Abstract
Caloric restriction (CR) prolongs lifespan and retards many detrimental effects of aging, but its effect on brain mitochondrial function and neuronal activity—especially in healthy aging—remains unexplored. Here we measured rates of neuronal glucose oxidation and glutamate–glutamine neurotransmitter cycling in young control, old control (i.e., healthy aging), and old CR rats using
INTRODUCTION
Mitochondrial oxidative phosphorylation of glucose is the predominant mode of energy generation (adenosine triphosphate (ATP) production) in mammalian species. The mammalian brain has the highest energy demands of any organ based on its size, 1 and majority of this energy is used to support neuronal activity and functional processes. 2 Mitochondrial function declines with age in the brain and has been proposed to be a major factor in the loss of brain function with aging. The metabolic decline is even more rapid and profound in neurodegenerative disorders, such as Alzheimer's disease. 3 As a result, preserving brain mitochondrial integrity and metabolism with age could be critical for maintaining healthy brain function, often referred to as healthspan, and for extending lifespan. 4
Caloric restriction (CR) without malnutrition is one of several other interventions that have been introduced to preserve metabolism in aging process, and moreover CR has been shown to increase the lifespan of a broad range of species.5,6 Several studies suggest that CR-induced increase in lifespan arises because of increased capacity for oxidative phosphorylation from elevated mitochondrial respiration.
7
Although CR effects on isolated mitochondria have been extensively studied, we posited that the effect of CR on brain mitochondrial function could be because of altered neuroenergetics. In this study, we determined whether CR could mitigate the declines of these measures in aging brain. We measured fluxes of neuronal tricarboxylic acid (TCA) cycle (index of mitochondrial function) and glutamate–glutamine neurotransmitter cycling (index of neuronal activity) using nuclear magnetic resonance (NMR) spectroscopy
MATERIALS AND METHODS
Animal
Experiments were conducted with male Fischer 344 Brown-Norway F1 (F344BNF1) rats, which have shown to extend longevity under CR.
8
Rats were obtained from a CR colony at National Institute on Aging. At the CR colony of National Institute on Aging, all rats were fed
Age, gender, diet, body weight, blood glucose, and ATP production of the rats
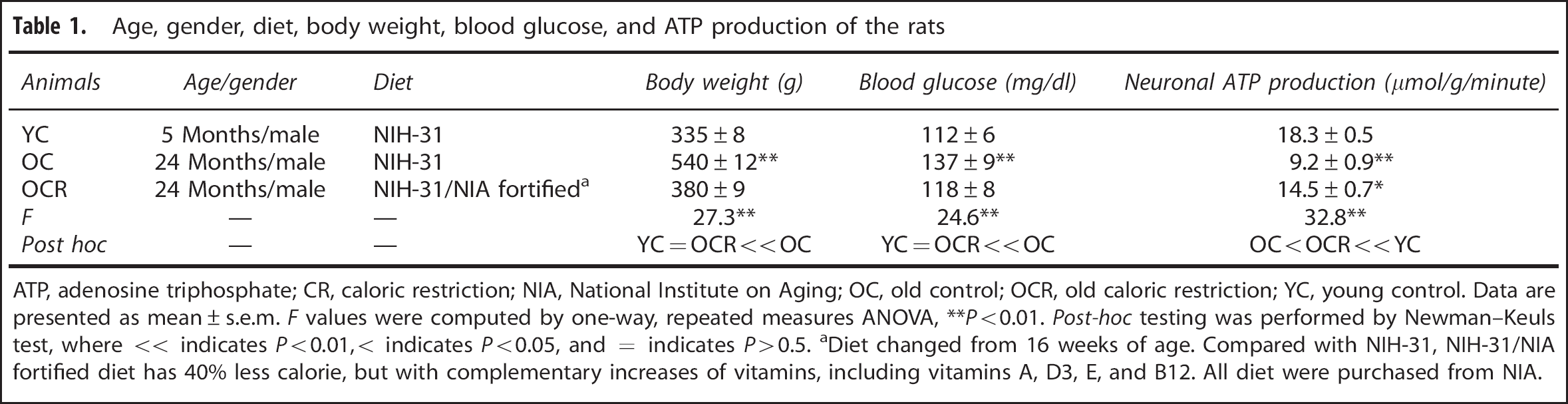
ATP, adenosine triphosphate; CR, caloric restriction; NIA, National Institute on Aging; OC, old control; OCR, old caloric restriction; YC, young control. Data are presented as mean ± s.e.m.
