Abstract
Intravenous immunoglobulin (IVIg) is currently evaluated in clinical trials for the treatment of various disorders of the central nervous system. To assess its capacity to reach central therapeutic targets, the brain bioavailability of IVIg must be determined. We thus quantified the passage of IVIg through the blood–brain barrier (BBB) of C57Bl/6 mice using complementary quantitative and qualitative methodologies. As determined by enzyme-linked immunosorbent assay, a small proportion of systemically injected IVIg was detected in the brain of mice (0.009±0.001% of injected dose in the cortex) whereas immunostaining revealed localization mainly within microvessels and less frequently in neurons. Pharmacokinetic analyses evidenced a low elimination rate constant (0.0053 per hour) in the cortex, consistent with accumulation within cerebral tissue.
INTRODUCTION
The poor brain bioavailability of immunoglobulins has limited their use for treating diseases of the central nervous system (CNS), either for passive or active immunotherapy. To enter the brain, Ig must cross the blood–brain barrier (BBB), which regulates the exchange of compounds between blood and brain. Endothelial cells sheathing brain capillaries differ from those in the rest of the body by the absence of fenestrations, limited pinocytic vesicular transport, prevalent adherens, and tight junctions.
1
The BBB normally permits water, ions, and small lipophilic substances to diffuse freely along their concentration gradient.
1
Nutrients such as glucose and amino acids enter the brain via transporters whereas receptor-mediated transcytosis allows the uptake of larger molecules including insulin, leptin, and transferrin.
1
Alternatively, epithelial cells of the choroid plexuses and of the arachnoid membrane indirectly allows the transport of plasma-borne molecules to the cerebrospinal fluid (CSF), which bathes the cerebral interior and exterior surfaces of the brain parenchyma.
2
IgG concentration in the CSF is routinely measured clinically with a normal steady-state CSF/serum ratio of 0.0027.
3
Although the protein movement across the blood–CSF barrier is limited, the tight junctions at the choroid plexus are considerably more permeable than those of capillaries of the BBB.3, 4 However, the presence of IgG in the CSF remains thus a poor assessment of its brain bioavailability,
4
because exchange of large molecules between brain cells and the CSF is limited.
4
For technical reasons, however, the passage of drugs into the brain is scarcely studied in humans. Because clinical efficacy of any drug mostly relies on interaction with its target, the need in determining brain bioavailability of therapeutic Ig is inescapable. Yet, few
Intravenous Ig (IVIg) is a blood-derived therapeutic preparation composed of over 98% polyclonal human IgG (hIgG). This heterogeneous product is used for the treatment of immune deficiencies, acute infections, autoimmune diseases and for neurologic disorders.5, 6 Interestingly, IVIg treatment has been associated with a lower risk of developing Alzheimer's disease (AD) 7 and cognitive improvement has been reported for AD patients treated with IVIg. 8 Moreover, phase 2 and 3 clinical trials are undergoing in AD (http://www.ClinicalTrials.gov). However, treating a 70-kg individual with IVIg (0.4 g/kg every 2 weeks) 8 would require the use of plasma fractionated from over 150 blood donations every year. 6 As more than 24 million people are affected with AD worldwide, 9 the utilization of IVIg for such a prevalent disease would most certainly lead to a shortage. Although indirect evidence of IVIg reaching the parenchyma exists,10, 11, 12 it is still unclear if the penetration of IVIg is a prerequisite for its clinical effects, because no quantification of the passage into the CNS has been performed. Therefore, to develop alternatives, it is essential to first determine the extent by which IVIg can access cerebral tissues. In this study, we investigated the concentrations of IVIg in the blood, peripheral tissues, and brain structures after systemic administration in mouse models. Our data provide a quantitative and qualitative assessment of the brain bioavailability and pharmacokinetic parameters, suggest that therapeutically relevant concentrations can be reached in cerebral tissues in the absence of BBB disruption, and support a role for brain IgG in the treatment of CNS diseases.
MATERIALS AND METHODS
Animals and Intravenous Immunoglobulin Treatment
For pharmacokinetic experiments, C57Bl/6 mice (The Jackson Laboratory, Bar Harbor, ME, USA) were used. Triple-transgenic mice (3xTg-AD) and age- and sex-matching nontransgenic (NonTg) littermates (mixed C57Bl/6 × 129S genetic background) produced in our animal research facility as well as B6129SF1/J (C57Bl/6 × 129S, F1) mice (also designated NonTg) purchased from The Jackson Laboratory were used. The 3xTg-AD mouse harbors three mutant genes coding for the following: beta-amyloid precursor protein (APPswe), presenilin-1 (PS1M146V), and tau (tauP301L), 13 reproduces the main pathologic features of AD patients and is used to model AD neuropathology and behavioral defects. 14 The animal research committee of Université Laval approved all procedures according to the guidelines from the Canadian council on animal care in science.
