Abstract
The therapeutic benefits of bilateral capsulotomy for the treatment of refractory obsessive compulsive disorder (OCD) are probably attributed to interruption of the cortico-striato-thalamo-cortical circuitry. We evaluated resting brain metabolism and treatment response in OCD patients using positron emission tomography (PET) imaging. [18F]-fluoro-deoxy-glucose PET was performed in eight OCD patients precapsulotomy and postcapsulotomy. We determined metabolic differences between preoperative images in patients and those in eight age-matched healthy volunteers, and postoperative changes and clinical correlations in the patients. The OCD patients showed widespread metabolic increases in normalized glucose metabolism in the bilateral orbitofrontal cortex and inferior frontal gyrus, cingulate gyrus, and bilateral pons/cerebellum, and metabolic decreases bilaterally in the precentral and lingual gyri. Bilateral capsulotomy resulted in significant metabolic decreases bilaterally in the prefrontal cortical regions, especially in the dorsal anterior cingulate cortex (ACC) and in the medial dorsal thalamus and caudate nucleus. In contrast, metabolism increased bilaterally in the precentral and lingual gyri. Clinical improvement in patients correlated with metabolic changes in the bilateral dorsal ACC and in the right middle occipital gyrus after capsulotomy. This study underscores the importance of the internal capsule in modulating ventral prefrontal and dorsal anterior cingulate neuronal activity in the neurosurgical management of OCD patients.
Keywords
INTRODUCTION
Obsessive compulsive disorder (OCD) is a chronic anxiety disorder marked by recurrent, intrusive, and distressing thoughts (obsessions) or repetitive behaviors (compulsions). It is the world's most common mental disorder with a lifetime prevalence of 2% to 3% in both pediatric and adult population, with up to 80% of all cases having their onset in childhood and adolescence. 1 Despite its high prevalence and the attendant morbidity, the pathophysiology of OCD remains unclear.
As a disabling mental illness that tends to be chronic unless treated effectively, it affects patients’ social and professional lives. Some patients have a severely reduced quality of life and at least 30% to 40% of patients with OCD fail to respond to first-line pharmacologic or psychotherapeutic treatments.2,3 For treatment-refractory OCD, neurosurgery such as capsulotomy, cingulotomy, limbic leucotomy, subcaudate tractotomy, and electrostimulation are sometimes used.4–6 In capsulotomy, lesions are placed in the anterior limb of the internal capsules (IC), and the evidence on the efficacy of capsulotomy in OCD is based on a number of case series reporting significant clinical improvement in 53% to 73% of patients.6–8
The cortico-striato-thalamo-cortical circuitry involving the orbitofrontal cortex (OFC) has been repeatedly implicated in the neuropathophysiology of OCD. Functional brain imaging with positron emission tomography (PET) and [18F]-fluoro-deoxyglucose (18F-FDG) has been used to identify specific brain regions that contribute to disease onset and underlying therapeutic effects in OCD patients. 9 The most consistent neuroimaging findings in patients with OCD symptomatology alone are relative hypermetabolism in OFC and anterior cingulate cortex (ACC) that can be reduced by pharmacologic or behavioral therapy. 10 [18F]-fluoro-deoxy-glucose PET has also been used to study the mechanism of anterior cingulotomy11,12 and anterior capsular electrostimulation.13,14 However, the underlying mode of action by which bilateral capsulotomy suppresses the dysfunctional OCD circuitry is largely unknown. In a clinical study on long-term efficacy of this procedure, 15 we have reported preliminary data on metabolic changes in a few hyperactive brain regions using a volume of interest (VOI) analysis. In this retrospective study, we determined region-specific metabolic effects of bilateral capsulotomy using voxel-based univariate brain mapping analysis over the whole brain. Our primary goal was to establish an unbiased data-driven approach to confirm the validity of the key hypothesis developed in earlier studies and to reveal functional differences in areas not previously considered.
MATERIALS AND METHODS
Subjects
All subjects described in this study took part in a clinical neurosurgical protocol established at Huashan Hospital affiliated with Fudan University in Shanghai, China. Thirty-five treatment-refractory patients with OCD (mean age ± s.d., 29.6 ± 10.6 years; duration of illness 10.8 ± 4.5 years) were originally enrolled under this protocol. 15 All patients who were registered met the clinical criteria for OCD according to the Diagnostic and Statistical Manual of Mental Disorders, fourth edition (DSM-IV). 16 The patient and their legal guardian gave consent to participate in the protocol that involved both capsulotomy and 18F-FDG PET imaging before and after the neurosurgery. Informed consent was delivered both in written form and orally, emphasizing in particular the potential health risks associated with neurosurgical procedures and radiation exposure in imaging studies. The signed consent form contained a description of the patient's rights in agreement with the Helsinki Declaration. Patients were considered to have refractory OCD and selected to undergo the neurosurgery if they met the following criteria: (1) the patient had been treated with medication, psychotherapy, or cognitive behavioral therapy for >5 years, and clinical symptoms had not improved or had worsened; (2) the disorder affected the quality of life of the patient, preventing their participation in normal activities such as work or study; (3) the patient had no cognitive deficits, severe heart disease, or clotting disorders; and (4) the patient showed relative orbitofrontal hypermetabolism on preoperative (Pre-Op) PET, a positive indication of OCD. All procedures were conducted in accordance with the institutional regulations and ethical guidelines.
In the present study, we selected eight right-handed patients with OCD (female/male 5:3; age 22.0 ± 5.2 years, range 16 to 29 years; duration of illness 8.6 ± 2.3 years, range 6 to 12 years) from the original cohort. This sample of three adolescents (age 16 to 18 years) and five young adults (age 20 to 29 years) came from our early patients who had no comorbid depressive symptoms was operated by the same neurosurgeon and scanned on the same PET camera before and after the surgery. They had high-quality 18F-FDG images available for voxel-based analysis. Only right-handed patients and controls were selected to remove the confounding effects of handedness on the results. This retrospective study had been approved by the institutional review board at Huashan Hospital of Fudan University. Demographic and clinical information were provided in Table 1. Clinical symptoms of all patients were quantitatively assessed before and after capsulotomy using Yale-Brown Obsessive Compulsive Scale (Y-BOCS) and Hamilton Depression Scale (HAM-D). These scores were recorded by an independent psychiatrist at the time of each PET study described below.
