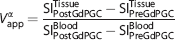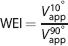Abstract
Insufficient vascular reserve after an ischemic stroke may induce biochemical cascades that subsequently deteriorate the blood–brain barrier (BBB) function. However, the direct relationship between poor cerebral blood volume (CBV) restoration and BBB disruption has not been examined in acute stroke. To quantify BBB integrity at acute stages of transient stroke, in particular for cases in which extravasation of the standard contrast agent (Gd-DTPA) is not observed, we adopted the water exchange index (WEI), a novel magnetic resonance image-derived parameter to estimate the water permeability across the BBB. The apparent diffusion coefficient (ADC) and R2 relaxation rate constant were also measured for outlining the tissue abnormality, while fractional CBV and WEI were quantified for assessing vascular alterations. The significantly decreased ADC and R2 in the ischemic cortices did not correlate with the changes in CBV or WEI. In contrast, a strong negative correlation between the ipsilesional WEI and CBV was found, in which stroke mice were clustered into two groups: (1) high WEI and low CBV and (2) normal WEI and CBV. The low CBV observed for mice with a disrupted BBB, characterized by a high WEI, indicates the importance of CBV restoration for maintaining BBB stability in acute stroke.
INTRODUCTION
Disruption of the blood–brain barrier (BBB) leads to devastating cerebral vasogenic edema and hemorrhagic transformation; hence, the functional status of the BBB is a crucial determinant of patient prognosis in ischemic stroke. In particular, as various treatments such as thrombolysis and maintenance of systemic blood pressure have been used to increase the cerebral perfusion in acute stroke patients, greater attention is paid to accurate measurement of stroke-related BBB alterations for preventing the reperfusion-related complications. 1 Among various methods adopted to evaluate BBB damage, the most widely used are those that show the leakage profiles of various staining dyes, isotopes, or imaging contrast agents as shown in Figure 1A (left panel).2–5 Such techniques can identify the location and extent of the BBB breakdown when the BBB disruption leads to obvious extravasation of exogenous contrast agents. However, this approach is inadequate to assess BBB integrity in cases of early or mild BBB deterioration, in which the amount of leaked contrast material is insufficient to show BBB damage.
Illustration of two different strategies for detecting an impaired blood–brain barrier (BBB) (
Based on a compartmentalized tissue model (Figure 1A), we have previously showed the feasibility of using a water exchange index (WEI) method to evaluate the BBB status even in the absence of overt extravasating compounds. 6 Specifically, this WEI technique relies on the magnetic resonance image (MRI) signal intensity affected by both the presence of an intravascular contrast agent and water exchange across the BBB (Figure 1A, right panel). Recent studies using the WEI technique have shown that it is an effective biomarker for quantifying water permeability as a means to assess subtle BBB damage, which is unquantifiable by less sensitive conventional methods.7–10 The high sensitivity of the WEI method is particularly useful for examining stroke models in which the ischemic burden on the BBB is too low to be detected by the leakage of exogenous contrast materials.6,9,11,12
In addition to measuring the degree of BBB disruption, it is important to identify the biophysical conditions that trigger BBB damage, as intervention of the causative factors should prevent further breakdown of the BBB. Magnetic resonance image-derived vascular and tissue parameters, particularly the cerebral blood volume (CBV), apparent diffusion coefficient (ADC), and transverse relaxation rate constant R2, have been rigorously investigated to noninvasively identify the parameters associated with impaired BBB integrity. According to previous study results, alterations in CBV, ADC, and R2 were positively predictive of BBB disruption, as all of these parameters significantly decreased in ischemic stroke characterized by extravasation of contrast materials.13–17 However, under such heavy ischemic stresses that lead to overt vascular disruption, changes in these MRI parameters may be derived not only from the weakened BBB itself, but also from other ischemia-affected tissue/vascular conditions including cytotoxicity, inflammation, and edema formation.3,18,19 Therefore, overly simplified dichotomous division of stroke cases according to the presence of apparent trans-BBB leakage does not definitively identify the causative factors in the BBB disruption. In this study of the WEI in transient ischemic mouse models, we theoretically validated the CBV and WEI quantification strategy using a computer simulation of a tissue model with a wide range of physiologic variables and also investigated the relation between the in vivo BBB integrity and the degree of CBV restoration. In particular, we aimed to quantify the severity of early BBB damage before extravasation of the standard contrast agent is observed, demonstrated by the lack of Gd-DTPA leakage, and to determine the MRI-derived parameters that are directly associated with changes in the BBB integrity.
MATERIALS AND METHODS
Animal Preparation
All experiments were conducted in accordance with the guidelines and regulations of the Subcommittee on Research Animal Care of Massachusetts General Hospital. A total of 21 male C57BL/6 mice (8 to 12 weeks old, ~25 g) were used in this study; 15 of these mice underwent transient occlusion of the left middle cerebral artery (MCA) and the remaining 6 mice were used as controls. Animals were anesthetized during the MCA occlusion surgical procedure with continuous inhalation of gas mixture (30% O2 and 70% NO2) with 1.5% isoflurane. The left external carotid artery was isolated and incised, and a 7-0 nylon monofilament covered by silicon (Doccol Corp., Sharon, MA, USA) was advanced via external and internal carotid artery for blocking the ostium of the MCA. The filament was then removed after 1 hour of MCA occlusion to induce reperfusion. Immediately after the start of reperfusion, the animals were prepared for MRI examinations. The mice were relocated to a stereotaxic frame designed to fix the head position. During the MRI acquisitions, the mice were anesthetized with 1.5% isoflurane in a 1:1 gas mixture of medical air and oxygen, while body temperature and oxygen saturation were continuously monitored.
