Abstract
Intravenous administration of iron oxide nanoparticles during the acute stage of experimental stroke can produce signal intensity changes in the ischemic region. This has been attributed, albeit controversially, to the infiltration of iron-laden blood-borne macrophages. The properties of nanoparticles that render them most suitable for phagocytosis is a matter of debate, as is the most relevant timepoint for administration. Both of these questions are examined in the present study. Imaging experiments were performed in mice with 30 minutes of middle cerebral artery occlusion (MCAO). Iron oxide nanoparticles with different charges and sizes were used, and mice received 300
INTRODUCTION
There is a profound inflammatory response to stroke that includes recruitment of blood-borne leukocytes to the brain. Because one of the roles of monocytes on their maturation is phagocytosis, it is generally believed that this subpopulation of leukocytes can take up contrast agents from the blood stream and bring them to the ischemic brain. For magnetic resonance imaging (MRI), superparamagnetic iron oxide nanoparticles (SPIOs) are widely used, reviewed in (refs 1–3). In general, they consist of an iron oxide core that can be encased in a variety of metabolizable monomers or polymers; the most common coating used is polysaccharides. The coating influences overall hydrodynamic diameter and surface charge, which in turn determines the pharmacokinetic properties of the particles such as blood half-life, uptake, and elimination.4,5 These properties are extremely important when attempting to use these agents to target blood-borne monocytes. Superparamagnetic iron oxide nanoparticles are generally grouped according to size. Micron-sized particles of iron oxides are the largest (0.7 to 3
USPIOs are the most commonly used nanoparticles to attempt to image the monocyte response to stroke. This is generally because their small size affords them some degree of protection from uptake by the mononuclear phagocyte, or reticuloendothelial, system; i.e., the macrophages in the lymph nodes, spleen, and liver (Kupffer cells). 6 Therefore, their blood half-life is slightly longer than that of SPIOs. The first preclinical study administered USPIOs 5 hours after permanent middle cerebral artery occlusion (MCAO) in the rat and observed signal loss within the first 2 days in T2-weighted images. 7 Prussian blue (iron stain)-positive cells were detected in the lesion boundary at 7 days. However, the same group was only able to observe a delayed (48 to 72 hours) hyperintense signal change in the ischemic hemisphere on T1- weighted images when the same strategy was used in a transient model of MCAO, 8 which is more consistent with the known timecourse of monocyte infiltration to the ischemic brain.9,10
These studies served as a launching platform for this technique and clinical studies began to emerge that showed a more heterogenous pattern of USPIO signal enhancement in stroke patients. When USPIOs were administered 6 days after symptom onset, a strong T2* signal decrease was observed on account of the blood-pool effect of the iron, which paralleled an increase in T1 contrast that was attributed to parenchymal accumulation.11,12 Earlier USPIO administration (between 24 to 96 hours of symptom onset) resulted in T1 enhancement in only one-third of patients. 13 This highlighted the need for further preclinical investigation with particular focus on delaying the timepoint of USPIO administration. However, studies that used this approach were less promising. The first preclinical study administered USPIOs for up to 3 days after transient MCAO in mice and no signal changes were observed in any of the images. 14 Comparable results were obtained by another group that administered USPIOs between 3 and 6 days after transient MCAO in rats. 15
Despite exhibiting a poor half-life when compared with USPIOs, SPIOs consistently exhibit much higher uptake by different type of macrophages
MATERIALS AND METHODS
Experimental Design
All experiments were approved by the Landesamt für Gesundtheit und Soziales under the license numbers G 0200/07 and A 0045/11, and performed in accordance with the German Animal Welfare Act. Animals were housed in a temperature (22°C ± 2°C), humidity (55% ± 10%), and light (12/12 hours light/dark cycle) controlled environment and given
To assess the potential of MRI to detect iron-containing macrophages in the brain, 42 male C57 BL6 N mice (8 weeks of age, Charles River, Sulzfeld, Germany) received 30 minutes of MCAO (
To determine blood iron content and leukocyte uptake of the nanoparticles closer to the time of injection in our specific model, 12 additional mice underwent MCAO and randomly received one of the three different types of iron oxide nanoparticles (300
To confirm that blood-borne macrophages are indeed capable of migrating to the ischemic brain, additional experiments were performed in bone marrow chimeras (see Supplementary Methods and Results).
MCAO Procedure
Animals weighed between 19 and 27 g (average 24.1 ± 1.4 g) at the time of the MCAO. Anesthesia was achieved using isoflurane in a 70:30 nitrous oxide:oxygen mixture, and core body temperature was maintained at 37.2 ± 0.8°C using an automated heat blanket with temperature feedback (Harvard Apparatus, Hugo Sachs Elektronik, March-Hugstetten, Germany). Transient occlusion of the MCA was performed using a modified intraluminal filament technique. Mice were placed in a supine position and the neck was shaved and cleaned. A midline incision was made in the neck, and the left sternomastoid muscle was retracted to expose the left carotid artery (CA). A silk suture was tied around the CA below the bifurcation into the internal and external carotid arteries, respectively. A loose suture was placed around the external carotid artery and secured externally. Another loose suture was placed on the internal carotid artery and a microclip was placed directly above it. A small incision was made in the CA and a 20-mm-long filament coated with 5 mm of silicone to a final diameter of 190
Magnetic Resonance Imaging Measurements
Anesthesia was also achieved using isoflurane. Magnetic resonance imaging experiments were conducted on a 7 T Pharmascan 70/16 (Bruker BioSpin MRI GmbH, Ettlingen, Germany) equipped with actively shielded gradient coils (300 mT/m, rise time 80 μs). Radio frequency transmission and reception were achieved with a 20-mm (inner diameter) quadrature mouse head volume resonator (RAPID Biomedical, Würzburg, Germany). During scanning, body temperature and respiration rate were monitored with an MRI compatible system (Small Animal Instruments, Stony Brook, NY, USA).