The animals were subjected to three IVIg regimens. First, a subacute treatment was used for basic pharmacokinetic analyses. Mice (
PK values were measured after a single intraperitoneal injection of 25 mg IVIg (mean±s.e.m. of
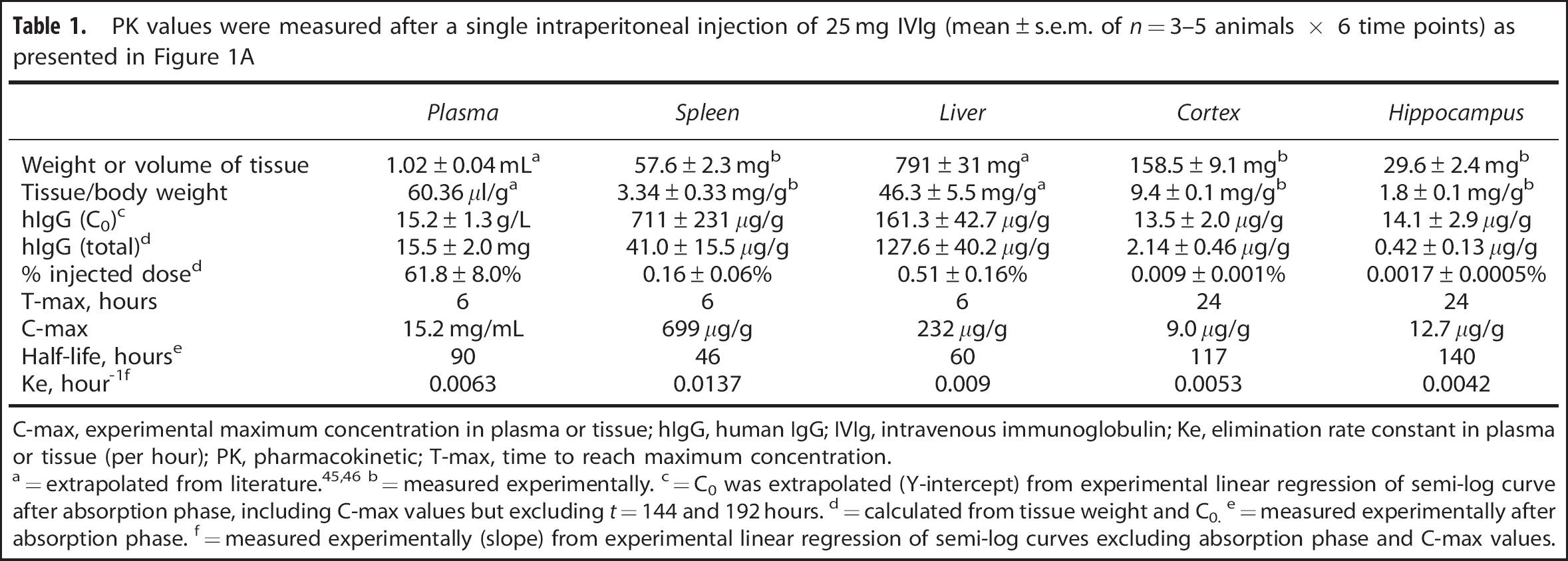
C-max, experimental maximum concentration in plasma or tissue; hIgG, human IgG; IVIg, intravenous immunoglobulin; Ke, elimination rate constant in plasma or tissue (per hour); PK, pharmacokinetic; T-max, time to reach maximum concentration.
=measured experimentally.
=C0 was extrapolated (Y-intercept) from experimental linear regression of semi-log curve after absorption phase, including C-max values but excluding
=calculated from tissue weight and C0.
=measured experimentally after absorption phase.
=measured experimentally (slope) from experimental linear regression of semi-log curves excluding absorption phase and C-max values.
In Situ Cerebral Perfusion
The brain transport coefficient (
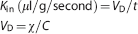
where
Tissue Preparation for Postmortem Analyses
For all analyses, mice were killed by intracardiac perfusion under deep anesthesia, with the exception of ISCP as described above. After transcardiac administration of 50 mL 0.1 mol/L phosphate-buffered saline (PBS) buffer (Bioshop Canada, Burlington, ON, Canada) containing protease and phosphatase inhibitors (SigmaFAST Protease Inhibitor Tablets, Sigma-Aldrich, St Louis, MO, USA with sodium fluoride and sodium pyrophosphate), the spleen, liver, and brain were collected (about 50 mg of tissue each). For IgG quantification, the hippocampus and the cortex were dissected, snap frozen on dry ice, and stored at −80°C until used. The cortex, hippocampus, spleen, and liver were homogenized in lysis buffer and processed as described for ISCP brain homogenates.