Patient characteristics and clinical information
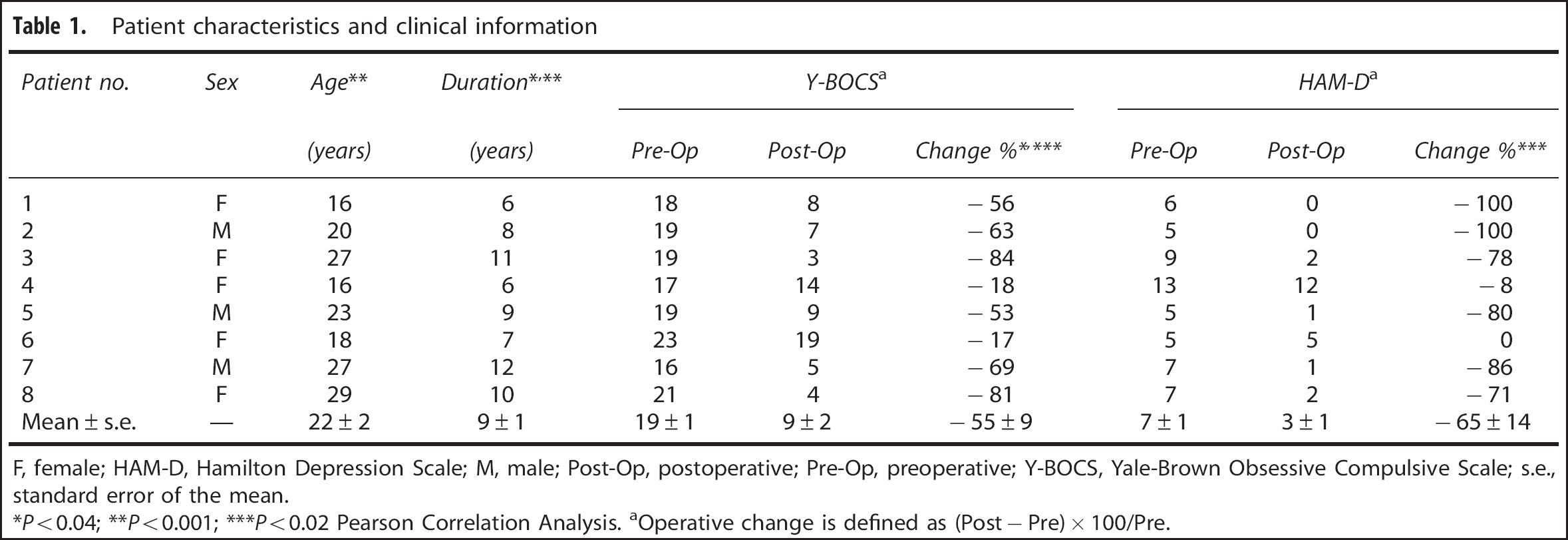
F, female; HAM-D, Hamilton Depression Scale; M, male; Post-Op, postoperative; Pre-Op, preoperative; Y-BOCS, Yale-Brown Obsessive Compulsive Scale; s.e., standard error of the mean.
*P < 0.04;
**P < 0.001;
***P < 0.02 Pearson Correlation Analysis.
aOperative change is defined as (Post – Pre) × 100/Pre.
Yale-Brown Obsessive Compulsive Scale has total maximal score of 40, 20 for obsessive symptoms and 20 for compulsive symptoms. Although the total score in our sample was in the range of moderate at baseline (16 to 23), they were actually considered as severe patients given the fact that the dominant symptoms of individual patients were either obsession or compulsion. Hamilton Depression Scale used in this study was the 17-item version with a maximal score of 54. Patients in our sample had no major symptoms of depression at baseline as their scores (5 to 13) were either normal or only in the mild range.
Eight age-matched female normal volunteers (age 21.5 ± 5.9 years; right handed) with 18F-FDG PET imaging on the same camera were also included for this study and written informed consent was obtained from each volunteer. The normal volunteers had no history of neurologic or psychiatric diseases and had no abnormalities on brain magnetic resonance imaging (MRI).
Target Designation and Operative Technique
For MRI-guided target localization, the Leksell Series G stereotactic system (Electra, Atlanta, GA, USA) was positioned under local anesthesia. 15 For MRI targeting, a 1.5 Tesla Sigma Unit (GE Healthcare, Beijing, China) was used with T2- and proton-weighted echo sequences in the axial and coronal planes. Images (slice thickness = 2 mm) were obtained parallel to one of the axes of the frame and parallel or orthogonal to the anterior commissure-posterior commissure line. Both the target's coordinates and the angles of electrode penetration were calculated in the MRI workstation using the SurgiPlan software system (Electra 2.1). The target for the anterior limb of IC was located as follows: 22 to 25 mm anterior to the middle of the anterior commissure-posterior commissure line, horizontal to the anterior commissure-posterior commissure, and 18 to 20 mm lateral to the midline. Electrodes for the capsulotomy were then placed on the skull, one on each side.
After the placement of each electrode in the target, 100 Hz stimulation was applied and rigidity was measured to confirm the position of the electrode relative to the IC and to avoid the electrode penetrating too deeply into the genu of the IC and posterior limb. Likewise, the position of the electrode to the optic nerve bundle was confirmed by checking for blurred vision or loss of the visual field. After confirming the targets, lesions were generated using radio-frequency energy (Radionics, Burlington, MA, USA) at 70°C and 80°C for 60 seconds each. Three lesions were created on each side using this procedure and were located in the anterior capsule at approximately one third of the way anterior and ventral based on postoperative (Post-Op) MRI (see Supplementary Figure 1).