The entire imaging protocol was selected to be completed within 30 minutes (FOV: 25 × 25 mm2, matrix: 256 × 256, 12 contiguous 0.6-mm-thick slices). It consisted of a spin-echo multislice multiecho T2 sequence (TR/TE: 3000/10.5 ms, 16 echoes, 9 minutes), a fast low-angle shot T1- weighted sequence (TR/TE: 150/3.5 ms, flip angle 30°, 4 minutes), a rapid acquisition with relaxation enhancement T1-weighted sequence (TR/TE: 800/13.2 ms, 2 averages, rapid acquisition with relaxation enhancement factor 2, 3 minutes), and a fast low-angle shot T2*-weighted sequence (TR/TE: 1000/7.2 ms, flip angle 30°, 4 minutes) that was not strongly T2*-weighted to avoid strong endogenous susceptibility effects.
Contrast Agents
One of three different types of iron oxide nanoparticles (for details regarding nanoparticle synthesis and characterization, see Supplementary Methods) were administered (300
MR-Image Analysis
Quantitative T2 maps were fitted on a voxelwise basis using a monoexponential decay function in Paravision version 4 software (Bruker BioSpin). Subsequently, all data were exported to ImageJ version 1.44p freeware (National Institutes of Health, Bethesda, MD, USA). An unblinded observer used the Sync Windows Analyze Tool to draw two circular (1.2 mm diameter) regions of interest (ROIs) on the slice image that was located 0.14 mm from bregma: 20 intact and ischemic striatum. T2 (ms) was measured in these ROIs from the T2 maps and data were expressed as a ratio of the intact to ischemic hemisphere. An additional large rectangular ROI (24 × 4 mm) was also drawn above the brain on the T1- and T2*- weighted images. Subsequently, signal-to-noise ratios in the striatum were calculated by dividing the signal intensity of each ROI by the standard deviation of the noise in the large rectangular ROI. These data were also expressed as a ratio of the intact to ischemic hemisphere.
Magnetic Particle Spectroscopy
Quantification of nanoparticle iron content in blood samples from 12 MCAO mice was determined immediately after nanoparticle injection using a commercial MPS (Bruker BioSpin MRI GmbH). 21 Primarily, this device is dedicated to quantify the performance of magnetic nanoparticles as tracers for a novel imaging modality called magnetic particle imaging. 22 Magnetic particle spectroscopy detects specifically the nonlinear magnetic response of magnetic nanoparticles exposed to an oscillating magnetic field. Biological tissue and paramagnetic blood iron do not contribute to the MPS signal.
Blood samples (between 50 and 100
Blood Leukocyte Harvest
Terminal blood samples were collected from all mice and processed to retrieve the leukocyte fraction. The samples were adjusted to equal volumes (5 mL) with 1 mol/L phosphate-buffered saline and embedded on 2.5 mL of Histopaque-1083 (Sigma-Aldrich). They were subsequently centrifuged for 30 minutes at 850
Histology
Mice that underwent MRI for up to 5 days after MCAO were euthanized with intraperitoneal chloral hydrate (4% in water), perfused with physiological saline, and the brains, livers, and spleens were snap frozen in −40°C methylbutane, sectioned to 20
RESULTS
Overall mortality within the primary study was low (1/42), two mice were euthanized prematurely because of excessive weight-loss in accordance with our animal care guidelines and one animal was excluded on the basis that no MCAO was observed in the T2- weighted images. Group assignments and drop outs are presented in Table 1. Only one of the additional 12 mice that were used for blood nanoparticles iron content analysis by MPS was euthanized prematurely and another died after particle injection.