For brain cell dissociation, mice (
For immunofluorescence and immunohistochemistry experiments, mice (
Enzyme-Linked Immunosorbent Assay and Immunoblot
The concentrations of human and mouse IgG (mIgG) were determined by species-specific enzyme-linked immunosorbent assay using IgG Fc-specific antibodies for capture and the corresponding HRP-conjugated antibodies for detection (Jackson ImmunoResearch Laboratories). For immunoblots, proteins from 5 × 106 dissociated brain cells were heated at 95°C for 5 minutes in Laemmli loading buffer, separated by SDS-Page on a 10% polyacrylamide gel, transferred on a polyvinylidene difluoride membrane (Immobilon-P, EMD Millipore, Billerica, MA, USA) and blocked in 5% non-fat dry milk, 0.5% bovine serum albumin, 0.1% tween 20 in PBS buffer as previously described. 12 Proteins were detected using the following primary antibodies: anti-NeuN, anti-PDGFR (receptor for platelet-derived growth factor), anti-GFAP (Sigma-Aldrich), anti-oligodendrocyte specific antibody (Abcam, Toronto, ON, Canada), anti-collagen IV (Chemicon), and anti-actin (ABM, Richmond, BC, Canada) followed by HRP-labeled secondary antibodies and chemiluminescence reagents (Lumiglo Reserve, KPL, Gaithersburg, MD, USA). Immunoblots were analyzed with a KODAK Imaging Station 4000MM Digital Imaging System (Molecular Imaging Software version 4.0.5f7, Carestream Health, Rochester, NY, USA).
Pharmacokinetic and Statistical Analyses
Pharmacokinetic analyses were performed in accordance with the standard procedure
17
and using Prism 5.0d software (GraphPad Software, La Jolla, CA, USA). Group means are presented with standard error of the mean (s.e.m.). A Bartlett test was executed on all data to test for equal variances. Statistical differences were determined using the appropriate one-way analysis of variance or Kruskal–Wallis nonparametric tests and
RESULTS
Pharmacokinetic Profile of Intravenous Immunoglobulin in Mouse Models
The use of IVIg in mouse models of human diseases has been widely reported.10, 11, 12 However, scarce pharmacokinetic data are available. In humans, the half-life of IVIg in plasma is evaluated to be 35 days 6 but the quantification of tissue distribution, specifically in the brain, remains technically challenging. We first undertook to evaluate the time-related distribution of human IVIg in plasma and tissues from young C57Bl/6 mice after a single i.p. injection of 25 mg IVIg (Figure 1A, Table 1). In the plasma, a half-life of 90 hours was determined experimentally with a T-max of 6 hours. We also investigated the time course of hIgG distribution in mouse tissues after removal of contaminating blood by intracardiac perfusion. This procedure efficiently removes blood from cerebral tissue but is generally less complete with highly irrigated organs, such as the liver or spleen. Similar to plasma, the IVIg reached a maximum concentration at around 6 hours in the liver and the spleen, suggesting a high vascularization and fast distribution equilibrium (Figure 1A, Table 1). However, the blood-to-brain IVIg transport was delayed in comparison with peripheral tissues (Figure 1A, Table 1) with a maximum concentration (C-max) being reached 24 hours after IVIg administration. Accordingly, a low brain elimination rate constant (Ke) was measured from experimental data (Table 1). From these results, we also calculated the percentage of the injected dose retained in each type of tissue under analysis. In the brain, we evaluated that 0.009±0.001% and 0.0017±0.0005% of systemically administered IVIg were distributed to the cortex and hippocampus of injected animals (Table 1). These results provide a quantitative assessment of the amount of hIgG in the brain after a systemic injection of IVIg and suggest a limited but significant accumulation of IVIg in the brain.
