Abstract
SMTP-7 (Stachybotrys microspora triprenyl phenol-7), a small molecule that promotes plasminogen activation through the modulation of plasminogen conformation, has excellent therapeutic activity against cerebral infarction in several rodent models. Detailed evaluations of SMTP-7 in a primate stroke model are needed for effective, safe drug development. Here we evaluated SMTP-7 in a monkey photochemical-induced thrombotic middle cerebral artery (MCA) occlusion model (n=6), in which MCA occlusion was followed by recanalization/reocclusion. SMTP-7 (10 mg/kg, intravenous infusion) significantly increased the postinfusion MCA recanalization rate (32.5-fold, P=0.043) and ameliorated the post-24-h neurologic deficit (by 29%, P=0.02), cerebral infarct (by 46%, P=0.033), and cerebral hemorrhage (by 51%, P=0.013) compared with the vehicle control animals. In normal monkeys, SMTP-7 did not affect general physiologic or hemostatic variables, including coagulation and platelet parameters. Investigations in rodent models of transient and permanent focal cerebral ischemia, as well as arterial thrombosis and bleeding tests, suggest a role for SMTP-7's regulated profibrinolytic action and neuroprotective properties in the monkey MCA occlusion model. In conclusion, SMTP-7 is effective in treating thrombotic stroke in monkeys. SMTP-7 is thus a promising candidate for the development of alternative therapy for ischemic stroke.
INTRODUCTION
Strokes are the third most common cause of mortality and often cause serious disability. 1 The only strategy for treating an acute-phase stroke is to restore blood flow within the ischemic area using a thrombolytic agent, tissue-type plasminogen activator (t-PA). Although thrombolytic therapy using t-PA is powerful, it occasionally leads to harmful outcomes such as intracerebral hemorrhage (6.4% compared with 0.6% in the placebo group in the NINDS trial), 2 and only a small percentage (1.8% to 8.9%) of patients with stroke can benefit from t-PA-induced thrombolysis.3, 4 The need for new strategies to combat stroke is urgent. Efforts to develop such new strategies include clinical trials of tenecteplase (an engineered recombinant t-PA) 5 and desmoteplase (a recombinant plasminogen activator obtained from a vampire bat species). 6
We have discovered a family of small molecules from the fungus Stachybotrys microspora, designated as SMTP (Stachybotrys microspora triprenyl phenol). 7 SMTP promotes plasminogen activator-catalyzed plasminogen activation by modulating its conformation.7, 8 SMTP-7, a potent SMTP congener,8, 9 effectively ameliorates embolic stroke in rodent models.10, 11 The excellent therapeutic activity of SMTP-7 is partly attributable to its enhancement of thrombolysis8, 12 As SMTP-7 is a plasminogen modulator, its action depends on the endogenous supply of plasminogen activators, and SMTP-7 action may thus be under the control of the physiologic regulatory system. 7 Along with this regulated profibrinolytic activity, our previous observations suggest the involvement of an anti-inflammatory mechanism in SMTP-7 action.11, 12, 13, 14
Recent clinical trials of several other drugs have suggested that in addition to rodent studies, studies in higher-order species are necessary to extrapolate animal data to humans.15, 16 A detailed evaluation of SMTP-7 in a primate stroke model is needed for effective, safe drug development. As both the neurologic function and cerebral vascular anatomy of monkeys closely resemble their human counterparts, 17 we investigated the therapeutic potential of SMTP-7 in monkeys. In this experiment, we employed a photochemically induced thrombotic middle cerebral artery (MCA) occlusion model in which arterial thrombosis progressively reduced the cerebral blood flow. This resulted in recanalization and rethrombosis (cyclic flow reductions),18, 19 which might occur in infarct progression in stroke patients. 20
Our results demonstrate that in the monkey thrombotic MCA occlusion model, SMTP-7 is effective for reducing cerebral infarction, neurologic deficits, and hemorrhage in the infarct area. The results in rodent models of transient and permanent focal strokes, thrombosis, and bleeding support the hypothesis that both regulated thrombolytic activity and, possibly, neuroprotective properties of SMTP-7 contribute to this drug's therapeutic efficacy.
MATERIALS AND METHODS
Stachybotrys microspora Triprenyl Phenol-7
SMTP-7 was produced by S. microspora 21 and was converted to sodium salt (lot SM8501). In the bleeding assay in mice, the triethanolamine salt of SMTP-7 was used. In all treatments, SMTP-7 and vehicle (saline) were administered intravenously (at a final volume of 5 mL/kg) as follows: 10% of the total dose was injected for 5 seconds and the remaining dose was infused for 30 minutes.
Animals
All of the animal protocols, including that of the primate model, were approved by the institutional animal experiment committees (the Tokyo Noko University Animal Experiment Subcommittee, based on the Regulation of Animal Experiments; the TMS Animal Experiment Committee, based on the Regulation of Animal Experiments; and the Hamamatsu Pharma Research Animal Experimental Committee, based on the Animal Experiment Ethics Guideline). All of the animal protocols conformed to the ARRIVE (Animal Research: Reporting In Vivo Experiments) guidelines. We took adequate steps to ensure that animals did not suffer unnecessarily at any stage of an experiment.