Experimental groups and hemispheric lesion volume
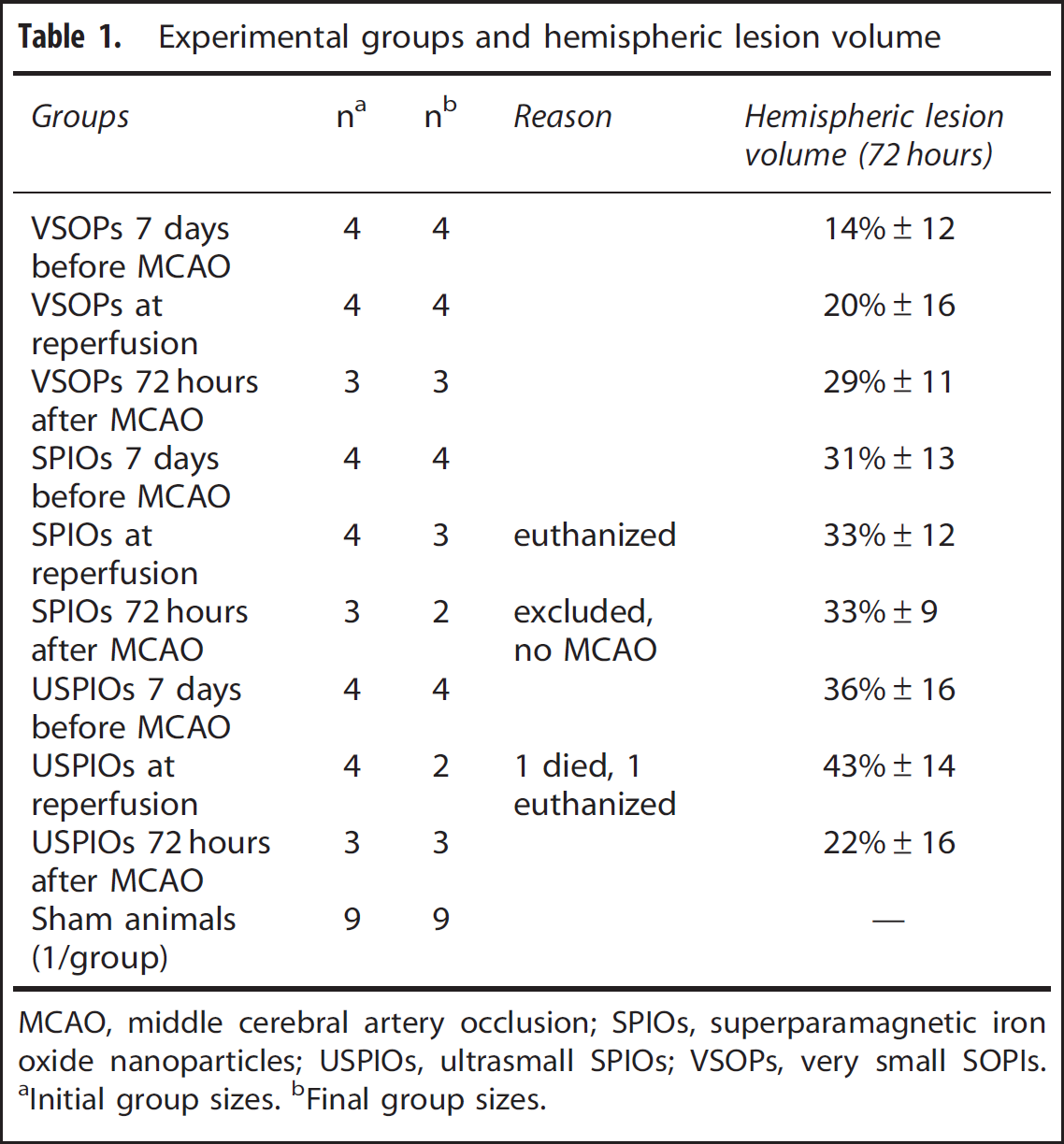
MCAO, middle cerebral artery occlusion; SPIOs, superparamagnetic iron oxide nanoparticles; USPIOs, ultrasmall SPIOs; VSOPs, very small SOPIs.
Initial group sizes.
Final group sizes.
MR-Image Analysis
Repetitive MRI of mice for up to 5 days after MCAO did not reveal any focal regions of hypointensity in T2- or T2*-weighted images, or hyperintensity in T1-weighted images, that could be attributed to any of the three used iron oxide nanoparticles (Figure 1). In the animals that received nanoparticles at 72 hours after MCAO (Figure 1, bottom panel), the infusion was performed while the mice were inside the MRI so that the blood-pool effect could be observed with pre (72 hours) and post contrast (72.5 hours) images. An overall signal decrease, which was particularly prominent in large blood vessels, was observed in T2*-weighted images after infusion of VSOPs, SPIOs, and USPIOs (Figure 1, bottom row). However, this effect was no longer present the following day (96 hours after MCAO, Figure 1, bottom row) and no additional hypointensities were observed at any other timepoint. Blood–brain barrier breakdown was also evident in the animals that received iron at 3 days after MCAO as the ischemic region became hyperintense in T1-weighted images after Gadolinium diethylenetriaminepentaacetic acid infusion (Figure 1, bottom panel).
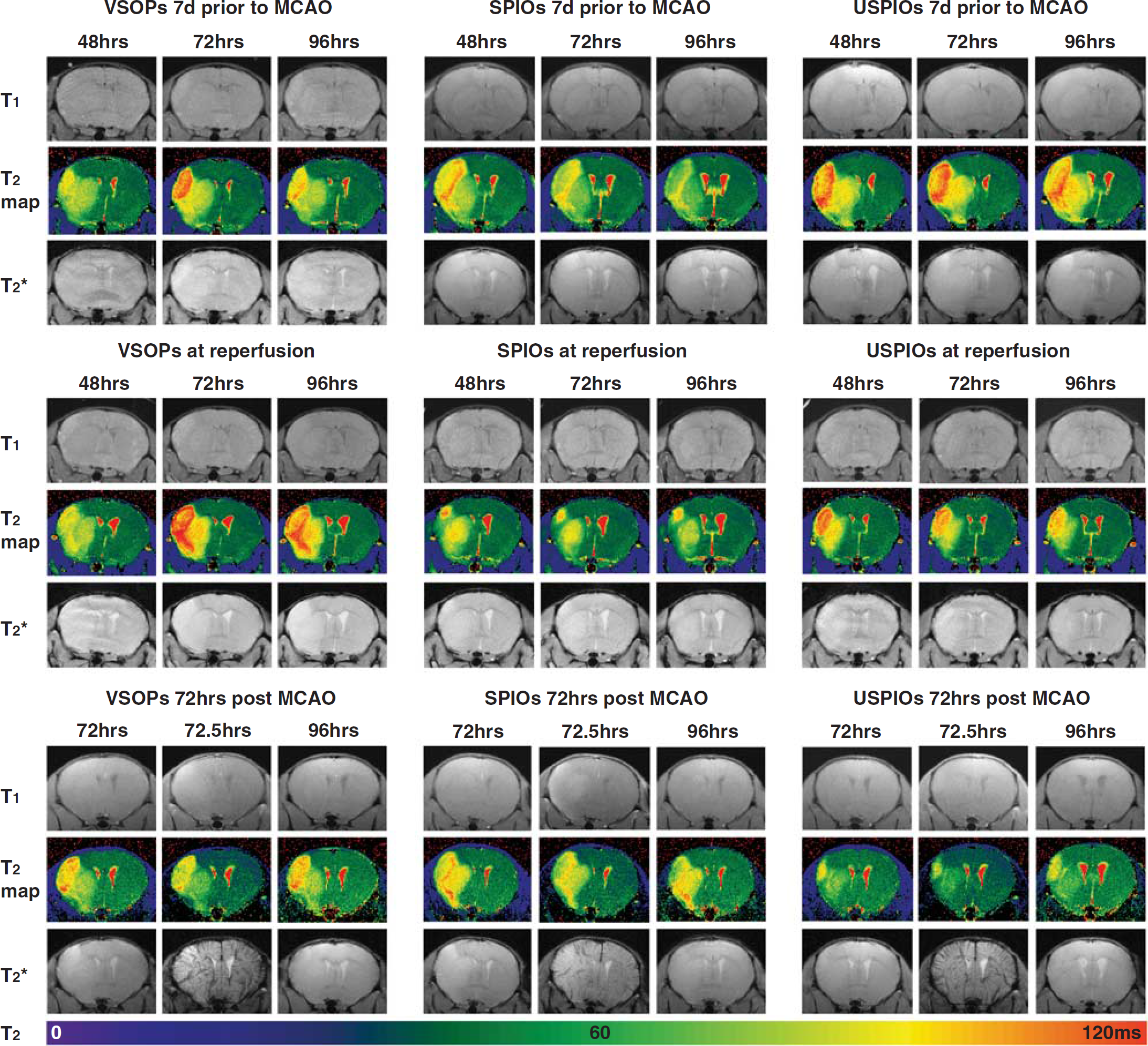
Images from animals treated with intravenous contrast agents at different timepoints with respect to middle cerebral artery occlusion (MCAO). A timecourse of T1-weighted images, T2 maps, and T2*-weighted images from a representative animal in each of the treatment groups (left panel—VSOPs, middle