Abstract
INTRODUCTION
Neural stem cells are able to self-renew and to differentiate into neurons, astrocytes, or oligodendrocytes.
1
Many endogenous compounds regulate proliferation and differentiation of NPCs both
Endocannabinoids were reported to induce proliferation of NPCs in a process that involves both CB1 and CB2 receptors.8–10 The naturally occurring endocannabinoids anandamide and 2-arachidonoylglycerol inhibit differentiation of NPCs.
11
The novel endocannabinoid-like compound
Traumatic brain injury leads to an increase in cell proliferation starting in the first two weeks after injury and is evident up to one year later, followed by recruitment of NPCs to the injured cortex.16–22 Brain injury also induces differentiation of the subventricular zone cells towards a glial fate, which is the default for postnatal differentiation, resulting in the formation of a glial scar in the injured cortex.18,23–25 As the effects of cannabinoids are central to cell survival 26 and may also influence differentiation, the present study was designed to investigate the effect of AraS on proliferation and differentiation of NPCs and to explore whether AraS has long-term effects after TBI.
METHODS
Animals
The study was performed according to Institutional Animal Use and Care Committee guidelines and was approved by the institution's Animal Care and Use Committee. Male Sabra mice (strain of the Hebrew University, Harlan; Jerusalem, Israel) aged 6 to 7 weeks and weighing 35 to 45 g was used in this study. No variations in dominant/submissive characteristics were observed with this strain.
Cell Culture