We obtained male cynomolgus monkeys (2 to 3 years old), male Sprague–Dawley rats (8 weeks old), and male ICR mice (6 to 7 weeks old) from Japan SLC (Hamamatsu, Japan). In the experiments using monkeys and rats, animals were allocated to experimental groups so that the differences in the average body weight among groups were minimized. In the experiments using mice, whose body weights were relatively uniform, the animals were randomly allocated to each experimental group. The criteria for exclusion and the results of the exclusion for each animal test are shown in Supplementary Table 1.
Photochemical Thrombotic Middle Cerebral Artery Occlusion in Monkeys
The transorbital approach to the right MCA was performed as described.18, 19 A photoirradiation probe was placed on the MCA and a Doppler flow meter was placed on the distal side of the MCA to measure blood flow. When a steady baseline flow was obtained, a 20-min photoirradiation (540 nm, 1.4 × 106 lux) was applied on the MCA trunk with a concomitant injection of the photosensitizer Rose Bengal (20 mg/kg, intravenously for 6 minutes). One hour later, SMTP-7 (10 mg/kg, n=6) or saline (n=6) was administered intravenously. The MCA blood flow was continuously monitored for 2 hours from the start of the photoirradiation, to measure the time to the initial cessation of blood flow (time to occlusion) and the occlusion time intervals. The animals were then allowed to fully recover from the anesthesia after wound closing. During the experiments, the animals' rectal temperature was maintained with a heating pad at 37.0 to 38.5 °C, which is comparable to the normal range (37.6±0.76 °C) reported for cynomolgus monkeys. 22
Neurologic deficits were evaluated 24 hours after photothrombosis with the following subcategorized parameters: consciousness, sensory system, motor system, and skeletal muscle coordination. 17 The brains were removed and cut into 6-mm-thick coronal sections in the direction of the forehead to the occiput. The occipital side of each section was photographed for the assessment of hemorrhagic transformation. The sections were then stained with 2% TTC (2,3,5-triphenyltetrazolium chloride). Digital images of the stained occipital side were recorded to quantify the infarct area. The degree of edema was calculated as follows: edema percent=(volume of postischemic hemisphere−volume of contralateral hemisphere)/volume of contralateral hemisphere × 100. The infarct size was corrected for the degree of edema. All of the evaluations and drug administrations were performed in a masked manner.
Physiologic and Hemostatic Parameters in Normal Monkeys
Physiologic variables including heart rate, blood pressure, blood gas, blood pH, hematocrit, blood glucose, and rectal temperature were measured with normal cynomolgus monkeys under anesthesia (isoflurane and pancuronium). SMTP-7 (10 mg/kg, n=6) or saline (n=6) was infused for 30 minutes. Variables were measured just before the infusion, as well as 0.5, 1, and 2 hours after the initiation of the infusion.
Hemostatic parameters, including activated partial thromboplastin time, fibrinogen, plasminogen, α2-antiplasmin, D-dimer, platelet aggregation, and thromboelastographic variables, were determined using blood drawn from conscious monkeys (n=8) 24 hours before and 30 minutes (for platelet aggregation and thromboelastography) or 90 minutes (for the other parameters) after the initiation of SMTP-7 infusion (10 mg/kg for 30 minutes).
Transient Focal Cerebral Ischemia in Rats
Transient focal cerebral ischemia was performed as described, using rats. 23 The external carotid artery and left common carotid artery were ligated, and a monofilament surgical suture coated with silicone at the distal 3 mm (0.38 to 0.58 mm in whole diameter) was introduced into the internal carotid artery to pass through the origin of the MCA, inducing ischemia in the MCA territory. SMTP-7 (1, 3, and 10 mg/kg, n=10 each) or saline (n=10) was infused for 30 minutes from 85 minutes after ischemia. At 90 minutes after ischemia, the inserted suture was withdrawn to restore blood flow. During the experiments, the animals' rectal temperature was maintained with a heating pad at 37.0 to 38.5 °C, which is comparable to the normal range (37.87±0.35 °C) reported for rats. 24 Neurologic deficits were evaluated 24 hours after the MCA occlusion as described. 25 The brains were coronally sliced at 2-mm intervals at +6 to −6 mm from the bregma, and the resulting six slices were stained with TTC for the evaluation of infarct size. The infarct size was corrected for the degree of edema. The experiments were performed in a masked manner.
Permanent Focal Ischemia in Rats
Permanent MCA occlusion in rats was carried out as described. 26 Subtemporal craniotomies were performed, and the left MCA proximal to the olfactory tract was permanently occluded by bipolar electrocoagulation and transected distal to the olfactory tract to avoid recanalization. SMTP-7 (10 mg/kg) or saline was then infused for 30 minutes. After 24 hours, neurologic deficits and infarct size were determined as described above. The experiments were performed in a masked manner.
Ferric Chloride-Induced Thrombosis in Rats
Rats were anesthetized with intravenous pentobarbital (50 mg/kg), and the left common carotid artery was carefully exposed and held in a semicircular plastic tube (5 mm long, 1.5 mm in internal diameter). A cuff-type Doppler flow probe was placed on the distal side of the artery. After obtaining steady blood flow, a filter paper (2.5 × 4.0 mm) soaked with 1.5 μL of ferric chloride (35%, w/w in saline) was wrapped around the artery at the site of the plastic tube. After 4 minutes of treatment, the filter paper was removed, the artery was washed with 0.5 mL of saline, and the blood flow was continuously monitored for 30 minutes. SMTP-7 (1, 3, and 10 mg/kg, n=10 each) or t-PA (0.55 mg/kg, n=10) was administered intravenously immediately after the ferric chloride treatment. For the t-PA injection, a dose corresponding to 0.1 mg/kg was administered for 5 seconds, and the remaining dose corresponding to 0.45 mg/kg was infused for 30 minutes. (The dose of t-PA was determined based on our unpublished observation in which a total of 1.0 mg/kg infused over 60 minutes, with 10% of the total dose administered as an initial bolus, was effective. As the blood flow was monitored for 30 minutes after the dosing, the dose at the end of this experiment became 0.55 mg/kg.)