panel—SPIOs, and right panel—USPIOs) after MCAO. The top panel contains animals that received iron oxides 7 days before MCAO, the middle panel: at the time of reperfusion, and the bottom panel: at 72 hours after MCAO. T2 values (ms) are indicated in the color scale bar below the images. Note the blood-pool effect of the iron oxides after infusion (72.5 hours) in the T2*-weighted images in the bottom most row. SPIOs, superparamagnetic iron oxide nanoparticles; USPIOs, ultrasmall SPIOs; VSOPs, very small SPIOs.
Semiquantification of the signal-to-noise ratio in T1- and T2*- weighted images, and T2 (ms), was performed in the intact and ischemic striatum to try and elucidate subtle changes in signal. Subsequent values were expressed as a ratio of the intact to ischemic hemisphere; thus, a value of 1 would indicate that there is no difference in signal-to-noise ratio, or T2, between hemispheres. Both T1 and T2* ratios in animals treated with VSOPs at 7 days before MCAO or at the time of reperfusion remained unchanged (around 1) at all measured timepoints after MCAO (Figure 2A and C). However, the T2 ratios of these animals were ~0.6 within the first 48 hours of MCAO (Figure 2B), which is not surprising given that T2 values are increased in the ischemic territory because of vasogenic edema. As edema resolves T2 values will decline slightly, which is visualized by the gradual increase in T2 ratios over time in the animals treated with VSOPs at 72 hours after MCAO (Figure 2B). In these same animals, no changes in T2 or T2* ratios were observed directly after injection of the iron (72 to 72.5 hours; Figure 2B and C). However, a decrease in the T1 ratio was observed at this timepoint because of the Gadolinium diethylenetriaminepentaacetic acid injection (Figure 2A).