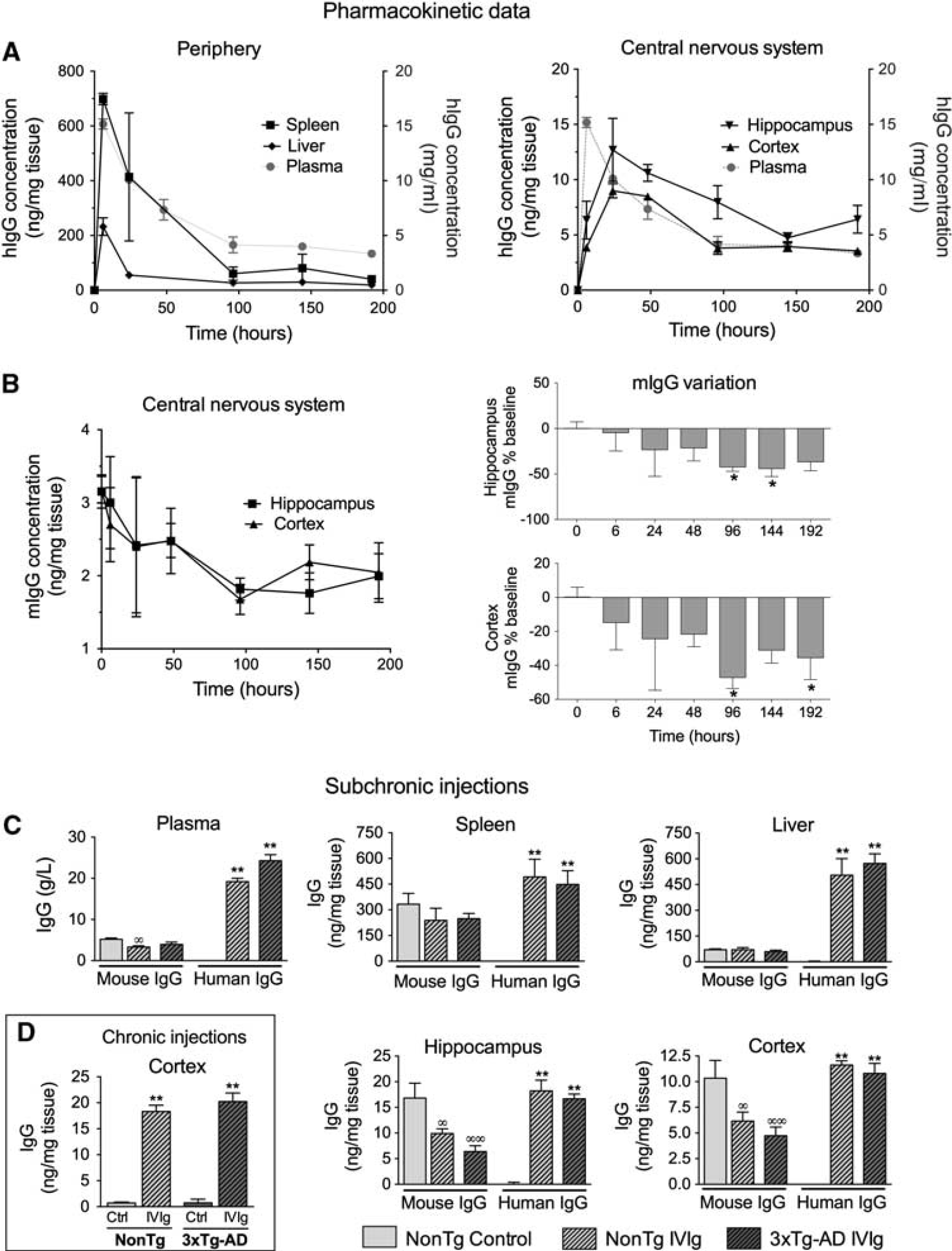
Biodistribution of human and mouse immunoglobulin G (IgG) in the plasma and tissue homogenates. (
Apparent Competition Between Human Immunoglobulin G and Endogenous Mouse Immunoglobulin G to Enter the Brain
Interestingly, subsequent to the injection of IVIg, mIgG was reduced by 47% in the cortex and 42% in the hippocampus after 96 hours, suggesting that IVIg competes with mIgG to reach the brain (Figure 1B). To characterize the accumulation of IVIg in the brain and peripheral tissues after repeated injections in 3xTg-AD mice, hIgG concentration was evaluated and compared with endogenous mIgG after a subchronic treatment. The expression of AD-related transgenes in the 3xTg-AD mouse did not influence the entry of IVIg in the brain and peripheral tissues. The concentration of hIgG in the brain rose to 11.6±0.4 and 10.8±1.0
Brain Uptake of Intravenous Immunoglobulin is a Saturable Process
Furthermore, we quantified the IVIg brain uptake using an ISCP method (Figure 2A).
