Abstract
Chronic cerebral hypoxia leads to a strong vascular remodeling response, though little is known about which part of the vascular tree is modified, or whether this response includes formation of new arterial vessels. In this study, we examined this process in detail, analyzing how hypoxia (8% O2 for 14 days) alters the size distribution of vessels, number of arteries/arterioles, and expression pattern of endoglin (CD105), a marker of angiogenic endothelial cells in tumors. We found that cerebral hypoxia promoted the biggest increase in the number of medium to large size vessels, and this correlated with increased numbers of alpha smooth muscle actin (α-SMA)-positive arterial vessels. Surprisingly, hypoxia induced a marked reduction in CD105 expression on brain endothelial cells (BECs) within remodeling arterial vessels, and these BECs also displayed an angiogenic switch in β1 integrins (from α6 to α5), previously described for developmental angiogenesis. In vitro, transforming growth factor (TGF)-β1 also promoted this switch of BEC β1 integrins. Together, these results show that cerebral hypoxia promotes arteriogenesis, and identify reduced CD105 expression as a novel marker of arteriogenesis. Furthermore, our data suggest a mechanistic model whereby BECs in remodeling arterial vessels downregulate CD105 expression, which alters TGF-β1 signaling, to promote a switch in β1 integrins and arteriogenic remodeling.
Introduction
Blood vessel remodeling is an integral part of many diseases including ischemic stroke (Chen et al, 1994; Hayashi et al, 2003), multiple sclerosis (Holley et al, 2010), and neoplasia (Folkman, 1995). While the manipulation of blood vessel formation could be a useful therapeutic tool to treat these and many other diseases, the lack of a complete understanding of the mechanisms regulating this process makes this currently unattainable. Three main mechanisms contribute to vascular growth: (1) vasculogenesis, defined as the formation of blood vessels from hematopoietic precursor cells, (2) angiogenesis, in which preexisting blood vessels sprout new capillaries, and (3) arteriogenesis, which describes the enlargement of preexisting vessels into mature arteries (Persson and Buschmann, 2011).
In conditions of chronic mild hypoxia, a strong angiogenic response occurs as part of the physiological adaptive response to counter the effects of low oxygen (LaManna et al, 2004). However, while it is well accepted that this results in an increased vascular density, little is known about which parts of the vascular tree are affected. In other words, is this effect mediated primarily by increasing the number of small capillaries or does it also involve an increased production of arteries or arterioles? This is an important question, because to achieve effective vascular remodeling after stroke will require more than just increasing the number of capillaries; rather this will require remodeling at all levels of the vascular tree, including the formation of new arteries/arterioles. Arteriogenesis is a normal part of vascular development, and also occurs in the adult, either by outgrowth of collateral arterioles due to occlusion of a major conductance artery, or by conversion of capillaries into arterioles (arteriolization) (Persson and Buschmann, 2011). Unlike angiogenesis, which is regulated by hypoxia and a cascade of downstream mediators (Folkman, 1995), arteriogenesis is driven by hemodynamic factors, such as stretch or flow shear stress (Persson and Buschmann, 2011).
Endoglin (CD105) is a transmembrane protein that is an auxiliary receptor for members of the transforming growth factor (TGF)-β family (Cheifetz et al, 1992). CD105 is predominantly expressed on endothelial cells and commonly used as a marker of blood vessels (Ge and Butcher, 1994). Genetic mutations of CD105 in humans result in hereditary hemorrhagic telangiectasia, a disease characterized by disordered vascular remodeling events and arteriovenous malformations (AVMs), which can lead to blood vessel rupture and organ hemorrhage (McAllister et al, 1994). Genetic knockouts in mice have confirmed an important role for CD105 in vascular stability by showing that global CD105 knockout mice die at mid gestation due to a failure of effective blood vessel formation (Bourdeau et al, 1999). Furthermore, endothelial cell-conditional CD105 knockout mice display defective vascular integrity, reduced vascular remodeling, and AVMs (Mahmoud et al, 2010). More recently, CD105 has attracted a lot of interest because it is reportedly expressed at higher levels on angiogenic endothelial cells in tumor-associated vessels, relative to stable vessels in normal tissue (Minhajat et al, 2006; Yao et al, 2005). These results show an important role for CD105 in the formation, stabilization, and remodeling of blood vessels, and further suggest that CD105 may be a useful prognostic marker for the development of immature blood vessels in tumors.