Cerebral cortical cultures of NPCs were prepared from 14-day mouse embryos. Brains were removed and the cortices were separated from the hemispheres and added to 0.025% trypsin solution for 10 minutes in 37°C humidified 5% CO2 incubator. After removal of trypsin, tissues were added with MEM supplemented with 10% horse serum, 2 mmol/L L-glutamine, 0.35% glucose, and 0.5% penicillin—streptomycin and triturated with a pasteur pipette until clear solution was obtained. Cells were centrifuged for 10minutes at 1,100r.p.m. and re-suspended in N2 media (Dulbecco's modified eagle medium/F12 supplemented with 0.5 mg/mL bovine serum albumin, 100 μg/mL apo-transferrin, 300 ug/mL L-glutamine, 0.11 mg/mL sodium pyruvate, 2.5 μg/mL gentamycin sulfate, 25 μg/mL bovine insulin, 6 ng/mL progesterone, 16 ug/mL putrescine, 5.2 ng/mL sodium selenite, and 10 ng/mL D-biotin). The culture was added with 20 ng/mL basic fibroblast growth factor and 10 ng/mL epidermal growth factor daily to avoid differentiation.
Size of Neurospheres
After 3 days
Propidium Iodide Uptake
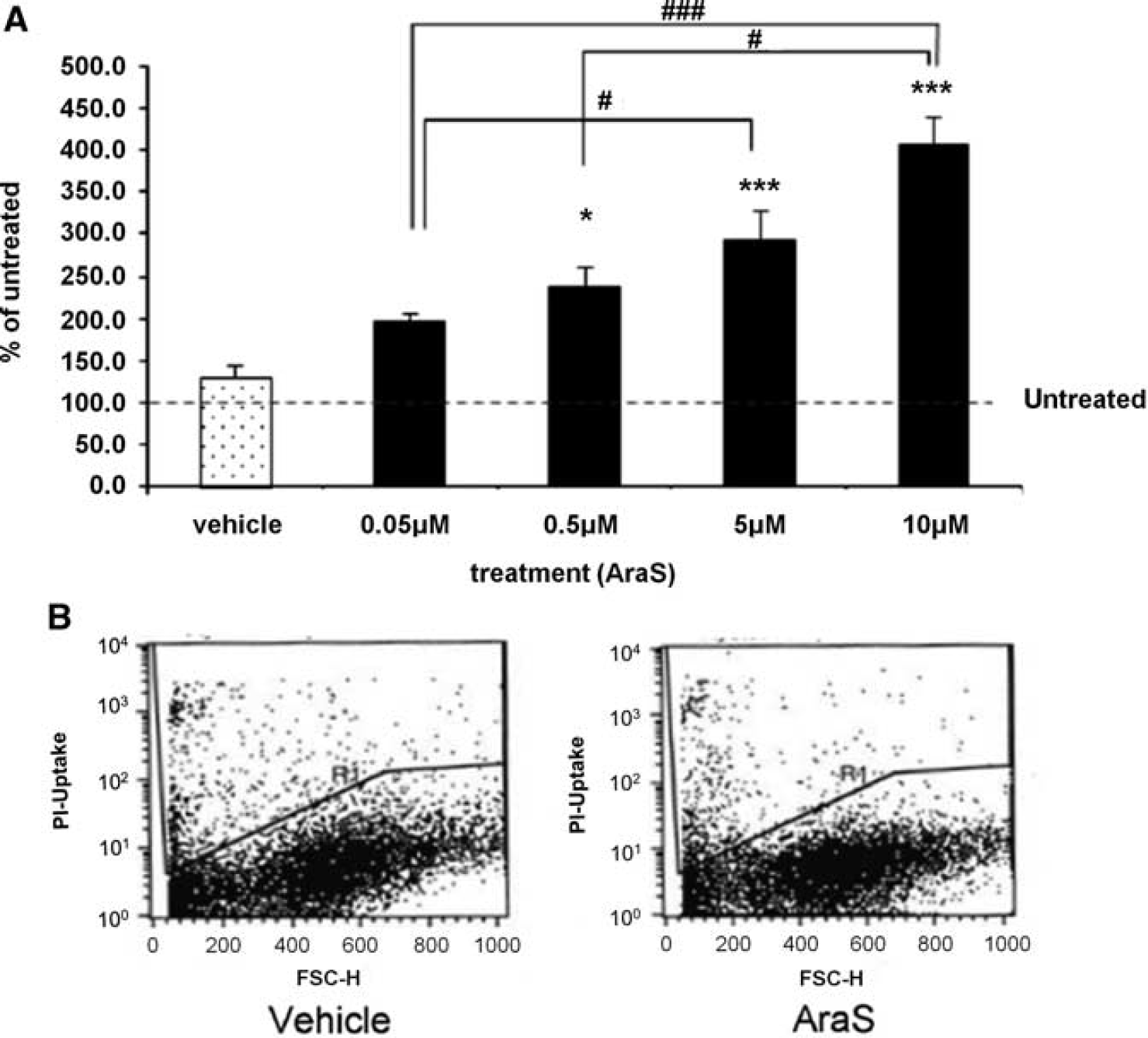
Four days after addition of 10 μmol/L AraS to culture, neurospheres were collected, centrifuged for 5 minutes at 1,100 r.p.m. and dismounted to single cells by triturating with a 200mL tip. Cells were re-suspended in phosphate-buffered saline before the addition of propidium iodide (1 mg/mL) and analyzed by flow cytometry (FACSCalibur and Cellquest Software Becton Dickinson, Istanbul, Turkey) using the FL3 channel. Propidium iodide-positive cells are considered dead cells.
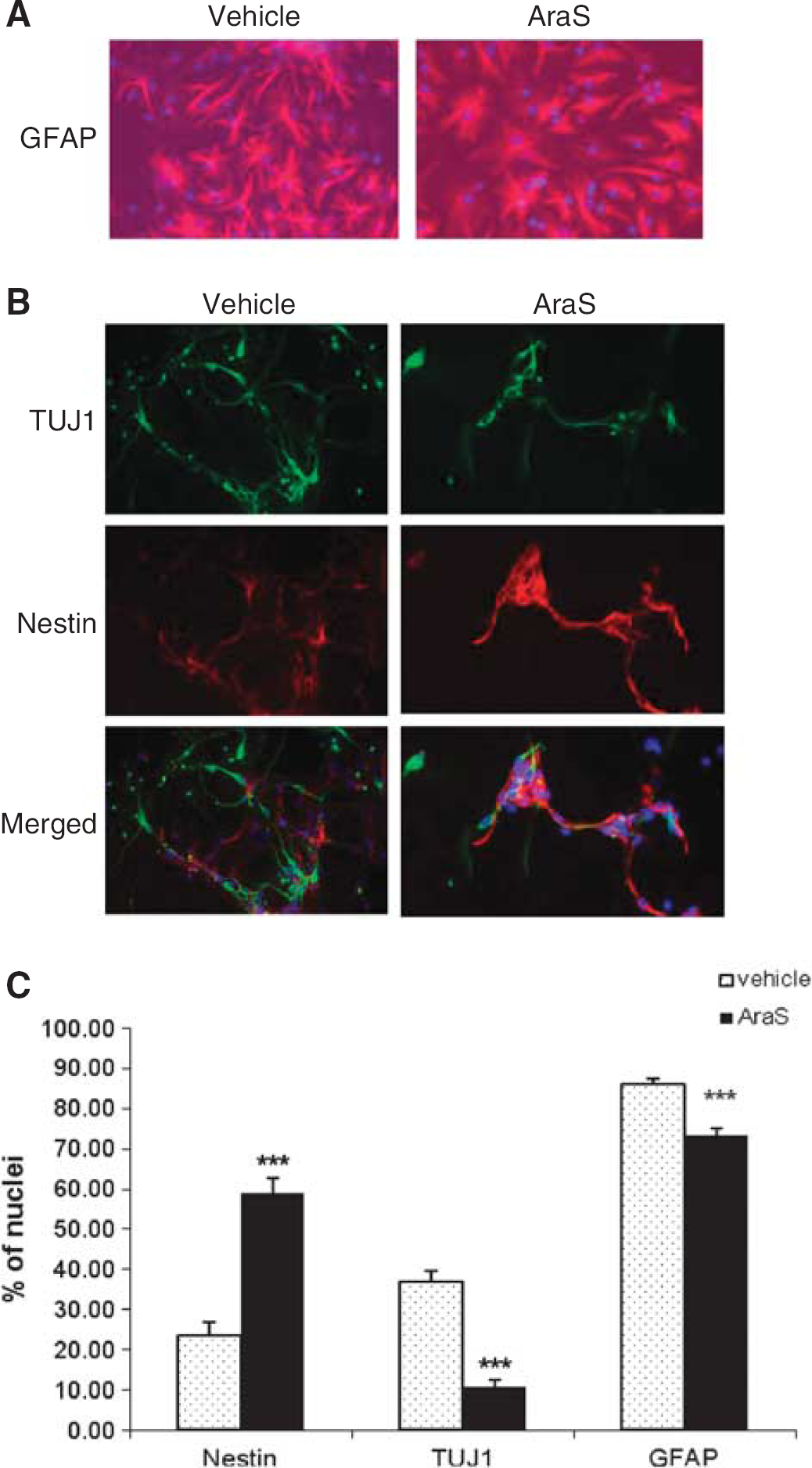