15
This technique was performed by injecting IVIg directly into the carotid artery, allowing almost 100% of the substance under study to quickly reach the cerebrovasculature. As expected, we measured a dose-dependent increase of IVIg concentration in the brain (Figure 2B). However, increasing the concentration was associated with a decreased percentage of hIgG reaching the brain (Figure 2C), evidencing a saturation of IVIg passage through the BBB. The
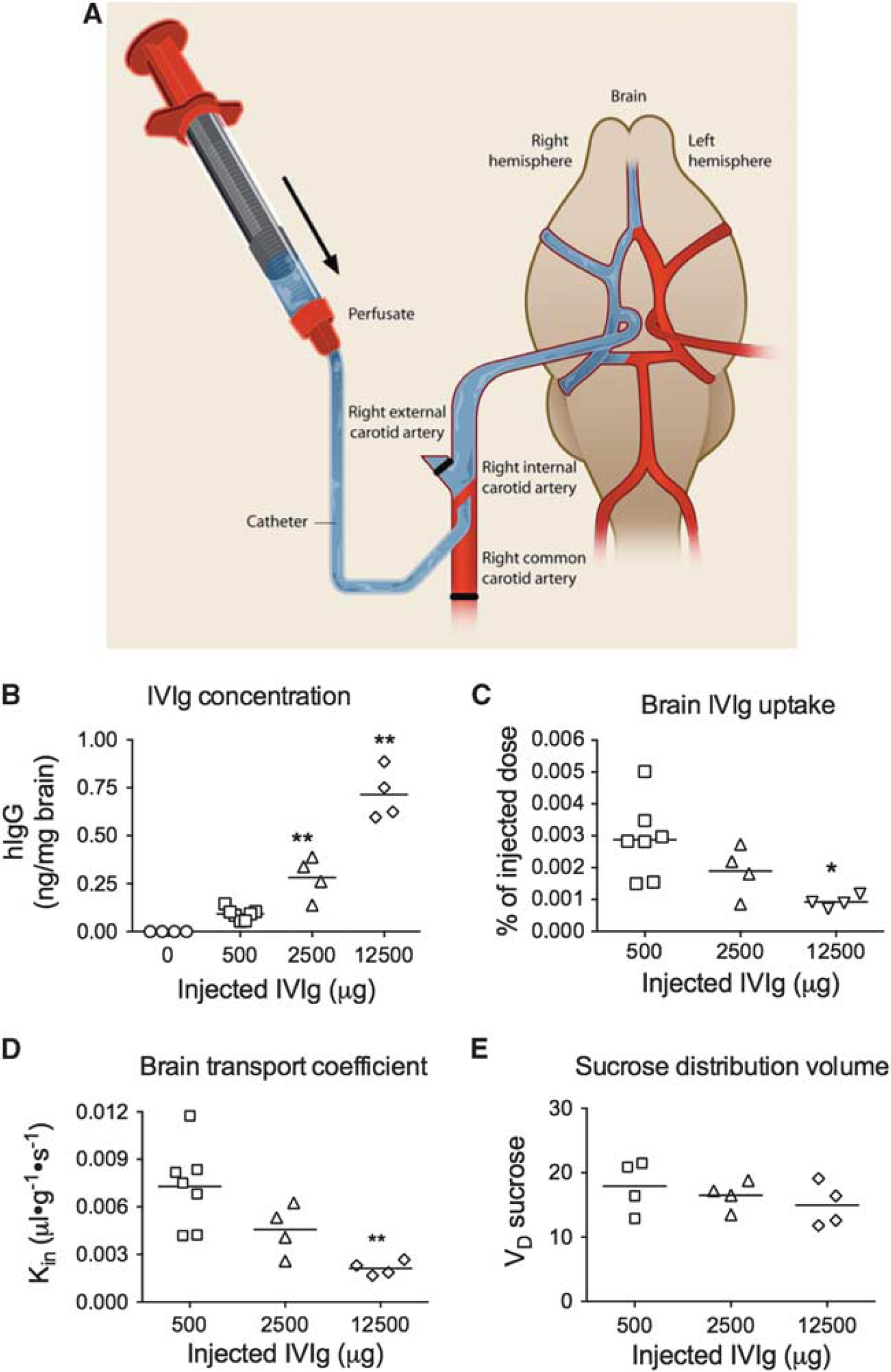
Quantification of intravenous immunoglobulin (IVIg) brain uptake: saturation of blood–brain barrier (BBB) transport. Mice were deeply anesthetized and (
Localization of Intravenous Immunoglobulin in Brain Microvessels and Cells after Systemic Administration
Immunohistochemical staining was used to localize IVIg in the CNS. We observed significant immunoreactivity for hIgG throughout the brain parenchyma (Figure 3A) after a subchronic treatment compared with controls, despite the fact that it was largely excluded from the white matter in 3xTg-AD and NonTg animals. Interestingly, an important proportion of hIgG-immunopositive cells were noticeable in the cortex of treated mice. To confirm hIgG localization in the brain, confocal microscopy was also performed. Intravenous immunoglobulin staining (Figure 3B) was strongly colocalized with the endothelial cell marker CD31. Remarkably, hIgG immunostaining also colocalized with NeuN, a neuronal marker. Results obtained in 4- to 9-month-old 3xTg-AD and age-matched NonTg mice were similar, despite the fact that amyloid plaques were clearly detectable at that age (Figure 3C). 13 Taken together, our findings indicate that IVIg crosses the BBB reaching the brain parenchyma, and hIgG-immunopositive endothelial cells and neurons were identified in 3xTg-AD and NonTg mice. As non-specific immunostaining was detected in the vehicle animals, mostly in the circumventricular organs, these regions were excluded from all subsequent analyses and photomicrographs (Figure 3A).