In our laboratory, we study the vascular remodeling events induced by chronic cerebral hypoxia. Chronic cerebral hypoxia (8% oxygen for 2 weeks) promotes an active and robust vascular remodeling response, which culminates in ∼50% increased vascular density across all areas of the central nervous system (CNS) (LaManna et al, 2004; Milner et al, 2008a). The value of this model is underscored by the finding that mice exposed to chronic cerebral hypoxia (hypoxic preconditioned) are subsequently protected from focal cerebral ischemia (Miller et al, 2001). In light of the obvious need for arteriogenesis as part of an effective remodeling response after ischemic stroke, and the interesting expression pattern of CD105 on angiogenic vessels, the object of this study was twofold: first, to determine if chronic hypoxia promotes the formation of new arterial vessels, and second, to determine if CD105 preferentially labels angiogenic endothelial cells during hypoxic-induced vascular remodeling.
Materials and methods
Animals
The studies described have been reviewed and approved by The Scripps Research Institute Institutional Animal Care and Use Committee. Wild-type C57Bl/6 was maintained under pathogen-free conditions in the closed breeding colony of The Scripps Research Institute.
Chronic Hypoxia Model
Wild-type C57Bl/6 littermate mice, 8 to 10 weeks of age, were housed four to a cage, and placed into a hypoxic chamber (Biospherix, Redfield, NY, USA), maintained at 8% oxygen for periods of 4, 7, and 14 days. Littermate controls were kept in the same room under similar conditions except that they were kept at normal oxygen levels (normoxia) for the duration of the experiment. Every few days, the chamber was opened for cage cleaning and food and water replacement as needed.
Immunohistochemistry and Antibodies
Immunohistochemistry was performed as described previously (Milner and Campbell, 2002) on 10 μm frozen sections of cold phosphate-buffer saline perfused brains taken from mice subject to either normoxia (control) or hypoxia for 4, 7, and 14 days. Each slide contained mouse brains representing the four different time points of hypoxia, to ensure consistent antibody incubation times across different time points. The following monoclonal antibodies were obtained from BD Pharmingen (La Jolla, CA, USA): rat monoclonal antibodies reactive for CD31 (clone MEC13.3), CD105 (clone MJ7/18), and the integrin subunits α5 (clone 5H10-27 (MFR5)), α6 (clone GoH3). Other antibodies used included: rat anti-β1 integrin (clone MB1/2; Chemicon, Temecula, CA, USA), mouse anti-α-SMA-Cy3 conjugate (clone 1A4; Sigma, St Louis, MO, USA), and hamster anti-CD31 (clone 2H8; Abcam, Cambridge, MA, USA). Secondary antibodies used included goat anti-Armenian hamster-DyLight 594 (Biolegend, San Diego, CA, USA), and anti-rat Alexa Fluor 488 (Invitrogen, Carlsbad, CA, USA). For flow cytometry analysis, α5 and α6 integrins were analyzed using the same rat monoclonals listed above, conjugated to PE (BD Pharmingen), and β1 integrin was examined using the β1 monoclonal listed above, followed by PE-conjugated donkey anti-rat (BD Pharmingen).
Image Analysis
Images were taken using a × 20 objective on a Zeiss Imager M1.m (Thornwood, NY, USA). Analysis was performed in the frontal lobe and medulla regions of the brain. For each antigen, three images were taken per region at × 10 or × 20 magnification and the mean calculated for each subject over three different sections. All data analysis was performed using Perkin Elmer Volocity software (Waltham, MA, USA). This includes quantification counts of alpha smooth muscle actin (α-SMA)-positive vessels, low-intensity CD105 vessels, and vessel size variation. To quantify the expression levels of the α5 and α6 integrin subunits, Volocity software was used to measure the fluorescent intensity of each vessel, and thereby calculate the mean vessel intensity of each integrin subunit per field of view for each condition. In each field of view, mean intensity measures were obtained for all vessels, and for the α-SMA-positive vessels. Each experiment was performed with three different animals per condition, and the results expressed as the mean±s.e.m. Statistical significance was assessed by using Student's t-test, in which P<0.05 was defined as statistically significant.
