Abstract
After cerebral ischemia or trauma, secondary neurodegeneration may occur in brain regions remote from the lesion. Little is known about the capacity of cerebral gliomas to induce secondary neurodegeneration. A previous study showed that cis-4-[18F]fluoro-D-proline (D-cis-[18F]FPro) detects secondary reactions of thalamic nuclei after cortical infarction with high sensitivity. Here we investigated the potential of D-cis-[18F]FPro to detect neuronal reactions in remote brain areas in the F98 rat glioma model using ex vivo autoradiography. Although the tumor tissue of F98 gliomas showed no significant D-cis-[18F]FPro uptake, we observed prominent tracer uptake in 7 of 10 animals in the nuclei of the ipsilateral thalamus, which varied with the specific connectivity with the cortical areas affected by the tumor. In addition, strong D-cis-[18F]FPro accumulation was noted in the hippocampal area CA1 in two animals with ipsilateral F98 gliomas involving hippocampal subarea CA3 rostral to that area. Furthermore, focal D-cis-[18F]FPro uptake was present in the necrotic center of the tumors. Cis-4-[18F]fluoro-D-proline uptake was accompanied by microglial activation in the thalamus, in the hippocampus, and in the necrotic center of the tumors. The data suggest that brain tumors induce secondary neuronal reactions in remote brain areas, which may be detected by positron emission tomography (PET) using D-cis-[18F]FPro.
INTRODUCTION
After cerebral injury in cortical areas, such as ischemia or trauma, secondary neurodegeneration may occur in brain regions remote from the site of the lesion.1,2 This phenomenon occurs mainly in the thalamus as thalamic neurons project to target neurons in the neocortex and are characterized by a marked sensitivity to neocortical injury.1,2 Experimental studies suggest that thalamic atrophy results primarily from retrograde degeneration, because of injury of the thalamocortical pathway.3–5
The phenomenon of retrograde thalamic reaction is well known from classic neuroanatomical experiments, and has also been observed with modern imaging methods in vivo. After cerebral infarction in the territory of the middle cerebral artery, secondary degeneration in the ventral nuclei of the thalamus was shown by magnetic resonance imaging in patients as regions of slightly lower signal on proton density or T2-weighted images, starting at approximately 6 weeks after stroke.6,7 Using the receptor ligand [ 3 H]PK11195, autoradiographic studies in rats after focal cortical infarctions and positron emission tomography (PET) studies using [ 11 C]PK11195 in humans with ischemic stroke revealed increased tracer uptake, indicating secondary reaction of thalamic nuclei.8,9 PK11195 binds to peripheral-type benzodiazepine receptors and its accumulation is associated with microglial activation. 10
In a previous autoradiographic study in rats, the amino-acid tracer cis-4-[ 18 F]fluoro-D-proline (D-cis-[ 18 F]FPro) was able to indicate secondary thalamic reaction already 3 days after cortical infarction and with a considerably higher lesion to background contrast than [ 3 H]PK11195. 11 The uptake mechanism of D-cis-[ 18 F]FPro in brain areas with secondary reaction is not yet clearly understood, but it was shown that the D-isomer of cis-4-[ 18 F]fluoro-proline is transported preferably across the blood–brain barrier (BBB) compared with its L-isomer. 12 Furthermore, no accumulation of D-cis-[ 18 F]FPro was observed in a large oligodendroglioma (WHO grade II), which was positive on a corresponding PET scan using O-(2-[ 18 F]fluoroethyl)-L-tyrosine. 12 Although the remote secondary thalamic reaction after primary ischemic or traumatic cortical damage is a well-known phenomenon, there are only very few reports on secondary reactions in those remote brain areas caused by brain tumors.13,14
This study was undertaken to investigate whether D-cis-[ 18 F]FPro is able to show secondary reactions in remote brain areas induced by brain tumors and to further explore the uptake behavior of this tracer in neoplastic lesions. [6- 3 H]Thymidine ([ 3 H]thymidine) was used as a reference, which allows a direct comparison of D-cis-[ 18 F]FPro uptake in relation with cell proliferation in the tumors. Different histological stainings were performed to identify anatomically the remote brain areas with focally increased D-cis-[ 18 F]FPro accumulation or areas directly affected by the tumor. In addition, corresponding parallel sections were analyzed by immunofluorescent imaging to identify specific cell types possibly involved in the process of D-cis-[ 18 F]FPro uptake and neurodegeneration.