Bleeding Time in Mice
Bleeding time was measured by a mouse tail tip amputation assay.8, 27 Briefly, male ICR mice were anesthetized with isoflurane. Saline (control), SMTP-7 (1, 10, 30, and 100 mg/kg), t-PA (0.1, 1, and 10 mg/kg), and tranexamic acid (100 mg/kg) in combination with either SMTP-7 or t-PA was administered intravenously for 30 minutes (n=5 to 8). Mice tails were immersed in a water bath at 37 °C for at least 5 minutes before tail amputation. Fifteen minutes after the drug administration, tails were cut 5 mm from the end with a razor blade. The tails were immersed immediately in prewarmed saline (37 °C), and the time required to stop visual spontaneous bleeding was determined.
Thromboelastography
Thromboelastography was carried out on a ROTEM system (Pentapharm, Munich, Germany) using whole blood drawn from normal monkeys (n=8) before (24 hours) and after an infusion of SMTP-7 (10 mg/kg). The spontaneous clotting of the blood was measured without any additions. In another assay, human citrated whole blood was used to assess the effects of SMTP-7 at high concentrations in vitro. To 240 μL of citrated blood, 30 μL of SMTP-7 (final concentration of 200 to 400 μM) and 30 μL of 133 mM CaCl2 were added. The thromboelastogram was recorded immediately after the recalcification.
Determination of Plasmin–α 2-Antiplasmin Complex and Tissue-Type Plasminogen Activator–Plasminogen Activator Inhibitor-1 Complex
Plasmin–α2-antiplasmin complex (Pm–AP), a marker of physiologic plasmin generation, was determined as described. 8 Male ICR mice anesthetized with isoflurane received an intravenous infusion (30 minutes) of SMTP-7 (10, 30, and 100 mg/kg), t-PA (0.1, 1, and 10 mg/kg), or saline (n=5 each). After 60 minutes of the initiation of the infusion, blood was collected from the inferior vena cava in 13 mM sodium citrate. Plasma was rapidly prepared and subjected to zymography 8 on gels containing 5 mg/mL casein.
An in vitro assay for Pm–AP was carried out to examine the effects of SMTP-7 on Pm–AP formation in vivo. Mouse citrated plasma (50%, v/v in saline) supplemented with human α2-antiplasmin (250 nM) was incubated with plasmin (62.5 nM) in the absence or presence of SMTP-7 (30, 100, 300, and 1,000 μM). After incubation at 37 °C for 30 minutes, the mixture was subjected to zymography on casein gels to determine Pm–AP.
We assessed the effect of SMTP-7 on the formation of t-PA–plasminogen activator inhibitor-1 complex (t-PA–PAI-1) in vitro using a zymographic method similar to that described above. Mouse citrated plasma (35%, v/v in saline) supplemented with PAI-1 (50 nM) was incubated with t-PA (31 nM) in the absence or presence of SMTP-7 (10, 100, and 1,000 μM). After incubation at 37 °C for 30 minutes, the mixture was subjected to zymography on gels containing 2 mg/mL casein and 22 nM plasminogen to determine the t-PA–PAI-1 level.
Data Analysis
Results are expressed as the mean±s.d. Unpaired Student's t-test was employed to determine the statistical differences between two groups. Variables before and after SMTP-7 treatments were compared using paired Student's t-test. The multiple comparison methods are described in the figure legends. P-values<0.05 were considered significant. The results of the post-hoc statistical power calculation are shown in Supplementary Table 2.
RESULTS
Photochemical Thrombotic Middle Cerebral Artery Occlusion Model in Monkeys
To test SMTP-7 for its therapeutic potential in primates, we employed a photochemical-induced thrombotic MCA occlusion model in cynomolgus monkeys. The MCA blood flow in this model was stopped by developed clots after an average of 6.9 minutes (Figures 1A and 1B), and this initial occlusion was followed by repeated recanalization and reocclusion (cyclic flow reductions; Figure 1A). Sixty minutes after the photoirradiation, either saline or SMTP-7 (10 mg/kg, based on experiments with rodent models)10, 11, 12, 13, 14 was infused for 30 minutes, which resulted in plasma SMTP-7 concentrations of 35 to 68 μM during the infusion period (Supplementary Figure 1). The postinfusion recanalization rate during the 30-minute monitoring period was 32.5 times higher in the SMTP-7 group compared with the saline-treated control monkeys (43.3±40.5% versus 1.3±2.9%, P=0.043), demonstrating the clot clearance potential of SMTP-7 (Figure 1C).