Magnetic Resonance Examinations
Magnetic resonance image examinations were performed on a 9.4 T scanner (Bruker Biospin, Billerica, MA, USA). A protected graft copolymer covalently linked to Gd-DTPA residues (Gd-PGC), with a molecular weight of ~560,000 Da and a hydrostatic diameter of ~10 nm, was used as an intravascular contrast agent (University of Massachusetts; Laboratory of Molecular Imaging Probes). 20 Before and after the intravenous administration of Gd-PGC (17.5 μmol Gd/kg), 3D-spoiled gradient echo images were obtained using the following parameters: matrix size = 64 × 64 × 128; field of view = 1.6 × 1.6 × 3.2 cm; repetition time/echo time = 40/3.1 ms; number of average = 1; flip angles = 10°, 20°, 40°, 60°, and 90°. For the post Gd-PGC MRI, images were acquired ~5 minutes after the intravenous Gd-PGC administration.
Diffusion-weighted images and multislice multispin echo images were obtained before administration of Gd-PGC to measure the ADC and R2, respectively. Diffusion-weighted image data were acquired using a spin echo sequence (repetition time/echo time = 4,000/30 ms; b value = 0, 310, 720, and 1,620 s/mm2; field of view = 1.5 × 1.5 cm2; matrix size = 72 × 72; number of slices = 9). The image acquisition parameters of multislice, multiecho images were as follows: repetition time = 3,000 ms; echo trains = 12; echo time = 10 to 120 ms with the interval of 10 ms; number of repetition = 1; field of view = 1.5 × 1.5 cm2; matrix size = 96 × 96; number of slices = 16. Finally, to verify that the vascular permeability to standard low molecular weight contrast agents is not elevated, 0.1 mmol/kg of Gd-DTPA (Magnevist, Bayer Schering Pharmaceuticals, Berlin-Wedding, Germany) was administered followed by the acquisition of additional 3D-spoiled gradient echo T1-weighted images every 5 minutes up to 15 minutes after injection. The acquisition parameters were identical to those used for Gd-PGC-enhanced images, but with a fixed flip angle of 60°.
Cerebral Blood Volume and Water Exchange Index
Assuming no exchange between intravascular and extravascular compartments, we calculated the apparent fractional blood volume (Vαapp) as follows: 6
where a is the flip angle and SI is the signal intensity. Depending on the flip angle, the calculated apparent blood volume (Vapp) could either reflect the true CBV or be overestimated because of the transvascular water exchange across the BBB. Specifically, application of a small flip angle tended to intensify such overestimation of CBV, whereas the use of a high flip angle (i.e., 90°) estimates the true CBV value.6,9 Therefore, the absolute CBV is defined as (V90app), and WEI representing the rate of water exchange across the BBB can be determined as follows:6,9
Simulation of Cerebral Blood Volume Measurement at Variable Flip Angles
We simulated the water exchange-dependent CBV profile with varying flip angles, as described previously in other published studies.6,7,9,11 Both the water exchange rate (WER) constant and CBV were varied for the simulation of Vαapp using the following parameters: repetition time= 40 ms, precontrast R1 of gray matter = 0.57/s, precontrast R1 of blood = 0.50/s, DR1 in blood induced by Gd-PGC = 10/s and flip angles = 10°, 20°, 40°, 60°, and 90°.
Analysis of In Vivo Measurement
Regions of interest (ROIs) were selected from the infarct cortex in the ipsilateral hemisphere and area-matched normal cortex in the contralateral hemisphere. To minimize partial volume effects, only blood pools (i.e., venous sinus) at least three voxels in width and more than three slices in length were used for selection of the venous blood ROI. We then measured CBV, WEI, ADC, and R2 in both the ipsilateral and contralateral cortices for comparison. Apparent diffusion coefficient and R2 were calculated using voxelwise monoexponential fitting of the signal intensities from diffusion-weighted images and multislice multi spin echo images, respectively. In addition, using ADC maps, we measured the percent total infarct volume by calculating the volume ratio of the entire infarct tissue over the total brain volume. The percent cortical infarct volume was also estimated by calculating the volume ratio of infarct tissue in the cortex over the total volume of the ipsilateral cortex.
In control mice, the left cortex was assigned as ipsilateral, and the right cortex as contralateral because the left MCA was occluded in stroke mice. The 95% confidence interval (CI) of the ipsilateral/contralateral ratio of the WEI in control mice was then calculated. Using this 95% CI as a threshold, the 15 stroke mice were divided into high and normal WEI groups: those with WEIs above 95% CI were grouped in the high WEI group, and those with WEIs within the 95% CI into the normal WEI group. In each mouse, we performed paired comparisons of CBV, WEI, ADC, and R2 in the ipsilateral and contralateral cortices. We then compared CBV, WEI, ADC, R2, and (%) infarct volume of each cortex across the high and normal WEI groups. Finally, the normalized ipsilateral/contralateral ratios of the CBV, WEI, ADC, and R2 for high and low WEI groups were compared.
Percent signal change maps were calculated from images before and after Gd-DTPA administration to delineate any signal enhancement. For detecting the extravasation of Gd-DTPA, signal enhancement is considered as ’positive’ (i.e., leaky) when the spatially averaged ROI percent signal in the ipsilateral cortex is greater than that taken from the contralateral ROI by a standard deviation (acquired from the control group).
Statistical Analysis
Statistical analyses were performed using GraphPad Prism version 5 (GraphPad Software Inc., San Diego, CA, USA). We used the Mann–Whitney U test for unpaired comparisons and the Wilcoxon test for paired comparisons. The correlations of the normalized CBV, ADC, and CBV with WEI were evaluated using Spearman's correlation coefficient. Statistical significance was accepted when the P value was <0.05.