Brain Endothelial Cell Culture
Pure cultures of mouse brain endothelial cells (BECs) were obtained as described previously (Milner et al, 2008b), with the modification that puromycin (4 μg/mL; Alexis GmbH, Grunberg, Germany) was included in culture media between days 1 and 3 to remove contaminating cell types. Endothelial cell purity was >99% as determined by CD31 in flow cytometry, and was used only for the first passage. BECs were cultured on collagen I-coated six-well plates (Nunc, Naperville, IL, USA) in the absence or presence of TGF-β1 (R&D Systems, Minneapolis, MN, USA) at two different concentrations (2 and 10 ng/mL) for 2 days.
Flow Cytometry
Integrin expression of BEC was examined as described previously (Milner et al, 2008b). The fluorescent intensity of labeled cells was analyzed with a Becton Dickinson FACScan machine (San Diego, CA, USA), with 10,000 events recorded for each condition. For each integrin subunit, the mean fluorescent intensity of BEC cultured at the two different doses of TGF-β1 was compared with cells grown in the absence of TGF-β1. Each experiment was repeated three times and the data expressed as the mean±s.e.m. Statistical significance was assessed by using Student's t-test, in which P<0.05 was defined as statistically significant.
Results
Cerebral Hypoxia Promotes the Appearance of Vessels that Show Reduced Levels of Endoglin (CD105) Expression
Chronic cerebral hypoxia promotes an active and robust vascular remodeling response, which culminates in increased vascular density across all areas of the CNS (LaManna et al, 2004). Two well-defined markers of endothelial cells are PECAM-1 (CD31) and endoglin (CD105). As previous studies show that CD31 and CD105 show differential expression profiles during angiogenesis (Minhajat et al, 2006; Yao et al, 2005), we performed dual-immunofluorescence (dual-IF) to examine whether BEC in the actively remodeling hypoxic CNS shows any difference in the expression of these two markers. Consistent with previous studies, CD31 was uniformly expressed by every vessel at all stages of the hypoxic response. In the normoxic CNS, CD105 expression totally overlapped with CD31, with uniform expression on all vessels. However, in the hypoxic CNS, clear changes became evident in the CD105 staining pattern, with two distinct levels observed: bright on linear vessels, but much weaker on what appeared to be loosely aggregated vascular structures (Figure 1). Interestingly, the morphology of the low-CD105 vascular structures was strikingly similar to that of the high-α5 integrin expressing angiogenic endothelial cell aggregates we previously described in the developing CNS (Milner and Campbell, 2002), suggesting that the low-CD105 structures may represent areas of remodeling BEC. This is well illustrated in Figure 1B, which displays a linear compact vessel expressing equivalent high levels of CD31 and CD105, as well as another vessel, which appears to be loosely organized, indicative of remodeling, expressing markedly reduced levels of CD105. To determine how hypoxia influences the appearance of low-CD105 vessels, we next quantified the appearance of low-CD105 vessels in two different areas of the brain: frontal lobe and medulla, at different time points of hypoxia, including normoxia (control), 4, 7, and 14 days hypoxia. This revealed that in both areas of the brain, compared with normoxic conditions, the percentage of low-CD105 vessels was significantly increased after 4 and 7 days hypoxia, but after 14 days hypoxia had started to decline back toward the normoxic level (Figure 1C). This temporal profile closely resembles the angiogenic response previously characterized in the chronic hypoxia model (Li et al, 2010), consistent with the idea that remodeling BECs transiently downregulate CD105 expression.