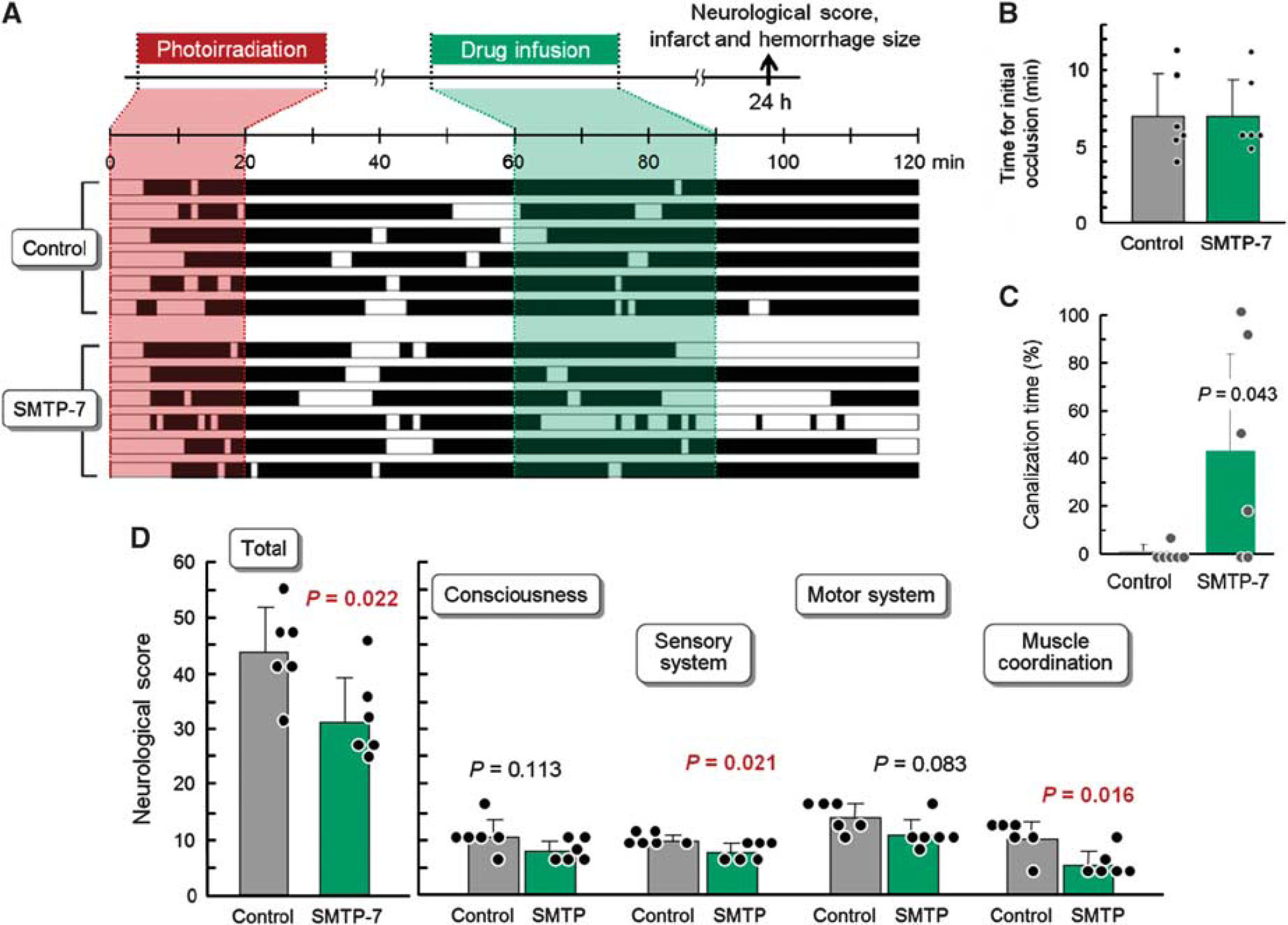
Effects of SMTP-7 (Stachybotrys microspora triprenyl phenol-7) on blood flow and neurologic deficits in a monkey thrombotic middle cerebral artery (MCA) occlusion model. (
The neurologic deficits in the SMTP-7-treated monkeys, determined 24 hours after photothrombosis, were milder than those in the saline-treated control animals. In the SMTP-7 group, the total of the neurologic deficit scores, comprising deficits in consciousness, the sensory system, the motor system, and skeletal muscle coordination, was significantly lower than that in the saline group (31.2±7.9 points versus 43.7±8.1 points, P=0.022; Figure 1D). Among the four subcategories, muscle coordination was markedly improved by the SMTP-7 treatment (10.0±3.1 points versus 5.3±2.4 points in the saline group, P=0.016; Figure 1D).
The cerebral infarct was distributed mainly over the basal ganglia and cerebral cortex in the saline-treated monkeys (Figure 2A), resulting in 2675±950 mm3 of the total infarct volume (corrected for edema). In the SMTP-7-treated monkeys, the total infarct size was reduced to 54% of the size in the saline-treated control (1449±570 mm3, P=0.033; Figure 2B). In particular, the infarct size in the basal ganglia in the SMTP-7 group was significantly smaller than that in the saline-treated monkeys (65%, P=0.027; Figure 2B). As observed in human ischemic stroke, the photochemical thrombotic stroke in monkeys was associated with hemorrhagic transformation (Figure 2C). The treatment with SMTP-7 resulted in a significant reduction in hemorrhage size (49% compared with the saline group, P=0.013; Figure 2D).