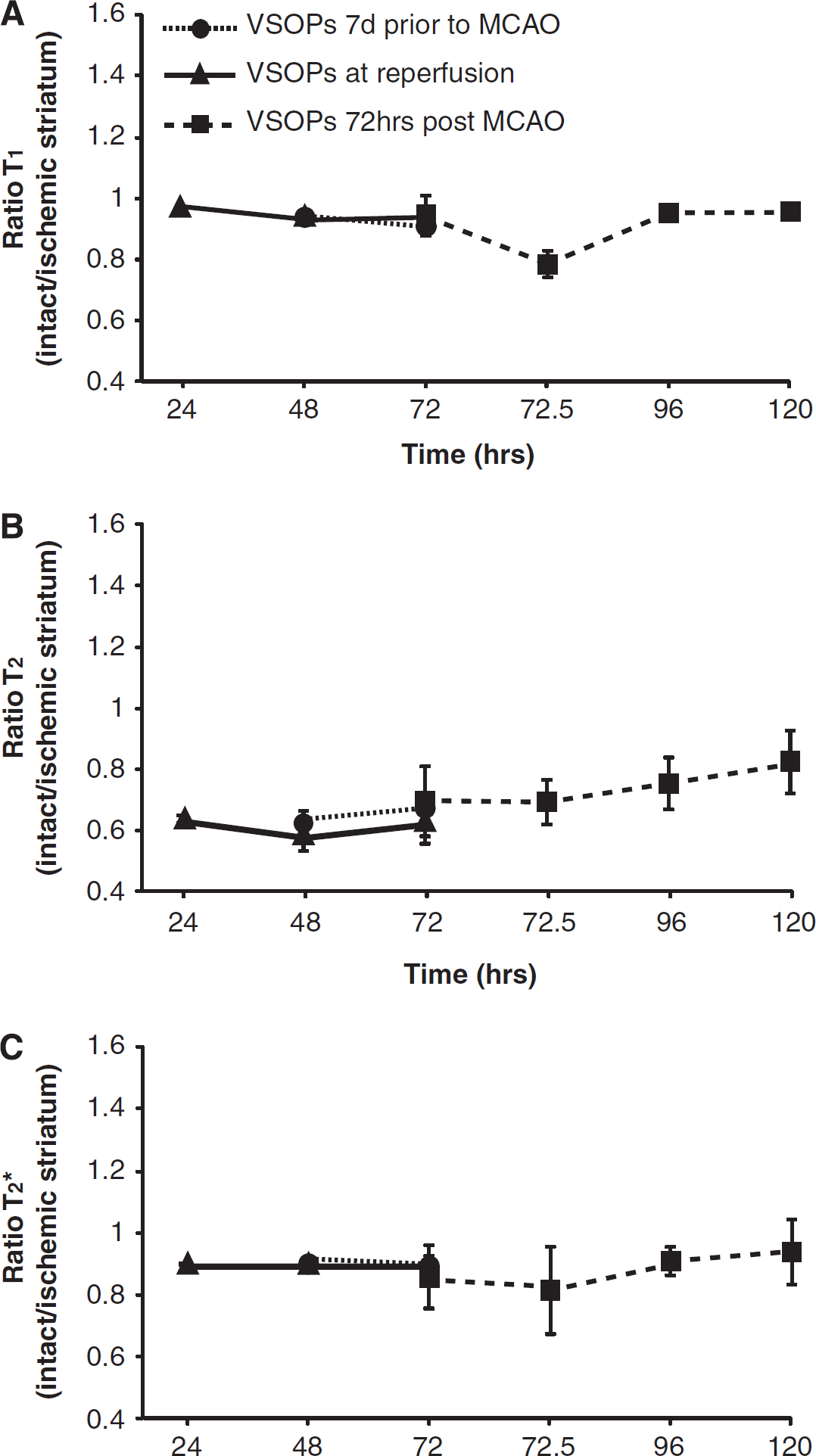
Signal intensity changes in the images over time in VSOP-treated animals. Semiquantitative analysis (means ± s.d.) of the signal-to-noise ratio in the striatum, expressed as a ratio of the intact to ischemic hemisphere in T1 (
Similar results were obtained from the animals treated with SPIOs and USPIOs (Figure 3 A–C and D–F, respectively). No changes in T2* ratios were observed over time in either SPIO (Figure 3C) or USPIO (Figure 3F) treated animals when nanoparticles were infused at 7 days before MCAO, at reperfusion, or at 72 hours after MCAO. This was also the case for T1 ratios. However, there was a decrease in the T1 ratio at 72.5 hours after Gadolinium diethylenetriaminepentaacetic acid injection in the animals treated with SPIOs at 72 hours after MCAO (Figure 3A). This effect was not observed in the animals treated with USPIOs at 72 hours after MCAO (Figure 3D). Animals in this group did not exhibit such severe lesions, as is indicated by the lower T2 values and thus T2 ratio of only 0.8 (Figure 3E). T2 ratios of the other USPIO, and SPIO-treated groups, were comparable (beginning ~0.6 and increasing to 0.8 by 120 hours after MCAO).