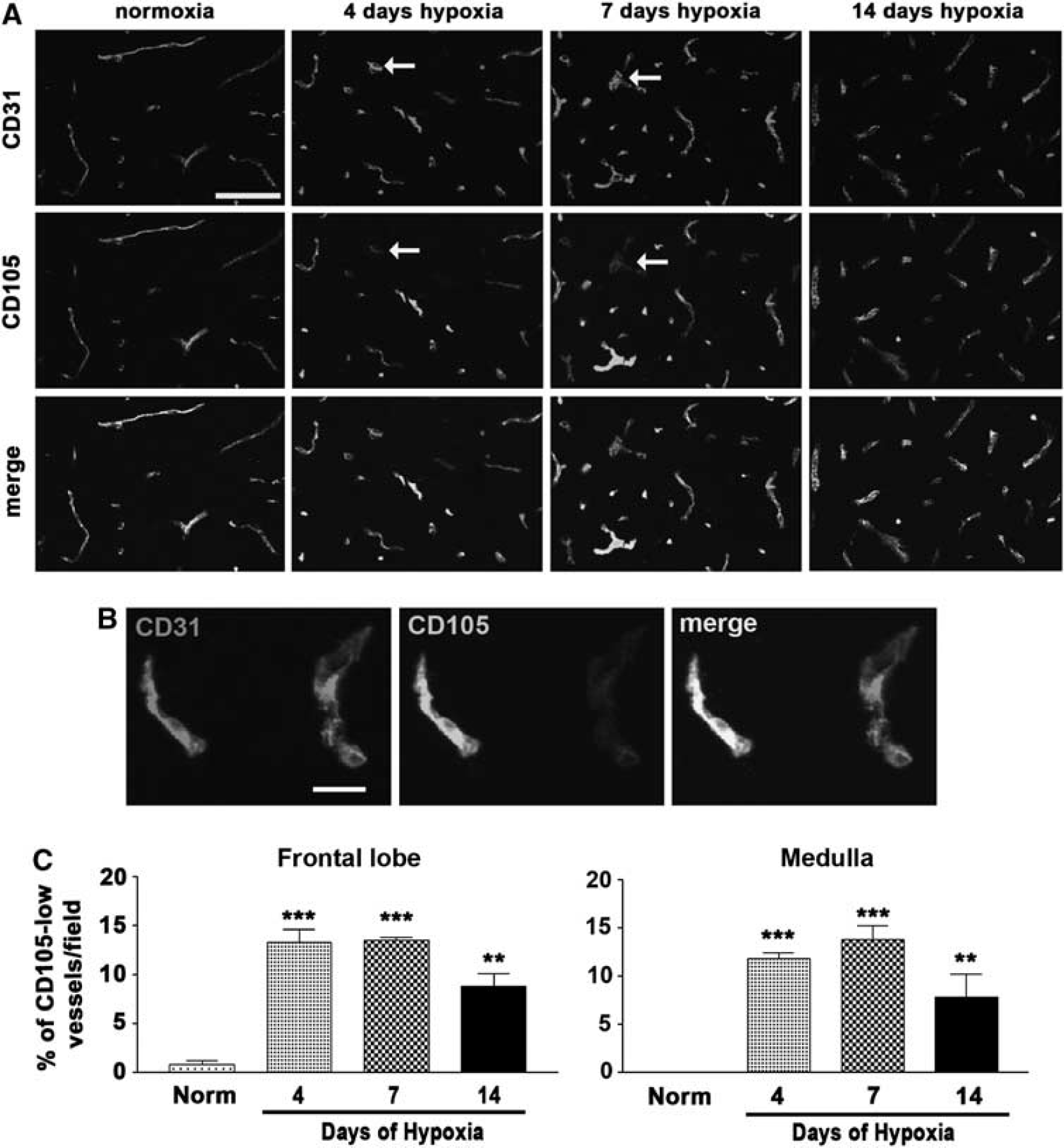
Differential expression profiles of CD31 and CD105 on blood vessels in the hypoxic central nervous system (CNS). (
Chronic Cerebral Hypoxia Preferentially Increases the Number of Large Vessels
Until recently, the increased vessel density induced by chronic hypoxia was assumed to be mainly due to an increase in the number of small caliber vessels, thus bringing about a physiological adaptation to reduce the average vessel-neuron distance in the hypoxic brain (LaManna et al, 2004). However, a recent study made the surprising observation that cerebral hypoxia significantly increases the number of large vessels while producing no major increase in the number of smaller vessels (Freitas-Andrade et al, 2011). To seek confirmation of this finding and to thoroughly examine how cerebral hypoxia alters the number of vessels of different sizes, we performed a detailed size distribution analysis of CD31-positive vessels in the normoxic and 4-, 7-, and 14-day hypoxic brains. As shown in Figure 2, in response to 14 days hypoxia, the number of vessels in the frontal lobe with an area of 0 to 100 μm2 appeared to show a small, though nonsignificant decrease, vessels between 100 and 200 μm2 showed an upward trend, but interestingly, larger vessels with areas of 200 to 500 μm2, or >500 μm2 both showed a significant increase in number. Similar changes were also observed in the medulla. This confirms the finding of Freitas-Andrade et al (2011), who showed that cerebral hypoxia promotes a preferential increase in the number of large vessels, while having no obvious change in the number of small vessels.