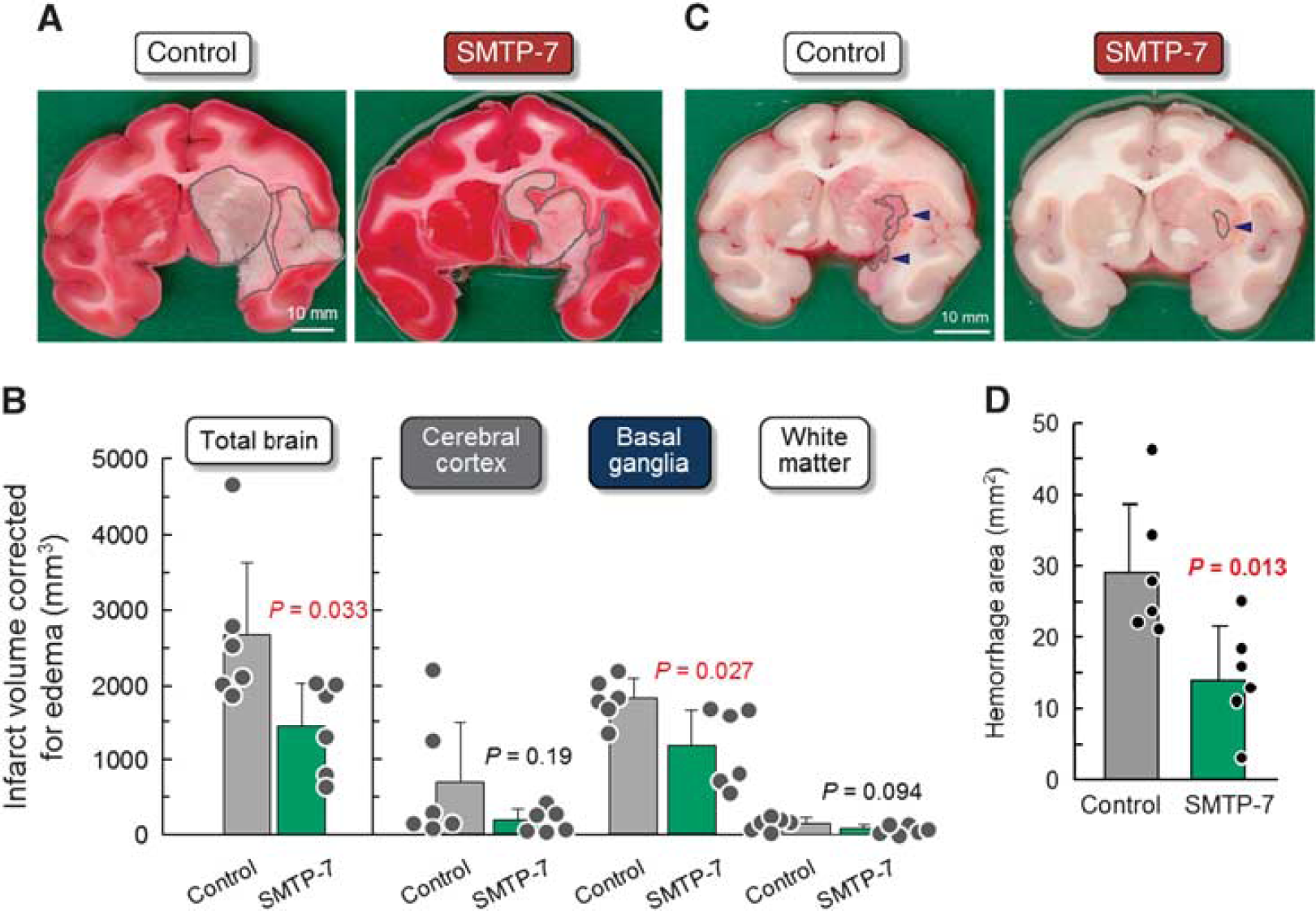
Effects of SMTP-7 (Stachybotrys microspora triprenyl phenol-7) on cerebral infarction and hemorrhage in a monkey thrombotic middle cerebral artery (MCA) occlusion model. (
Physiologic and Hemostatic Parameters in Normal Monkeys
To investigate the physiologic and hemostatic systems that may affect ischemic stroke progression and recuperation, we measured several parameters in normal monkeys before and after SMTP-7 treatment. The infusion of SMTP-7 at 10 mg/kg did not affect heart rate, blood pressure, body temperature, blood gas, blood pH, hematocrit, or blood glucose levels (Supplementary Figure 2). Hemostatic parameters in normal monkeys, determined at both pre- and post-SMTP-7 infusion (10 mg/kg), did not change significantly about the following variables: activated partial thromboplastin time, plasma levels of fibrinogen, plasminogen, α2-antiplasmin, and D-dimer, as well as platelet aggregation and thromboelastographic blood coagulation parameters (Supplementary Figure 3).
Transient and Permanent Focal Ischemia Models in Rats
We studied the impact of SMTP-7 on ischemia/reperfusion-induced brain damage in a rat transient focal ischemia model, in which a silicone-coated suture was inserted into the MCA and removed after 90 minutes to cause ischemia/reperfusion in the MCA territory. The infusion of SMTP-7 at 10 mg/kg significantly ameliorated neurologic deficits in the affected animals (21% reduction in the neurologic deficit score, P<0.05; Figure 3A). Cerebral infarcts in this model were found predominantly in the cerebral cortex (Figure 3B). SMTP-7 significantly reduced the infarct size in the cerebral cortex (by 38%, P<0.05), resulting in a reduction in total infarct size (by 34%, P<0.05; Figure 3B). These results imply neuroprotective properties of SMTP-7 as part of its efficacy under mechanical ischemia/reperfusion conditions, whereas thrombolysis may minimally contribute.