RESULTS
Simulation of Cerebral Blood Volume Measurement at Variable Flip Angles
The CBV calculated at high flip angles approximated the true CBV, whereas the CBV values were overestimated at low flip angles (Figure 1B). Based on the simulation, a flip angle of 90° was expected to accurately predict the true CBV independent of the baseline WER. As shown in Figure 1C, the simulation also showed that the WEI is only very weakly dependent on the baseline CBV for the physiologically relevant ranges observed in the current study (see Table 1), in which only ~6% underestimation of WEI (at WER = 7 Hz) is expected when the fractional CBV changes by 300% from 0.5% to 2%; and thus, the WEI can be used to consistently measure the actual WER.
Water exchange index, cerebral blood volume, apparent diffusion coefficient, and (%) infarct volume
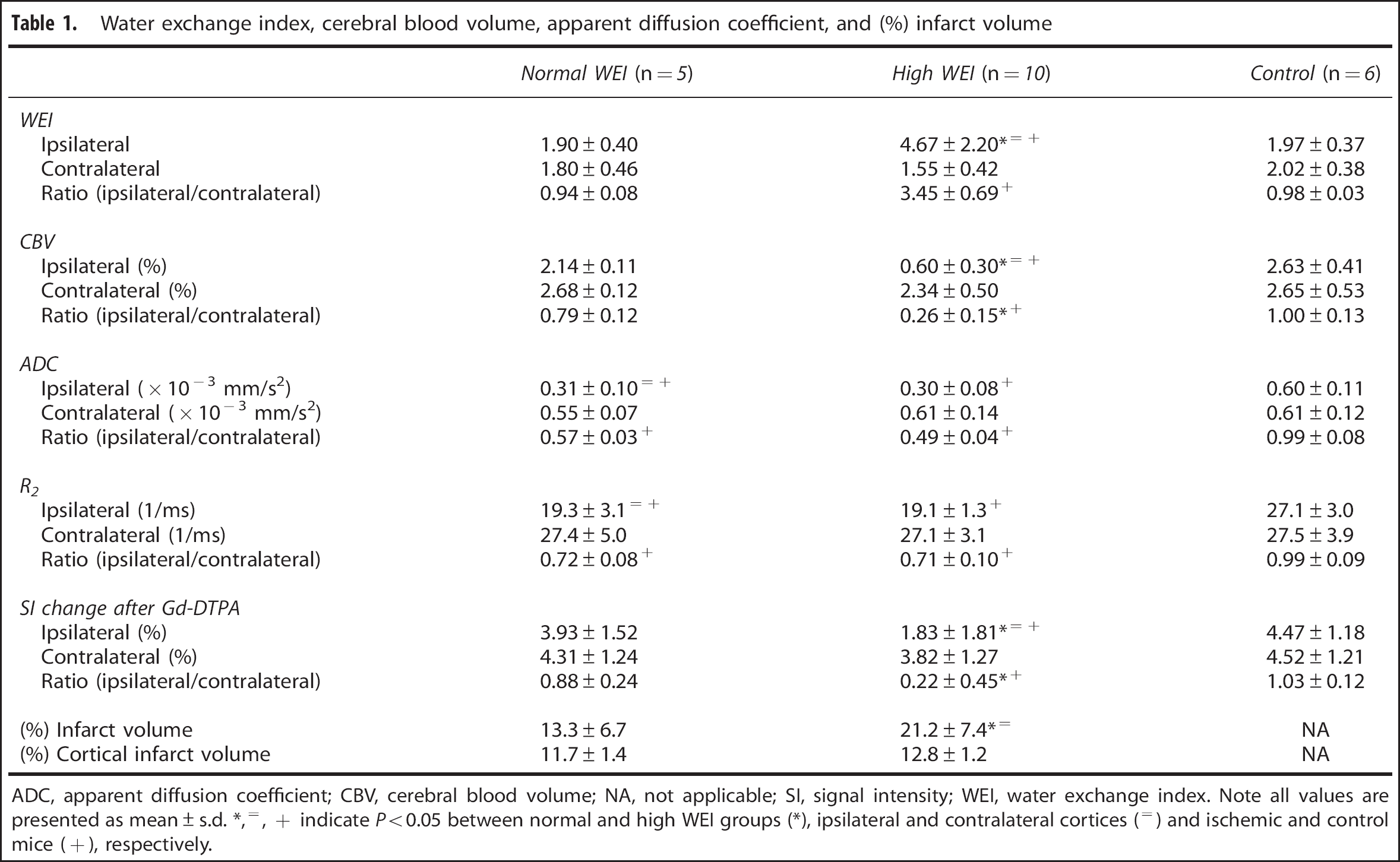
ADC, apparent diffusion coefficient; CBV, cerebral blood volume; NA, not applicable; SI, signal intensity; WEI, water exchange index. Note all values are presented as mean ± s.d. *,=, + indicate P < 0.05 between normal and high WEI groups (*), ipsilateral and contralateral cortices (=) and ischemic and control mice (+), respectively.
In Vivo Study
In all stroke mice, MR angiography using a 3D SPGR sequence and a maximum intensity projection map showed reopening of the MCA after ischemia/reperfusion (see Figure 2A). In addition, gradient echo images obtained after Gd-DTPA administration displayed no contrast agent leakage in any mice (Figure 2C). Stroke mice exhibited ischemic lesions in the ipsilateral cortices, which showed significantly lower ADCs than nonischemic tissues in the contralateral cortices (Figure 2B). In ADC maps, the percent cortical infarct volume was nearly constant (~12%), but the percent total infarct volume varied widely (9% to 35%) because of variation in the infarct volume of the subcortical area.
