Abstract
The blood—brain barrier (BBB) selectively controls the exchanges between the blood and the brain: it is formed by tight junctions (TJs) between adjacent microvascular endothelial cells. The transmembrane protein claudin-5 is known as a key TJ protein at the BBB, although, the molecular mechanisms by which it regulates TJ tightness are poorly understood. To identify putative claudin-5 partners that contribute to TJ integrity, claudin-5-enriched membrane microdomains were prepared by cell fractionation, using the human brain endothelial cell line hCMEC/D3 and claudin-5 immunoprecipitates were submitted to tandem mass spectrometry. Because a high concentration of mannitol is known to transiently destabilize TJs, this analysis was performed in basal conditions, after mannitol treatment, and after recovery of TJ integrity. We here demonstrate that the G-protein subunit αi2 (Gαi2) interacts with claudin-5 and that association is correlated with TJ integrity in hCMEC/D3 cells; also, a selective expression of Gαi2 is observed in human brain vasculature
Introduction
The blood—brain barrier (BBB) is a selective interface between the blood and the brain, which maintains ionic homeostasis within the brain microenvironment (Abbott et al, 2010). The BBB is present at the level of brain microvascular endothelial cells, sealed by tight junctions (TJs), which constitute the most apical intercellular junctional complex. Tight junctions closely link the plasma membranes of adjacent cells, restricting the paracellular diffusion of macromolecules and polar solutes between brain endothelial cells (Tsukita et al, 2001), as well as between epithelial cells in other physiological barriers, such as the choroid plexus or the intestinal barrier. Tight junctions are constituted by a number of transmembrane and cytosolic proteins assembled in a multiprotein complex. Among the transmembrane proteins involved, the tetraspan proteins occludin (Furuse et al, 1993) and claudins (Tsukita et al, 2001) have a crucial role in TJ integrity; they interact with several cytosolic scaffolding proteins such as zonula occudens (ZO) proteins, MUPP-1, and Cingulin, which link TJs to the actin cytoskeleton and recruit signaling proteins (Tsukita et al, 1999; Wolburg and Lippoldt, 2002).
Claudins are a multigene family of> 20 members: brain endothelial cells predominantly express claudin-5 and claudin-3, possibly also claudin-12 (Morita et al, 1999). Importantly, claudin-5-deficient mice present an altered BBB with higher permeability to small molecules (Nitta et al, 2003), whereas exogenous expression of claudin-5 in cultured rat brain endothelial cells strengthens BBB properties (Ohtsuki et al, 2007): both observations point to claudin-5 as a key component of the BBB. It has been established that claudin-5 acts in TJs through a number of protein—protein interactions. Indeed, the second extracellular loop of claudin-5 was shown to mediate claudin/claudin
The aim of the present study was to identify claudin-5 partners, which contribute to the regulation of TJ integrity at the BBB. We worked with the hCMEC/D3 cell line, a validated
We hypothesized that changes in permeability might be associated with an alteration of claudin-5-containing TJ complexes. Using a hypertonic concentration of mannitol—used in clinics to transiently open the BBB for therapeutic purposes (Rapoport, 2000)—we induced a rapid and reversible increase in hCMEC/D3 permeability to small molecules. Claudin-5 was immunoprecipitated from hCMEC/D3 cells that were either left untreated or treated with mannitol followed or not by a recovery period. For each experimental condition, coimmunoprecipitated claudin-5 partners were identified by liquid chromatography coupled with tandem mass spectrometry (nano-LC-MS/MS) analysis. Here, we identify the heterotrimeric G-protein subunit αi2 (Gαi2) that associates with claudin-5 in control conditions, dissociates after mannitol treatment, and reassociates after the recovery period. We show that Gαi2 is expressed by human brain vascular endothelium
Materials and methods
Cell Culture Conditions
The human brain microvessel endothelial cell line hCMEC/D3 (Weksler et al, 2005) was cultured on culture inserts (0.4 μm pore size; Corning, Lowell, MA, USA) coated with 150 μg/mL rat tail collagen type I (R&D Systems, Minneapolis, MN, USA). The seeding density was 50,000 cells/cm2. The culture medium contained endothelial basal medium-2 (EBM-2) (Lonza, Walkersville, MD, USA) supplemented with 5% fetal bovine serum ‘Gold,’ 10 mM hydroxyethyl-piperazineethane sulfonic acid (HEPES) (PAA Laboratories GmbH, Pasching, Austria), 1% Penicillin—Streptomycin, 1% chemically defined lipid concentrate (Invitrogen Ltd, Paisley, UK), 1.4 μM hydrocortisone, 5 μg/mL ascorbic acid, and 1 ng/mL basic fibroblast growth factor (bFGF) (Sigma-Aldrich, St Louis, MO, USA). Cells were grown for 6 days at 37°C in a humidified incubator in 5% CO2 and the medium was changed 3 days after seeding. When indicated, confluent hCMEC/D3 cells were incubated for 30 minutes at 37°C in a serum-free culture medium supplemented with 1 M
Small Interfering RNA Experiments
Small interfering RNA transfections were performed using various Stealth RNAi duplexes (Invitrogen) against claudin-5 (#HSS186370 and #HSS144294), Gαi2 (#HSS104225 and #HSS178468) or nontargeting siRNAs. Cells were plated onto culture inserts (#3450, Corning) or E-plates for xCELLigence assays (Roche, Basel, Switzerland) in culture medium without antibiotics. One hour after cell seeding, the transfection mix containing Lipofectamine RNAiMAX, 50 nM of siRNAs, and Opti-MEM Reduced Serum Medium (Invitrogen) was added to the culture medium according to the manufacturer's instructions. Cells were incubated for 6 days at 37°C in a CO2 incubator and the culture medium was replaced at day 3 by standard medium (cells on E-plates) or by fresh siRNA-supplemented transfection medium (cells on culture inserts).