Differentiation of Neural Progenitor Cells
Trauma Model
Mice were subjected to closed head injury (CHI) under isoflurane (2%) anesthesia, using a weight-drop device that falls over the left hemisphere, as described elsewhere. 28 In brief, after a longitudinal scalp incision, mice were immobilized under a cylindrical calibrated weight-drop device. A tipped teflon cone was placed (upside down) 2 mm lateral to the midline and 1 mm caudal to the left coronal suture, and a metal rod (94 g) was dropped down on the cone from a height of 11 to 14cm (adjusted to body weight, to ensure the severity of injury 28 required to produce CHI). Less than 10% of the injured mice were excluded from the study, mostly because of death by apnea within minutes of injury. After recovery from anesthesia, the mice were returned to their home cages with free access to food and water.29,30
Evaluation of Functional Outcome
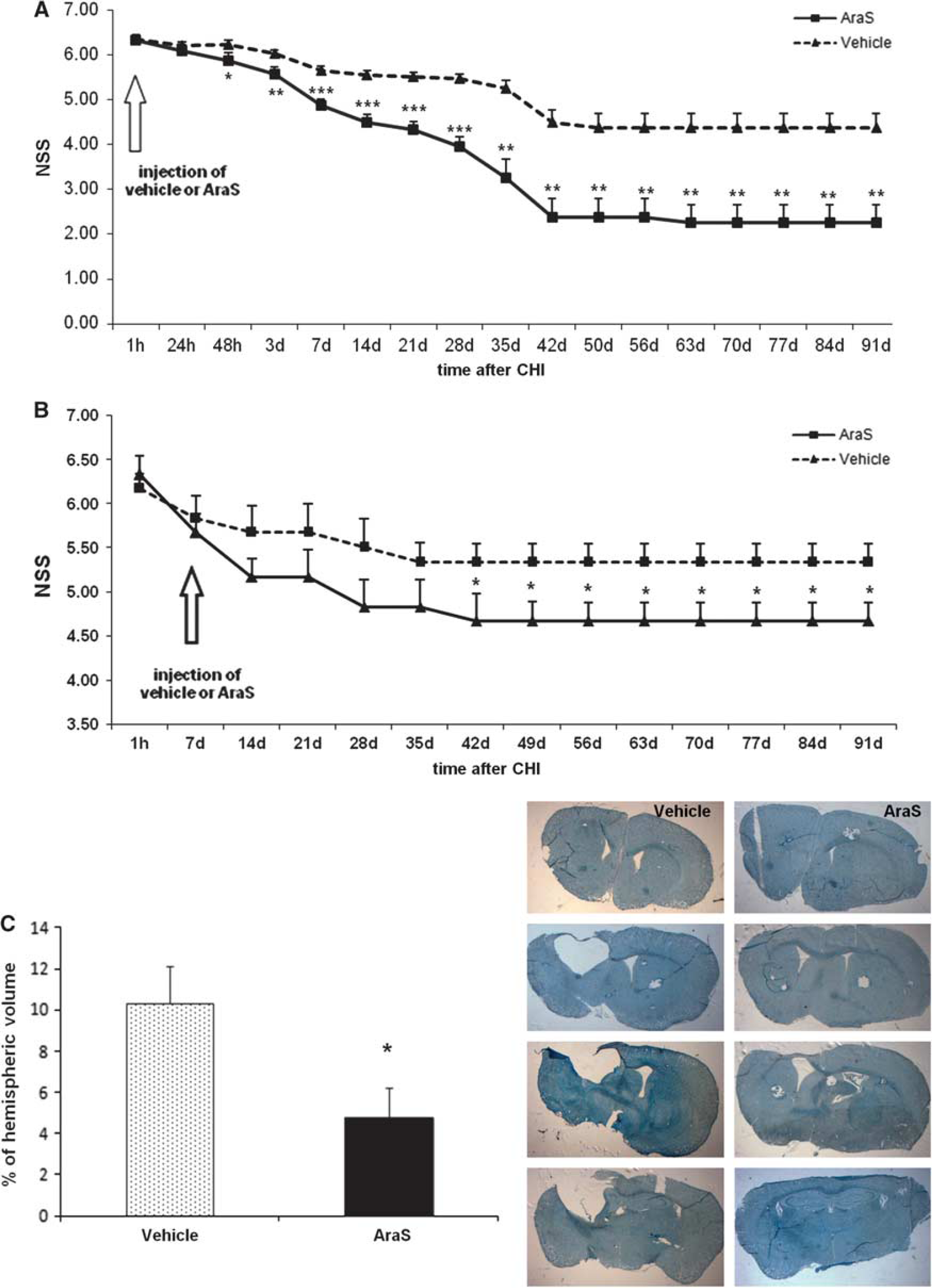
One hour after CHI, the functional status of the mice was evaluated according to a set of 10 neurobehavioral tasks (neurologic severity score, NSS), which examine reflexes, alertness, coordination, motor abilities, and balancing. 31 Failure to perform a task scores 1 point and a success scores 0. Hence, normal animals score 0, reflecting healthy mice, whereas a score of 10 reflects maximal neurologic impairment. Only mice with NSS 6 to 8 at 1 hour after injury (NSS 1 hour) were included in the study. The extent of improvement (ANSS) was calculated by subtracting the NSS at a specific time point from that achieved 1 hour after CHI. A NSS follow-up was performed weekly for 91 days.