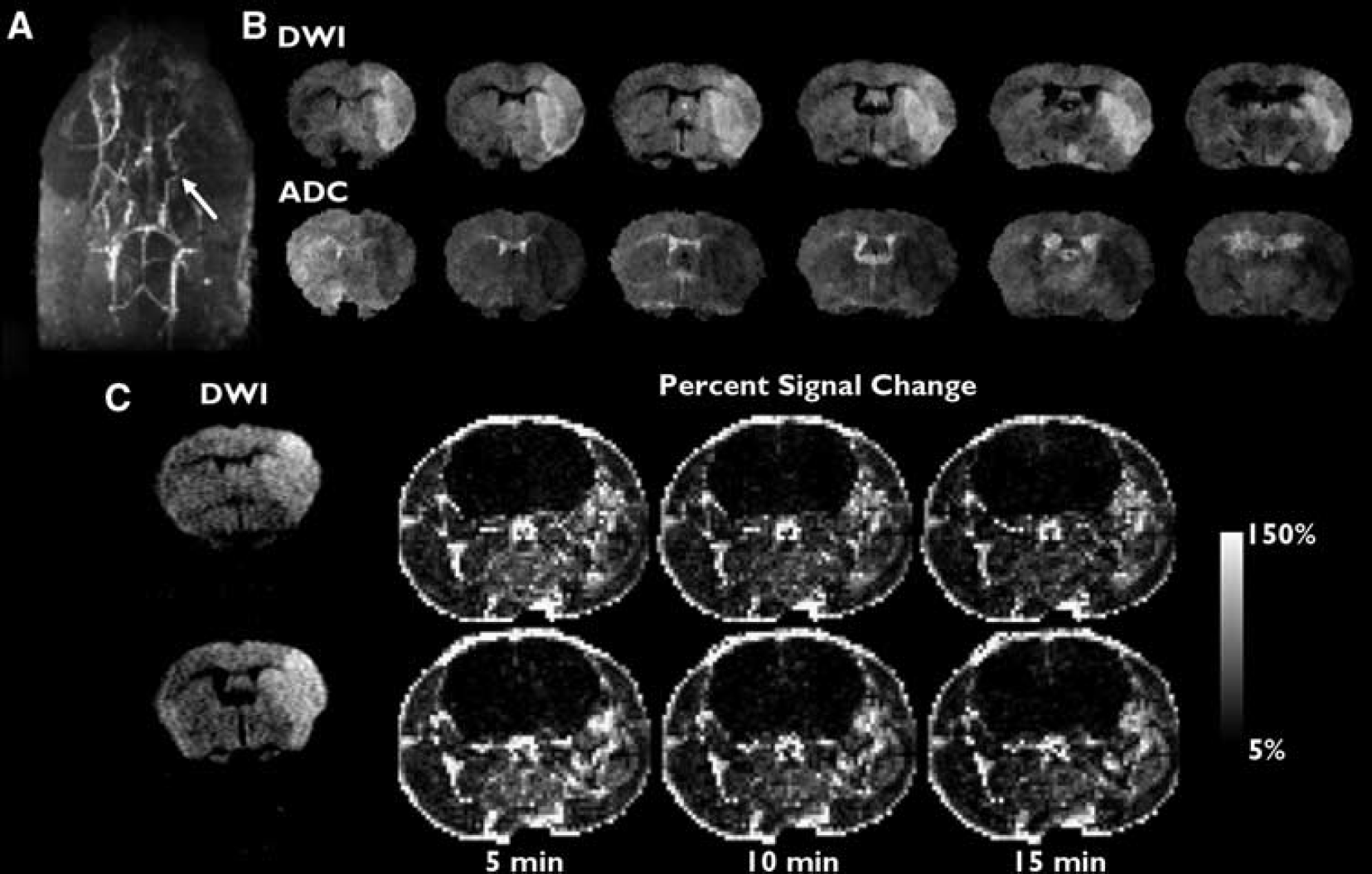
Representative angiogram using maximum intensity projection before Gd-PGC injection (
The CBV, WEI, ADC, R2, and percent signal enhancement after Gd-DTPA and percent total infarct volume are summarized in Table 1, while representative maps of these MRI-derived parameters are shown in Figure 3. The 95% CI of the normalized WEI in control mice was 0.72 to 1.29. According to the group division based on the CI, of the 15 mice with stroke, 10 were assigned to the high WEI group, while the remaining five were assigned to the normal WEI group. No mouse exhibited a normalized WEI below the normal WEI range. While the WEI in the ipsilateral cortex varied greatly (1.25 to 9.25), the WEI in the contralateral cortex (1.12 to 2.90) and bilateral cortices of control mice (1.21 to 2.06) showed relatively narrow WEI ranges (Figure 4). However, there was no significant correlation between ADC and CBV in both high and normal WEI groups according to Spearman's correlation analysis (P > 0.05). As for the signal enhancement after Gd-DTPA administration, contralateral changes in percent signal were always greater than those measured in the ipsilateral cortex for all animal groups (see Table 1). In general, the normalized percent signal increase appeared to match the normalized CBV; however, no statistically significant correlation was found between the percent signal increase and any other parameters including CBV.