Immunoblotting Assays
Confluent hCMEC/D3 cells, untreated or treated with siRNAs, were washed with phosphate-buffered saline (PBS). Cells were incubated on ice for 10 minutes with ice-cold Laemmli lysis buffer. Cell lysates were heated at 100°C for 5 minutes before sodium dodecyl sulfate polyacrylamide gel electrophoresis; proteins were then transferred onto a nitrocellulose membrane (Watman GmbH, Dassel, Germany). After saturation in blocking buffer (Tris-Buffer Saline, 3% nonfat dry milk, 0.05% Tween-20) for 30 minutes, membranes were blotted overnight with antibodies against claudin-5 (Invitrogen), Gαi2 (clone T-19, Santa Cruz Biotechnology, Heidelberg, Germany), VE-cadherin (clone BV6, Santa Cruz Biotechnology) or actin (Sigma-Aldrich) diluted in the blocking buffer. Immunoreactivity was revealed by secondary antibodies coupled to 680 nm fluorophores using the Odyssey LI-COR infrared fluorescent scanner (ScienceTec, Les Ulis, France).
Fluorescence Microscopy
hCMEC/D3 cells, untreated or treated with siRNAs, were cultured on insert filters. Cells were fixed either with ice-cold ethanol for 10 minutes at −20°C or with 4% paraformaldehyde for 10 minutes at room temperature, respectively, for claudin-5 or Gαi2 staining. Cells were permeabilized with 0.1% Triton X-100 in PBS for 10 minutes at room temperature, incubated in 3% bovine serum albumin in PBS (blocking buffer) for 30 minutes and stained with the indicated primary antibodies, diluted in the blocking buffer overnight at 4°C. After three washes with PBS, cells were incubated for 1 hour with fluorophore-conjugated secondary antibodies (Jackson Immunoresearch Laboratories, West Grove, PA, USA). Labeled preparations washed with PBS were and mounted in Glycergel medium (Dako Inc., Carpinteria, CA, USA). Fluorescence microscopy was performed with a Zeiss Axio Observer Z1 microscope, using a × 40 Oil Objective. Acquisitions were made with the MetaMorph 7 software (Molecular Devices, Sunnyvale, CA, USA).
Permeability Assays
hCMEC/D3 cells were grown on culture inserts for 6 days. Culture medium was removed and replaced by Hank's buffered salt solution (HBSS) transport buffer containing 10 mM HEPES, and 1 mM sodium pyruvate (Invitrogen Ltd, Paisley, UK) in the lower chamber. In the upper chamber, the transport buffer was supplemented with 50 μM Lucifer Yellow (LY) salt, 50 μM fluorescein isothiocyanate (FITC)-Dextran/4 kDa