Diet changed from 16 weeks of age. Compared with NIH-31, NIH-31/NIA fortified diet has 40% less calorie, but with complementary increases of vitamins, including vitamins A, D3, E, and B12. All diet were purchased from NIA.
Animal Preparation
All rats were fasted for 12 hours before scanning to decrease the plasma glucose level (to ˜5 to 6 mmol/L) such that upon infusion of 13C-labeled glucose the plasma level was approximately doubled (to ˜10 to 12 mmol/L) and hence the 13C-enrichment was ˜50% with [1,6-13C]-D-glucose. The animals were initially anesthetized with isoflurane (1% to 2%), tracheotomized, and ventilated (30% oxygen; ˜68% nitrous oxide). After surgery anesthesia was maintained with
in vivo NMR Spectroscopy
We used
(
During the scan, each rat received 1.5 mL of 0.75 mol/L [1,6-13C]-D-glucose infusion. The determination of glutamate and glutamine turnover required rapid attainment of a high and steady fractional enrichment of [1,6-13C]glucose in the blood throughout the time period of the experiment. The infusion protocol was optimized by adapting the 2-deoxy-D-[14C]-glucose method described by Patlak and Pettigrew
11
to accommodate the large amount of [1,6-13C]-glucose infused during the experiment. The infusion rate, which was modified manually every 30 seconds, followed a decreasing exponential function during the first 8 minutes and was constant for the remaining period.
10
Total blood glucose concentration was measured from arterial blood samples (50 to 100
Preparation of Blood Plasma and Brain Extracts
Plasma samples were prepared for further POCE spectral analysis by adding 200
Ethanol extracts were prepared from the frozen frontoparietal cortex (100 to 150 mg wet weight) using the procedure described by Patel
Metabolic Modeling
The time courses of brain amino acid 13C enrichments of Glu-C4, Glu-C3, Gln-C4, and the steady-state labeling of Gln-C3 (from extracts) were fitted to a two-compartment (neuron–astrocyte) metabolic model 13 using the plasma time courses of 13C-enriched glucose as the input. The goal of the metabolic modeling was to derive the rates of the neuronal TCA cycle (VTCA,N) and the glutamate–glutamine neurotransmitter cycling (Vcycle). The relationship between 13C enrichments of plasma glucose and brain metabolites was described by coupled differential equations with restrictions of mass and isotope balance, using CWave 3.0 software (GF Mason, New Haven, CT, USA). The differential equations were solved in MATLAB 7.1 (Natick, MA, USA) using a first-order Runge-Kutta algorithm and fitting optimization was achieved with the Levenberg–Marquardt algorithm. 13 In this model, glutamate was assigned 10% to glia and 90% to neurons, whereas glutamine was assigned 100% to glia, the astrocytic TCA cycle flux (VTCA,A) was set to 15% of total TCA cycle flux, the anaplerotic flux through pyruvate carboxylase (VPC) was set to 20% of the rate of glutamine synthesis (Vgln), 14 and rates of the neuronal TCA cycle (VTCA,N) and glutamate–glutamine cycling (Vcycle) were iterated using CWave for optimal fits of the model to the data. 13 The ATP production rate in neurons (VATP,N) was calculated assuming the steady-state oxygen-to-glucose index of 5.515,16 and that 34 ATP is generated per mole of glucose oxidation, 2
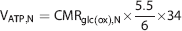
where CMRglc(ox),N is the rate of neuronal glucose oxidation and is the hexose unit representation of the neuronal TCA cycle flux. Because one glucose molecule is converted to two pyruvate molecules to generate two acetyl-CoA moieties to enter TCA cycle, CMRglc(ox),N is given by ½VTCAN.