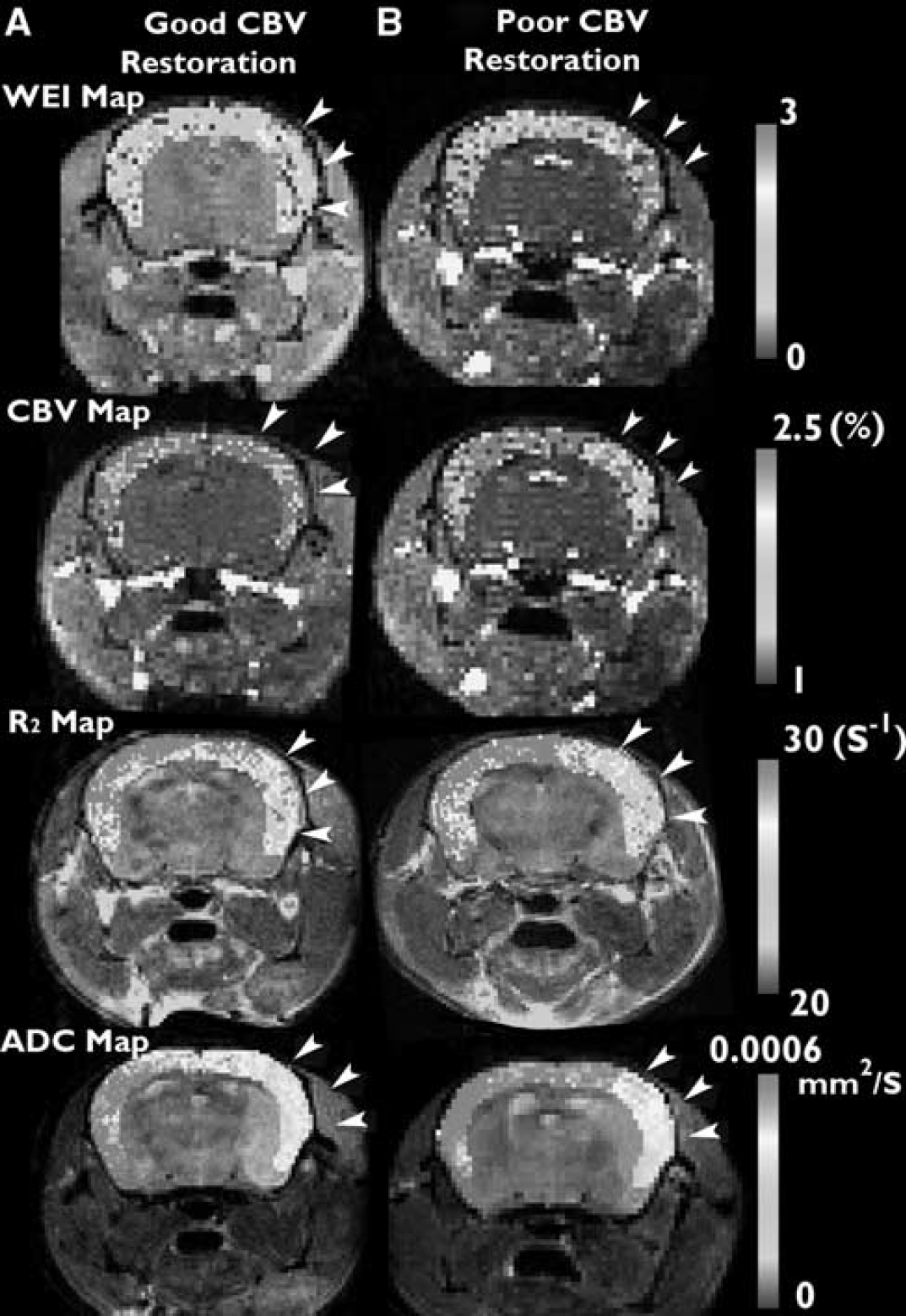
Representative voxelwise cortical color maps of cerebral blood volume (CBV), water exchange index (WEI), R2, and apparent diffusion coefficient (ADC) in mice with good (
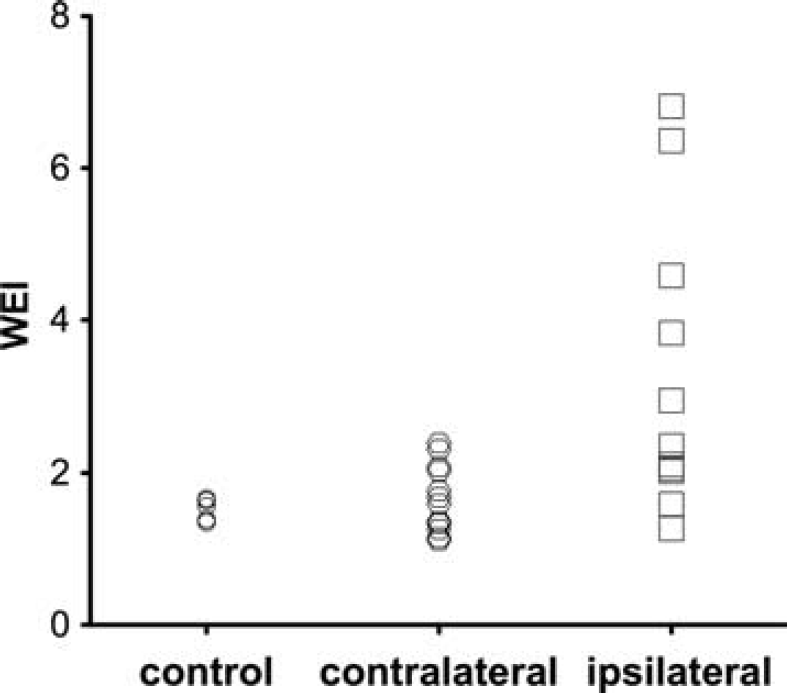
Distribution of water exchange index (WEI) values in the ipsilateral and contralateral cortices of stroke and control mice. The WEI distribution range was much greater in the ipsilateral cortex than that in the contralateral and control cortex. CBV, cerebral blood volume.
In the ipsilateral cortex of stroke mice, the WEI was higher and the CBV was lower in the high WEI group than in the normal WEI group (P < 0.01 for both comparison), although ADC and R2 were not different between the two groups (P > 0.05 for both comparison). In the contralateral cortex, all of the MRI parameters, WEI, CBV, ADC, and R2 were similar across the high WEI, normal WEI, and control groups (P > 0.05). When normalized to the contralateral cortex, the ipsilateral WEI (ipsi/contra) was significantly higher, and the normalized CBV was lower in the high WEI group than those measured in the normal WEI group (P < 0.05 for both comparisons). The percent total infarct volume was also greater in the high WEI group than in the normal WEI group (P = 0.042), while the percent cortical infarct volume was similar between the two groups (P = 0.05). The normalized ADC and R2 were not differentiable between the high and normal WEI groups (P > 0.05 for both comparisons).