or 20 μM FITC-Dextran/70 kDa (Sigma-Aldrich), as indicated. Incubations were performed in triplicates at 37°C, 95% humidity, and 5% CO2 as previously described (Weksler et al, 2005). Samples were analyzed using a fluorometer (Fusion, Packard Bioscience Company, Meriden, CT, USA). Results are expressed as Permeability coefficients (Pe: 10−3 cm/min) or as the percentage of control permeability (corresponding either to untreated cells or to control siRNA-treated cells, as indicated).
xCELLigence Assays
The xCELLigence system (Roche) is an instrument that measures in real-time electrical impedance across gold microelectrodes integrated in the bottom of culture E-plates. hCMEC/D3 cells were seeded in standard culture medium without antibiotics at density of 15,000 cells/well onto E-plates coated with 150 μg/mL rat tail collagen type I. One hour after cell adhesion, the transfection SiRNA mix was added (as described above) and the cells were maintained in culture for 7 days. As indicated, at
Proteomic Analysis (Sample Preparations and Protein Identification)
Chromogenic Immunohistochemistry
Three individuals who died accidentally with no brain involvement, collected in Forensic Medicine, were examined to determine the
Modeling by Ingenuity Pathway Analysis
Nano-LC-MS/MS results were analyzed using ingenuity pathway analysis (Ingenuity Systems, Inc., USA, http://www.ingenuity.com). A network is a graphical representation of the molecular relationships between molecules. We selected only networks scoring ≥ 2, with
Statistical Analysis
Data are presented as the mean values ± standard errors (s.d.). Statistical analysis was performed by using Student's
Results
Claudin-5 Contributes to the Restriction of Paracellular Permeability in Brain Endothelial Cells
In the first series of experiments, we assessed whether claudin-5 has a predominant role in the barrier feature of the human brain endothelial cell line, hCMEC/D3 which recapitulates
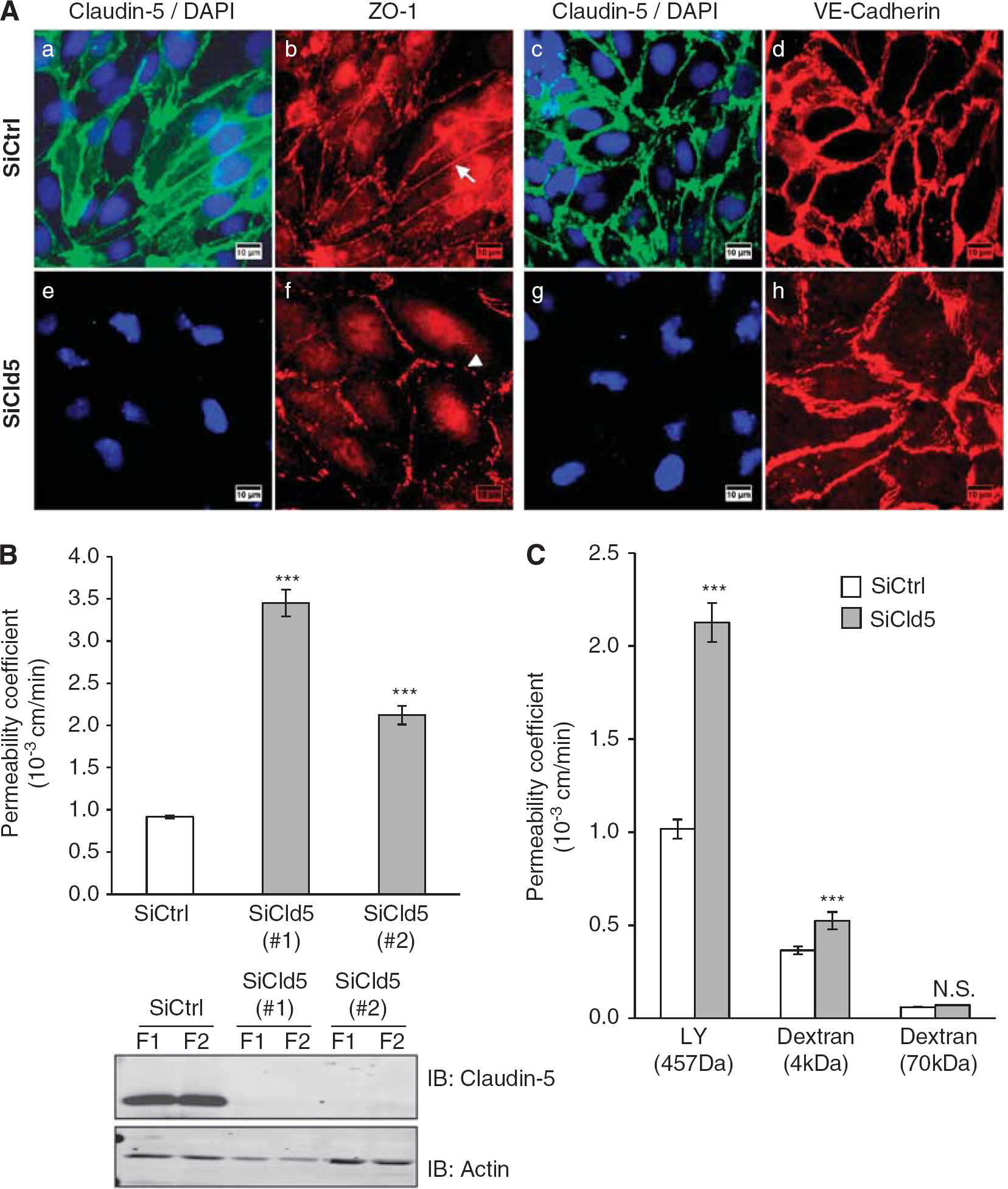
Claudin-5 knockdown increases paracellular permeability in hCMEC/D3 cells. (
Because in claudin-5-deficient mice, the BBB permeability to small molecules (compared with larger molecules) was selectively affected (Nitta et al, 2003), permeability assays were further performed with larger molecules such as 4 and 70 kDa FITC-dextrans (Figure 1C). Claudin-5 knockdown increased the permeability coefficient value to LY and 4 kDa FITC-dextrans by 2- and 1.5-fold, respectively. No significant change in the permeability to 70 kDa FITC-dextran was detected, confirming here