MATERIALS AND METHODS
Animal Experiments
Ten male Fischer 344 rats (220 to 370 g; Charles River Wiga Deutschland GmbH, Sulzfeld, Germany) were implanted with syngeneic intracerebral F98 tumors. F98 tumors have been described histologically as anaplastic or undifferentiated glioma and exhibit an invasive growth pattern and a weak immunogenic response. Usually, large tumors exhibit central necrotic core areas. The characteristics of this cell line closely simulate those of human glioblastoma. 15
All animal experiments were performed according to the German Law on the Protection of Animals (LANUV NRW Recklinghausen, Germany, no. 8.87-50.10.35.08.229) and the Animal Research: Reporting In Vivo Experiments (ARRIVE) guidelines. The rats were housed under standard conditions with free access to food and water. For stereotaxic tumor implantation, animals were sedated in a 2% to 5% atmosphere of isoflurane and anesthetized subsequently with an intraperitoneal injection of a mixture of ketamine (100 mg/kg bodyweight) and xylazine (10 mg/kg bodyweight). The head was fixed in a stereotactic frame and the skin of the skull was incised to expose the cranial bone. According to a stereotaxic brain atlas, 16 a hole was drilled into the skull with a diameter of 0.9 mm using a microdrill. The F98 rat glioma cells (30,000 to 100,000 cells in 5 to 10 μL) were injected intracerebrally inserting a microliter syringe into the brain at various positions (caudate nucleus, motor, somatosensory, visual cortex, or hippocampus) (Table 1). The cells were injected slowly over a time period of 10 minutes. After injection, the syringe remained in the brain for 10 minutes to minimize the reflux of cells. After removal the incision was sutured.
Summary of data on animals with F98 gliomas
alv, alveus of the hippocampus; B, basal nucleus; CA1, area CA1 of the hippocampus; CA3, area CA3 of the hippocampus; cc, corpus callosum; Cg, cingulate cortex; cg, cingulum; chp, choroid plexus; Cl, centrolateral thalamic nucleus; CPu, caudate putamen (striatum); cst, commissural stria terminalis; Cx, cortex;
Substructures with the exception of area CA3 are not mentioned separately; em-dash indicates that the corresponding brain area was not affected by the tumor or that no significant
Radiotracers
The synthesis of the radiotracer D-cis-[ 18 F]FPro was performed by aminopolyether-mediated no-carrier-added nucleophilic 18 F-fluorination starting from (2R,4S)-N-Boc-4-(p-toluolsulfonyloxy)proline methylester, as described previously. 11 [ 3 H]Thymidine was obtained commercially (Perkin-Elmer, Waltham, MA, USA) as an aqueous solution with a specific activity of 533 GBq/mmol and a concentration of 37 MBq/mL.
Dual Tracer Autoradiography
At 7 to 14 days after tumor implantation, rats were sedated and anesthetized as described above. The different timing of the animals being killed was because of methodological reasons, such as poor general condition of the animals or irregularities in tracer production. A mixture of the radiotracers D-cis-[ 18 F]FPro (70 MBq) and [ 3 H]thymidine (22 MBq) was injected intravenously into the tail vein. At 2 hours after tracer injection, the animals were killed, the brains were removed immediately, and frozen in 2-methylbutane at −50°C. Coronal sections (thickness 20 μm) were prepared using a cryomicrotome (CM 3050S; Leica Microsystems GmbH, Wetzlar, Germany). Every 10th section was placed on phosphor imaging plates (Raytest-Fuji, Straubenhardt, Germany) along with industrial 3 H-activity standards (Microscales; Amersham Biosciences, Little Chalfont, UK) and with in-house calibrated 18 F liver paste standards. The remaining parallel brain sections were stored at −80°C for histologic staining and immunostaining. The first exposition to depict distribution of the 18 F-radioactivity was started within 4 hours after tracer injection for 14 hours. This exposition was performed on imaging plates that were insensitive to β-particles of 3 H (BAS-SR 2025; Raytest-Fuji). After decay of 18 F (10 half-lives), the brain slices were exposed again to 3 H-sensitive imaging plates for 72 hours to obtain the 3 H distribution (BAS-TR 2025; Raytest-Fuji). On exposure, the imaging plates were scanned with a high-performance imaging plate reader (BAS 5000 BioImage Analyzer; Raytest-Fuji). Quantitative autoradiograms were generated (Bq/mg wet weight of tissue) using the software provided by the manufacturer (AIDA Version 4.50; Raytest-Fuji) and the known radioactivity concentrations of the standards. Tracer uptake was quantified by lesion-to-brain ratios (L:B) using a small circular region of interest (ROI) placed on the areas with high D-cis-[ 18 F]FPro accumulation, i.e., thalamic nuclei, hippocampus, the necrotic center of the tumor, and in proliferating parts of the tumor according to the [ 3 H]thymidine autoradiogram (range of ROI size, 0.1 to 3.7 mm2). A larger reference ROI was placed in the contralateral hemisphere in the normal grey matter (range of reference ROI size, 0.7 to 5.6 mm2). Lesion-to-brain ratios were calculated by dividing the average uptake in the ROI of the lesion by the average uptake in the ROI of the normal brain.