In the WEI-CBV scatter plot (Figure 5), the normalized (ipsi/contra) WEI and CBV in stroke mice were widely spread (WEI, 0.84 to 6.07; CBV, 0.09 to 1.18), whereas the control group showed a much narrow distribution of these parameters (WEI, 0.84 to 1.21; CBV, 0.95 to 1.05). In addition, the normalized WEI and CBV showed markedly strong negative correlation (Spearman's rho, −0.9164; P < 0.01). In contrast, there was no significant correlation between either the normalized ADC and WEI or the normalized R2 and WEI (P > 0.05). The WEI-CBV scatter plot showed a clear separation between two distinct groups: those with high WEI and low CBV and those with relatively normal WEI and CBV.
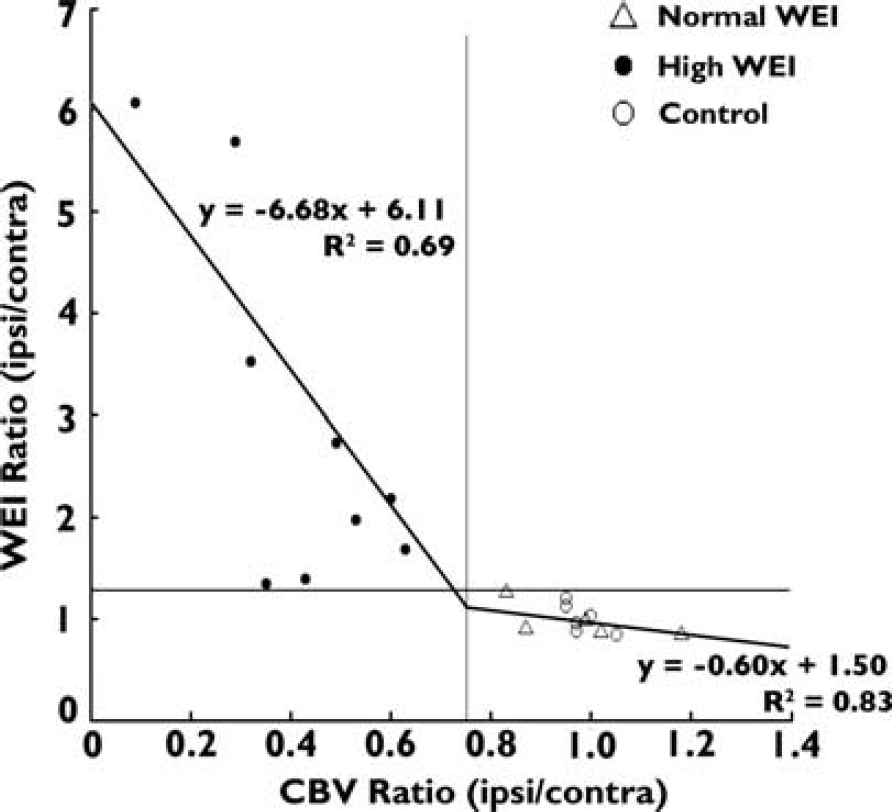
Scatter plot of the normalized cerebral blood volume (CBV) and water exchange index (WEI) (ipsilateral/contralateral cortex) and linear fits using the normal (Δ) and high (∗∗∗•) WEI group data. The high WEI group corresponds to poor CBV restoration, as defined by low CBV values. A CBV ratio of 0.75 represents the critical threshold for maintaining the integrity of the blood–brain barrier (BBB).
Correlation analyses within each WEI group revealed the strong negative correlation between normalized WEI and CBV in both high and normal WEI groups (Spearman's rho, −0.83; P < 0.01 for high WEI and Spearman's rho, −0.91 and P < 0.05 for normal WEI group), in which the slope of the linear fit within each WEI group was –6.68 and −0.61 for high and low WEI groups, respectively. The intercept between the linear fits of the two groups occurs for a CBV ratio (ipsi/contra) of 0.75. This intercept provides a CBV ratio threshold that needs to be achieved for restoration of the BBB integrity. In contrast to CBV and WEI, there was no difference in ADC and R2 between the ipsilateral and contralateral cortices for the high and normal WEI groups; both groups showed ADC and R2 values that were significantly lower in the ipsilateral cortex than in the contralateral cortex (P < 0.05 for both comparisons). The control group showed similar WEI, CBV, ADC, and R2 between the bilateral cortices.
DISCUSSION
Magnetic resonance image has been established as an important tool for diagnosing and, hence, improving prognosis in stroke patients. However, the direct link between affected MRI parameters and early disruption of the BBB—which may lead to vasogenic edema and subsequent hemorrhagic transformation, the most detrimental outcomes in stroke—has not been clarified. To specifically delineate the factors associated with early and nonobvious BBB disruption, we used acute transient stroke models afflicted by relatively low ischemic burden, which were characterized by no Gd-DTPA extravasation at the end of each experiment. We observed a highly heterogeneous distribution of significantly elevated WEI in the ischemic cortex (see Figure 4), suggesting that altered WEI serves as a biomarker to evaluate a wide spectrum of BBB alterations. The highly stable WEI we measured in the control mice also indicates that the current WEI method is a reliable tool for assessing BBB integrity. Furthermore, the simulation results showed that the strong linear correlation between the WEI and WER was nearly independent of the baseline CBV, validating the use of WEI for quantifying the subtle changes in the cerebrovascular integrity from the altered water exchange across the BBB. These results also indicate that the WEI technique has an advantage over other permeability measurement methods that are based on the leakage of exogenous agents, providing a highly sensitive biomarker for monitoring subtle alterations of the BBB in acute stroke.