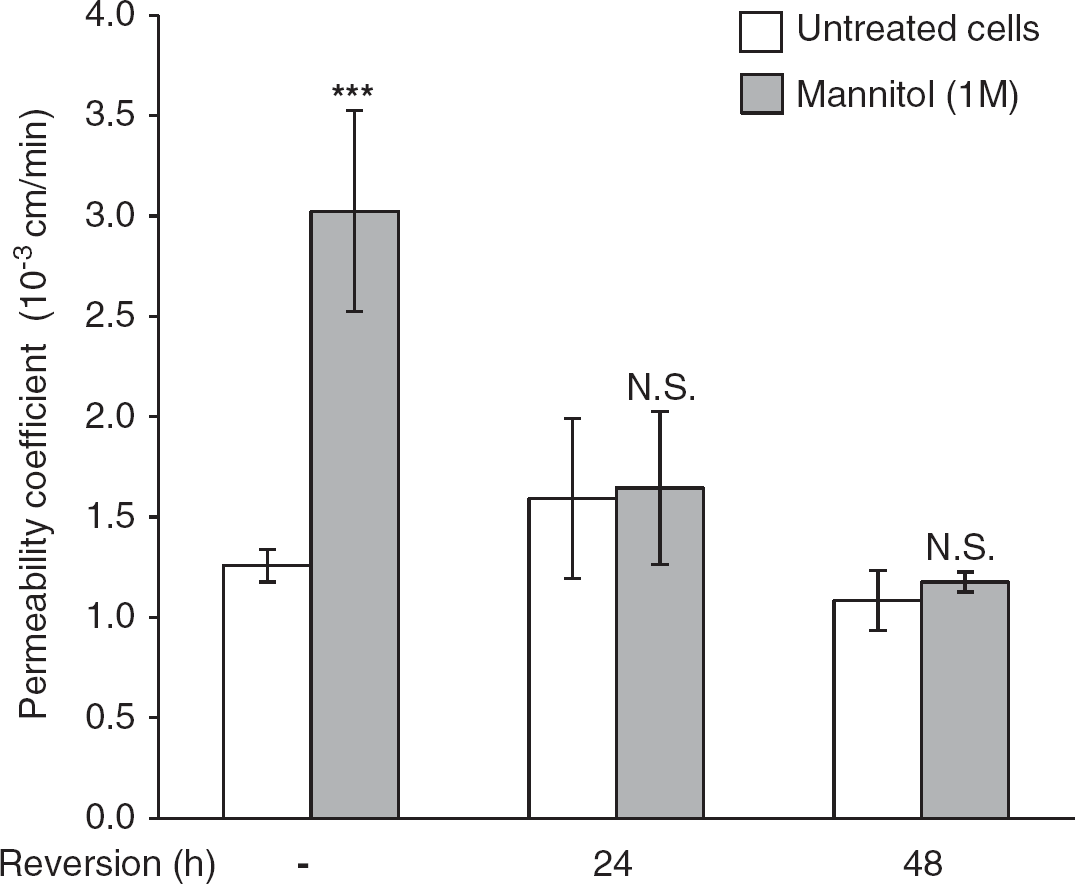
Hyperosmolar concentration of mannitol induces a transient increase of endothelial permeability to Lucifer Yellow (LY). hCMEC/D3 cells were grown at confluence on Transwell inserts. Cells were treated with 1 M mannitol for 30 minutes, then medium was changed to let the cells recover for up to 48 hours. Permeability to LY was measured directly after mannitol treatment (white bars) and after 24 or 48 hours recovery (gray bars), as indicated. Results are mean Pe values ± s.d. from three independent experiments performed in triplicates. ***
hCMEC/D3 cell fractionation on sucrose gradients was performed to obtain caveolae-enriched fractions known to incorporate TJ proteins (Dodelet-Devillers et al, 2009; Lambert et al, 2007). Claudin-5 was enriched, as expected, in these fractions (not shown) and was immunoprecipitated from fractions prepared from untreated hCMEC/D3 cells, cells treated with mannitol (1 M) or treated with mannitol plus a 48-hour recovery period. Coimmunoprecipitated claudin-5 proteins were identified by nano-LC/MS/MS tandem mass spectrometry analysis in each experimental condition. In addition to claudin-5
Claudin-5 partners in hCMEC/D3 cells identified by mass spectrometry
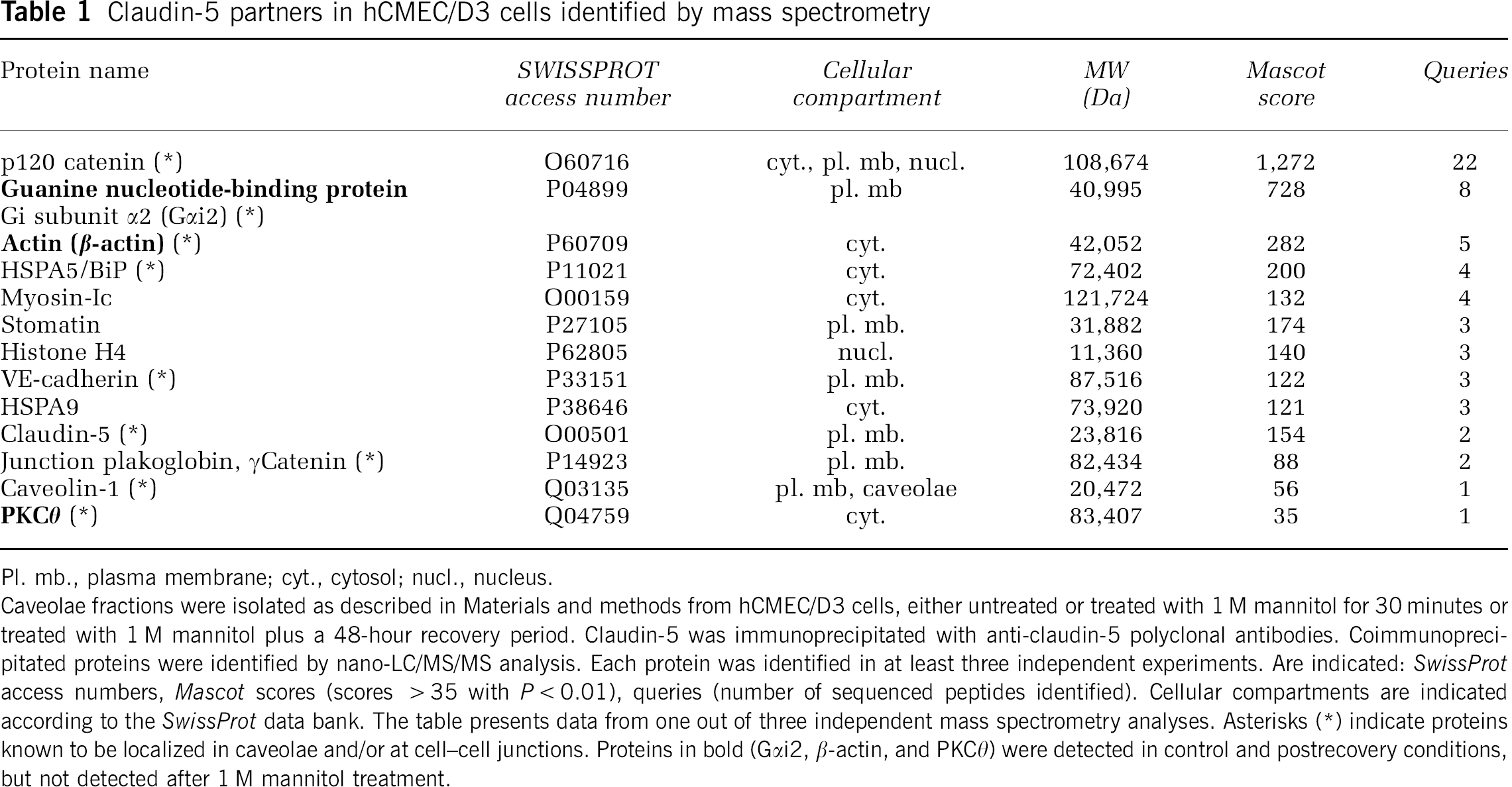
Pl. mb., plasma membrane; cyt., cytosol; nucl., nucleus. Caveolae fractions were isolated as described in Materials and methods from hCMEC/D3 cells, either untreated or treated with 1 M mannitol for 30 minutes or treated with 1 M mannitol plus a 48-hour recovery period. Claudin-5 was immunoprecipitated with anti-claudin-5 polyclonal antibodies. Coimmunoprecipitated proteins were identified by nano-LC/MS/MS analysis. Each protein was identified in at least three independent experiments. Are indicated:
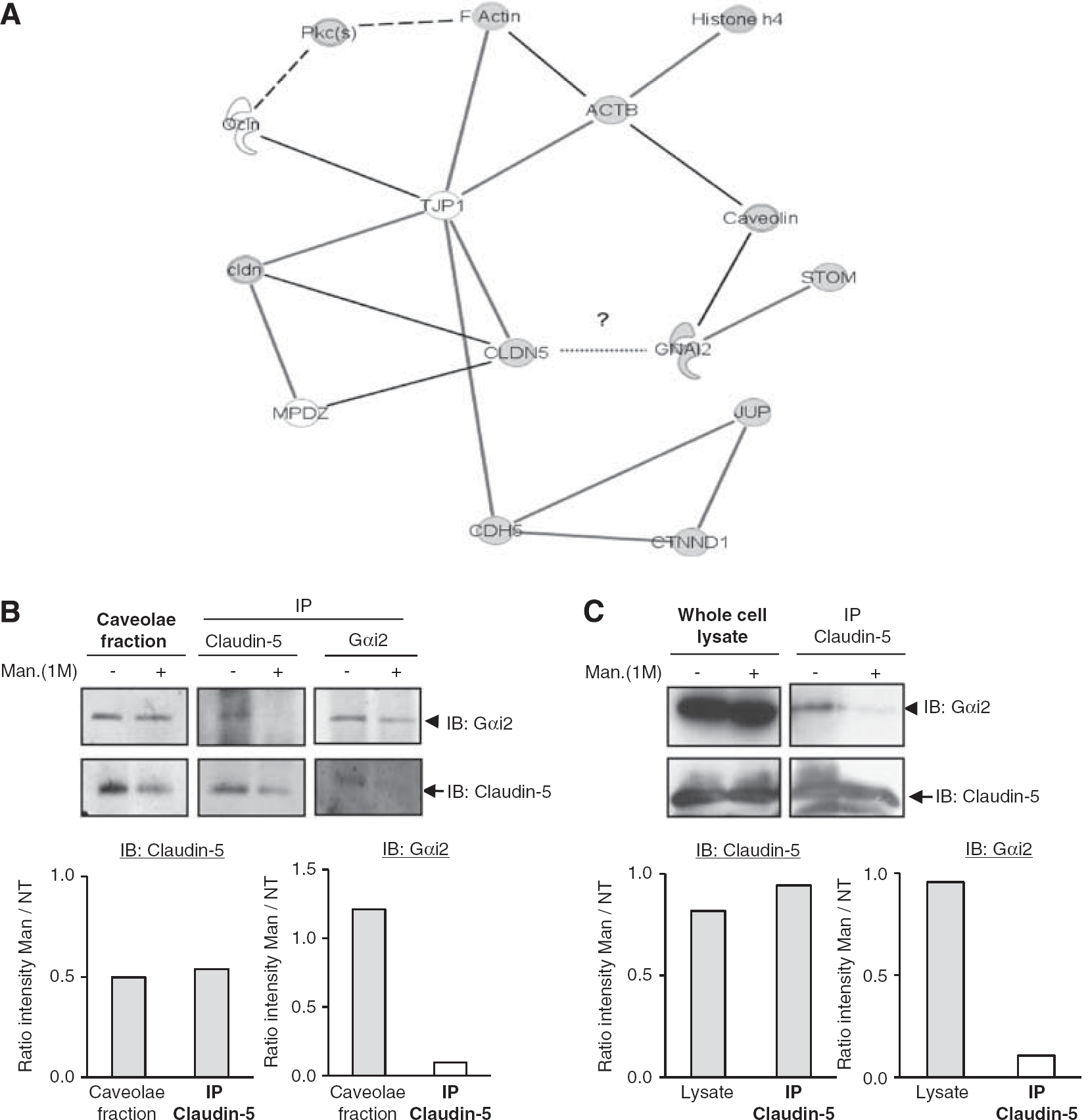
G-protein subunit αi2 (Gαi2) interacts with claudin-5 and belongs to a multiprotein complex. (