Statistics
Data are presented as mean ± s.e.m. One-way, repeated measures analysis of variance with
RESULTS
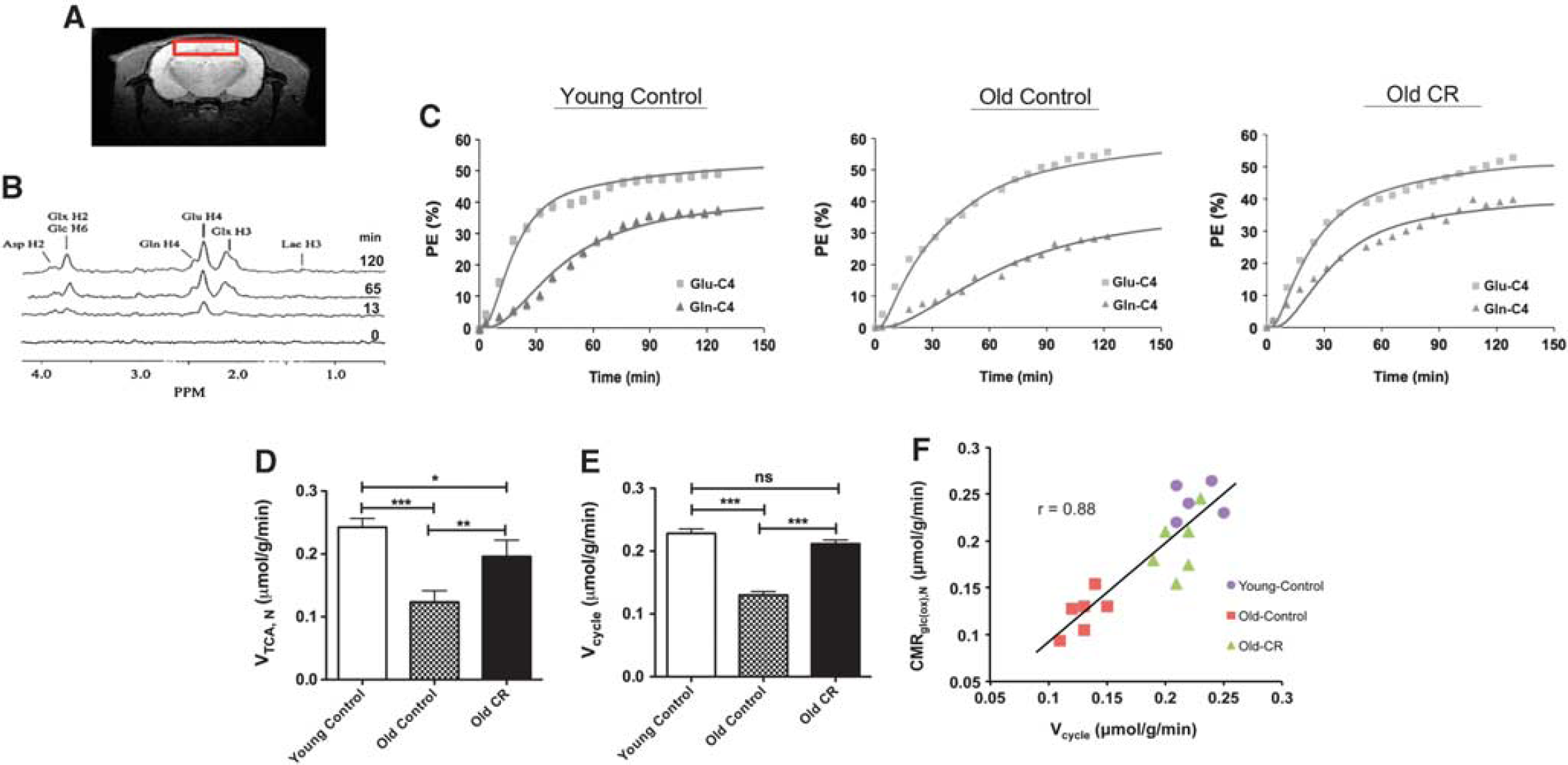
Figure 1A shows the cortical region from which we obtained the POCE measurements and Figure 1B shows an example from spectral time course of 13C labeling over the 2-hour infusion (only spectra from four time points are shown here for presentation). It is observed that 13C labeling of Glu-C4 appears faster than that of Gln-C4, indicating 13C-glucose was mainly oxidized in neuronal compartment, and Gln-C4 was labeled after Glu-C4 via the glutamate–glutamine neurotransmitter cycling. Figure 1C depicts time courses of cortical 13C enrichments of Glu-C4 and Gln-C4 for a typical young control, old control, and old CR rats during [1,6-13C]-glucose infusion. The solid lines reflect the best fits of the constrained two-compartment metabolic model to the
Previously, it has been found that, when VTCA,N is plotted in hexose units (i.e., CMRglc(ox),N) against Vcycle, there is a close to 1:1 relationship between increments in glucose oxidation over an isoelectric baseline state and increments in glutamate–glutamine cycling. In the metabolic model, CMRglc(ox),N was expressed as ½VTCA,N because one glucose molecule makes to two pyruvate molecules and which in turn generates two acetyl-CoA molecules, etc. This result has been interpreted as indicating a high energetic cost for neuronal function that is approximately constant throughout the normal activity range, especially in the awake state.16,17 Figure 1F shows a plot of CMRglc(ox),N versus Vcycle for each of the rats in all three groups, where the slope of the line is 1.01, showing that the best fit is highly consistent with the previous findings.16,17
DISCUSSION
We showed that CR impeded the age-related decline of brain mitochondrial respiratory function—old CR rats had comparable VTCA,N, CMRglc(ox),N, and ATP production rate relative to the young control rats. Although we were not able to determine whether this is because of mitochondrial biogenesis using POCE techniques in the study, our finding is consistent with the literature that long-term CR preserves mitochondrial function and induces bioenergetic efficiency (in isolated mitochondria), possibly because of decreased oxidant emission, increased antioxidant scavenging, and/or minimized oxidative damage to DNA and protein.7,19