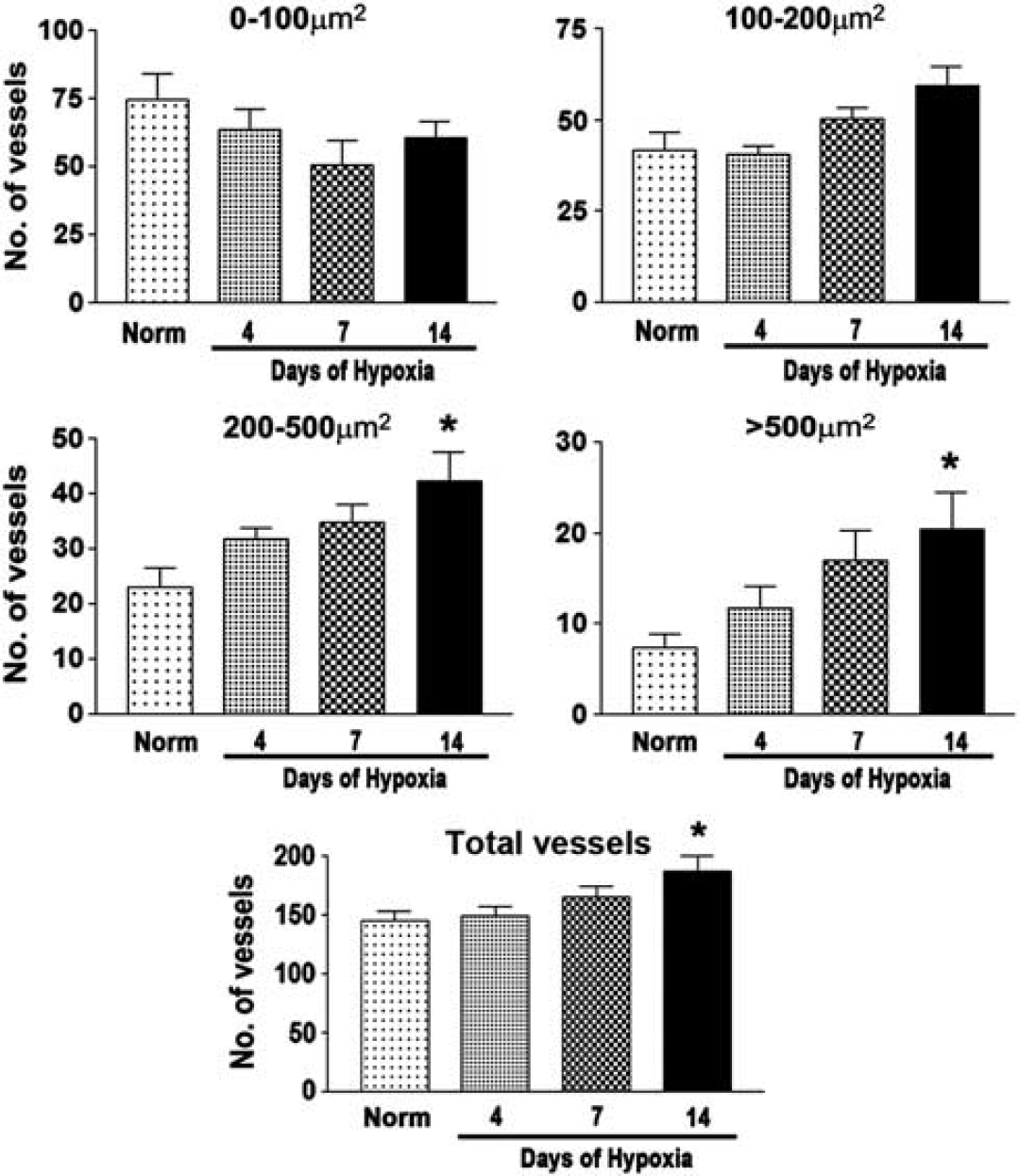
The influence of cerebral hypoxia on vessel size distribution. Frozen sections of frontal lobe taken from mice exposed to normoxia or 4, 7, or 14 days hypoxia were immunostained for CD31, photographs taken, and vessel size distribution analysis performed using Volocity software. All points represent the mean±s.e.m. of three subjects. Note that 14 days hypoxia led to a downward trend in the number of vessels μm2 but in contrast, the number of larger vessels between 200 and 500 μm2 and those >500 μm2 both showed a significant increase. ∗P<0.05.