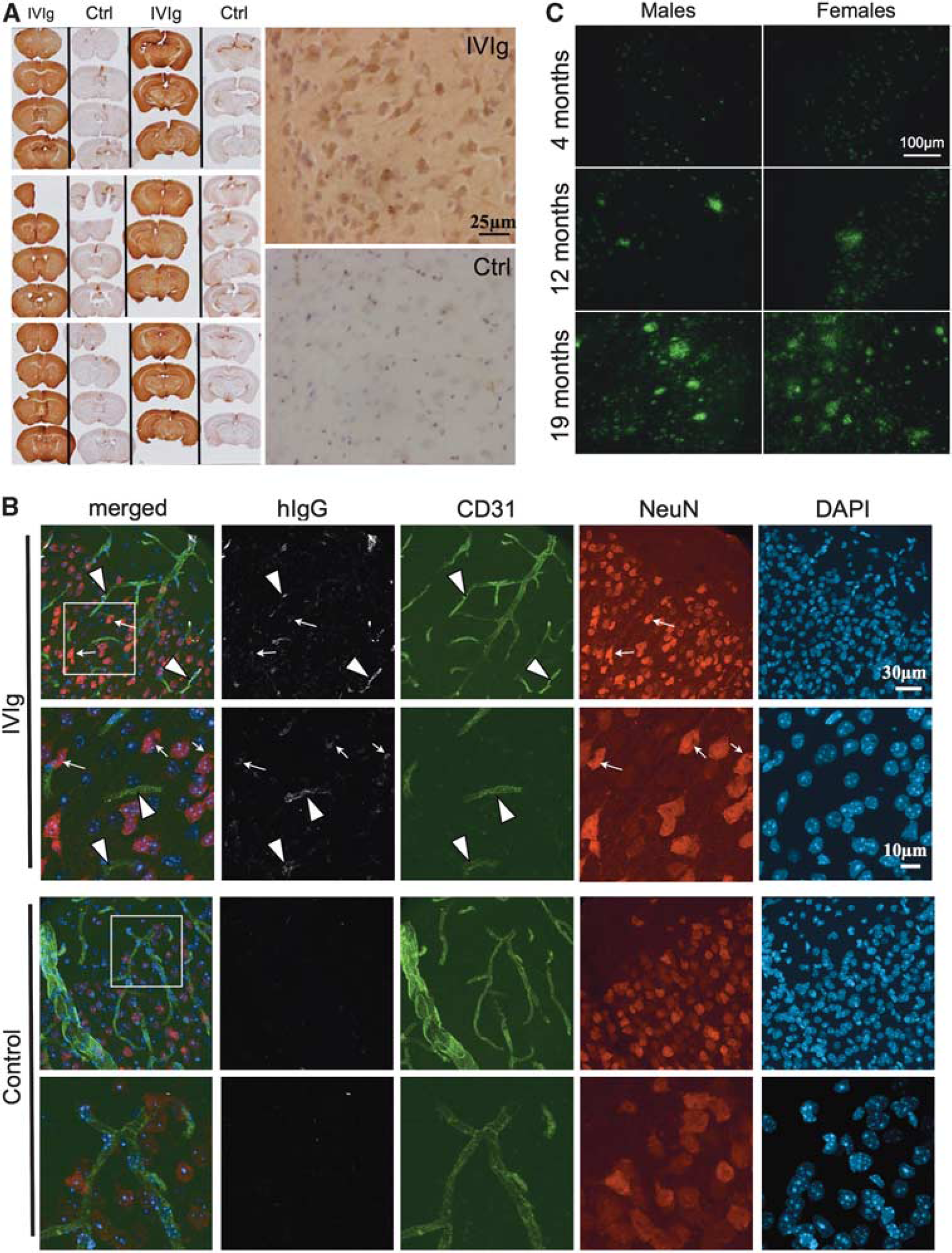
Intravenous immunoglobulin (IVIg) was detected in the capillary endothelial cells and neurons after systemic administration in mice. (
To confirm the immunolabeling results, we next dissociated brain cells from cerebral tissue of IVIg-treated mice and verified the isolation of astrocytes, oligodendrocytes, endothelial cells, pericytes, and neurons by immunoblot, thus validating the efficiency of the dissociation process (Figure 4A). To quantify the number of IVIg molecules per cell, we prepared cell lysates from 1 × 107 cells and performed a hIgG-specific enzyme-linked immunosorbent assay. In these conditions, 5842±2021 and 4236±803 molecules/cell were detected in NonTg and 3xTg-AD animals after a subchronic treatment (Figure 4B). Again, no statistical differences were observed between 3xTg-AD and NonTg mice. These data are supportive of a direct interaction between IVIg and brain cells.
