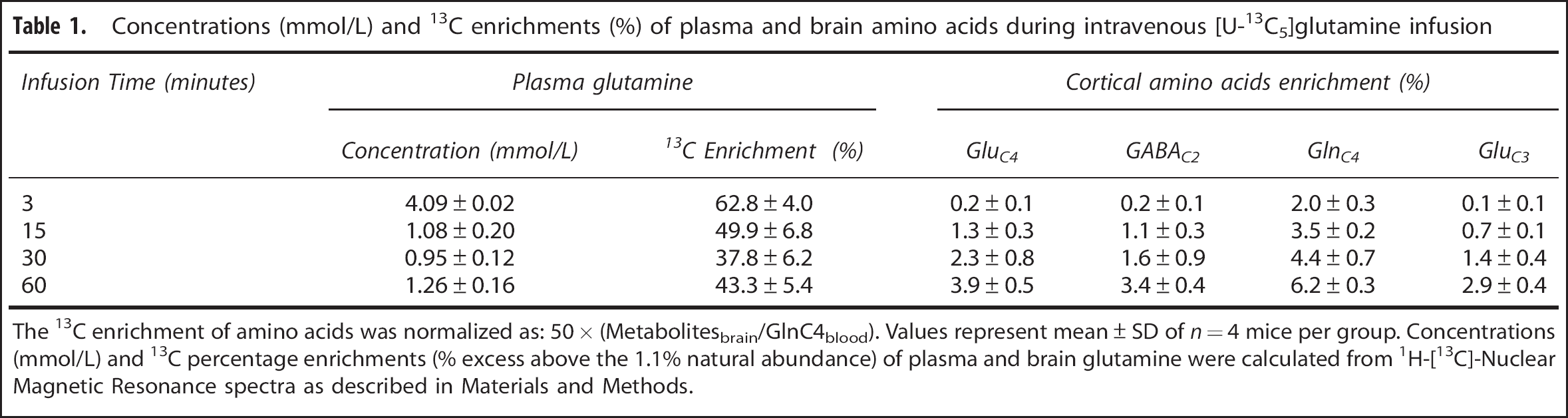
The 13C enrichment of amino acids was normalized as: 50 × (Metabolitesbrain/GlnC4blood). Values represent mean ± SD of
Steady-state 13C enrichment of cortical amino acids for different concentrations of plasma glutamine during [U-13C5]glutamine infusion
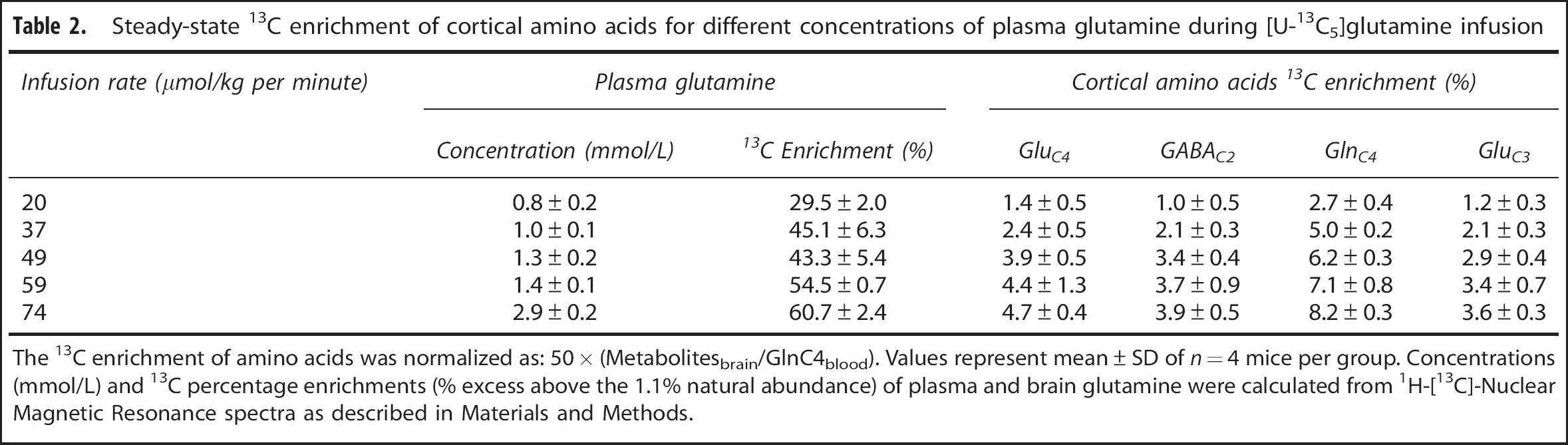
The 13C enrichment of amino acids was normalized as: 50 × (Metabolitesbrain/GlnC4blood). Values represent mean ± SD of
Effect of Plasma Glutamine Elevation on Concentrations of Cortical Metabolites
The concentrations of cortical metabolites were measured in the extract from the 1H-[13C+ 12C]-NMR spectrum (Figure 3A). The time course of metabolite data revealed that compared with baseline (~ 5.6 ± 0.4
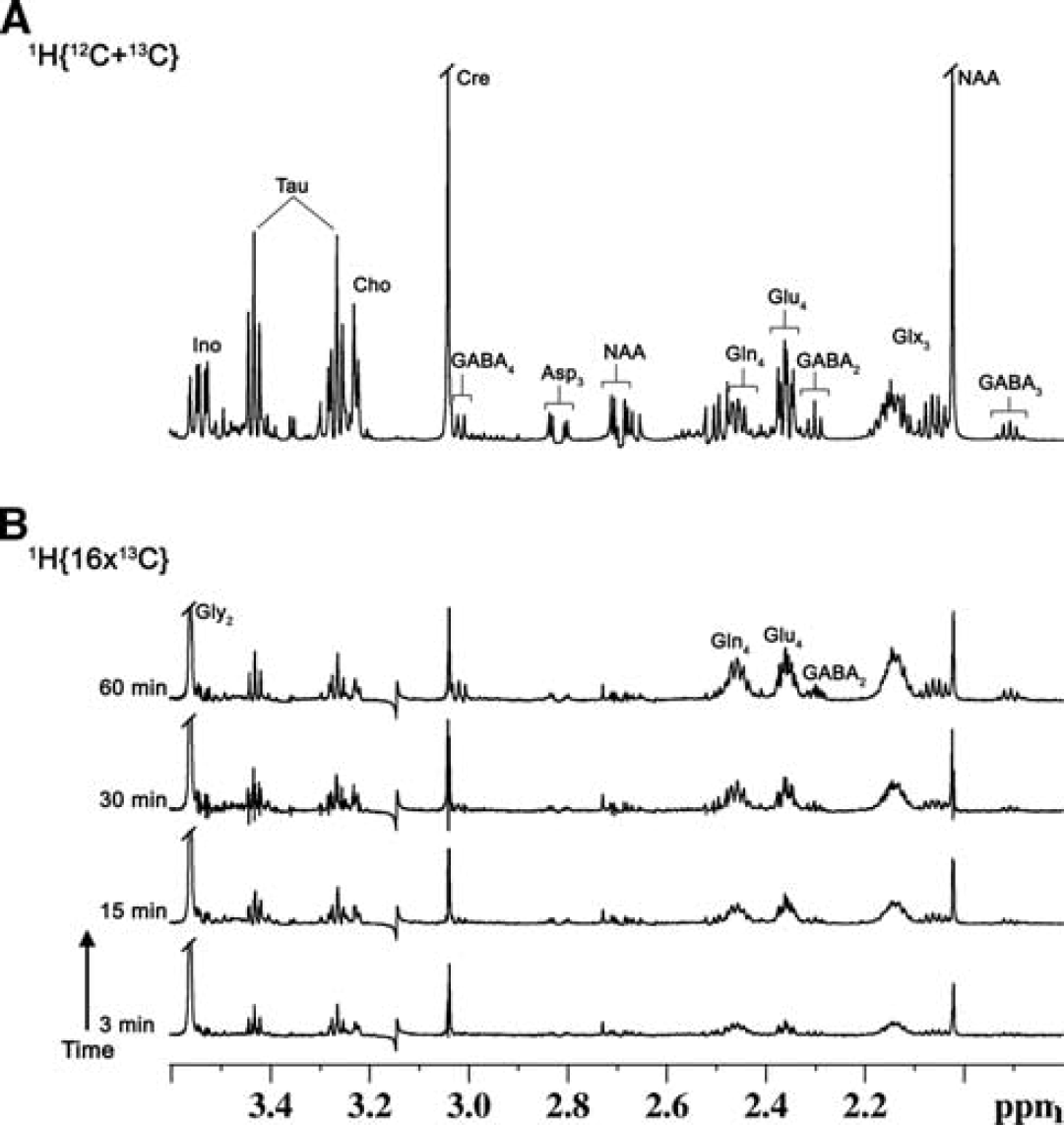
Representative 1H-[13C]-Nuclear Magnetic Resonance (NMR) spectra of cortical tissue extracts of mouse cerebral cortex after timed infusions of [U-13C5]glutamine. (
Effect of Plasma Glutamine Elevations on the Time Course of Brain Metabolite Enrichments