Cerebral Hypoxia Increases the Number of α-Smooth Muscle Actin-Positive Vessels, and Low-CD105 Expression Colocalizes with these Vessels
As chronic cerebral hypoxia promotes an increased number of large vessels, it was logical to next examine whether cerebral hypoxia actually increases the number of arterial vessels. This is an important question, because although it is well established that hypoxia promotes a strong angiogenic response, whether arteriogenesis occurs under hypoxic conditions has yet to be addressed. To answer this question, we examined the distribution of arterial vessels using α-SMA, a marker of smooth muscle cells, which preferentially labels arteries and arterioles, as these vessels contain the highest number of smooth muscle cells. As expected, in the normoxic CNS, α-SMA was expressed predominantly on larger caliber vessels, being expressed by ∼10% of cerebral vessels (Figure 3A). Interestingly, cerebral hypoxia induced a marked increase in the number of α-SMA-positive vessels. In both areas of brain examined, the frontal lobe and the medulla, the number of α-SMA-positive vessels significantly increased throughout hypoxia (Figure 3B). This shows that cerebral hypoxia induces not only angiogenesis as previously described (LaManna et al, 2004), but also strongly promotes the formation of new arterial vessels, and suggests that some small vessels may be actively remodeled into larger α-SMA-expressing arterial vessels.
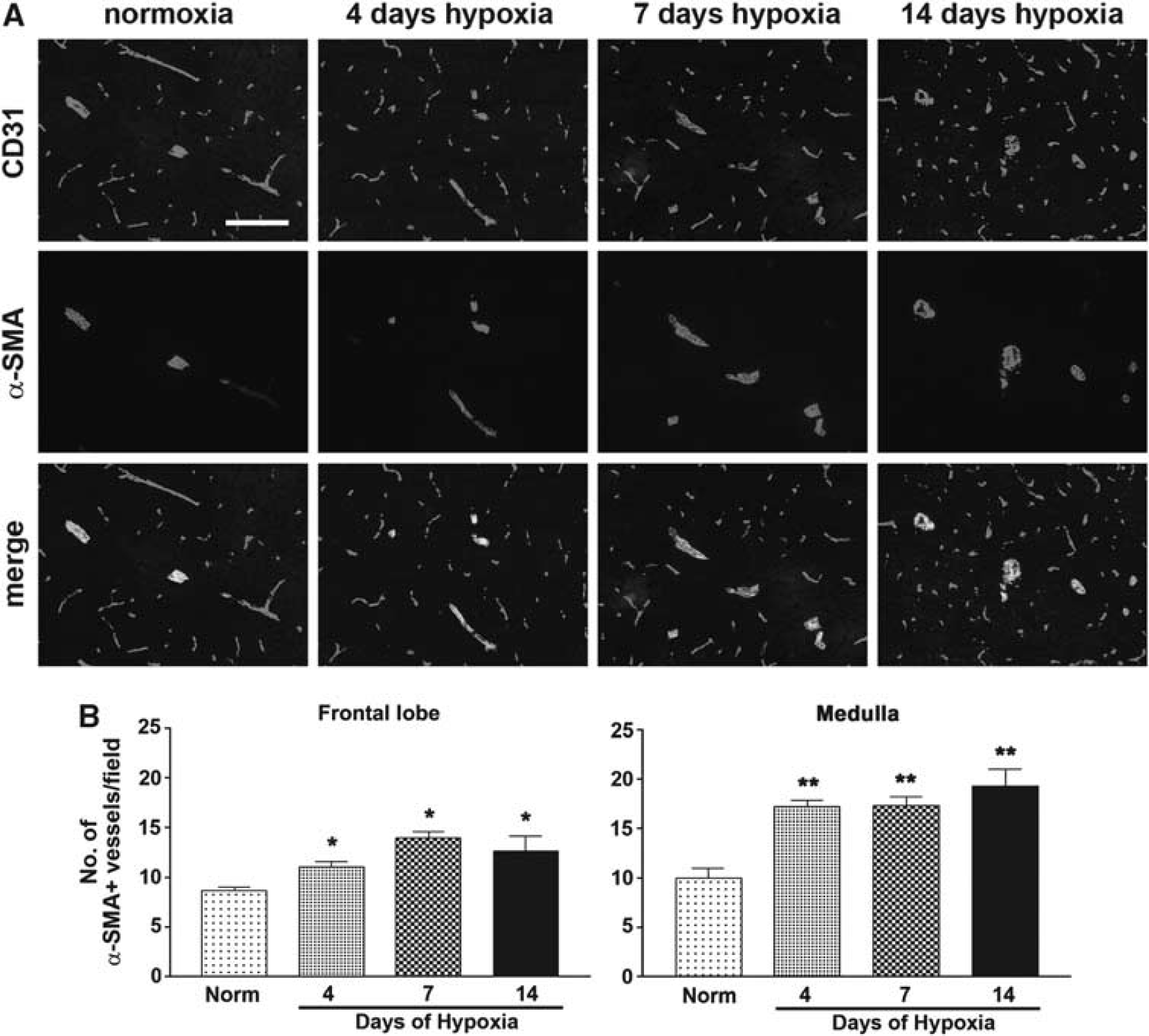
Hypoxia stimulates cerebral arteriogenesis. (
To examine the potential link between the formation of new arterial vessels and the endothelial remodeling events identified by low-CD105 staining, we next performed dual-IF with CD105 and α-SMA (Figure 4). This revealed that under hypoxic conditions, α-SMA-positive vessels expressed much lower levels of CD105. Under