To confirm the interaction between endogenous Gαi2 and claudin-5 proteins in hCMEC/D3 cells, coimmunoprecipitation assays were performed using either untreated cells or cells treated with mannitol from caveolae-enriched fractions (Figure 3B) or whole cell lysates (Figure 3C). G-protein subunit αi2 was detected in claudin-5 immunoprecipitates (Figures 3B and 3C); conversely, claudin-5 was detected in Gαi2 immunoprecipitates (Figure 3B). After mannitol treatment, although claudin-5 level was decreased in some experiments for unknown reason (Figure 3B), we observed in all cases that the interaction between claudin-5 and Gαi2 was disturbed. Altogether, these results firmly establish that Gαi2 and claudin-5 constitutively interact with each other, either directly or indirectly through caveolin, β-actin, and ZO-1, which may link the two proteins in a multiprotein complex (Figure 3A).
G-Protein Subunit αi2 Is Expressed in Human Brain Microvessels and Contributes to the Integrity of the Cell—Cell Junctions
To get further insight into the physiological relevance of our findings, we investigated whether claudin-5 and Gαi2 might be expressed in the same vascular beds in human brain tissue. For this purpose, immunohistological staining of claudin-5 or Gαi2 was performed on three post-mortem adult human brains and five sections were examined, from brain cortex, thalamus, brain stem, and cerebellum. Gαi2 localization was found to be similar to claudin-5 localization, with a specific labeling of endothelial cells in small capillaries (5 to 8 μm) and larger vessels (25 to 100 μm), in brain cortex (Figures 4Aa and 4Ac) and white matter (Figures 4Ab and 4Ad). Unlike claudin-5, Gαi2 was also detected in neurons (data not shown) as previously described (Khan and Gutierrez, 2004). No claudin-5 or Gαi2 immunostaining was observed in glial cells. These observations reveal that Gαi2 and claudin-5 are expressed by the endothelium of the same vascular beds in human brain. We suggested that Gαi2 may have a role in the integrity of TJs of brain endothelial cells.
Experiments were performed to investigate first whether G-protein αi subunits (hCMEC/D3 cells express the three known subunits: Gαi1, Gαi2, and Gαi3, not shown) are involved in the tightness of cell junctions. hCMEC/D3 cells were treated with PTX (pertussis toxin), a well-known nonselective inhibitor of the GTPase activity of Gαi subunits, for 6 or 24 hours before LY permeability assay. A limited but significant permeability increase was observed (Figure 4B), suggesting that functional Gαi subunits are involved in maintaining the endothelial barrier integrity. This assay, however, could not discriminate between Gαi2 and the two other Gαi subunits (Gαi1 and Gαi3).