The infusion of [U-13C5]glutamine led to a significant 13C enrichment of brain amino acids, as shown in 1H-[13C]-NMR difference spectra of cortical extracts measured
The time courses of 13C enrichment of cortical glutamine-C4 and glutamate-C4/C3 are shown in Figure 4A. Glutamine-C4 13C enrichment rose within 3 minutes to ~2% (percentage enrichment normalized to blood enrichment) followed by a more gradual rise to ~6% at 60 minutes. Glutamate-C4 13C enrichment lagged glutamine-C4 at all infusion time points reaching to ~4% at 60 minutes, consistent with blood glutamine uptake in astroglia and metabolism to glutamate in neurons. Glutamate-C3 enrichment rose slower than glutamate-C4 (Figure 4A), despite the use of uniformly labeled glutamine precursor, suggesting an initial processing of glutamine (glutamate) destined for neurotransmitter glutamate synthesis in the neuronal TCA cycle where dilution with glucose-derived acetyl-CoA occurs.
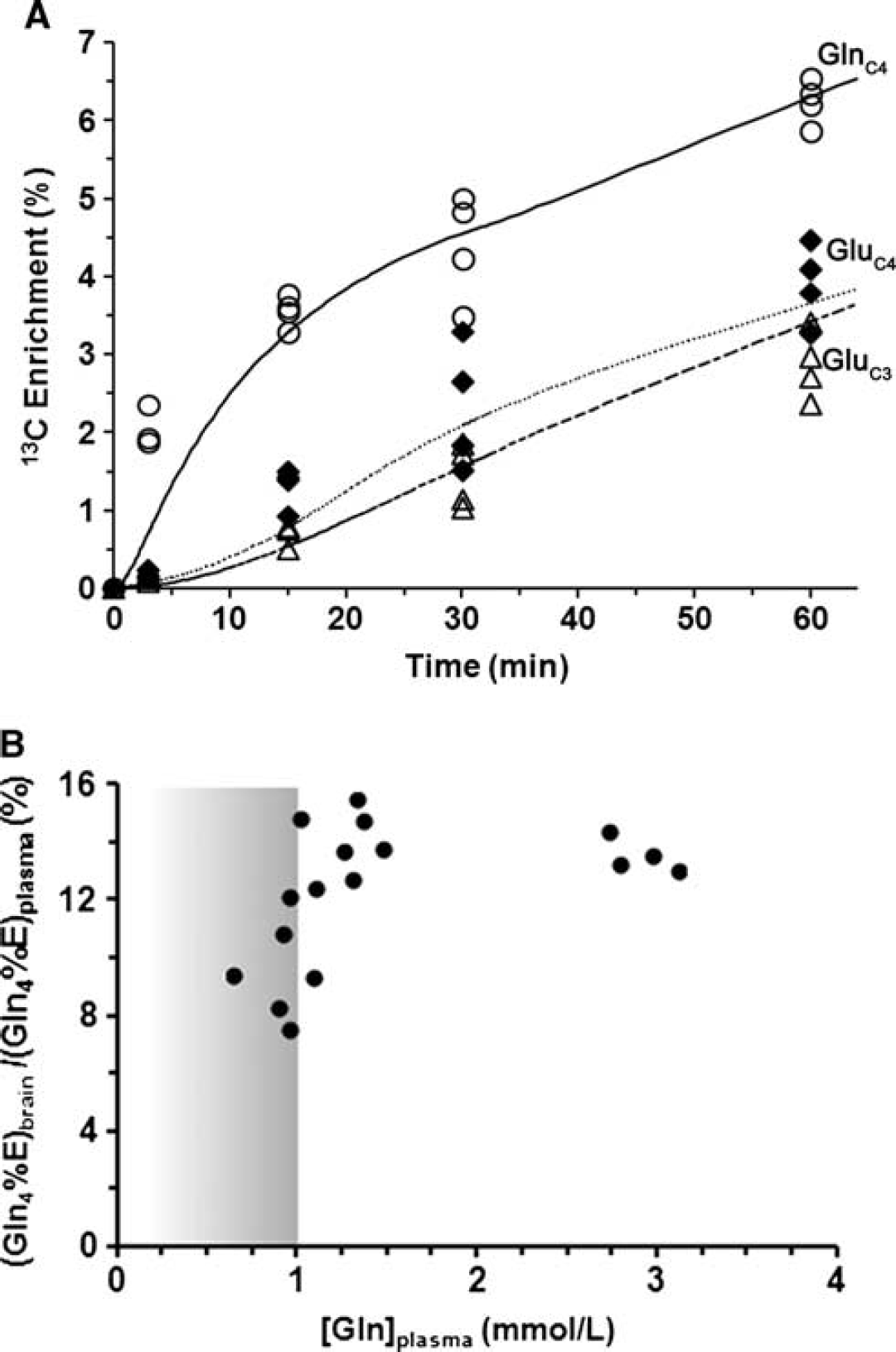