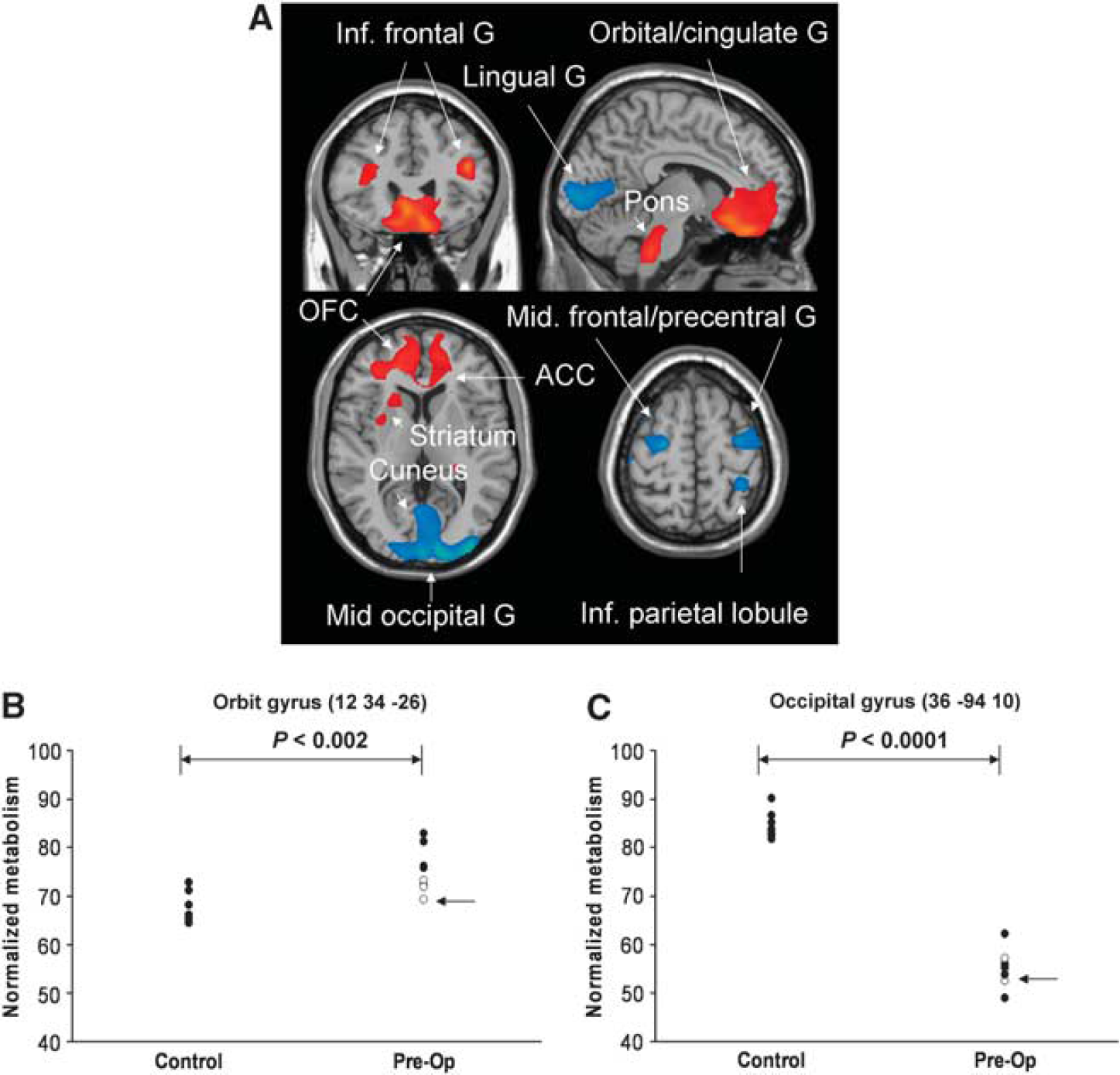
Brain regions with significant metabolic differences in obsessive compulsive disorder (OCD) patients compared with normal subjects. (
Positron Emission Tomography Imaging Procedure
In a fasting state, all patients who received bilateral capsulotomy also underwent 18F-FDG PET before and after the surgery. The Pre-Op PET was conducted in all patients ~3.0 ± 1.1 days before surgery (range 2 to 5 days). The Post-Op study was performed in all patients ~6.4 ± 0.2 months after surgery (range 6.1 to 6.6 months). All patients were asked to withdraw medications for at least 12 hours before PET imaging. The levels of serum glucose before FDG injection were in normal range (average 5.1 ± 0.3 mol/L, range 4.7 to 5.5 mmol/L). The PET studies were performed with an ECAT EXACT HR+ tomography (CTI, Knoxville, TN, USA) operating in three-dimensional mode. This scanner acquires 63 image slices with an interslice spacing of 2.43 mm, a total axial field of view of 15.5 cm, and an axial and transverse intrinsic resolution of 4.1 and 4.3 mm at the center of the field of view. After a 10-minute transmission scan, a PET scan of 10 minutes duration was performed 30 minutes after an intravenous bolus injection of 18F-FDG. Injected dose per unit of body weight was 0.15 to 0.20 mCi/kg in adults but reduced to ≤0.10 mCi/kg in children (<18 years old), well within the dosimetry guidelines published by the Society of Nuclear Medicine (The Procedure Guideline for FDG-PET Brain Imaging v1.0 available online; http://www.snm.org/). Images were reconstructed using a three-dimensional filtered-backprojection method with Hanning filters. No arterial blood samples were taken since we measured relative changes in regional glucose metabolism. All studies in patients and normal controls were performed using the same imaging protocol with the subject's eyes open in a dimly light room with minimal auditory stimulation.
Data Analysis
The PET images were preprocessed with statistical parametric mapping (SPM5, Wellcome Department of Cognitive Neurology, London, UK) running in Matlab 6.5.1 (Mathworks, Sherborn, MA, USA). Scans from each patient were realigned between precapsulotomy and postcapsulotomy, and spatially normalized into MNI (Montreal Neurological Institute) brain space with linear and nonlinear three-dimensional transformations via the mean image after the realignment. The normalized PET images for patients and healthy volunteers were filtered with a Gaussian kernel of 10 mm (full width at half maximum) in the x, y, and z axes, respectively. Mean signal differences over the whole brain were removed by proportional scaling in each individual subject.
Changes in regional glucose metabolism before and after bilateral capsulotomy.
To characterize Pre-Op metabolism in OCD patients versus. healthy subjects, we performed a two-sample t-test in SPM5 according to the general linear model at each voxel. The effects of bilateral capsulotomy on regional brain metabolism were also estimated using a paired t-test. To evaluate significant differences, we set the peak threshold at P = 0.001 (uncorrected) over whole brain regions, a criterion used in several previous studies.13,17 For hypothesis generation, we only reported clusters that survived a false discovery rate (FDR) correction at P < 0.025 and with an extent threshold of 40 voxels (corresponding to a tissue volume of 320 mm3). Significant regions were localized by Talairach Daemon software after using an MNI-to-Talairach conversion tool. 18 The SPM maps for increased or decreased metabolism were overlaid on a standard T1-weighted MRI brain template in stereotaxic space.
To quantify regionally specific metabolic changes, we constructed a 4-mm radius spherical VOI within the image space, centered at the peak voxel of clusters that were significant in the unpaired and paired t-tests, respectively. We then calculated the relative glucose metabolism (i.e., globally normalized) in normal, Pre-Op, and Post-Op PET images with the VOI option in SPM5.
Correlations between clinical score and glucose metabolism before and after bilateral capsulotomy.
Using SPM5 we correlated Pre-Op Y-BOCS scores with PET images to evaluate whether specific brain regions would correlate with the measures of clinical severity in OCD patients. To determine the relationship between clinical outcome and operative metabolic changes, we also correlated the percentage changes in Y-BOCS score with the subtraction images between globally normalized Pre-Op and Post-Op PET images across the patient cohort. As exploratory analyses, these hypothesis-driven searches were conducted and considered significant at P < 0.01 (uncorrected), with an extent threshold of 40 voxels and significant at P < 0.05 at the cluster level. The SPM maps of correlations between Pre-Op Y-BOCS scores and regional metabolism or their operative changes were overlaid on the MRI brain template.