Drug Application
Treatment at 1 hour
The dose selected for treatment was based on our previous studies.
13
Mice were injected intraperitoneally with 3 mg/kg AraS dissolved in ethanol:cremophor:saline 1:1:18 or vehicle immediately after NSS evaluation at 1 hour. Based on the NSS at 1 hour, mice were assigned to treatment groups such that similar severity of injury (NSS 1 hour) was ensured in all groups (
Treatment at 7 Days
After CHI and NSS evaluation at 1 hour, NSS was re-evaluated 7 days after the injury and immediately afterwards, the mice were assigned to treatment groups such that similar neurologic status (NSS 7 days) was ensured in all groups (
In Vivo Proliferation and Differentiation
At 90 days post TBI, mice were perfused with 4% paraformaldehyde, decapitated, and the brains were frozen immediately on dry ice and kept at − 80°C until further use. Before staining, brains were cut to 10 μm slices from bregma 0 to bregma − 2 mm using a Leica CM1850 Cryostat (Leica Microsystems GmbH, Wetzlar, Germany). Slices were thawed, fixed with 4% paraformaldehyde solution for 20 minutes, washed, and incubated in 2 N HCl solution for 25 minutes (excluding staining for doublecortin) in a 37°C oven. Slices were washed and blocked for 2 hours at room temperature in 5% normal donkey serum followed by an overnight incubation at 4°C with the primary antibodies (BrdU 1:100 (Sigma-Aldrich, St Louis, MO, USA) + nestin 1:100 (Abcam), glial fibrillary acidic protein 1:200 (Dako, San Diego, CA, USA) or NeuN 1:100 (Millipore, Billerica, MA, USA)). After washes, the slices were incubated for 2 hours at room temperature with the appropriate secondary fluorescent antibodies (donkey antirat 488, donkey anti-mouse 488, donkey anti-mouse 555, donkey anti-rabbit 488, or donkey anti-rabbit 555; Invitrogen, Carlsbad, CA, USA) followed by incubation with 4'6-diamido-2-phenylindole solution (Invitrogen) for 5 minutes and mounting. A group of five sections was counted every 150 μm (
Lesion Volume
Lesion volume was evaluated using Giemsa staining, as described elsewhere.
25
Briefly, the mice were subjected to CHI followed by the different treatments at 7 days after the injury (
Western Immunoblotting
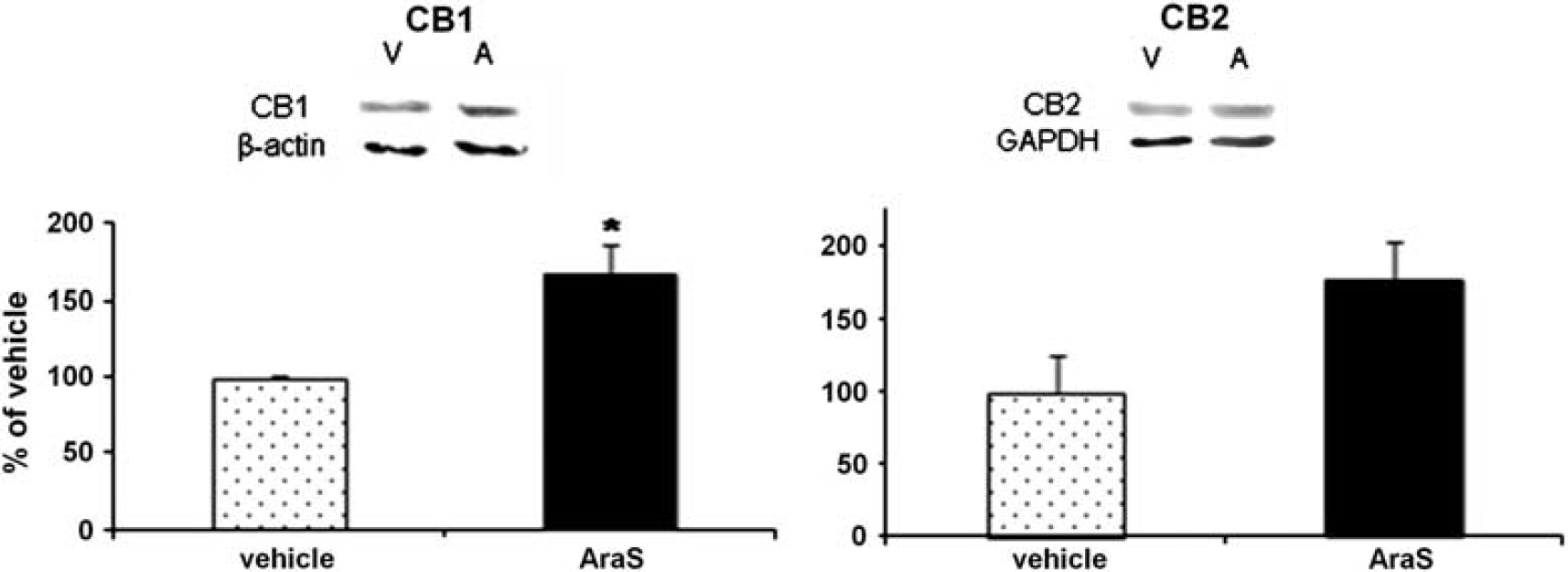