(A) Time courses of 13C labeling of cortical glutamine-C4 (***◯) and glutamate-C4 (♦), C3 (Δ) measured
In another experiment, 13C labeling of brain glutamine was measured at 60 minutes (steady state) for different plasma glutamine levels achieved by adjustment of the [U-13C5]glutamine infusion rate. A plot of the normalized steady-state brain glutamine-C4 13C enrichment against increasing plasma glutamine concentration revealed a steep dependence between 0 and ~1 mmol/L followed by the saturation of glutamine enrichment at ~11.8% for plasma glutamine concentration ≥ 1.0 mmol/L (Figure 4B). Extrapolating the plasma glutamine concentration proportionally downward to the baseline (physiologically normal) value of 0.59 mmol/L, we estimate that brain glutamine-C4 enrichment would be ~7% (11.8% × 0.59 mmol/L/1.0 mmol/L). Considering that the ‘dilution’ of glutamine-C4 enrichment relative to glutamate-C4 at isotopic steady state during infusion of [1-13C] or [1,6-13C2]glucose amounts to ~30% (see Discussion), for plasma glutamine at physiologic concentration, glutamine influx from blood then could account for ~20% of this dilution. This is likely an upper limit considering that glutamine in plasma would compete with other nonpolar amino acids for transport into the brain. 23
Blood-to-Brain Glutamine Influx
The rate of glutamine influx from blood,
The value of
DISCUSSION
In this study, we have shown that blood-borne labeled glutamine enters into brain in a concentration and time-dependent manner, reaching saturation for plasma glutamine concentration above ~ 1.0 mmol/L Cortical glutamate and GABA were labeled from plasma [U-13C5]glutamine in the order (glutamine-C4 > glutamate-C4 > GABA-C2), consistent with glutamine flow from astrocytes to neurons in the glutamate/GABA cycle. Further, glutamine influx from blood at physiologic blood glutamine level can account at most for ~ 20% of the previously observed steady-state isotopic dilution of glutamine-C4 (relative to glutamate-C4) from [1-13C]glucose.