Histologic Staining and Double Immunofluorescence Labeling
Parallel sections of the autoradiographed brain sections were stained with 4′,6-diamidino-2-phenylindole dihydrochloride (DAPI; nuclear stain), cresyl violet (Nissl stain), or silver nitrate (myelin stain).
For nuclear staining with DAPI (Sigma-Aldrich Chemie GmbH, Taufkirchen, Germany), the rat brain sections were fixed in a 4% paraformaldehyde solution and washed with phosphate-buffered saline (PBS) for 10 minutes. The fixed sections were stained for 10 minutes with DAPI (1 μg/mL). Afterwards, the sections were washed 3 × 5 minutes in phosphate-buffered saline and coverslipped with a nonfluorescent aqueous mounting medium (Aqua-Poly/Mount, Warrington, PA, USA).
For Nissl staining, the rat brain sections were fixed for 30 minutes in a neutral-buffered formalin solution. Then, the sections were washed with demineralized water and stained in a cresyl violet solution for 30 minutes at 60°C. After staining, the sections were washed again and dehydrated in ascending alcohol solutions (70%, 96%, and 100% propanol) for 2 × 5 minutes each. Finally, the sections were cleared 2 × 5 minutes in HS200 (xylene substitute; National Diagnostics, Charlotte, NC, USA) and mounted in DPX (Sigma-Aldrich Chemie GmbH).
Myelin staining was performed by a modification of the Gallyas 17 technique. Briefly, rat brain sections were fixed overnight in a neutral-buffered formalin solution. Subsequently, sections were rinsed in demineralized water (20 to 30 minutes) and pretreated with a 2:1 pyridine/acetic anhydride solution for 60 minutes in the dark and rinsed again for 20 to 30 minutes. After washing the sections were incubated in an ammonium silver nitrate solution for 30 minutes in the dark and washed two times in 1% acetic acid for 5 minutes. The sections were transferred to a developer solution for 15 to 30 minutes and rinsed for 5 minutes. The development was terminated by incubating the sections in 1% acetic acid for 5 minutes. Subsequently, the brain sections were washed again for 20 to 30 minutes and fixed in a T-Max fixer solution (Kodak, Paris, France) for 2 minutes in the dark. The sections were rinsed for 5 minutes and dehydrated in ascending alcohol solutions (70%, 96%, and 100% propanol) for 2 × 5 minutes each. Finally, the sections were cleared 2 × 5 minutes in HS200 and mounted in DPX.
Double immunofluorescence labeling was performed to identify cells possibly involved in the process of tracer uptake. Reactive astrocytes were detected by staining for glial fibrillary acidic protein (GFAP) using rabbit anti-rat GFAP polyclonal antibodies (1:1000; Abcam, Cambridge, UK). Microglia was detected with mouse anti-rat CD11b monoclonal antibodies (1:50; AbD Serotec, Oxford, UK). As secondary fluorochrome-conjugated antibodies, goat-anti-rabbit Alexa Fluor 488 and goat-anti-mouse Alexa Fluor 568 (1:300; Invitrogen, Carlsbad, CA, USA) were used. In all slices, cell nuclei were counterstained with DAPI.