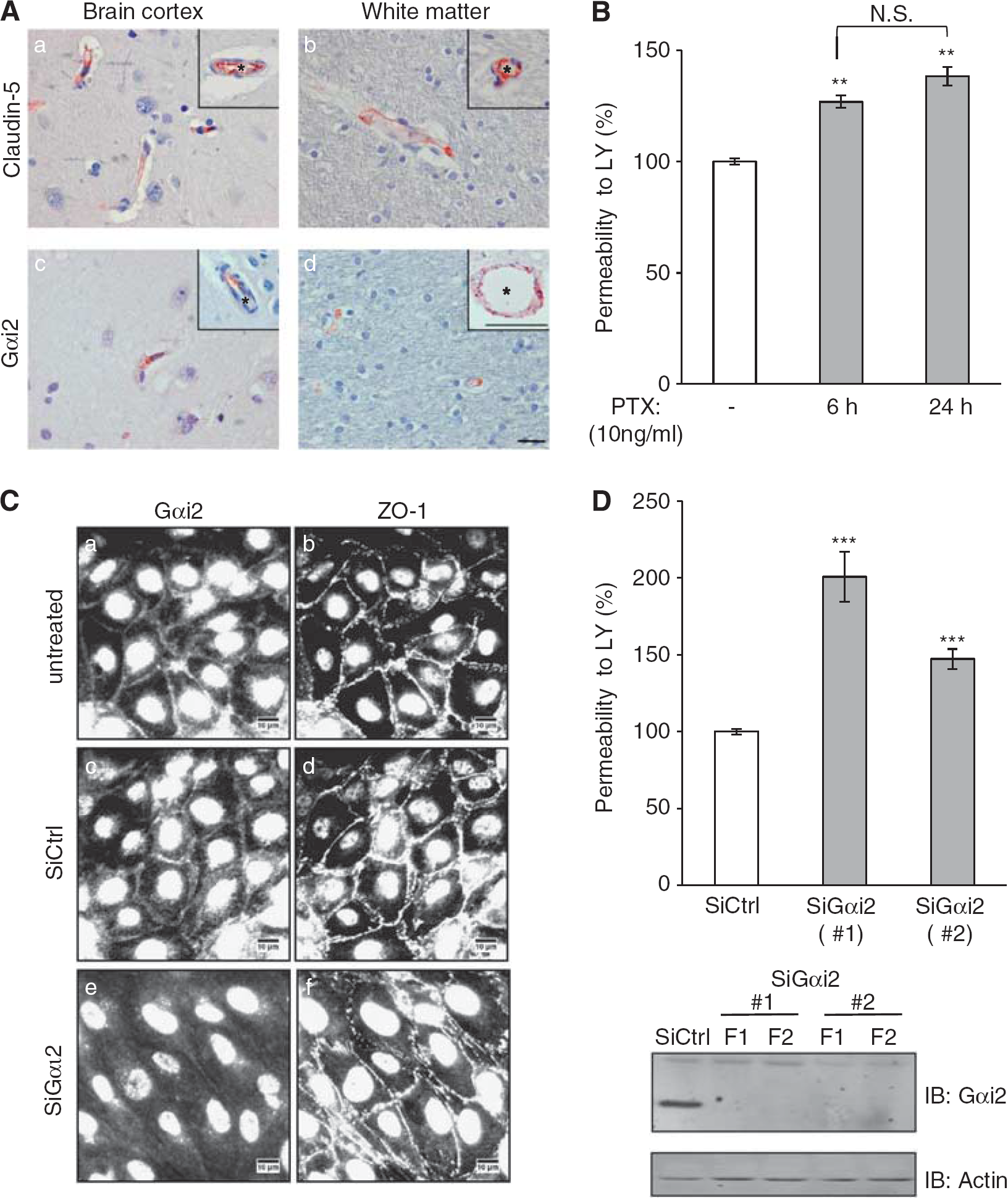
G-protein subunit αi2 (Gαi2) is expressed in human brain endothelium
Instead, RNA interference approaches were used to investigate the specific role of Gαi2 in the restriction of permeability to LY by hCMEC/D3 cells. First, immunofluorescence analysis confirmed that Gαi2 is expressed at the plasma membrane and at cell—cell junctions in untreated cells or siRNA control-treated cells (Figures 4Ca and 4Cc). We observed that Gαi2 depletion did not prevent cells from getting to confluence, with no visible change in ZO-1 (Figures 4Ce and 4Cf) or VE-cadherin (not shown) accumulation at cell junctions; however, Gαi2-depleted cells appeared more elongated than siRNA control cells. Interestingly, two individual siRNAs that efficiently silenced the expression of Gαi2 (Figure 4D, lower panel) significantly increased the LY permeability coefficient by 1.5- to 2-fold (Figure 4D, upper panel). Altogether, these results clearly demonstrate a functional role of Gαi2 for the maintenance of junction integrity in brain endothelial cells.
G-Protein Subunit αi2 Is Important for Tight Junctions Reassembly After Mannitol Treatment
To address the role of Gαi2 in TJ formation in brain endothelial cells, the behavior of hCMEC/D3 cells during the recovery period after mannitol treatment was monitored in real time by impedance measurement using the Roche xCELLigence system; values expressed as ‘cell index’ are maximal (about 10 arbitrary units, a.u.) with tight monolayers and minimal when cell—cell junction integrity is severely affected (0 to 1 a.u.).
As shown in Figures 5A–5D, hCMEC/D3 cells were depleted in claudin-5 or Gαi2 by siRNAs and compared with cells treated with control siRNA (siCtrl); in addition, codepletion of these proteins was performed by simultaneous treatment with both individual siRNAs and compared with cells treated with control siRNA used at a twofold higher concentration (siCtrl × 2). Experiments were performed for 7 days and the cell index was monitored on-line: hCMEC/D3 cells were cultured until confluence, after 70 hours, they were treated with 1 M mannitol for 30 minutes (asterisk) and medium was changed to remove mannitol for recovery for 4 additional days. Control (siCtrl) hCMEC/D3 cells reached confluence at 56 hours associated with a high cell index plateau (10.4 a.u.). As expected, mannitol treatment induced a dramatic decrease of cell index and after medium change, the impedance gradually increased to reach the plateau value at the end of the experiment (Figure 5A), likely reflecting TJ reassembling during the recovery period. These observations confirmed that the measure of impedance is a good read-out for assessing the integrity of cell—cell junctions. We then used this experimental device to further evaluate the functional consequences of Gαi2 and/or claudin-5 depletion in hCMEC/D3 cells.