We also showed that CR impeded the age-related decline of neuronal activity—old CR rats had similar Vcycle compared with the young control rats. This is consistent with prior observations that metabolic and neuronal functions were highly associated;3,4 that metabolic reserve was a determinant of cognitive aging. 20 Metabolic reserve has been proposed as the ability of neuronal circuits to respond adaptively to perturbations in energy metabolism because of aging and disease processes, thereby maintaining their ability to support neuronal circuits and preventing declines in cognition. In line with this, old rats treated with CR (same strain used in the current study) have been previously shown to have enhanced memory compared with the age-matched controls. 21 Moreover, rodents with CR had lower incidence of Alzheimer's disease phenotypes. 22 Our findings suggest that CR is beneficial to aging brain by preserving metabolic and thus neuronal functions.
In addition to brain function, we showed that CR preserved other physiologic functions, including body weight and blood glucose level. The dramatic changes of these measures in normal aging rats may imply a developing pre-diabetic symptom. Collectively, these observations may provide a rationale for CR-induced extended healthspan and lifespan.
It has to be pointed out that CR-induced extended longevity may not be universal. Recent studies have showed that the lifespan response to a single level of CR (e.g., 40% CR) exhibits wide variation in mice with different genetic backgrounds. 23 They also showed that there are cases where CR can shorten lifespan in inbred mice. However, brain metabolism and function was not examined in these studies. It will be important for future studies to identify whether CR has adverse effects on brain metabolism and functions during aging in rodent strains where deleterious effects on lifespan are observed.
In summary, we used NMR to show that during aging CR preserves mitochondrial energy production, energy demand, and neuronal activity with a long-lived rodent model. These results provide a rationale for CR-induced sustenance of brain health with extended lifespan. Understanding of nutritional effects on brain function may have profound implications in human aging and other age-related neurodegenerative disorders.
Footnotes
The authors declare no conflict of interest.
ACKNOWLEDGMENTS
The authors would like to thank Dr Graeme Mason and Dr Robin de Graaf of Yale University for their valuable comments and help with POCE experimental design and metabolic modeling.