normoxic conditions, CD105 was expressed at equivalent levels by α-SMA-positive and α-SMA-negative vessels. However, after 4 or 7 days hypoxia, CD105 expression levels on α-SMA-positive vessels were markedly reduced, to the point of being barely visible on some vessels. This effect was transient because by 14 days hypoxia, CD105 expression on α-SMA-positive vessels had largely returned to prehypoxic levels. Thus, during the hypoxic response, there was a strong colocalization between α-SMA expression and the appearance of low-CD105 expressing BEC. This indicates that cerebral hypoxia promotes vascular remodeling events leading to the formation of new arterial vessels, and that BEC in these remodeling vessels can be identified by low-CD105 expression.
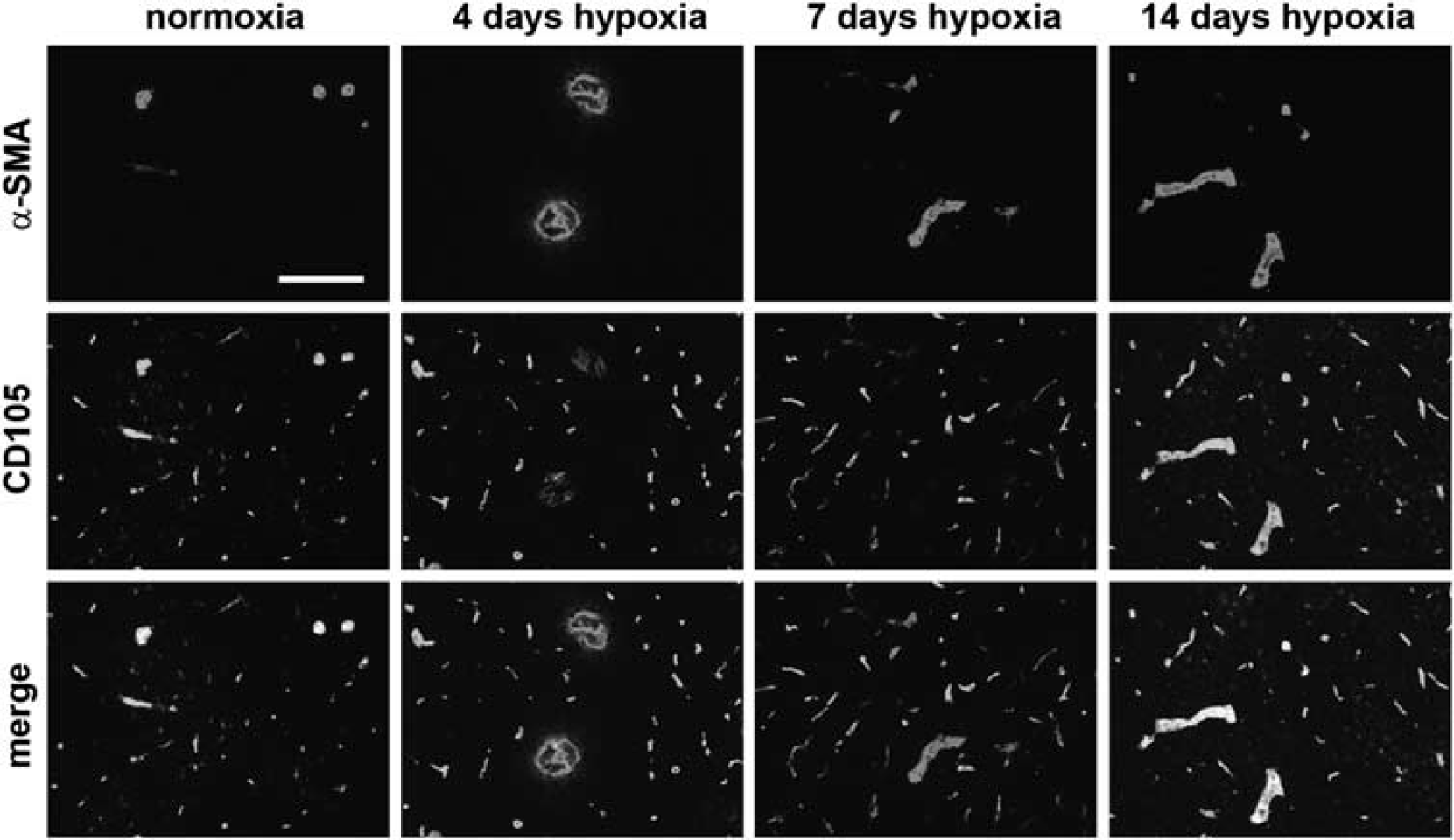
Arteriogenic centers downregulate CD105 expression during cerebral hypoxia. Dual-immunofluorescence (Dual-IF) was performed on frozen sections of the frontal lobe from mice exposed to normoxia or 4, 7, or 14 days hypoxia using antibodies specific for alpha smooth muscle actin (α-SMA) (Cy-3, red) or CD105 (AlexaFluor-488, green). Scale bar=100 μm. Note that in the normoxic CNS, CD105 was expressed at equivalent levels by α-SMA-positive and α-SMA-negative vessels. However, after 4 or 7 days hypoxia, CD105 expression on α-SMA-positive vessels was markedly reduced, to the point of being barely visible on some vessels. This is well illustrated in the merged figures, which under normoxic and 14 day hypoxic conditions, which show α-SMA+ vessels as yellow, but under 4 and 7 day hypoxic conditions, as being only red. The color reproduction of this figure is available on the Journal of Cerebral Blood Flow and Metabolism journal online.
Angiogenic Brain Endothelial Cell Shows a Switch in Expression of β1 Integrins