Anatomic Correlations
Brain areas affected by the tumor as shown by [ 3 H]thymidine uptake and distant areas with focally increased D-cis-[ 18 F]FPro accumulation were anatomically assigned by comparing the autoradiograms and the different histologic stainings with maps of a stereotaxic rat brain atlas, 16 as described previously. 11 In short, the localization of sections was identified on the basis of anatomic landmarks in the histologic sections (DAPI, cresyl violet, and myelin staining). Thereafter, an ROI circumference was drawn along the borders of the coronal brain slices in the anatomic map. After adapting the size of the corresponding autoradiogram to that ROI, additional ROIs were drawn around the site of abnormal tracer uptake in the tumor areas or in remote brain areas. The set of the two ROIs was reprojected on the stereotaxic map, which allowed an identification of the brain regions affected by the tumor, the thalamus, or an area in the hippocampus with increased D-cis-[ 18 F]FPro uptake. In addition, ROIs were drawn along the borders of the tumor according to cresyl violet stainings and projected on all relevant histologic sections and autoradiograms.
Statistical Analysis
Values are expressed as mean ± s.d. Statistical methods used were t-test or Mann–Whitney rank-sum test for group comparisons. Correlation analyses were performed with Pearson's correlation coefficient. A probability value of <0.05 was considered significant. Statistical analysis was performed using the SigmaPlot software (SigmaPlot Version 11.0; Systat Software, San Jose, CA, USA).
RESULTS
Tracer Uptake in F98 Gliomas
Large F98 tumors were present in all animals (mean diameter, 4.4 ± 1.7 mm). As expected, tumor growth was not strictly limited to the implantation site but varied depending on the individual spread of the tumor in individual animals. An overview of the sites of implantation and the corresponding affected brain areas are shown in Table 1.
In all animals, the proliferating solid tumor mass of the F98 gliomas exhibited strong accumulation of [ 3 H]thymidine but no relevant D-cis-[ 18 F]FPro uptake (L:B—28.2 ± 8.8 vs. 1.4 ± 0.3, P < 0.001; Table 2). In seven animals the histologic stainings as well as the [ 3 H]thymidine autoradiograms displayed parts of very low cell density in the center of the tumors, indicating the typical finding of a central necrosis. These necrotic core areas partially showed focal D-cis-[ 18 F]FPro uptake that was significantly higher than that of [ 3 H]thymidine (L:B—9.0 ± 3.0 vs. 4.5 ± 1.4, P < 0.01; Table 2). Cis-4-[ 18 F]fluoro-D-proline uptake in the necrotic center of the tumors was accompanied by CD11b-expressing cells, whereas only single GFAP-positive reactive astrocytes could be observed in those areas. CD11b-expressing cells in the periphery of the tumors, however, showed no D-cis-[ 18 F]FPro uptake, suggesting that D-cis-[ 18 F]FPro is not linked to activated microglia. A representative example is shown in Figure 1.
Quantified tracer uptake on animals with F98 glioma
CPu, caudate putamen (striatum);
Coronal sections of F98 tumor-bearing rat brain (rat no. 2). Red dashed lines indicate the position of the tumor. [
3
H]Thymidine autoradiography (
Cis-4-[ 18 F]Fluoro-D-proline Uptake in Remote Brain Areas
Prominent D-cis-[ 18 F]FPro uptake was noted separately from the solid tumor mass in 9 out of 10 animals in brain areas that showed no tumor tissue in the histologic stainings and no [ 3 H]thymidine uptake (L:B—6.5 ± 4.0 vs. 1.3 ± 0.5, P < 0.01; Table 2). Cis-4-[ 18 F]fluoro-D-proline accumulation was noted in the thalamus, subthalamic nucleus, hippocampal area CA1, subiculum, caudate putamen, and in the retrosplenial cortex. Details on D-cis-[ 18 F]FPro-labeled brain areas are given in Table 1.