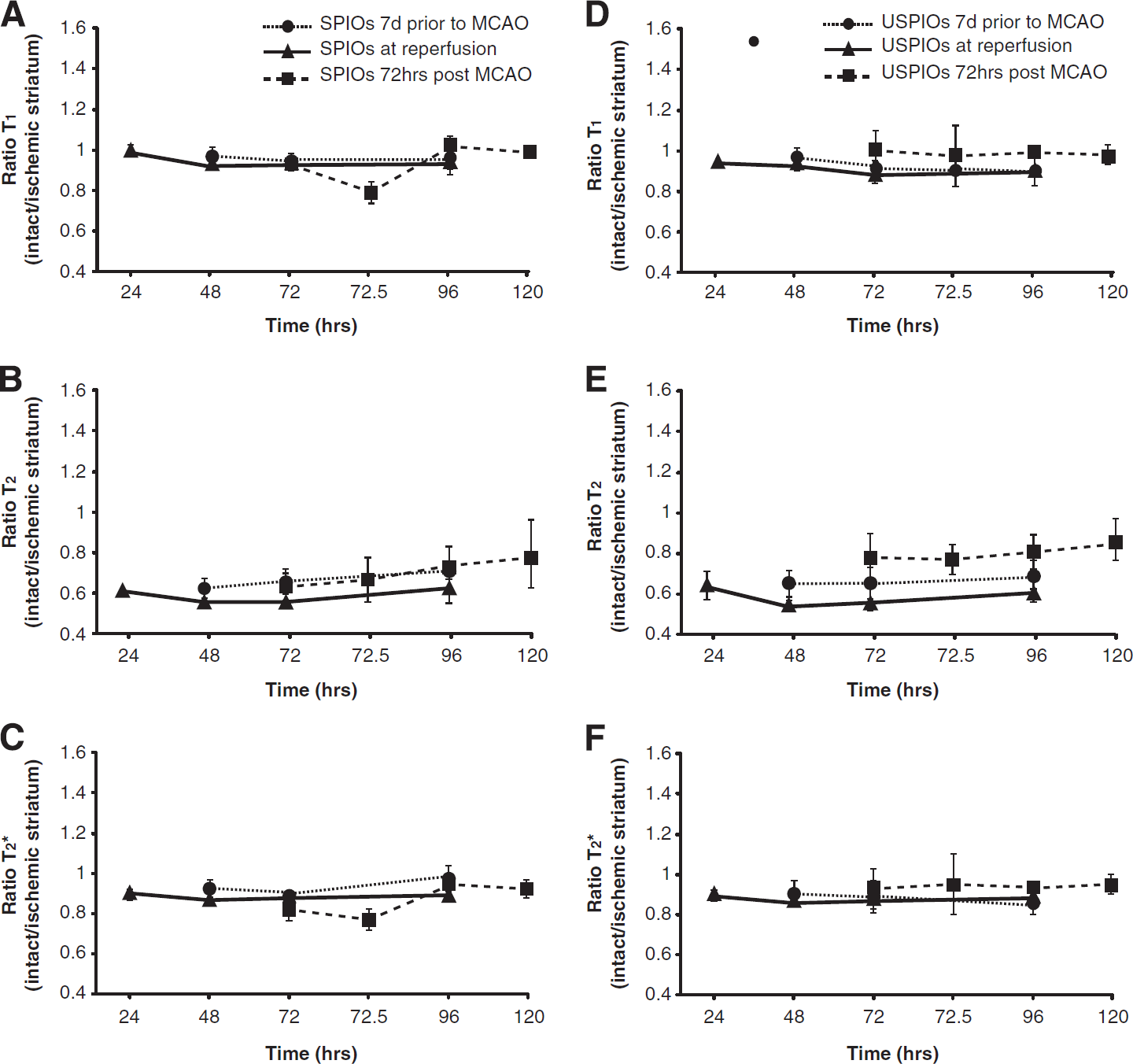
Signal intensity changes in the images over time in SPIO- and USPIO-treated animals. Semiquantitative analysis (means ± s.d.) of the signal-to-noise ratio in the striatum, expressed as a ratio of the intact to ischemic hemisphere in T1- (
Magnetic Particle Spectroscopy
Magnetic particle spectroscopy revealed that the concentration of magnetic nanoparticles in the blood was negligible before injection. However, the nanoparticle iron concentration increased dramatically in all three groups within the first 2 minutes of the infusion (between 100 and 200 ng Fe/mg of blood) (Figure 4). This response began to decline as early as 5 minutes after injection and was nearly at baseline levels by 45 minutes, and this pattern was similar for all three nanoparticle formulations.
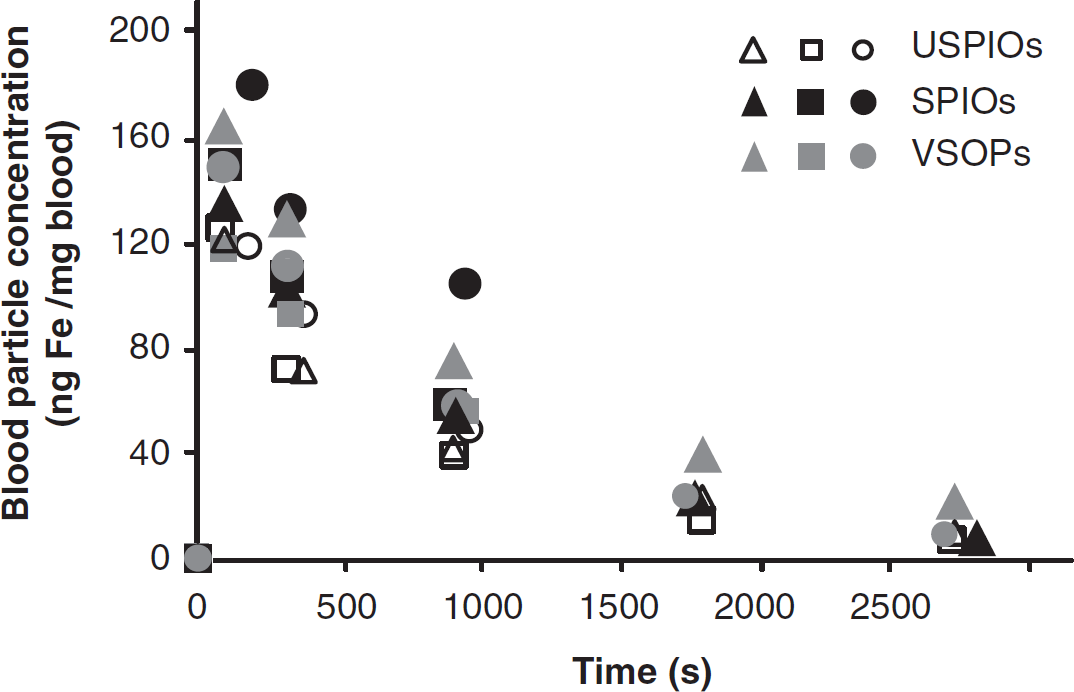
Nanoparticle iron concentration (ng Fe/mg blood) in the blood as measured by magnetic particle spectroscopy before (0 second) and up to 45 minutes (2700 seconds) after injection in animals treated with VSOPs (gray symbols), USPIOs (empty symbols), and SPIOs (black symbols). Note: each symbol corresponds to a different animal (
Hearts, lungs, and kidneys also contained a negligible amount of nanoparticle iron. However, the portions of the spleens and livers (respectively) from all three groups contained high amounts of nanoparticle iron: VSOPs (362 ± 56 and 197 ± 82 ng Fe/mg of tissue), UPSIOs (223 ± 76 and 172 ± 53 ng Fe/mg of tissue), and SPIOs (41 ± 3 and 58 ± 21 ng Fe/mg of tissue). When this was corrected for total organ weight, assuming homogenous distribution, the liver contained ten times more nanoparticle iron than the spleen in VSOP- and USPIO-treated animals, and 30 times more in the SPIO-treated animals. Overall particle concentration, in general, was lower in the SPIO-treated group.