To determine the levels of CB1 and CB2 in NPC, neurospheres were treated with AraS or vehicle and collected 4 days afterwards. The spheres were centrifuged (1,100 r.p.m. × 5 minutes) and soup was removed. Cells were lysed with cold lysis buffer (50 mmol/L Tris-HCl, pH 7.5, 150 mmol/L NaCl, 1 mmol/L EDTA, 1 mmol/L ethylene glycol tetraacetic acid, 1 mmol/L Na3VO4, 50 mmol/L NaF, 10 mmol/L sodium β-glycerophosphate, 10 μmol/L sodium pyrophosphate, 1 mmol/L phenylmethylsulfonyl fluoride, supplemented with 0.1% (v/v) NP-40, 0.1% (v/v) 2-β-mercaptoethanol and protease inhibitor cocktail). Protein content in the supernatant was determined according to Bradford method and the manufacturer's instructions. A total 15 μg of the protein extract from each sample was mixed with SDS-sample buffer and boiled at 95°C for 5 minutes before loading. The proteins were separated by 10% sodium dodecyl sulfate polyacrylamide gel electrophoresisand transferred onto trans-blot nitrocellulose membranes. The membranes were blocked in 5% nonfat dry milk in TBS, pH 7.4, with 0.1% Tween 20 (TBS–Tween) for 1 hour at room temperature. Primary antibodies (antiCB1 1:500, antiCB2 1:500, β-actin 1:1000, Glyceraldehyde-3-phosphate dehydrogenase 1:2,000) were diluted in 5% bovine serum albumin solution, and the membranes were incubated overnight at 4°C. The primary antibody was removed, and the blots were washed in TBS–Tween and incubated for 1 hour at room temperature with horseradish peroxidase-conjugated secondary antibodies (1:5,000, Jackson Immunoresearch, Soham, Cambridgeshire, UK). Reactive proteins were visualized using enhanced chemiluminescence reagent (Biological Industries, Beit Haemek, Israel) by direct chemiluminescence using FUJIFILM Luminescent Image Analyzer camera LAS-3000 (Tokyo, Japan). The optical density was determined using ImageJ 1.40 g software (National Institutes of Health, USA).
Statistical Analysis
Statistical analysis was performed using SigmaStat 2.03 software (SPSS, Chicago, IL, USA). The data are presented as the mean ± s.e.m. The statistical significance of differences between means was evaluated by student's
RESULTS
AraS Increases the Size of Neurospheres of NPCs In Vitro
To determine the involvement of AraS in neurogenesis, the role of AraS in NPC proliferation was investigated using an
AraS Reduces Differentiation of NPC In Vitro