Out of 10 rats, 7 exhibited D-cis-[ 18 F]FPro uptake in the thalamus ipsilateral to the tumor. Tracer accumulation in specific nuclei of the thalamus varied with the cortical areas affected by the tumor. According to the reciprocal thalamocortical or corticothalamic fibers known from the literature thalamic nuclei with D-cis-[ 18 F]FPro uptake could be assigned to their corresponding known target regions in the cerebral cortex. Thus, it was observed that D-cis-[ 18 F]FPro uptake in the ventrolateral thalamic nucleus, ventral anterior thalamic nucleus, ventromedial thalamic nucleus, and mediodorsal thalamic nucleus occurred when the corresponding motor and medial prefrontal cortex was affected by a tumor. Cis-4-[ 18 F]fluoro-D-proline uptake was found in the ventral posterolateral thalamic nucleus, ventral posteromedial thalamic nucleus, and posterior thalamic nuclear group when the somatosensory cortex contained the tumor. Cis-4-[ 18 F]fluoro-D-proline uptake in the laterodorsal thalamic nucleus was observed when the retrosplenial cortex was affected, and in the lateral posterior thalamic nucleus when the visual cortex was affected by the tumor. A representative example of D-cis-[ 18 F]FPro accumulation in the thalamic nuclei is shown in Figure 2.
Coronal sections of F98 tumor-bearing rat brain (rat no. 10). Red dashed lines indicate the position of the tumor. Cis-4-[
18
F]fluoro-D-proline (D-cis-[
18
F]FPro) autoradiography (
In one animal D-cis-[ 18 F]FPro accumulation was also observed in the subthalamic nucleus. The subthalamic nucleus is known to be connected with the cortex, 18 and the respective cortical site was infiltrated by F98 tumor cells in that animal (Table 1).
At 13 to 14 days after tumor implantation, in 3 out of 10 animals thalamic nuclei with increased D-cis-[ 18 F]FPro uptake were strongly positive for CD11b and slightly for GFAP staining, indicating microglial activation and reactive astrogliosis (rat nos. 1, 2, and 10). A representative example of glial activation in thalamic nuclei is also shown in Figure 2.
In 2 out of 10 animals, F98 tumor cells were implanted into the hippocampus of the left hemisphere, both resulting in ipsilateral D-cis-[ 18 F]FPro uptake in the hippocampal area CA1, particularly in the radiatum layer, which was not infiltrated by the tumor (Figures 3 and 4). In addition, one animal exhibited simultaneously tracer uptake in the subiculum and thalamus (Figure 3). Of note, both accumulating areas, CA1 and subiculum, were located caudal to the tumor mass (Figure 3). In the hippocampal area CA1, increased uptake of D-cis-[ 18 F]FPro was accompanied by strong immunoreactivity for CD11b (rat no. 10). In addition, GFAP-positive cells were observed in that area which, however, exhibited a symmetric pattern in the contralateral hemisphere with no D-cis-[ 18 F]FPro uptake (Figure 4).

Coronal serial sections of F98 tumor-bearing rat brain analyzed by cis-4-[
18
F]fluoro-D-proline (D-cis-[
18
F]FPro) autoradiography (
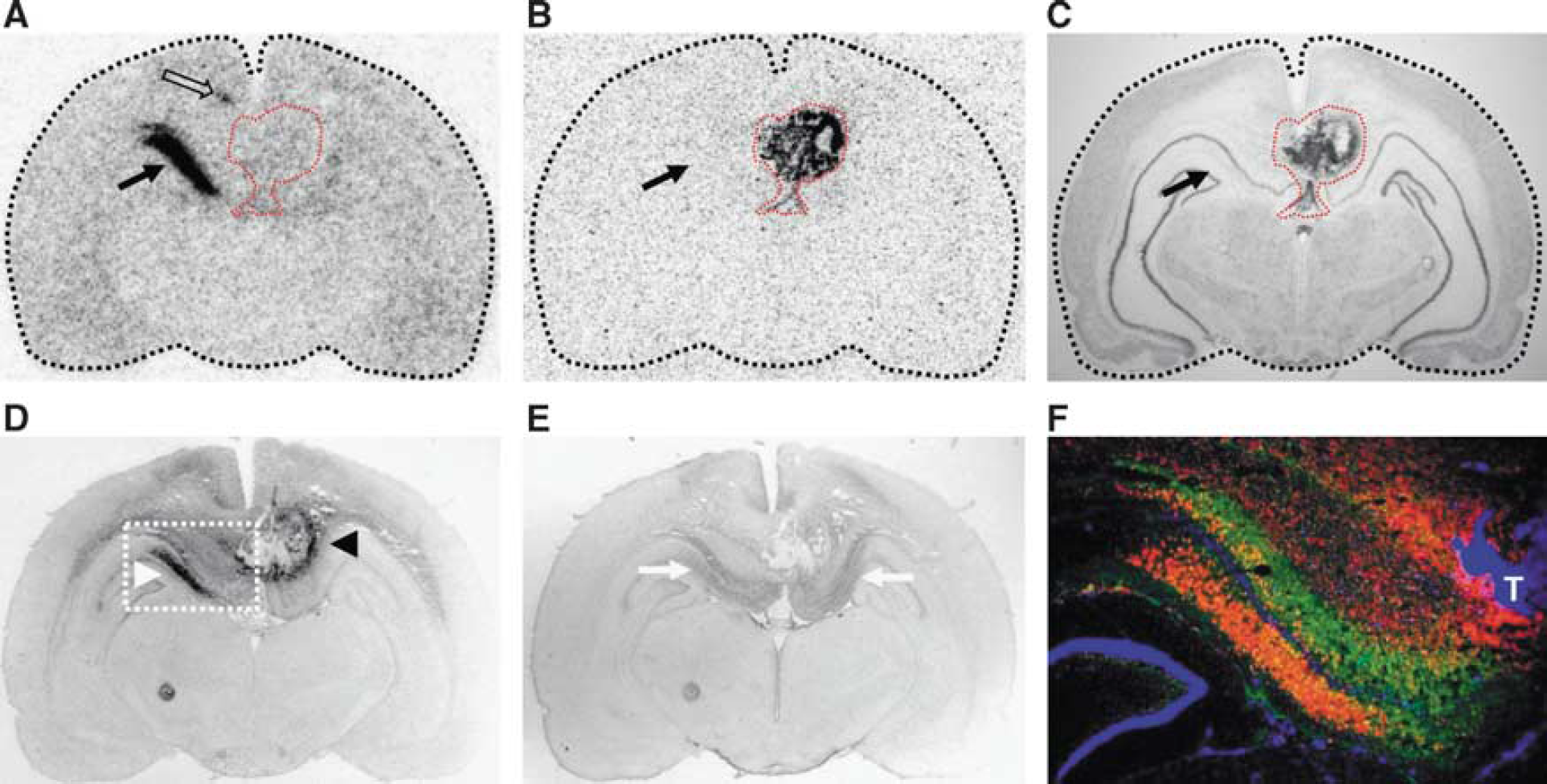
Coronal sections of F98 tumor-bearing rat brain (rat no. 10). Red dashed lines indicate the position of the tumor. Cis-4-[
18
F]fluoro-D-proline (D-cis-[
18
F]FPro) autoradiography (
DISCUSSION