We also calculated the relative metabolism in Pre-Op and subtraction PET images using a 4-mm radius VOI within the image space, surrounding the voxel of maximal significance in the corresponding correlation analysis. Correlations between Pre-Op clinical scores and metabolic values or their interval changes were assessed using Pearson correlation coefficients and were considered significant at P < 0.05.
RESULTS
Clinical Response after Bilateral Capsulotomy
The clinical characteristics of the OCD patients were presented in Table 1 and Supplementary Figure 2. There were significant reductions in clinical scores of Y-BOCS (mean/s.e.: 55 ± 9%, P < 0.001) and HAM-D (65 ± 14%, P < 0.002) 6 months after the operation. While six of the patients (75%) were regarded as clinically improved (Y-BOCS decreased by 53% to 84%; HAM-D decreased by 71% to 100%), two of them (25%) showed suboptimal improvement or no effect (Y-BOCS decreased by <18%; HAM-D decreased by <8%). Of note, the percentage reduction in Y-BOCS was correlated positively with that in HAM-D (R 2 = 0.65; P = 0.016), as well as negatively with disease duration (R 2 = 0.56; P = 0.033) and patient age (R 2 = 0.64; P = 0.018) at Pre-Op baseline. In addition, there was a positive correlation between disease duration and patient age at the time of operation (R 2 = 0.88; P < 0.001).
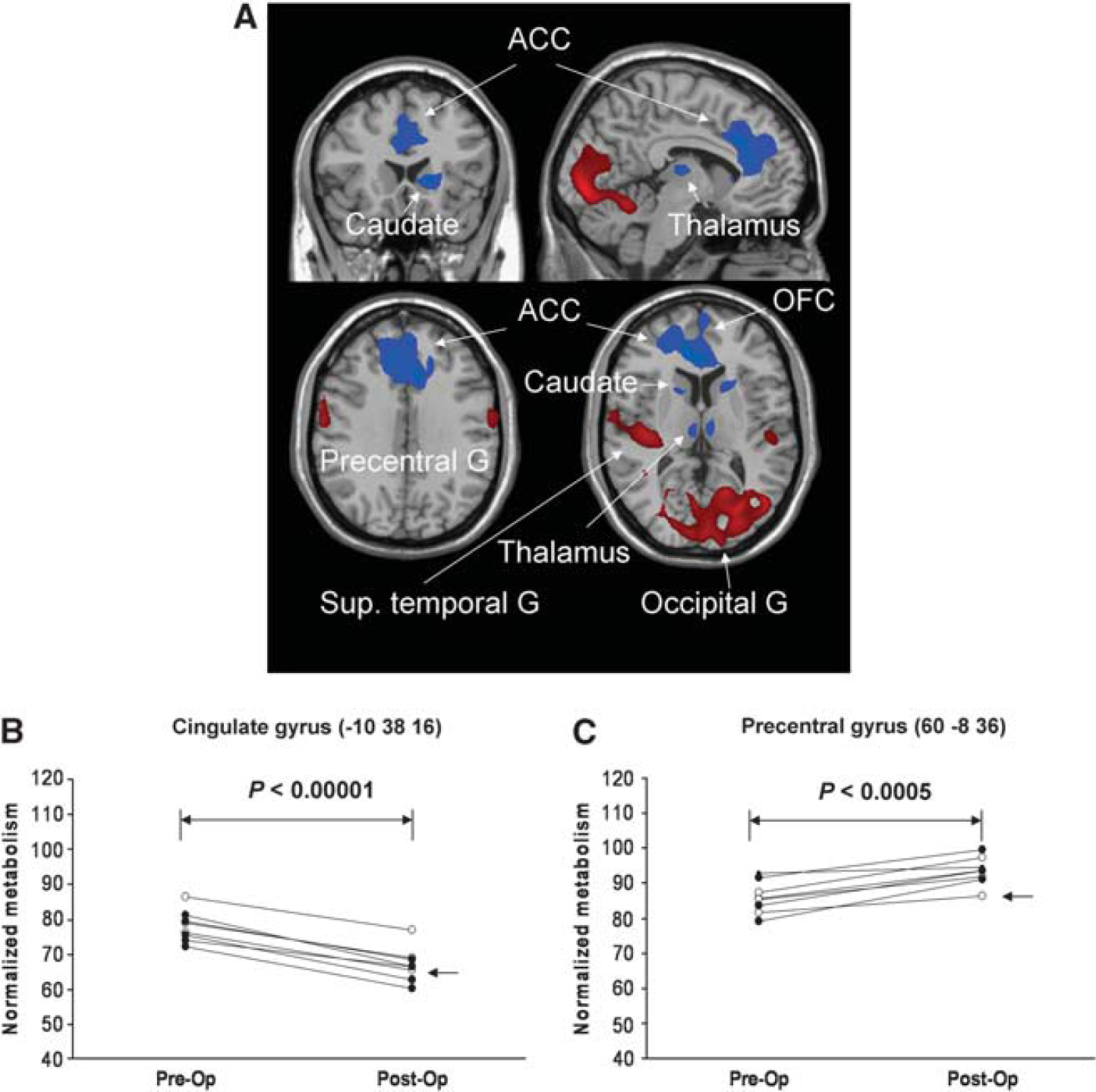
Brain regions with significant metabolic changes in obsessive compulsive disorder (OCD) patients after bilateral capsulotomy. (
Preoperative Glucose Metabolism
Compared with healthy volunteers, the OCD patients showed significant and widespread differences in normalized regional glucose metabolism at Pre-Op baseline in both cortical and subcortical brain regions (Table 2; Figure 1). Relative hypermetabolism occurred in the bilateral prefrontal cortex (including rectal/orbital gyri, ACC, as well as inferior frontal gyrus and its triangular parts), left posterior cingulate cortex (PCC; Brodmann's area (BA) 31), and parahippocampal gyrus, and bilaterally in the middle temporal gyrus (BA 38), pons and posterior cerebellar tuber. The hypermetabolic cluster enclosing the OFC also extended bilaterally to the ventral caudate and IC, and to a smaller area located in the left putamen. In contrast, relative hypometabolism was evident in the left middle frontal (BA 6) and bilateral precentral gyri (i.e., supplementary motor area (SMA)), right inferior and bilateral superior (BA 7) parietal lobe, and right middle occipital gyrus (BA 19) extending to bilateral cuneus/lingual gyrus. All these regions also survived at FDR-corrected P < 0.01. This pattern of abnormal regional metabolism was evident in exploratory single case analysis in which each of the OCD patients was compared separately with the same sample of healthy volunteers using SPM5 (Supplementary Figure 3).