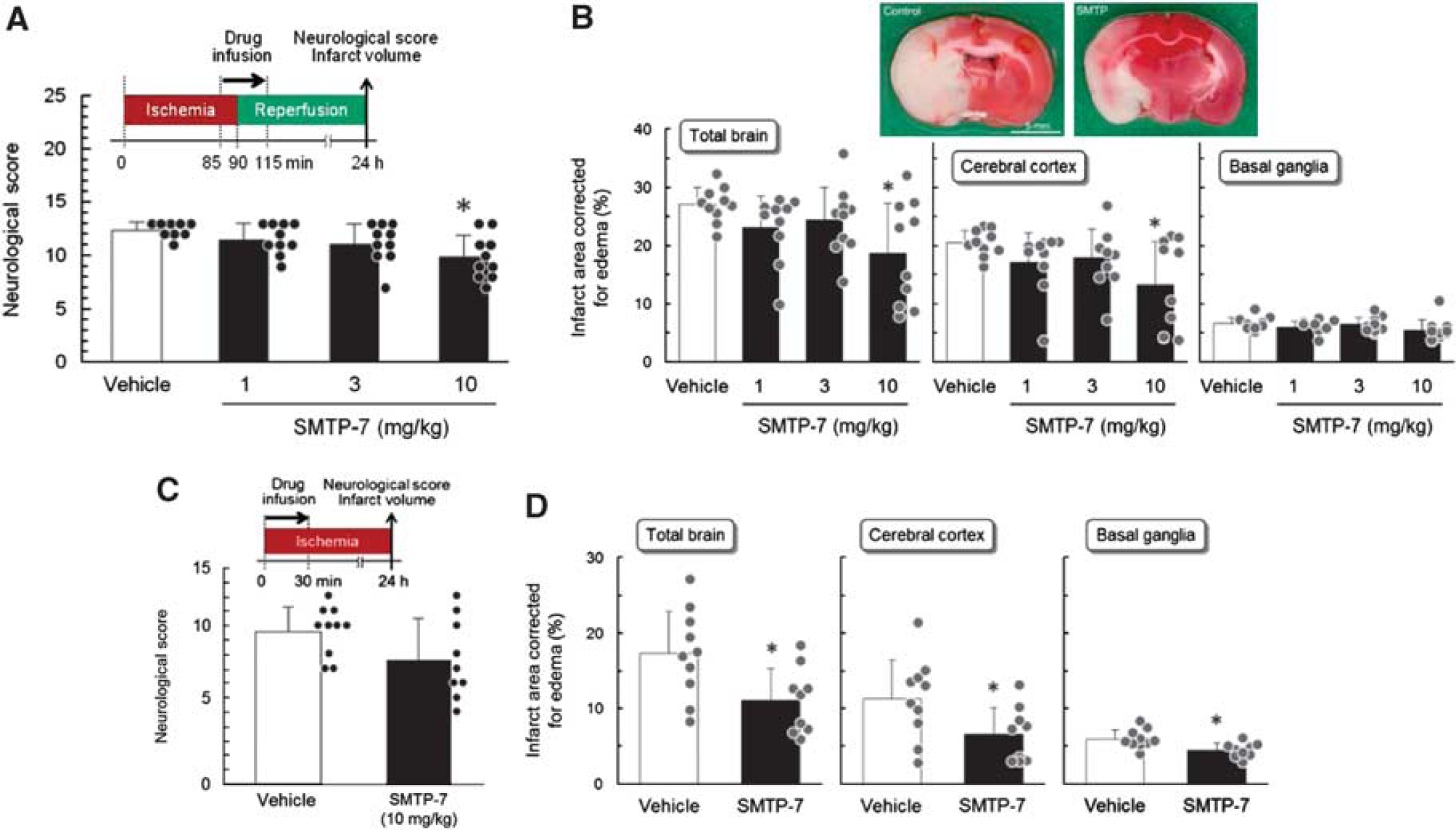
Effects of SMTP-7 (Stachybotrys microspora triprenyl phenol-7) on neurologic deficits and cerebral infarct in transient and permanent focal cerebral ischemia models in rats. (
To further investigate the neuroprotective properties of SMTP-7, we tested the agent in a rat permanent focal ischemia model. Although the infusion of SMTP-7 at 10 mg/kg significantly ameliorated cerebral infarction (Figure 3D), the difference in the neurologic deficit scores was not significant (Figure 3C). Thus, the effect of SMTP-7 on neuroprotection is marginal in the permanent focal ischemia model, in which the MCA blood flow is completely blocked.
Thrombosis, Bleeding, and Plasmin Formation
To characterize the effect of SMTP-7 on the hemostatic system, we performed additional experiments using rodent models and in vitro assays. In a rat carotid artery thrombosis model, in which ferric chloride-induced vascular damage promoted thrombus formation, SMTP-7 (10 mg/kg) significantly prolonged the time required for initial occlusion (Figure 4A). SMTP-7 did not affect coagulation parameters in rodents (see below) in the same way as in monkeys (Supplementary Figure 3), suggesting that the prevention of thrombotic arterial occlusion can be attributed to the promotion of thrombolysis. The potential of SMTP-7 at 10 mg/kg was comparable to that of t-PA at 0.55 mg/kg (Figure 4A).