Histology
Three brain sections (1.42, 0.14, and −1.7 mm from bregma) from each of the animals were examined for the presence of Prussian blue-positive cells. No Prussian blue-positive cells were observed in any of the examined sections (Figure 5). However, the livers and spleens of all animals in the study were highly positive for iron. Iron in the spleen was not always homogenously distributed and appeared to be both intracellular and extracellular. Prussian blue staining was always observed inside the cells of the liver. The blood leukocyte fraction was also examined and none of the slides contained any Prussian blue-positive cells.
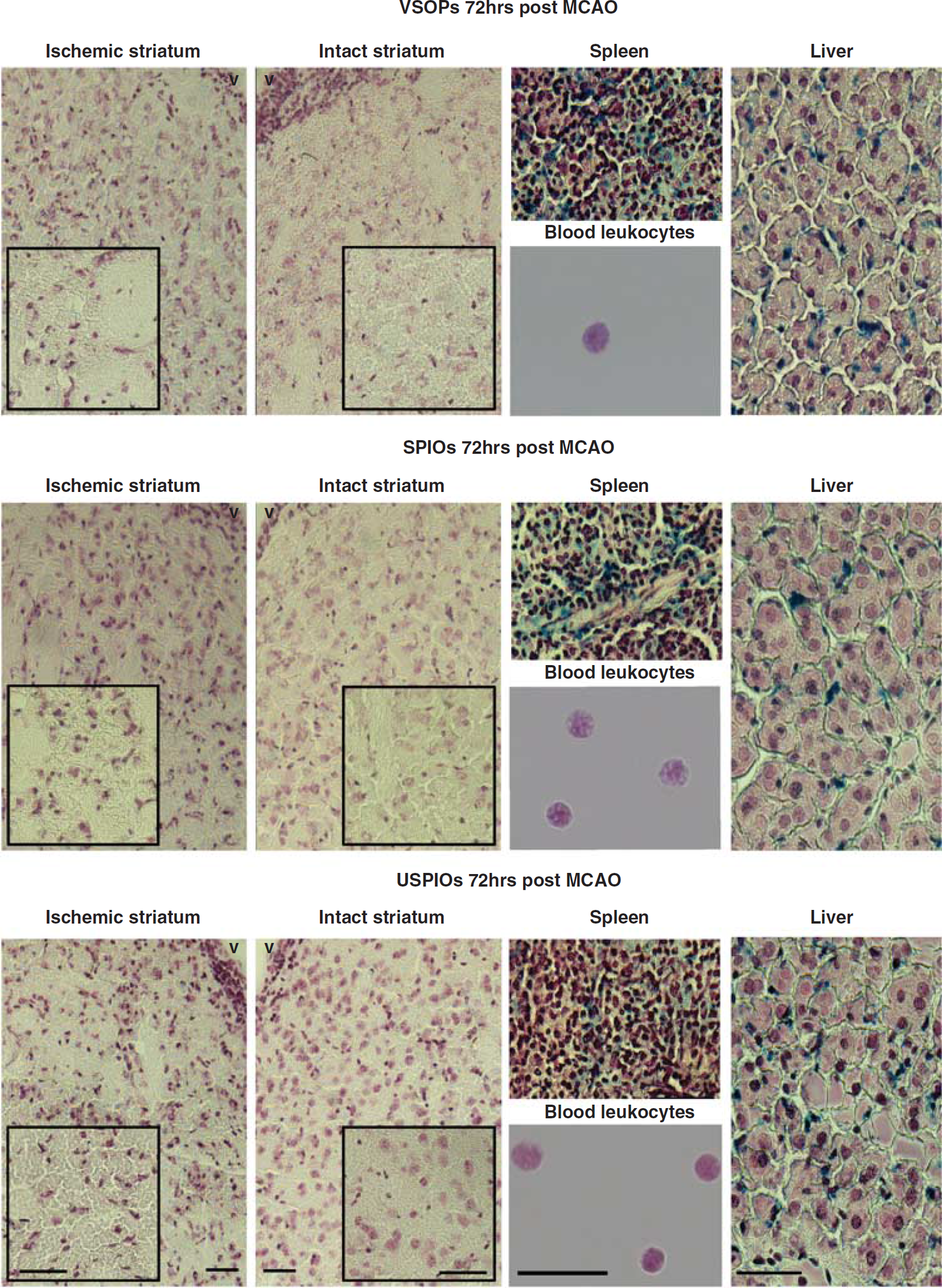
Prussian blue staining of the tissues and leukocyte fractions. Prussian blue sections from a representative animal treated with VSOPs (top row), SPIOs (middle row), and USPIOs (bottom row) at 72 hours after MCAO. The ischemic and intact striatum images contain higher magnification insets. Prussian blue-positive cells were only found in the spleens and livers. Note: scale bars in the leukocyte images correspond to 16
DISCUSSION
The present study administered three different types of iron oxide nanoparticles, each with a different size and coating, at three different timepoints with respect to MCAO. Interestingly, no regions of circumscribed signal change that could be attributed to iron were observed in T2*-, T2-, or T1-weighted images under any combination of conditions. A detailed examination of the corresponding tissue sections was unable to identify any Prussian blue-positive cells in the ischemic brains, although the livers and spleens from all groups, at all timepoints, contained high amounts of iron. We were also unable to find any Prussian blue-positive cells among the blood leukocytes that were harvested from terminal blood samples. Repetitive blood sample analysis in another group of ischemic animals revealed that all three particle types, irrespective of differences in formulations, rapidly disappeared from the blood within 45 minutes and at this time were only detectable in the livers and spleens and not the other organs or blood leukocytes. Taken together, these results suggest limited application for the use of these type of iron oxide nanoparticles to image the peripheral macrophage response to transient MCAO. We would like to make the controversial suggestion that the available literature actually supports our findings. Because negative results are excluded from the literature, researchers are under pressure to place a positive spin on their results. This means that even though the research community is aware that this technique has certain limitations, many publications still promote iron oxides as a useful tool to image blood-borne monocytes. With this article, we hope to bring this issue forward for discussion.