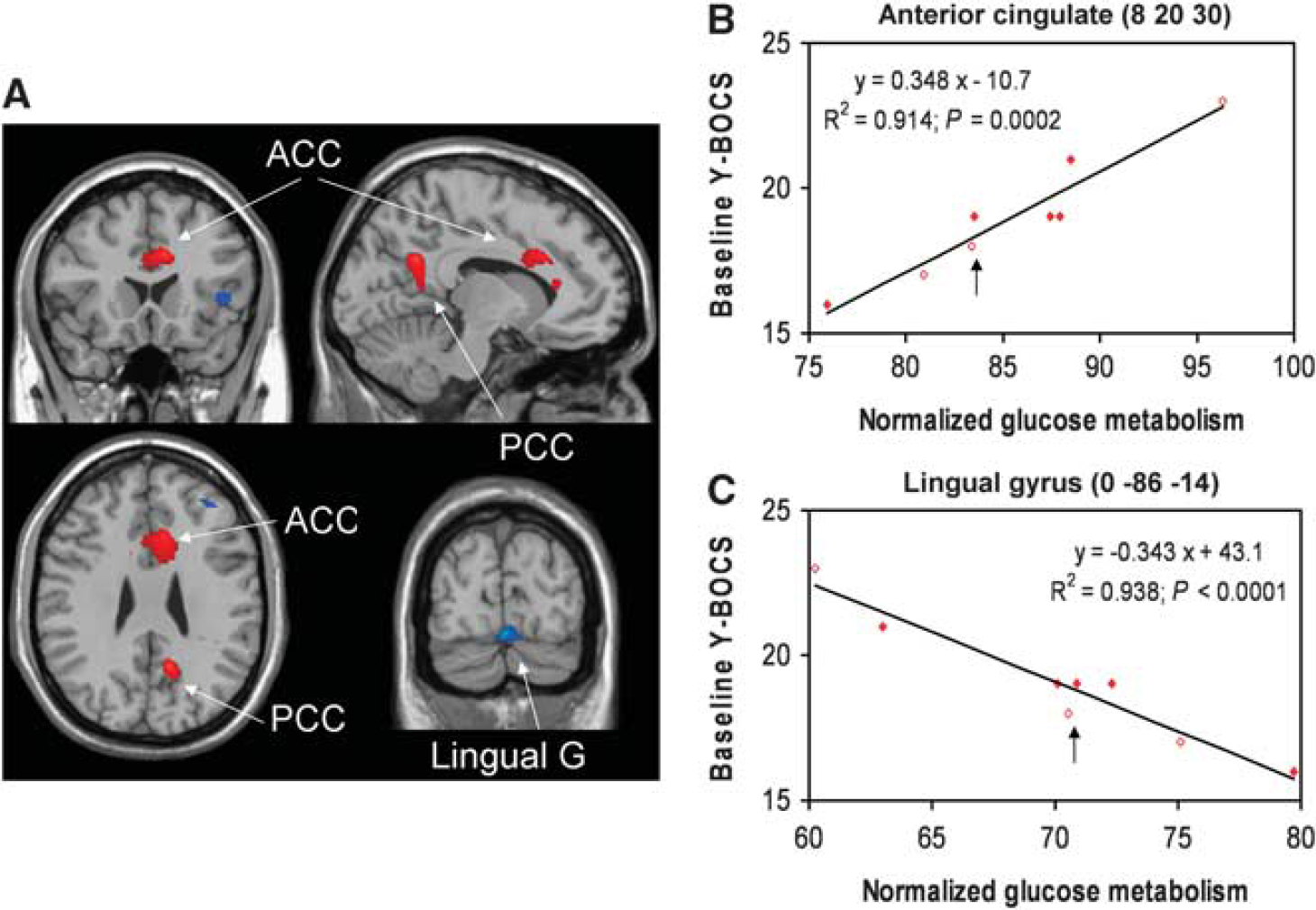
Brain regions with significant correlations between clinical scores and glucose metabolism at preoperative (Pre-Op) baseline. (
Brain regions with significant metabolic changes in OCD compared with normal subjects
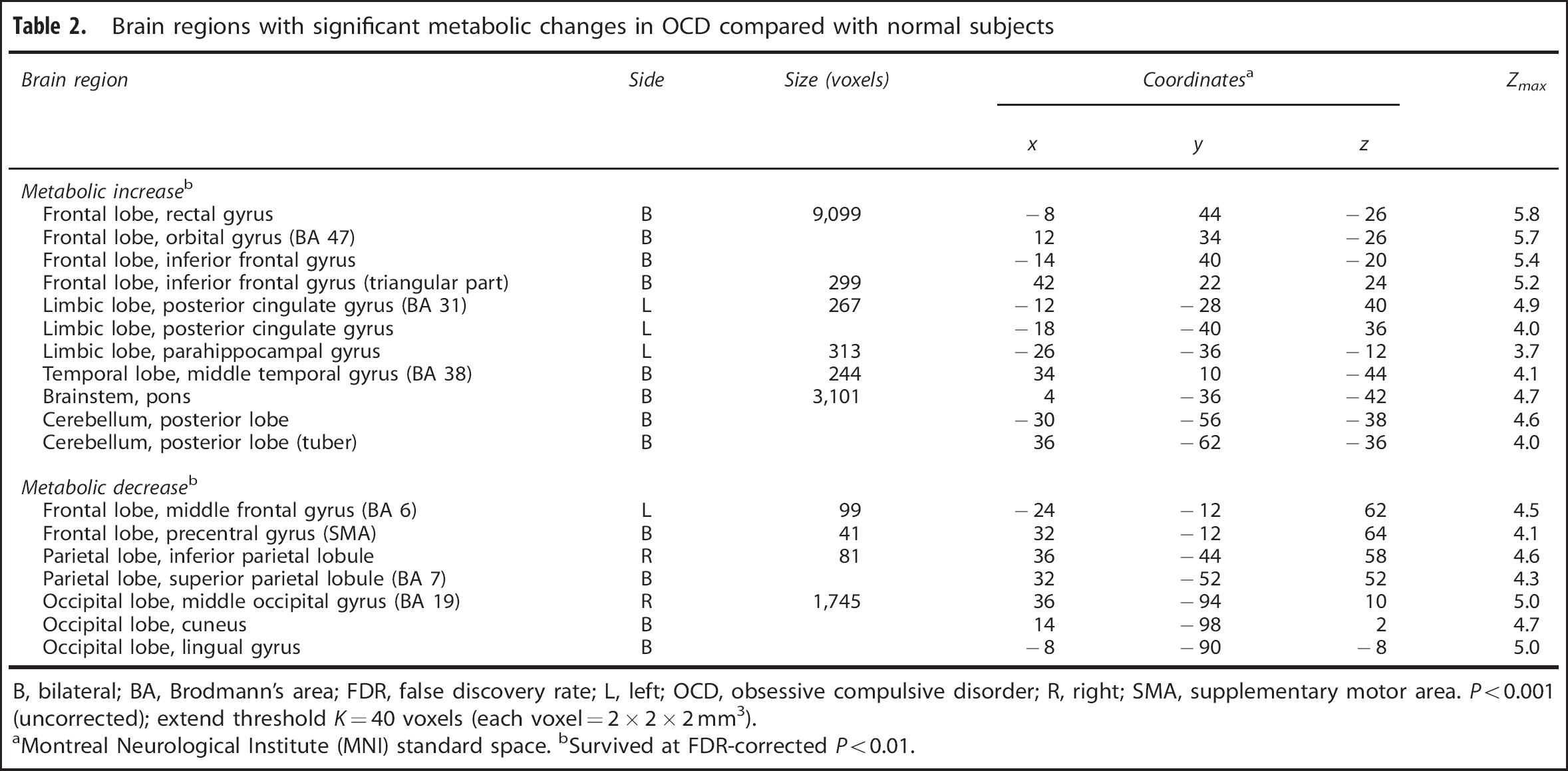
B, bilateral; BA, Brodmann's area; FDR, false discovery rate; L, left; OCD, obsessive compulsive disorder; R, right; SMA, supplementary motor area. P < 0.001 (uncorrected); extend threshold K = 40 voxels (each voxel = 2 × 2 × 2 mm3).