Comparison of Glutamine Influx with Previously Reported Values
The rate of glutamine influx (
Assuming that
Compared with the
Substrates Contributing to the Glutamine Dilution at Isotopic Steady State
Our findings indicate that blood-borne glutamine at physiologic levels provides at most 15% to 20% of the glutamine-C4 dilution observed using [1-13C]glucose as precursor. Because glutamine is synthesized in astrocytes and 70% to 80% of glutamine is derived directly from glutamate released by neurons, the remaining dilution of glutamine-C4 relative to glutamate-C4 must arise in astrocytes from unlabeled sources. Other potential sources of glutamine-C4 dilution could include influx of unlabeled amino acids arising from blood or the breakdown of endogenous proteins, which enter the TCA cycle at the level of acetyl-CoA or
In addition to the above substrates, a number of non-glucose substrates that can be metabolized to acetyl-CoA are supplied from blood, as well as acetyl groups arising through turnover of endogenous membrane lipids and proteins. A significant amount of free acetate is present in mouse blood (~0.2 to 0.9 mmol/L),
32
which can be readily oxidized in brain, mainly by astrocytes.
33
In the cerebral cortex of anesthetized rats, blood acetate is oxidized at a rate of 0.04
In the metabolic model used to evaluate the transport properties, glutamine efflux (
Limitations of the Study and Other Observations
There are some limitations in the present study that impact interpretation. First, glutamine transport between blood and brain involves several transporter proteins, all of which belong to one or more families of large neutral amino-acid transporters.26,38–40 Glutamine transport into brain and cerebrospinal fluid from the blood involves both System N, A and L-like transporters, and within brain parenchyma, glutamine transport into neurons and astrocytes involves mainly systems-A and N transporter proteins, respectively. Because the present study involved bulk cortical tissue measurements of glutamine and glutamate, and consequently did not differentiate between the different neural compartments, the measured value of
Finally, the current study was conducted in mice anesthetized with urethane, which produces a long-lasting steady level of anesthesia. Metabolic analysis performed under urethane and other anesthetics using [1,6-13C2]glucose shows similar differences in the labeling of glutamate-C4 and glutamine-C4 when accounting for depth of anesthesia (neural activity). Because different anesthetics do not appear to influence the difference in glutamate and glutamine-C4 13C labeling at steady state, it may be argued that anesthesia has little impact on the transport of glutamine from blood to brain, although further studies are needed to address this issue. The labeling of astroglial glutamine from [2-13C]acetate
5
or [2-13C]glucose
17
has been used to estimate the neurotransmitter cycling-to-TCA cycle flux ratio of glutamatergic and GABAergic neurons,
CONCLUSIONS
13C-Labeled glutamine is readily transported from the blood into the brain, first enriching brain glutamine and then glutamate and GABA, consistent with neuron–astrocyte neurotransmitter cycling. Glutamine influx showed saturation above blood concentrations of 1.0 mmol/L, reaching 14% enrichment with a unidirectional rate of glutamine influx of 0.036
Footnotes
The authors declare no conflict of interest.
ACKNOWLEDGMENTS
The authors would like to thank Dr Robin A de Graaf, Yale University for providing the 1H-[13C]-NMR sequence, Mr. Bhargidhar Babu for his assistance in conducting the animal studies. All NMR experiments were performed at the NMR Facility, CCMB, Hyderabad, India.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