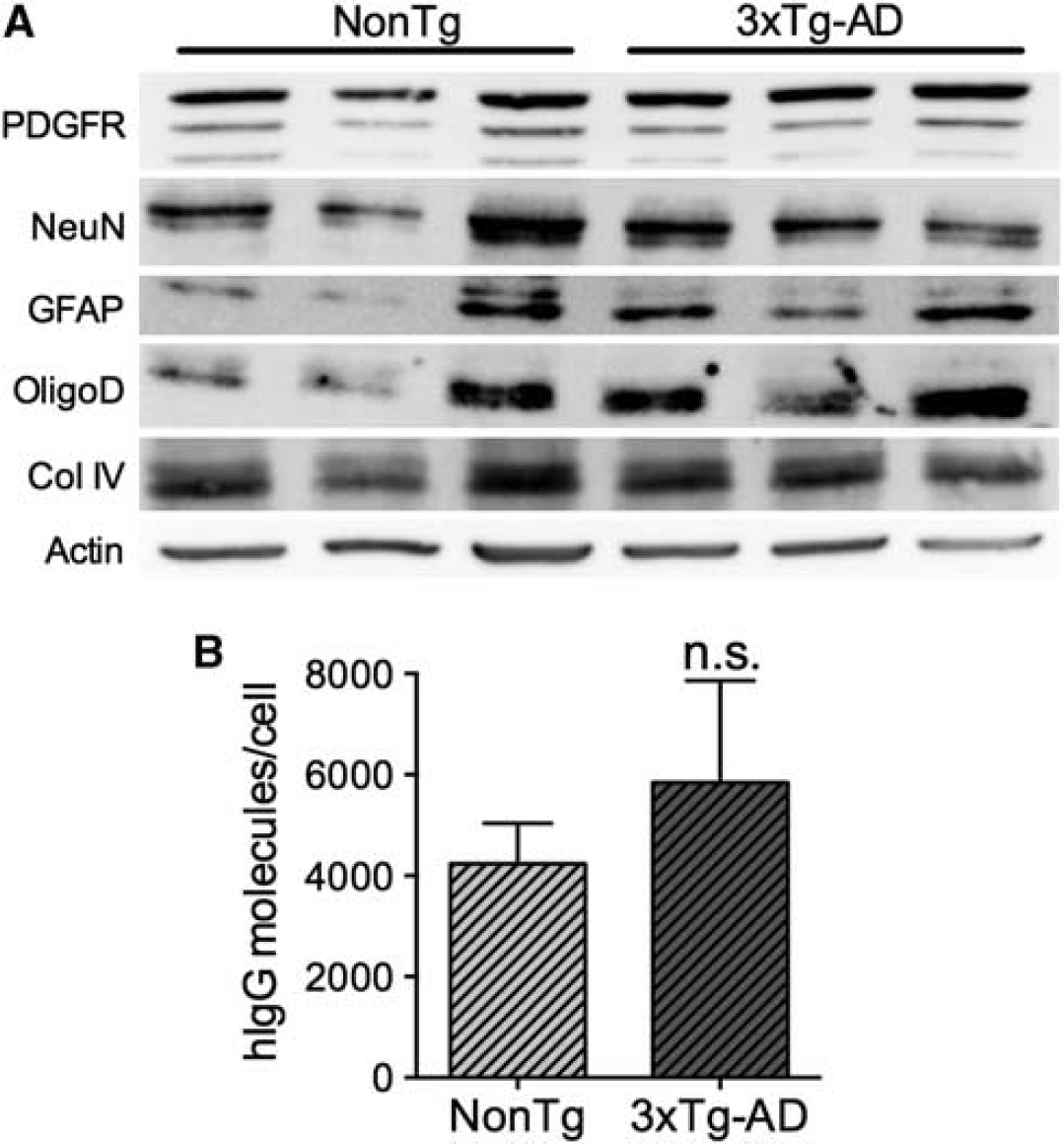
Quantification of intravenous immunoglobulin (IVIg) binding to brain cells.