aMontreal Neurological Institute (MNI) standard space. bSurvived at FDR-corrected P < 0.01.
Metabolic Effects of Bilateral Capsulotomy
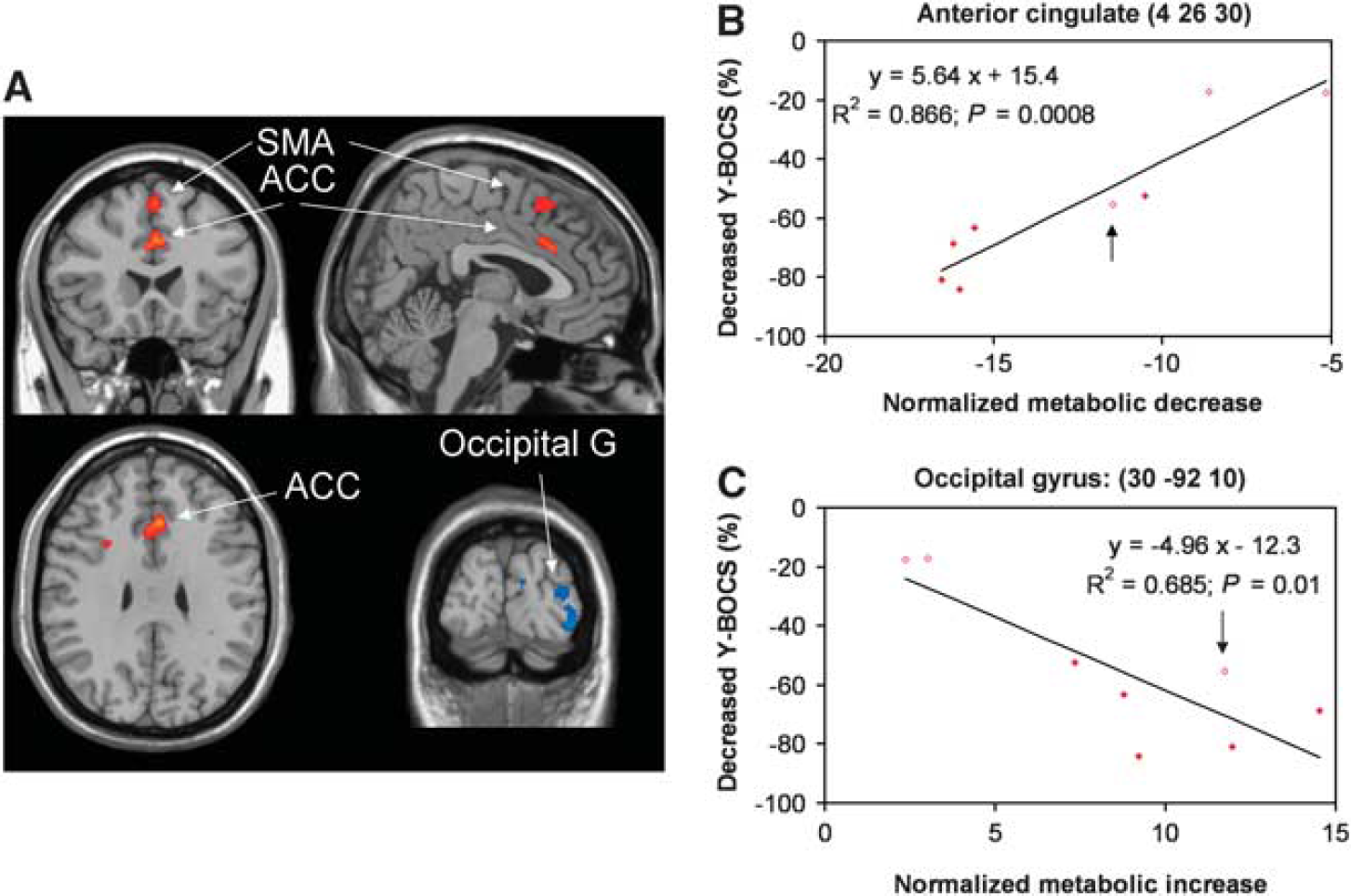
Bilateral capsulotomy led to widespread modulation in normalized regional glucose metabolism from the Pre-Op baseline (Table 3; Figure 2). Indeed, there was a highly significant cluster of decreased metabolism bilaterally in the ACC (BA 24/32) extending to the OFC. Metabolic reduction was also seen in the bilateral medial dorsal thalamus, caudate (spreading to the left IC and anterior putamen), right inferior frontal gyrus (BA 47) and posterior cerebellum (tonsil). However, increased metabolism was observed in bilateral precentral gyrus (BA 6), left inferior parietal lobule, and bilateral superior temporal gyrus (BA 41). Metabolism increased mainly in a large cluster encompassing the bilateral lingual gyrus (BA 18) and right posterior cerebellum (declive). All these regions also survived at FDR-corrected P < 0.025. The specific metabolic effects of the surgery were shown in select anatomical regions (Figure 2; Supplementary Figure 4). Of note, measures of mean global metabolism in all patients did not change before and after this neurosurgical intervention (P = 0.63).