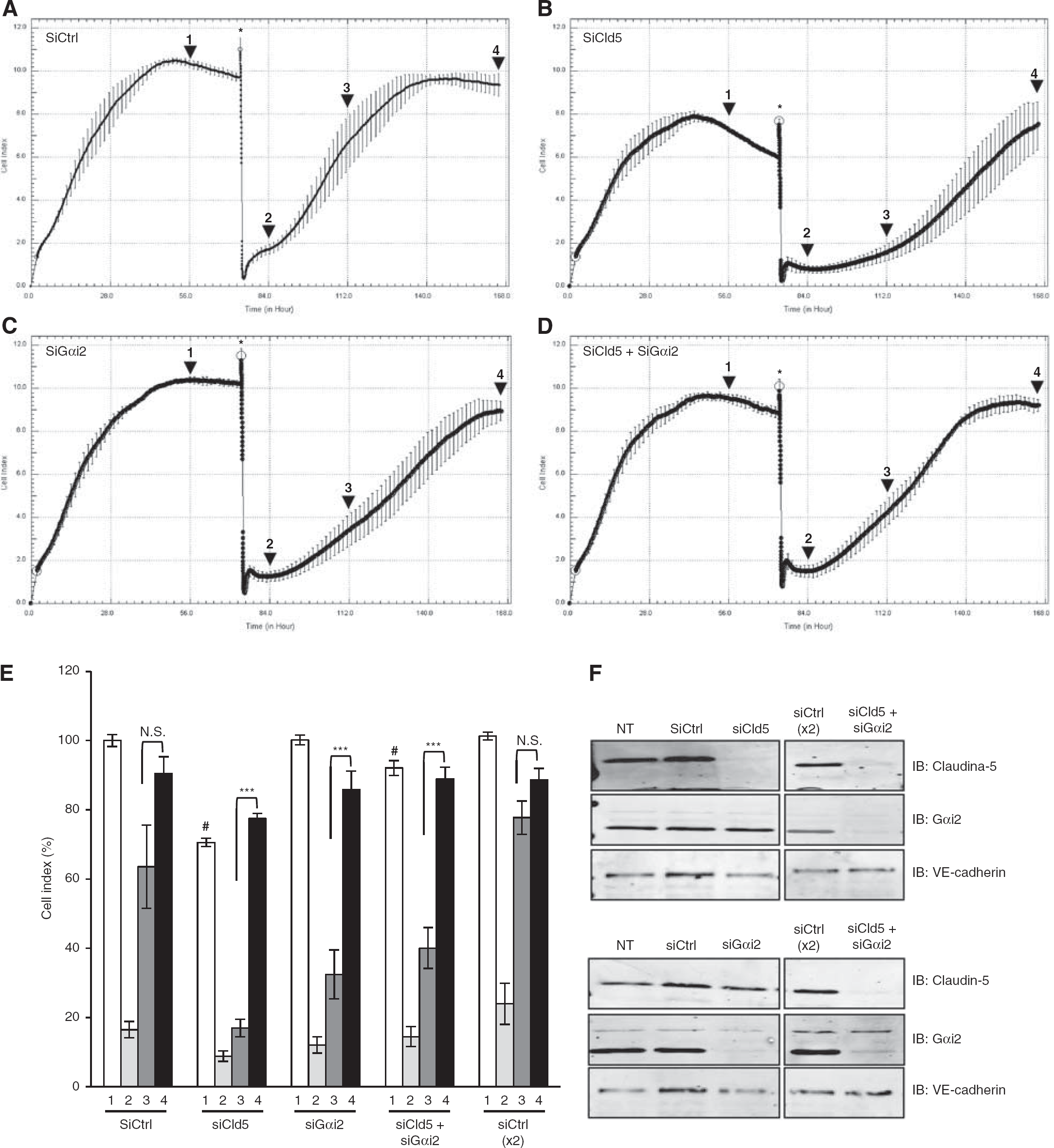
G-protein subunit αi2 (Gαi2) or claudin-5 depletion delays the recovery of cell—cell junction integrity after 1 M mannitol treatment. hCMEC/D3 cells treated with nontargeting small interfering RNAs (siRNAs) (siCtrl) or siRNAs against claudin-5 (siCld5), Gαi2 (siGαi2), or both siCld5 + siGαi2 were grown in a 96-well plate of an xCELLigence system (Roche). The impedance measurement (cell index: CI) was monitored in real time and plots were produced using the RTCA Software. At
Figure 5E presents impedance values in each experimental condition, with cell index normalization to the value of control cells at confluence. Impedance values are presented when cells get to confluence (
Figure 5F presents the expression levels of claudin-5 and Gαi2, as well as VE-cadherin as control, at the end of the experiment, when cell indexes reached 90% to 100% of the initial plateau for each condition (Figure 5E, black bars), indicative of a complete recovery: these data confirm that claudin-5 or Gαi2 depletion was maintained all over the experiments and did not affect the expression of each other, while VE-cadherin expression was not affected by any treatment. Altogether, these data further document that the absence of claudin-5 or Gαi2 clearly affect the integrity of cell—cell junctions in hCMEC/D3 cells by disturbing the process of junction reassembly during recovery after an osmotic shock.
Discussion
To date, TJ architecture in brain endothelial cells remains partly understood in contrast to our extensive knowledge of TJ molecular organization in epithelial cells. Although claudin-5 is known to directly interact with several membrane or membrane-associated proteins in epithelial cells, like occludin (Saitou et al, 1998), ZO proteins (Itoh et al, 1999), MUPP-1 (Poliak et al, 2002), and other members of the claudin superfamily (Coyne et al, 2003), the molecular mechanisms by which claudin-5 regulates BBB permeability are still incompletely understood.