Neurospheres were treated with 10 μmol/L AraS or vehicle and were then let to differentiate for 5 days without addition of growth factors, after which they were stained for different neuronal and glial markers. A reduction in the expression of the astrocytic marker glial fibrillary acidic protein (73.26 ± 1.60%) together with the neuronal marker TUJ1 (10.56 ± 1.94%) was detected in NPCs treated with AraS compared with vehicle (86.11 ± 1.26% and 36.79 ± 2.69%, respectively). In contrast, a 2.5-fold increase in the expression of the NPC marker nestin was detected in the treated culture compared with control (58.88 ± 3.65% and 23.40 ± 3.59%, respectively; Figure 2). Stainings for the marker for oligodendrocytes galactocerebroside did not show any differentiation to these cells. Taken together, these results indicate that AraS maintains NPCs in an undifferentiated state and reduces their terminal differentiation
CB1, CB2 and TRPV1 are Involved in the Proliferative Effect of AraS In Vitro
The mechanism involved in the proliferative effect of AraS was investigated
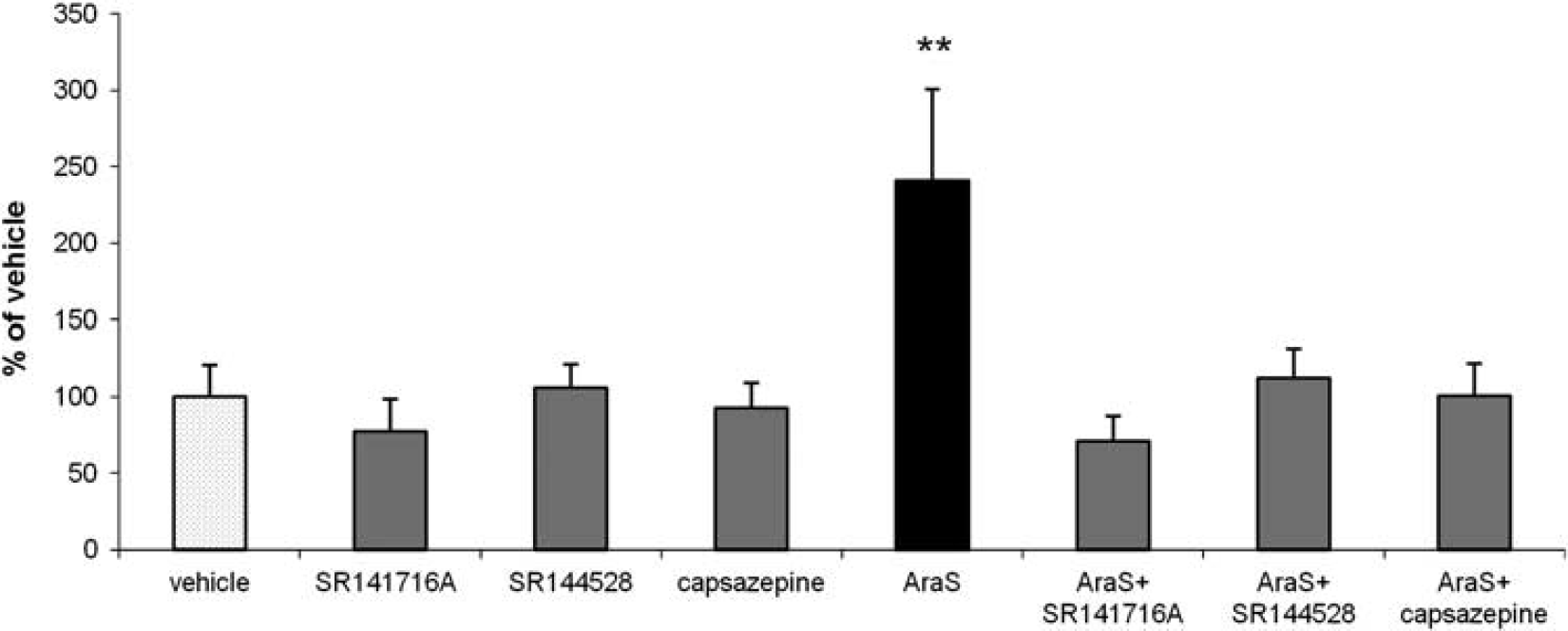
CB1, CB2, and TRPV1 antagonists abolish the proliferative effect induced by
AraS Improves Neurobehavioral Function, Increases Proliferation and Decreases Differentiation of NPC Into Astrocytes After Traumatic Brain Injury
To further investigate the neurogenic properties of AraS, and to discriminate between the early neuroprotective phase that was shown in our previous study13,15 and the later proneurogenic phase, AraS was injected in the current series of experiments 7 days after TBI. The beneficial effect of AraS, namely reduction of the behavioral deficits as evaluated by NSS, was only evident from 5 weeks after treatment and sustained up to 3 months thereafter (Figure 5B). A sham group was included in our previous experiments and showed no clinical deficits in the NSS test over time.13,33 It should be noted that when AraS was given 1 hour after TBI, the effect reached significance 48 hours later, and its extent was more pronounced (Figure 5A).
Injection of
The improved function may imply an induction of neurogenesis and remodeling processes. Indeed, treatment with AraS led to a 1.8-fold increase in the levels of proliferating cells (BrdU +) (10.94 ± 2.40 and 6.10 ± 0.77, respectively; Figure 6). Staining with the astrocytic marker glial fibrillary acidic protein together with the proliferation marker BrdU demonstrated a decrease in the differentiation of proliferating cells into astrocytes in the AraS-treated group compared with vehicle (21.5 ± 1.9% versus 32.8 ± 4.1% respectively;
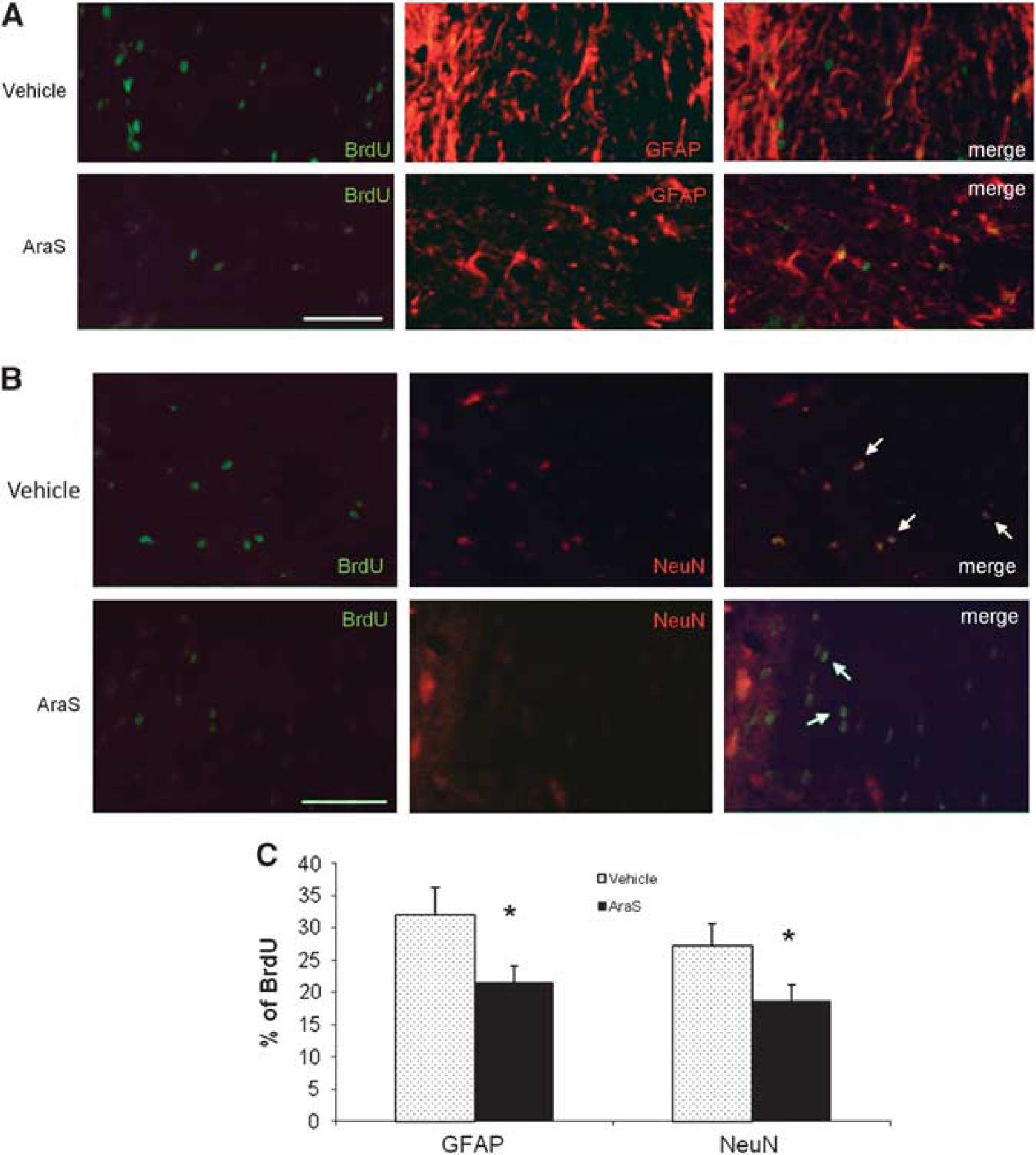