Brain regions with significant clinical correlations between changes in Yale-Brown Obsessive Compulsive Scale (Y-BOCS) score and those in glucose metabolism after bilateral capsulotomy. (
Brain regions with significant metabolic changes in OCD after bilateral capsulotomy
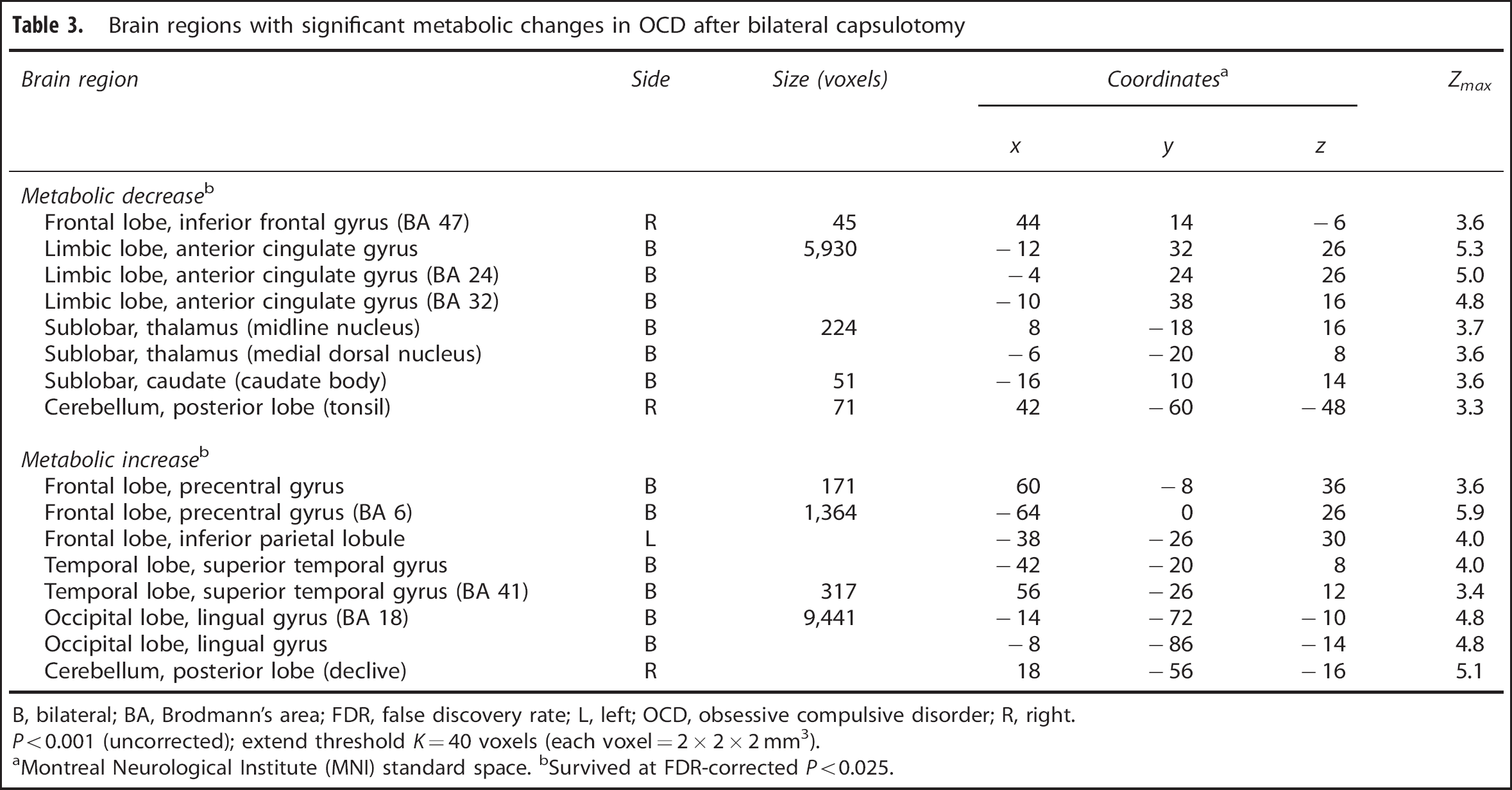
B, bilateral; BA, Brodmann's area; FDR, false discovery rate; L, left; OCD, obsessive compulsive disorder; R, right. P < 0.001 (uncorrected); extend threshold K = 40 voxels (each voxel = 2 × 2 × 2 mm3).
aMontreal Neurological Institute (MNI) standard space. bSurvived at FDR-corrected P < 0.025.
Correlation of Clinical Severity with Preoperative Metabolism
The Y-BOCS scores before surgery correlated significantly with normalized Pre-Op metabolism in a number of brain regions (Supplementary Table 1; Figure 3A). Positive correlation was found in the bilateral ACC, right PCC, and in the left middle temporal gyrus (BA 21) and insula (Figure 3B: R 2 = 0.91; P = 0.0002). Negative correlation was evident in the lingual gyrus and right posterior cerebellar tonsil (Figure 3C: R 2 = 0.94; P = 0.0001).
Correlation between Clinical Improvement and Metabolic Change after Bilateral Capsulotomy
The SPM analysis revealed significant correlations between decreases in Y-BOCS scores and operative changes in regional glucose metabolism (Supplementary Table 2; Figure 4A). Positive correlation was found between % reduction in Y-BOCS score and decreases in normalized metabolism in the bilateral superior frontal gyrus and ACC (Figure 4B: R 2 = 0.87; P < 0.001). Negative correlation was evident with metabolic increases in the right middle and inferior occipital gyri (Figure 4C: R 2 = 0.69; P < 0.01).
DISCUSSION
Capsulotomy is effective in treating refractory OCD but may carry a substantial risk of adverse effects. We found significant short-term clinical outcome in eight patients, especially in the six best responders showing a greater clinical improvement in symptoms of OCD and depression (Y-BOCS: 68 ± 5%; HAM-D: 86 ± 5%). The degrees of clinical improvement in these two domains agreed with those by other medical, behavioral, and neurosurgical interventions.11–13 We observed a few transient side effects in our small sample; one patient had urine incontinence over 5 days, three patients had fatigue for 1 month, and three others had confusion for 1 week.