The notion that USPIO-induced signal change in MR images is because of the infiltration of monocytes that have phagocytosed contrast agents from the blood has been losing momentum over the last few years. The landmark studies in rat and mouse7,23 were followed by results that suggested some of the observed signal change may be because of passive entry of the USPIOs into the brain and/or accumulation in the cerebral spinal fluid and interstitial space 24 and experiments using transient models were never as promising.8,14,15 The results of the present study using a transient model are in agreement, with the notable exception that we did not observe a delayed hyperintensity develop in T1- weighted images that was reported by one group. 8 While clinical studies have also reported T1 increases when USPIOs were administered 6 days after symptom onset,11,12 it is still not clear if this represents accumulation of iron-containing cells. The T1 effect can overcome the susceptibility effect when iron concentration is low25,26 or when the iron is extracellular as opposed to clustered intracellularly.27,28 Therefore, it is possible that the T1 hyperintensity could reflect low amounts of iron in the interstitial space.23,29
In contrast to USPIOs, very little work has been done with SPIOs on account of their short half-lives. One group attempted to overcome the restrictions imposed by poor circulation times by administering SPIOs on a daily basis, and were able to observe signal change in the ischemic brain; 18 unfortunately, we only administered SPIOs once per animal. Another group has suggested that administration of SPIOs 7 days before MCAO allows these particles to accumulate in the bone marrow progenitor cells; thus, any contrast observed in the brain after stroke would be from cells that originated in the bone marrow and not because of passive entry of contrast into the brain. 19 The preloading strategy was not successful in our hands, although it is not clear if SPIOs are capable of accumulating in the bone marrow. This property has generally been attributed to USPIOs on account of their half-life, and even USPIOs show relatively little bone marrow accumulation beyond 24 hours in rodents.30,31 The SPIOs used in the present study would be more suitable than most other formulations for this purpose, because they were coated with polyethylene glycol to increase circulation time. As we did not measure the bone marrow, we cannot exclude the possibility that the SPIOs accumulated there.
The fact that none of the particles appear to be suited to track blood-borne monocytes is not entirely surprising if we consider their historical development. The first-generation SPIOs (Endorem, Guerbet, Paris, France, Feridex, Amag Pharma, Lexington, MA, USA; coated in dextran with a neutral charge) were developed for iron replacement therapy. Subsequent formulations were coated with carboxy-dextran and possessed a negative charge (SH U 555 A/Resovist, Bayer Schering, Berlin, Germany) because this reduced side effects of administration. Both formulations are sterically stabilized, which means they are not stable for long
It is necessary to point out that there are several limitations with the present study. The first is that we did not include statistical analysis of our results on account of the small group sizes, which makes it difficult to make definitive conclusions. However, as the results were emerging as overwhelmingly negative, we could not justify the continued use of resources and animals. The second is that our experiments were conducted solely in mice, which is unfortunate as we cannot directly compare our findings with those that were obtained in rats. The third important limitation was that we chose to focus our attention on nanoparticle formulations that were consistent with those used in the published literature (namely, USPIOs and SPIOs). As was previously mentioned, the half-lives of these formulations are not conducive for labeling blood-borne leukocytes. While the same appears to be true for the VSOPs used in the present study, it is possible that similar particles with longer circulation times, such monocrystalline iron oxide nanocompound, 35 or crosslinked iron oxide nanoparticles, could result in uptake by blood-borne leukocytes.
In conclusion, certain formulations of iron oxide nanoparticles, at least the ones used in the present study, appear to have a limited application as agents to label blood-borne monocytes of bone marrow origin in a mouse model of transient focal ischemia.
DISCLOSURE/CONFLICT OF INTEREST
The authors declare no conflict of interest.
Footnotes
ACKNOWLEDGEMENTS
The authors wish to acknowledge Janet Lips and Marco Foddis for technical assistance.
Notes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