Brain lesions involving the cerebral cortex may induce secondary reactions in thalamic nuclei connected to the affected cortical areas via thalamocortical and corticothalamic fibers. This phenomenon has been reported in animal experiments, such as in rodents subjected to middle cerebral artery occlusion, 4 photothrombosis, 11 or traumatic brain injury.19,20 In contrast, little is known about the capability of cerebral gliomas to induce secondary reactions in remote brain areas. Experiments in slice cultures of the entorhinal cortex and dentate gyrus indicated that implanted F98 glioma cells caused not only local tumor growth but also cytotoxic damage to the infiltrated brain parenchyma. 21 Moreover, it is known that glioma cells release increased amounts of glutamate, which in turn triggers neuronal degeneration by excitotoxicity,22,23 a mechanism that was also reported as a driving force in thalamic retrograde reaction. Thus, a marked decrease in thalamic high-affinity uptake of glutamate and aspartate was observed after cortical ablation. 1 In humans, some case studies reported brain atrophy associated with germ cell tumors infiltrating the thalamus or the internal capsule, which in turn caused anterograde and/or retrograde reactions and subsequent atrophy.13,14
In a previous study we showed that the PET tracer D-cis-[ 18 F]FPro detects secondary thalamic degeneration in rats with cortical infarctions with high sensitivity and with a high lesion to background contrast. 11 In this study, we used this method to explore the effects of brain tumors in remote areas of the brain in the rat F98 glioma model. The cerebral cortex was affected by F98 tumors in all animals and prominent D-cis-[ 18 F]FPro uptake was detected in ipsilateral thalamic nuclei in 7 out of 10 rats (Figure 2 and Table 1). Notably, the specific thalamic nuclei accumulating D-cis-[ 18 F]FPro varied with the specific connectivity with the cortical areas affected by the tumor. According to these thalamocortical projections, thalamic nuclei with D-cis-[ 18 F]FPro uptake could be assigned in all cases to their corresponding specific target region in the cerebral cortex. 16 In addition, strong D-cis-[ 18 F]FPro accumulation was noted in the hippocampal area CA1 (Figures 3 and 4) in two animals with ipsilateral F98 gliomas involving hippocampal subarea CA3 rostral to that area (Figure 3 and Table 1), which is a remarkable finding and probably the consequence of a tumor-induced lesioning of corresponding fiber tracts. Various studies in rodents reported that the hippocampus is particularly vulnerable to brain injuries leading to time-delayed hippocampal degeneration. Thus, a brief period of global or focal ischemia is sufficient to trigger delayed neuronal death in the areas CA1 and CA3 of the hippocampus several days after reperfusion.24,25 Secondary degeneration in the areas CA1 and CA3 as well as in the dentate gyrus was also shown after traumatic brain injury.26,27 Axons of CA3 pyramidal cells project forward to area CA1 via Schaffer collaterals, particularly to the hippocampal radiatum layer of CA1. 28 Cis-4-[ 18 F]fluoro-D-proline uptake in the hippocampus of those animals was confined to the hippocampal radiatum layer of CA1 (Figures 3 and 4). Moreover, one animal exhibited additional D-cis-[ 18 F]FPro uptake in the subiculum (Figure 3), which forms a specific target of CA1 neurons. 