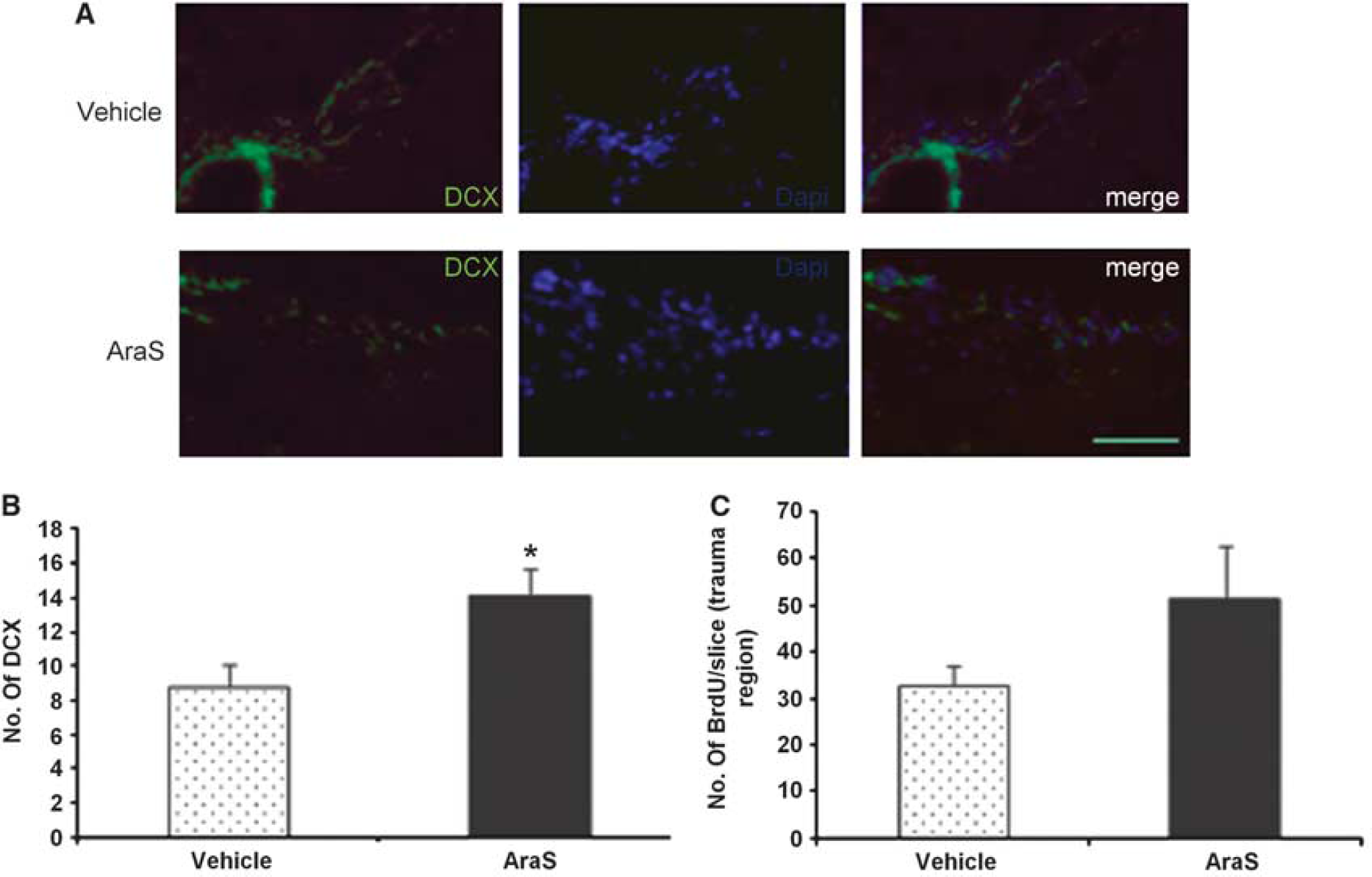
These results show that similar to its effects
AraS Reduces Lesion Volume Three Months After Brain Trauma
To further explore the effects of delayed (7 days post injury) administration of AraS, the lesion volume was measured 91 days after injury. While lesion hemispheric volume of 10.34 ± 1.79% was found in the vehicle-treated mice, it was only 4.76 ± 1.43% in the brains of AraS-treated mice (
DISCUSSION
This study is focused on the long-term recovery and rehabilitative effects of the endocannabinoid AraS, given at a single dose after injury. When the drug was given 1 hour post injury the neurobehavioral function continuously improved during 6 weeks. This time is long beyond the cellular prosurvival signaling, which we have reported earlier,
13
suggesting the involvement of other mechanisms, including activation of neural stem/progenitor cells. To dissociate the neuroprotective from the proliferative effects, mice were treated with AraS 7 days after injury and their functional outcome was followed for 3 months. In addition, NPC were prepared from 14-day mouse embryos and exposed to AraS
At different embryonic and postnatal stages of brain development, the endocannabinoid system is involved in the regulation of NPC differentiation, which occurs in parallel with CB1 receptor expression.9,11 CB2 receptor is present in progenitor cells from embryo origin and from adult brain. It mediates acceleration of neurogenesis and stimulates neural progenitor proliferation.8,34 In the present study, we found that the proliferative effect of AraS is mediated via the cannabinoid receptors CB1, CB2, and TRPV1, as treatment of neurospheres
AraS previously demonstrated neuroprotective effects in a CB2-related mechanism, 13 although it binds very weakly to this receptor. 12 As CB2 is a known direct mediator of NPC proliferation and indirectly mediates neurogenesis,8,34 it may be suggested that the increase in the levels of the neuroepithelial marker nestin induced by AraS is mediated, indirectly, by CB2. However, this suggested the mechanism needs to be elucidated.
AraS is shown here to induce proliferation of NPCs
It is important to mention that the lack of change in cell death