[18F]-fluoro-deoxy-glucose PET is ideally suited to map the dysfunctional cortico-striato-thalamo-cortical circuitry previously reported in OCD. In our study, the patients showed widespread increases in Pre-Op metabolism in the bilateral prefrontal (e.g., orbital gyrus and anterior cingulate gyrus) and striatal regions (including caudate), and in the left posterior cingulate gyrus (Figure 1). Prior baseline studies in OCD have found that hypermetabolism in orbital gyrus,19–22 caudate nuclei, 19 and cingulate gyrus23,24 compared with normal volunteers. We found more brain regions with increased metabolism, including bilateral middle temporal gyrus, brainstem, and cerebellum, generally in accordance with the results published previously.13,21 Furthermore, the worsening of clinical symptoms was positively related with increased metabolism in ACC and PCC as well as in middle temporal gyrus and insula (Figure 3). All these findings confirmed the unique roles of orbital gyrus and the associated hyperactive neurocircuitry in OCD.
We also reported relative hypometabolism in OCD patients in cortical regions such as middle frontal cortex and parietal-occipital lobe. Indeed, the severity of OCD symptoms in patients was found to correlate with decreased metabolism in the inferior-middle frontal gyri and occipital lobe, in line with early reports of decreased metabolism in the SMA 13 and parieto-occipital areas20,22 in OCD. Hence, the connections between these cortical association regions and striatal circuits may also be affected in OCD, which may well contribute to the deficits in OCD patients in cognitive functioning not thought to be dependent on orbito-fronto-striatal circuitry.
The validity of this ablative approach is supported by functional brain imaging studies showing that the brain substrate of untreated OCD patients is hypermetabolic21,23,25–28 and that regional metabolism is decreased by medication, behavioral therapy, or neurosurgery.11–13,29,30 In this study, we confirmed that bilateral capsulotomy decreased metabolism bilaterally in the ACC (BA 24/BA 32) and OFC, medial dorsal thalamus, caudate and in the right cerebellar tonsil (Figure 2), consistent with previous reports that Pre-Op hypermetabolism in the cingulate, thalamus, and caudate predicted favorable clinical outcome after effective treatments.11–13,17 In contrast, lesions increased metabolism bilaterally in the precentral (BA 6), superior temporal and occipital lingual gyri. In particular, metabolic changes in the superior-inferior frontal gyri, dorsal ACC, and occipital lobe (Figure 4) correlated with clinical improvement in patients after bilateral capsulotomy. Such an effect in the ACC agree with metabolic data on anterior cingulotomy 12 and capsular simulation. 13
The variations in our results can be better understood with Pre-Op patient characteristics. Our sample was younger and less severely affected patients (shorter duration and no comorbidity) than those reported by other investigators. Nonetheless, our results concur with the previous salient findings and confirm especially that Pre-Op hypermetabolism in OFC is a signature of OCD in the absence of comorbid major depression. The OFC clusters were connected to other regions in prefrontal, temporal, and parietal cortices and subcortical areas in the striatum and the midbrain. These unique patterns may contribute to the specific functional roles of OFC in OCD patients for reward processing, learning, and decision making. 31 Of importance, the distribution of abnormal metabolism in our group was also evident in each of the OCD patient before the surgery, in particular, orbital hypermetabolism, fulfilling the key criterion of Pre-Op imaging evaluation before capsulotomy.
Among the three adolescents, clinical symptoms improved markedly in one patient but only minimally in two others. Two of these adolescents had lower Y-BOCS scores at Pre-Op baseline; one was a responder with the other a nonresponder. The other adolescent was a nonresponder even with higher Y-BOCS score.
Although clinical improvements were robust in the five young adults, there were no differences in Pre-Op metabolic values or operative changes between these patients based on all post hoc plots in individual subjects. Of note, clinical-metabolism correlations in the patients between baseline measures or operative changes were not driven by the two adolescents who were nonresponders. These correlations remained significant (albeit with a reduced magnitude) after removing the two nonresponders in the analyses. These suggested that poorer clinical response was not likely to be dependent of patient age and symptom severity at baseline, but might be related to subtle differences in the restoration of functional network connectivity by the surgery.
The metabolic correlates of successful treatment in our study imply a central role for the dorsal ACC and posterior occipital cortex in the outcome of bilateral capsulotomy for refractory OCD. Internal capsule is a central gateway on white-matter tracks between cortical and subcortical pathways, involved in the regulation of the ventral prefrontal complex underlying mood and anxiety disorders. Lesions placed on the ventral portion of the anterior limb of the anterior capsule are purported to interrupt OFC, dorsal ACC, striatal, medial dorsal thalamic and pontine connections. 32 Involvement of brainstem and other frontal, temporal, and parietal cortices in this regional network have also been implicated in a recent study in primates.6,33 Neurosurgical interventions at the IC need to affect different combinations of thalamic and brainstem ventral prefrontal fibers. Our data generally support this neurobiologic model as preoperative hypermetabolism observed in the IC was suppressed by capsulotomy, leading to concurrent metabolic restoration in other related brain regions. However, the role of occipital cortex is not well understood but may involve a specific brain network with the retrolenticular part of the IC.
Several important issues need to be addressed with regard to our study design and analytical methodology. First, we examined OCD patients without comorbid major depressions and controlled rigorously for experimental factors such as age, IQ, and handedness of the participants. Second, we performed whole brain-based analyses using a voxel-level SPM method to localize more areas not examined in earlier studies based on an a priori hypothesis. Third, the sample size is limited in the current study that may imply a chance of false-positive findings. However, the threshold applied in the SPM analysis is one commonly used in the analysis of PET data and a correction for multiple comparisons was made in the major parts of our study to minimize FDRs. Fourth, the patients in our study had a 12-hour washout of medications (including selective serotonin reuptake inhibitors) before PET imaging, but plasma concentrations were not evaluated to ensure total clearance. Selective serotonin reuptake inhibitors may also relieve some OCD symptoms by a different downstream effect of serotonin transmission by inducing metabolic decreases in the ACC, middle temporal, precuneus, and parahippocampal gyri. 34 Nevertheless, the results reported in this study, particular those mediated by neurosurgery need to be replicated in a large sample of OCD patients after bilateral capsulotomy.
CONCLUSION
This is the first study to show metabolic modulation of regionally specific dysfunction in the cortico-striato-thalamo-cortical neurocircuitry by ablative capsulotomy in patients with OCD symptomatology alone using voxel-based analysis of 18F-FDG PET images. More brain regions besides orbitofrontal/cingulate gyri and caudate were found to have abnormal metabolism at Pre-Op baseline in our study, possibly due to differences in clinical population and analysis methods. In particular, decreased metabolism was detected in the parietal-occipital and frontal regions, likely reflecting inherent compensatory processes associated with the pathophysiology of OCD. Bilateral capsulotomy was able to restore abnormal regional metabolism to a great extent and emphasized the importance of dorsal anterior cingulate gyrus.
DISCLOSURE/CONFLICT OF INTEREST
The authors declare no conflict of interest.
Footnotes
ACKNOWLEDGEMENTS
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