28 Cis-4-[ 18 F]fluoro-D-proline uptake was also detected in the caudate putamen, the subthalamic nucleus, and in the retrosplenial cortex (Table 1), which is in agreement with the connections of these structures with the tumor-bearing cortical areas. 18 Comparable results in our previous study showed D-cis-[ 18 F]FPro uptake in the caudate putamen after cortical infarction involving the motor cortex. 11 Because of multiple connections to other brain areas, the relationship between D-cis-[ 18 F]FPro accumulation in the retrosplenial cortex (Figure 4) may also be explained by the same principle of retrograde or anterograde neuronal reaction.
Activated microglia and reactive astrocytes have been used as indicators of neuronal degeneration because of their pronounced reaction to neurologic injuries. In thalamic nuclei of rodents, for instance, activated microglia and reactive astrocytes were observed after cortical infarction induced by photothrombosis11,29 or traumatic brain injury. 20 In this study, glial immunoreactivity in thalamic nuclei in congruency with D-cis-[ 18 F]FPro uptake (Figure 2) could be detected in three animals, but D-cis-[ 18 F]FPro was also positive in four animals, which showed no immunoreactivity (Table 1). Also, D-cis-[ 18 F]FPro uptake in the hippocampus was accompanied by strong immunoreactivity for CD11b and GFAP in one animal (Figure 4).
These results suggest that brain tumors may induce reactions in remote thalamocortical relay neurons and other brain areas by destruction of their cortical target areas, similar to the effects observed in animals with cortical infarction or trauma. Although it is not an unexpected finding that gliomas may cause secondary reactions in remote brain areas similar to the observation with infarction and other brain lesions, to the best of our knowledge this process has never been shown before in an in vivo animal model or in humans. Furthermore, the results of this study and those of our previous report support the notion of a remarkable sensitivity of D-cis-[ 18 F]FPro for the detection of neuronal reactions in brain areas remote from the primary lesion, which offers the possibility to transfer this approach to in vivo imaging in humans using PET. Initial results in patients with akinetic-rigid parkinsonism indicate an association of D-cis-[ 18 F]FPro uptake in the striatum with neurodegeneration in Parkinsonion syndromes. 30
The molecular mechanisms of D-cis-[ 18 F]FPro uptake are not yet understood clearly. As mentioned above, the D-isomer of cis-4- 18 F-fluoro-proline is transported preferably across the BBB compared with its D-isomer. 12 Analysis of the rat brain homogenate after injection of D-cis-[ 18 F]FPro revealed a significant part of the radioactivity in the form of L-trans-[ 18 F]FPro, indicating that D-cis-[ 18 F]FPro is accepted by a racemase in the human brain and converted to the D-isomer. 12 Preliminary experiments in our laboratory have shown that D-cis-[ 18 F]FPro is transported by only one of the known proline transporters, i.e., PROT (unpublished results). PROT is expressed in excitatory nerve terminals but has not been observed at the BBB. 31 Therefore, the transport of D-cis-[ 18 F]FPro via the intact BBB remains unclear.