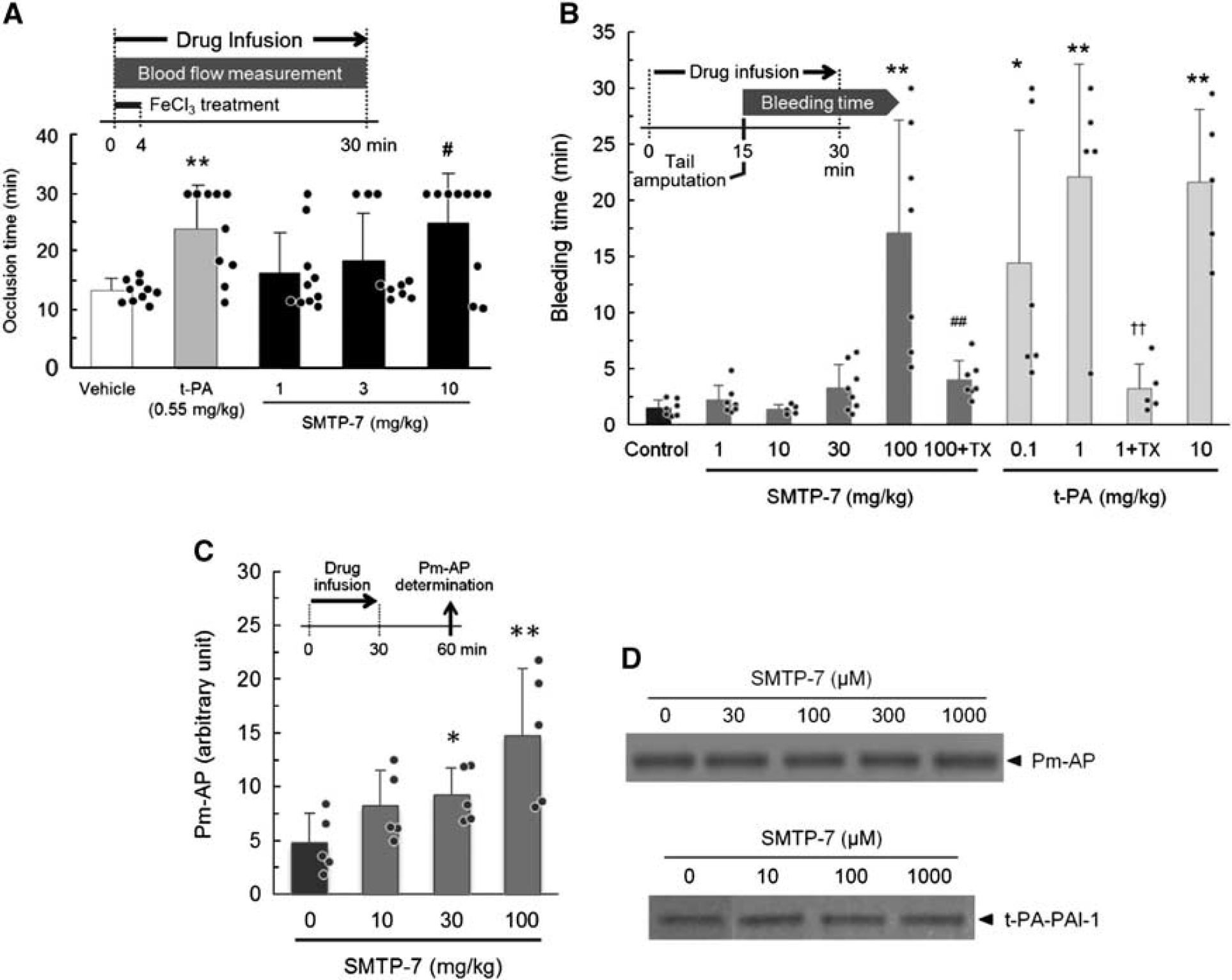
Effects of SMTP-7 (Stachybotrys microspora triprenyl phenol-7) on thrombotic carotid artery occlusion, bleeding, and plasmin formation in vivo. (
In the tail amputation bleeding assay in mice, SMTP-7 up to 30 mg/kg did not promote bleeding prolongation (Figure 4B). These results were in a striking contrast to the effect of t-PA, which prolonged bleeding at 0.1 to 10 mg/kg, a range comparable to its pharmacological doses (Figure 4B). However, a very high dose of SMTP-7 (100 mg/kg, 10 times its pharmacological dose) significantly prolonged the bleeding times. This effect was canceled by tranexamic acid, an inhibitor of fibrinolysis (Figure 4B). Similarly, bleeding in t-PA-treated mice was also abolished by tranexamic acid (Figure 4B).