The aim of the present study was to identify putative claudin-5 partners involved in TJ regulation in human brain endothelial cells. In line with an earlier report demonstrating that BBB was loosened in a size-selective manner in claudin-5-deficient mice (Nitta et al, 2003), we observed
To identify claudin-5 partners that might contribute to the regulation of TJ integrity, we reasoned that these proteins might associate with claudin-5 when TJ integrity is maintained and dissociate when TJs are disrupted. We chose a hypertonic concentration of mannitol used clinically to transiently destabilize brain endothelium TJs for drug delivery to the CNS (Rapoport, 2000), although the molecular mechanisms of mannitol-induced changes in cerebral endothelial cells are poorly understood. An earlier report had described that hyperosmolar concentration of mannitol may not only induce a mechanical shrinkage of the endothelial cells, but also the activation of signaling pathways such as src kinase-dependent phosphorylation of β-catenin and its dissociation from cadherin in cerebral endothelial cells (Farkas et al, 2005). In hCMEC/D3 cells, mannitol (1 M) treatment rapidly induced a marked increase in LY permeability, which, upon medium change, returned to control value after a 24- to 48-hour recovery period, confirming that treatment with mannitol could mimic a transient disruption of TJs tightness. We then took advantage of this observation to identify putative proteins associated with claudin-5 in hCMEC/D3 cells in basal conditions that dissociate after mannitol treatment and reassociate following a 48-hour recovery period. As a first step of our proteomic analysis of claudin-5-associated proteins, hCMEC/D3 cells were fractionated in detergent-free conditions on sucrose gradients and claudin-5 was shown to be enriched in light, caveolin-rich fractions. This observation was in agreement with previous reports, indicating that caveolae are membrane microdomains that incorporate TJ proteins, specific transporters, and adhesion molecules (Dodelet-Devillers et al, 2009). When claudin-5 immunoprecipitation was performed from these fractions, we detected and identified by nano-LC-MS/MS analysis only 12 proteins that constitutively associated with claudin-5. Interestingly, a majority of these proteins are known to be localized to plasma membrane and/or to be associated with junction complexes, such as caveolin-1, VE-cadherin, p120 catenin, Gαi2, and PKCθ, strongly suggesting that our proteomic approach was appropriate to identify membrane proteins with potential involvement in TJs. Surprisingly, ZO proteins, claudin-3, or MUPP-1, previously described as claudin-5 partners by coimmunoprecipitation studies or binding assays with the cytoplasmic tail of claudin-5 (Coyne et al, 2003; Itoh et al, 1999; Poliak et al, 2002), were not detected in our analysis. Interestingly, however, other proteomic studies using claudin-binding bacterial toxin, also failed to detect these proteins (Lohrberg et al, 2009).
Out of these 12 proteins, Gαi2, β-actin, and PKCθ were found to be associated with claudin-5 in basal conditions, to dissociate after mannitol treatment, and to reassociate after recovery, displaying the expected behavior according to our hypothesis. We focused on Gαi2 because several lines of evidence have suggested the involvement of heterotrimeric G-proteins in TJ maintenance and biogenesis in epithelial cells (Denker and Nigam, 1998; Denker et al, 1996). Indeed, MDCK cells expressing Gαi2 or a constitutive active form of Gαi2 developed a high
Here, we present evidence that Gαi2 coimmunoprecipitates with claudin-5 in hCMEC/D3 cells in culture conditions preserving TJ integrity (basal condition and postrecovery after a hyperosmolar shock), strongly suggesting that both proteins interact at TJs in brain endothelial cells. In this regard, we showed that, in adult human brains, Gαi2 and claudin-5 display a similar localization in endothelial cells of small capillaries and larger vessels. We describe Gαi2 as a regulator of barrier integrity in human brain endothelial cells by using RNA interference, which firmly demonstrated that Gαi2 knockdown in hCMEC/D3 cells significantly enhanced their permeability to LY, without affecting cell proliferation or monolayer formation. Because intracellular cAMP is widely thought to decrease brain endothelial permeability (Rubin et al, 1991), we investigated whether Gαi2 depletion might affect the intracellular cAMP level in hCMEC/D3 cells. Interestingly, the depletion of Gαi2 induced a slight increase in cAMP basal level (data not shown), strongly suggesting that the observed increased permeability to LY was independent of the cAMP pathway. This conclusion is in agreement with a previous report, which proposed that PTX-enhanced permeability of brain endothelial cells was mediated via a cAMP-independent pathway (Bruckener et al, 2003).
It is noteworthy that hCMEC/D3 cells also express Gαi1 and Gαi3, the expression of which was not affected by Gαi2 depletion (data not shown), confirming the unique role of Gαi2 in the regulation of permeability in the hCMEC/D3 cell line. Moreover, we showed by impedance measurement that Gαi2 knockdown significantly delayed TJ resealing after a hyperosmotic shock. Interestingly, no additive effect was observed in the absence of both claudin-5 and Gαi2, suggesting that both proteins might be involved in the same pathway. Altogether, these results clearly indicate that Gαi2, similarly to claudin-5, is important for the permeability restriction to small molecules displayed by brain endothelial cells, which reflects the integrity of their TJs.
On the basis of our knowledge of claudin-5 interactome (Figure 3A), we propose that claudin-5 and Gαi2 may control TJ integrity as components of a multiprotein complex, also including caveolin and ZO-1, linked to the actin cytoskeleton; this complex might also include occludin and MUPP-1. Whether the interaction between Gαi2 and claudin-5 is direct or indirect, notably through caveolin, β-actin, and ZO-1 remains to be investigated. It is now well established that TJs and AJs are functionally related and that AJs influence TJ organization. Whereas AJs precede TJs at cell—cell contacts (Gumbiner et al, 1988; Sedar and Forte, 1964), ZO-1 has been implicated in the crosstalk between these two junctional complexes (Ikenouchi et al, 2007). Our observation that claudin-5 or Gαi2 deletion delays junction recovery after a hyperosmotic shock strongly suggests that the claudin-5/Gαi2-containing multiprotein complex may actively contribute to the integrity of cell—cell junctions in human brain endothelial cells through a crosstalk with the VE-cadherin-containing AJ complex.
In conclusion, for the first time to our knowledge, the present study establishes that Gαi2, together with claudin-5, is a key protein controlling TJ integrity in human brain endothelial cells and suggests that both proteins may be present in a multiprotein complex localized at TJs. Further studies will focus on deciphering the functional crosstalk between Gαi2, claudin-5, and their partners for BBB regulation in physiological as well as pathophysiological situations.
Footnotes
Acknowledgements
The authors thank Drs BB Weksler, S Bourdoulous, J Gavard, and C Artus for helpful discussions. The authors are grateful to B Durel (Imaging Facility, Cochin Institute, Paris) and M Andrieu (Immunobiology Facility, Cochin Institute, Paris).
The authors declare no conflict of interest.