DISCUSSION
To unravel the mechanisms of action and identify potential pharmacologic targets for IVIg in AD and other neurological disorders, it is crucial to determine whether IVIg is limited to the periphery or is able to cross the BBB and reach therapeutic targets directly in the brain. Published evidence of systemically administered IVIg exerting CNS effects in animal models hints toward central bioavailability.10, 11, 12 However, BBB passage of IVIg remains poorly defined. In this work, we quantified the migration of systemically administered hIgG to the brain, in the absence of BBB-enhanced permeability.
Our results are consistent with an accumulation of IVIg in the brain, albeit in a small percentage of the injected dose. Previous experiments with monoclonal antibodies injected directly into the parenchyma in rats indicate a half-life of elimination in the brain of approximately 45 minutes.
18
In contrast, our findings rather show a low elimination rate constant in the hippocampus and cortex of IVIg-treated animals and, thus, a long half-life in the brain. Our experiments also demonstrate that the absorption rate of IVIg is slower in the brain than in peripheral tissues. In a previous study in the mouse, Bard
We determined the percentage of injected IVIg migrating to the CNS to provide a quantitative assessment of the capacity of IVIg to interact with therapeutic targets within the brain. We report that 0.009±0.001% and 0.0017±0.0005% of systemically administered IVIg reached the cortex and hippocampus of animals after a single injection. Surprisingly, although IgG are commonly considered for the treatment of CNS diseases, very few quantitative data on their brain bioavailability can be found in the literature. Statements of ∼0.1% of injected IgG reaching the brain in the mouse are often made,21, 22 referring to results mentioned as data not shown in a report published in 2000
23
or measured from relative % of injected dose per gram of brain from a single time point (1 hour post injection).
24
In most of these analyses, the concentration was obtained from biotinylated or radiolabeled injected Ig. As stated by Bard
Data generated from ISCP experiments evidenced a saturable brain uptake of IVIg, strongly suggesting that transporters are involved in its transfer across the BBB. Such observations are in agreement with previous investigation using polyclonal IgG in guinea pigs, in which the brain transport of Ig isolated from guinea pigs is saturated at blood physiologic concentrations 28 but contradicts other data showing absence of saturation when small doses (up to 30 mg/kg) of monoclonal mIgG were injected. 19 In our study, higher concentrations of IVIg in the cortex and hippocampus were also associated with a reduction of mIgG, further suggesting that IgG are transported through the BBB by a saturable mechanism, in a species-independent manner. In light of the determination of a resident half-life of 45 minutes for monoclonal IgG 18 and the observed reduction of brain mIgG after systemic injections of IVIg, our data suggest a regulated influx/efflux of IgG at the BBB.
Little is known about the transporters, receptors, or adhesion molecules implicated in IVIg entry into the CNS.
Differences between mouse and human antibodies, IgG subtypes for example, have been reported and should be taken into account when interpreting the present data. Indeed, in the mouse, the isotypes are IgG1, IgG2a (or IgG2c, depending on the strain used), IgG2b, and IgG3 whereas in humans, the four subtypes are as follows: IgG1, IgG2, IgG3, and IgG4. Although subtypes differ in their interactions with Fc
Although our
To analyze the distribution of IVIg in the mouse brain, we used a subchronic treatment, to visualize IgG molecules that enter the brain during the absorption (75 minutes time point) and elimination phases (96 hour) as well as those present in the brain at C-max (24 hours), in the same experiment. Using these conditions, IVIg was detected in the cerebral microvessels and in the brain parenchyma. With dissociated brains, we further demonstrated that a small proportion of IVIg was bound to brain cells further suggesting that IVIg can interact with therapeutic targets in the CNS. Interestingly, increased hIgG concentrations were observed after administration of a chronic regimen, consistent with an accumulation of IVIg in the brain.
The brain uptake of IVIg did not differ between 3xTg-AD and NonTg mice, consistent with previous data with purified anti-A
In summary, our data demonstrate that a significant fraction of systemically administered IVIg reaches the cerebral microvessels and enters the brain through a saturable transport across the BBB. To our knowledge, this study is the first to provide quantitative measurement of IVIg brain entry and suggests that IVIg may interact with therapeutic targets within the CNS, in the absence of BBB leakage. Finally, further analysis is warranted to identify specific subpopulations of purified hIgG molecules with the ability to reach the brain and to decipher the underlying transport mechanisms.
Footnotes
The authors declare no conflict of interest.
ACKNOWLEDGEMENTS
The authors are grateful to Catherine Routhier for preliminary work on IVIg quantification in the brain, to Mélissa Ouellet for providing the sketch for ISCP description and to Dr Vincent Emond and Dr Charlotte Delay for their valuable editing of this manuscript.
The authors have not received any royalties from Grifols or any other IVIg-producing companies and declare that they have no competing financial interests.