To test the possibility that SMTP-7 infusion increases plasmin formation in vivo, we assessed plasma levels of Pm–AP after SMTP-7 administration, as plasmin in vivo rapidly converts to the Pm–AP complex. The Pm–AP in normal mice treated with SMTP-7 increased dose dependently, with significant changes at 100 mg/kg (Figure 4C). As SMTP-7 (30 to 1,000 μM) did not affect Pm–AP formation from preexisting plasmin and α2-antiplasmin in vitro (Figure 4D), this increase in the plasma Pm–AP level in mice represented an increase in plasminogen activation to plasmin in vivo. In addition, SMTP-7 at these doses had no effect on t-PA–PAI-1 formation in vitro (Figure 4D), suggesting that the increase in Pm–AP was irrelevant to the effects of SMTP-7 on t-PA inactivation by PAI-1.
We assessed the effects of SMTP-7 on blood coagulation by thromboelastography using freshly drawn human whole blood. SMTP-7 (200 to 400 μM) did not affect any blood coagulation parameters, including reaction time (time for the initiation of clotting) and maximum amplitude (an index of the clot firmness; Supplementary Figure 4).
DISCUSSION
In this study, we demonstrated that SMTP-7 is effective in treating thrombotic stroke in a monkey model. In the photochemical-induced thrombotic MCA occlusion model, cyclic flow reductions, representing arterial occlusion and recanalization, occur spontaneously after the initial thrombotic arterial occlusion elicited by photoirradiation-induced endothelial injury.18, 19 Although the pattern of the cyclic flow reductions varied widely among the animals with respect to frequency and duration (Figure 1), the administration of SMTP-7 significantly increased the postinfusion recanalization rate (32.5-fold on average). This result is consistent with the previous findings that SMTP-7 promotes clot clearance in rodent models.8, 12 Accordingly, SMTP-7 improves brain infarction and neurologic deficits associated with ischemic stroke in the monkey model (Figures 1 and 2). These outcomes are compatible with the results with t-PA in monkeys, 18 demonstrating the impact of thrombolysis on the treatment of thrombotic stroke. In addition, SMTP-7 reduces hemorrhagic transformation in this model (by 51%, P=0.013; Figure 2). Hemorrhagic transformation in ischemic stroke is assumed to be caused by inflammation of the affected brain region.28, 29 Our previous results in the rodent model demonstrated that SMTP-7 suppresses inflammation in the ischemic brain.11, 12 In addition, a neuroprotective potential of SMTP-7 is suggested in a rat transient focal ischemia model under mechanical ischemia/reperfusion conditions in which thrombolysis may participate minimally (Figure 3A).
Regarding the physiologic and hemostatic parameters, no significant changes followed the SMTP-7 treatment (10 mg/kg) in heart rate, blood pressure, body temperature, blood gas, blood pH, hematocrit, or blood glucose, or in the activated partial thromboplastin time, fibrinogen, platelet aggregation, or thromboelastographic coagulation parameters (Supplementary Figure 3), suggesting no involvement of hemodynamic change and coagulation inhibition in the efficacy of SMTP-7 in the monkey stroke model. SMTP-7 (10 mg/kg) also caused no significant change in the fibrinolytic parameters plasminogen, α2-antiplasmin, and D-dimer in the normal monkeys, whereas the D-dimer level was twice the pretreatment level (Supplementary Figure 3). Thus, under normal conditions (without thrombosis), SMTP-7 may minimally mobilize the fibrinolytic system in monkeys. This observation is consistent with the result of the tail amputation bleeding assay in normal mice, in which 10 mg/kg SMTP-7 did not increase bleeding (Figure 4B), and this dose of SMTP-7 only slightly elevated the Pm–AP level in normal mice (Figure 4C). However, conversely, higher doses of SMTP-7 (100 mg/kg) significantly enhanced the bleeding and plasma Pm–AP levels (Figures 4B and 4C). Taken together, the dose of SMTP-7 at ∼10 mg/kg—which is pharmacologically active in various thrombosis and stroke models (Figures 1, 2, and 4A, as well as refs8, 10, 11, 12)—may minimally affect the fibrinolytic system in normal animals. However, t-PA suppressed thrombosis (Figure 4A) at 0.55 mg/kg, whereas a dose of t-PA as low as 0.1 mg/kg significantly augmented bleeding (Figure 4B). In this respect, SMTP-7 can be discriminated from t-PA. The difference can be explained by the fact that unlike t-PA, which directly activates plasminogen, SMTP-7 is a plasminogen modulator that alters the plasminogen conformation to be susceptible to proteolytic activation by endogenous plasminogen activators, and it acts through a physiologic on-demand system coping with thrombotic events.
In conclusion, SMTP-7 was effective in treating thrombotic stroke in a monkey model by its amelioration of neurologic deficits, infarct size, and hemorrhagic transformation. The results in rodent models and in vitro experiments suggest that the excellent efficacy of SMTP-7 is attributable to its regulated profibrinolytic action and possibly to its neuroprotective properties. However, caution is required with respect to the following points. It should be noted that a difference in body temperature affects the outcome of stroke, 30 although in the present study the animals' rectal temperature was kept at a normal range (37 to 38.5 °C) in the monkey and rat experiments. We should be careful about interpreting the present data as suggesting that SMTP-7 reduces hemorrhagic risk. Further studies, especially comparisons of SMTP-7 with t-PA concerning their effects on recanalization and hemorrhagic transformation, are required. In addition, as not all forms of hemorrhagic transformation have clinical impact, whether SMTP-7 can truly reduce the risk of symptomatic intracerebral hemorrhage awaits further study. It is also important to analyze the long-term outcomes of neurologic deficits.
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