In the analysis of relationships between MRI-derived parameters and BBB integrity, we found a strong negative correlation between normalized WEI and CBV (ipsilateral/contralateral). The main reason for normalizing the measured values is due to rather a high uncertainty in the WEI quantification even in the normal cortex of healthy mice (standard deviation ~15% of the average value: Table 1). Such uncertainties are most likely because of the variation of the applied flip angle across the subjects. Since we used a volume rf coil, the inaccuracy in flip angle directly relates to the global measurement uncertainty of WEI and CBV. Uncertainty in the MRI parameter control is also readily apparent even in the ADC and R2 measurements, in which the normalization considerably decreases the measurement uncertainty in the control mice (Table 1). Therefore, normalization mitigates the possible global MRI system uncertainty and increases the confidence when studying the relationship between the measured parameters. In this regard, the distribution pattern of normalized WEI and CBV served as the basis for dividing the stroke mice into two groups: one group that exhibited decreased CBV and elevated WEI, and the other with normal CBV and WEI. Based on such dichotomous alterations, we suggest a correlative relationship between the degree of CBV restoration and BBB impairment. Significant correlation between the normalized WEI and CBV within each stroke group (Pearson's correlation, P < 0.05) further reflects a solid link between CBV and WEI even under the different severity of pathophysiologic conditions. However, the slope of the linear fit of the data within each group is distinctive (see Figure 5), indicating that an underreperfused CBV becomes a significant factor in the deterioration of the BBB integrity only when a certain threshold is reached. Specifically, the minimum requirement of CBV restoration in the infarct tissue to maintain (or recover) normal water exchange across the BBB is ~75% of the normal CBV as displayed in Figure 5. Notably, the couple of anomalous data points in the high WEI group (i.e., low CBV and normal WEI: Figure 5) were probably because of the collapsed small arteries, prohibiting the distribution of the contrast agent, in which the CBV and WEI measurements would be confined to the reperfused vessels and relevant areas only. We hypothesize that this local exclusion of the contrast agent from affected downstream capillaries (high WEI) because the irrecoverably collapsed arterioles would limit the signal enhancement and result in a low CBV and normal WEI.
Physiologically, the failure of complete restoration of the CBV can result from the ’no-reflow’ phenomenon, that is, lingering obstruction of the downstream microvasculature, even after the main feeding artery is reopened. 21 This condition is initially induced by intravascular coagulation and endothelial swelling. 21 Subsequently, progressive lactic acidosis due to insufficient reflow exacerbates edema in the neurovascular unit, thereby aggravating vascular occlusion and interfering with the restoration of blood perfusion.21,22 Despite the direct relevance of cerebral blood flow for assessing the reperfusion status, we instead elected to quantify the steady-state CBV due to the technical convenience (e.g., no model fitting) and relatively high measurement accuracy and precision of the method. Since a strong correlation between the cerebral blood flow and CBV is almost always observed, we posited that the status of CBV restoration may reveal the degree to which the ischemic tissue is entrapped in a series of pathologic processes that inflict further BBB damage. In fact, our results showed that the restored CBV alone may be used to predict the normalization of vascular integrity. As such, CBV restoration influences the BBB status, and ’no-reflow’ is suggested to be the primary cause of stroke-related alterations in CBV stability and BBB integrity. Cessation of the ’no-reflow’ cascade seems imperative for preventing devastating stroke-related complications such as hemorrhagic transformation or severe vasogenic edema.
Several lines of molecular and histologic evidence also support that the deterioration of BBB integrity in transient stroke is directly related to incomplete restoration of CBV. First, poor CBV restoration after ischemia/reperfusion induces BBB damage by activating variable proteases such as tissue plasminogen activator, matrix metalloproteinases, cathepsins, and heparanases.23–25 These enzymes accelerate degradation of the extracellular matrix of the BBB, consequently irreparably damaging the BBB. Second, continuing hypoxia caused by poor CBV restoration stimulates integrin-mediated cell detachment from the extracellular matrix. The onset of this process aggravates apoptosis in the neurovascular unit, worsening the damage to the BBB.26,27 Third, CBV insufficiency evokes vascular conditions refractory to the immediate benefit of nitric oxide (NO), an important neuroprotective vasodilator produced in response to ischemia. The failed CBV restoration further induces excessive synthesis of NO, leading to heightened BBB impairment. Hazardous effects from nitrosative stress, which is well known to impair the neurovascular unit, become dominant over the neuroprotective function of vasodilation by NO. 28 Therefore, NO overproduction caused by poor restoration of CBV eventually aggravates disruption of the BBB.28–30 Other possible mechanisms for the elevated water exchange include, but are not limited to, the upregulation of AQP4 at a highly acute stage of reperfused mouse brains and disjointing of the endothelial tight junctions.31,32
Many previous studies have reported significantly decreased ADC and R2 in cases of overt BBB disruption, suggesting that these parameters may be correlative indices for describing stroke-induced BBB damage.13–17 On the contrary, in the current study, we observed similar infarct ADC and R2 values that were independent of the WEI. The discrepancy principally results from different scales of BBB injury between studies. Whereas our current study classified the degree of BBB impairment in the absence of extravasation, other previous studies categorized the BBB status by the permeability of leaky exogenous contrast agent through the BBB based on the dynamic contrast enhanced MRI method. 33 Therefore, considering that the previous correlation between BBB damage and stroke-induced ADC and R2 changes was determined only under the severe ischemic burden that accompanies overt BBB breakdown, ADC and R2 are not adequate parameters for evaluating mild or early BBB impairment caused by moderate ischemic burden. However, the experimental ischemia in this study yielded a wide range of percent total infarct volume, despite the lack of Gd-DTPA extravasation, implying variable ischemic damage caused by even a moderate ischemic burden. While the traditional ADC and R2 values that are spatially averaged over the infarct area in the ipsilesional cortex did not correlate with other MRI-derived parameters, the cortical WEI values correlate significantly with the percent total infarct volume. Therefore, we suggest that the WEI is not only useful as a biomarker for assessing the BBB integrity, but also has the range and sensitivity needed to predict the stroke outcome under moderate ischemic burden. Conversely, the percent total infarct volume may be used as an indirect measure for predicting the severity of the overall BBB impairment in acute transient stroke.