Further information on the biologic properties of this tracer can be derived from this study. On the one hand, the solid tumor mass of proliferating F98 cells exhibited no relevant D-cis-[ 18 F]FPro uptake but strong [ 3 H]thymidine accumulation, and, on the other hand, areas of neuronal reaction with prominent D-cis-[ 18 F]FPro accumulation distant to the tumors showed neither [ 3 H]thymidine uptake nor structural changes in the histologic stainings. Obviously, a proliferative process is not involved in the uptake process of D-cis-[ 18 F]FPro. These observations indicate a highly specific uptake mechanism of D-cis-[ 18 F]FPro, which appears to be linked to the process of secondary neurodegeneration. Furthermore, the absence of D-cis-[ 18 F]FPro in F98 gliomas suggests that a disruption of the BBB does not lead to unspecific accumulation of D-cis-[ 18 F]FPro. Blood–brain barrier permeability was not tested in our study but radioactive thymidine analogs do not cross the intact BBB and the high uptake of [ 3 H]thymidine indicates BBB disruption in the F98 gliomas. 32
Another interesting finding was focally increased uptake of D-cis-[ 18 F]FPro in the necrotic center of large F98 tumors. The histologic stainings displayed areas of very low cell density with low [ 3 H]thymidine uptake in the center of the tumors (Figure 1), indicating a central necrosis, which is a typical finding in large F98 gliomas and human glioblastoma. 15 These necrotic core areas showed focal D-cis-[ 18 F]FPro uptake accompanied by CD11b-expressing cells, indicating activated microglia (Figure 1). However, additional CD11b-positive cells forming a narrow zone at the tumor margin were found that exhibited no D-cis-[ 18 F]FPro uptake (Figure 1). Therefore, the hypothesis that D-cis-[ 18 F]FPro uptake reflects activated microglia must be questioned. Also, there is no obvious relationship of D-cis-[ 18 F]FPro uptake with reactive astrogliosis, which is in agreement with the observations in cortical infarctions. 11 The peritumoral tissue exhibited strong astrogliosis but no tracer uptake and there were nearly no GFAP-positive cells in the areas of focally increased D-cis-[ 18 F]FPro uptake in the center of the tumors (Figure 1). Thus, it appears that D-cis-[ 18 F]FPro accumulation in remote brain areas with secondary neurodegeneration and in the necrotic center of brain tumors indicates an uptake mechanism related to pathologic cell death. Analysis of secondary neurodegeneration of thalamic nuclei after cortical ischemia in rats suggested that cell death in the thalamus underlies both apoptosis and necrosis, 33 and neuronal death in area CA1 of the hippocampus after global cerebral ischemia was attributed to programmed necrosis. 34
Here we show that brain tumors may induce secondary reactions in remote thalamocortical relay neurons and other brain areas by destruction of their cortical target areas, similar to the effects observed in animals with cortical infarction or trauma. In contrast to other amino-acid tracers, D-cis-[ 18 F]FPro exhibits a high specificity for the detection of secondary neuronal reactions and does not accumulate in neoplastic lesions. Our results warrant a further clinical evaluation of D-cis-[ 18 F]FPro in various brain injuries or neurodegenerative diseases.
DISCLOSURE/CONFLICT OF INTEREST
The authors declare no conflict of interest.
Footnotes
ACKNOWLEDGEMENTS
The authors thank Stephanie Krause for assistance in histological staining, Jeyakamalini Prasath and Michael Schöneck for technical assistance, and Silke Grafmüller, Bettina Palm, Erika Wabbals, and Sascha Rehbein for the radiosyntheses of D-cis-[ 18 F]FPro.