To summarize this part of the study, AraS possesses properties, which may contribute towards remodeling the tissue using the existing unharmed cells, while maintaining the population of progenitors in the brain. This assumption may also explain, at least in part, the relatively late improvement in neurobehavioral function since such processes take a long time to establish.25,38
Behavioral function of injured mice, assessed during 3 months after trauma, was less impaired in the AraS-treated groups compared with vehicle. This functional improvement was achieved already at 48 hours, after a single dose of AraS was given 1 hour post injury and could be attributed to the neuroprotective processes induced by AraS. These involve increased levels of pAkt and the prosurvival factor Bcl-xL as well as reduced activation of the proapoptotic enzyme caspase 3. 13 These neuprotective effects may also account for the decrease in lesion volume reported in our previous study, from 14.4% in the vehicle-treated mice to 8.00% in the mice treated with AraS at 1 hour. 13 However, the neuroprotective cellular effects are short-term (2 to 24 hours) and cannot account for those achieved when AraS is given 7 days after injury. Thus, the late-onset benefits described here as improved functional outcome, yet much less pronounced than in the group treated 1 hour post injury, and reduced lesion volume, exclude the period described as the window for neuroprotection and could be derived from another property of the endocannabinoids, including AraS.
Handling, anesthesia and stress may affect neurogenesis and may have impacted our results. However, since similar manipulations were used in all groups including vehicle, and since sham-operated animals demonstrated no abnormalities in the NSS or neurogenesis, 13 these factors were not likely to impact the results shown in this work.
One of the secondary insults after TBI is cerebral vasospasm, which occurs in more than one-third of patients with TBI (for a review, see Werner and Engelhard
39
). The temporal profile and extent of hypoperfusion with posttraumatic vasospasm shows an onset, which varies from posttraumatic day 2 to 15 as a result of chronic depolarization of vascular smooth muscle.
40
In our earlier studies, (for a review see Shohami
In conclusion, we showed here for the first time that the novel endocannabinoid AraS, even when given as late as 7 days after injury, may still improve neurobehavioral function and reduce infarct volume. It exerts regenerative effects
DISCLOSURE/CONFLICT OF INTEREST
The authors declare no conflict of interest.