Considering the time course of ischemia/reperfusion and the variable degree of alteration in BBB integrity, our transient stroke models mimic stroke patients who undergo thrombolysis treatment for recovery of cerebral reperfusion. In this regard, we suggest that postthrombolytic CBV and WEI can serve as valuable surrogates for predicting the outcome of reperfusion treatment. Although cerebrovascular permeability measurements based on the leakage of contrast agents have been previously suggested to predict stroke patient outcomes,33,34 at premature stages of stroke progression, corresponding to no extravasation of standard contrast agents, elevated WEI and poor CBV restoration may provide an early warning sign of devastating postreperfusion complications. However, open factors such as biphasic opening of postischemic BBB and a relatively weak link between early permeability increase and eventual hemorrhagic transformation warrant investigations for elucidating technical advantage in WEI and involvement of other directly causative factors.35,36 While further verification with pathologic indices is required for confirming such prognostic potential, the proposed WEI method provides simultaneous assessment of water permeability and CBV, which are closely related to the endogenous sources accountable for vasogenic edema and hemorrhagic transformation.
As supportive or alternative treatment options to tissue plasminogen activator-using thrombolysis, various neuroprotective drugs are being investigated to strengthen the stability of the BBB and to improve CBV restoration. Such therapeutic agents are expected to reduce the risk of thrombolysis-related complications as well as the extent of ischemic tissue injury. For instance, simultaneous administration of tissue plasminogen activator and a matrix metalloproteinase inhibitor may decrease the extent and severity of stroke-induced BBB damage, 28 and inhibition of NO production is expected to improve the neurologic deficit score by reducing infarct volume and edema. 22 In this regard, we propose that the CBV and WEI may be useful for monitoring the effectiveness of therapeutic agents under development.
To evaluate ischemic damage in the cerebral vasculature, a number of studies have used relative CBV, quantified by analyzing time-intensity curves from dynamic susceptibility contrast MRI or perfusion computed tomography. Despite the popularity of MRI-derived relative CBV, it provides limited intrasubject and intersubject reliability because the measurement depends significantly on image-acquisition techniques, baseline tissue states, a possibly nonlinear relationship between CBV and tissue signal contrast, and analysis algorithms. 37 Unlike previous studies that measured relative CBV, this study estimated absolute CBV by calculating the degree of steady-state contrast enhancement, which is induced by the administration of Gd-PGC, a long-circulating intravascular agent. In contrast to relative CBV measured by dynamic contrast methods, absolute CBV provides a reliable measurement that is less affected by the variable neurovascular milieu and efficiently allows for crosssubject comparisons. However, it is notable that, despite the schematic simplicity, MRI signal, thus the absolute CBV measurement was significantly affected by the strong inflow of arterial blood in subcortical structures for the current study set-up. Some straightforward adjustments in the MRI acquisition method, the use of separate rf transmit over a larger region (e.g., body coil) and receive only head coil, will easily eliminate the unwanted inflow effects in rodent model studies using WEI.
A potential limitation of our study is that Gd-PGC is not clinically approved. Fortunately, another intravascular contrast agent, an albumin-binding Gd-chelate, has been recently approved and has become commercially available for clinical applications (Ablavar, Lantheus Medical Imaging, North Billerica, MA, USA). However, to use this relatively new intravascular agent to measure WEI and CBV, it is necessary to clarify its pharmacological properties such as the range of molecular size, clearance pattern, and stability. As another alternative, the use of small molecular Gd-based contrast agents, such as Gd-DTPA, can be considered. These agents have the advantage that they are already FDA approved and widely used in clinical practice. However, measurement of the WEI and CBV using relatively small molecular agents may be limited since they can be used only when there is no contrast leakage across the BBB. Moreover, because steady concentration of contrast agent is a prerequisite for measuring the WEI consistently, the effect of rapid clearance from the vascular space should be investigated. 38 In addition, we recently explored the use of clinically approved iron-oxide nanoparticles (Faraheme, AMAG Pharmaceuticals, Lexington, MA, USA) for quantifying the WEI in combination with ultrashort echo time imaging sequences. 39 The ultrashort echo time technique is particularly enticing since no correction for T2∗ relaxation, caused by almost any MRI contrast agents, is required. Although preliminary, the ultrashort echo time study results show that intravascular contrast agents commonly used to alter the magnetic susceptibility can also be used for quantifying the WEI.
In summary, using a transient stroke model exposed to a moderate ischemic burden, which does not allow extravasation of Gd-DTPA, we showed that the WEI and CBV are sensitive parameters for quantifying stroke-related vascular alterations immediately after reopening of the temporally occluded cerebral arteries. We also showed that the degree of baseline CBV restoration is a strong determinant factor for maintaining BBB integrity, which can be assessed via the WEI when contrast agents do not leak across the BBB. The study results provide an important basis for understanding and designing therapeutic strategies for the management of reperfused ischemic cerebral tissue.
DISCLOSURE/CONFLICT OF INTEREST
The authors declare no conflict of interest.
Footnotes
ACKNOWLEDGEMENTS
The authors gratefully acknowledge financial support from the American Heart Association (SDG #0835451N), the Basic Science Research Program (No. 2011-0028826), and Advanced Research Center (No. 2012-0008999) through the National Research Foundation of Korea (NRF) funded by the Ministry of Education, Science of Technology and the National Institutes of Health (K25-AG029415).