In previous studies, we described an angiogenic switch in the use of different β1 integrins expressed by BEC during developmental angiogenesis in the CNS, from high α5/low α6 during angiogenesis, to low α5/high α6 integrin in mature vessels (Milner and Campbell, 2002). Using low-CD105 expression as a way of identifying angiogenic BEC in the adult remodeling brain, we next examined whether BEC within remodeling foci in the hypoxic CNS shows a similar switch in integrin expression. Specifically, as low-CD105 expression tightly colocalized to α-SMA-positive vessels, we next examined the expression of the integrin subunits α5 and α6 in α-SMA-positive vessels. Dual-IF with α-SMA and the integrin subunits α5 or α6 was performed on normoxic and 7 day hypoxic brain sections (Figure 5). Within the normoxic CNS, all vessels including the α-SMA-positive vessels expressed the α6 integrin subunit at similar levels. During normoxia, the α5 integrin subunit was expressed at relatively low levels. Significantly, cerebral hypoxia induced marked changes in the relative expression of these integrins. After 7 days hypoxia, α-SMA-positive vessels showed a much weaker α6 integrin signal compared with surrounding vessels, while α5 integrin expression on α-SMA-positive vessels was noticeably enhanced. To characterize these changes, we quantified fluorescence intensity using the Volocity software program. This showed that in both areas of the brain studied, the frontal lobe and medulla, BEC within arteries in the 7-day hypoxic CNS expressed lower levels of α6 and higher levels of α5 integrin, relative to normoxic conditions. This shows that in a similar manner to angiogenic BEC in the developing CNS (Milner and Campbell, 2002), BEC within remodeling arterial vessels of the hypoxic CNS makes a switch in β1 integrin expression, downregulating the α6 subunit while upregulating the α5 integrin subunit.
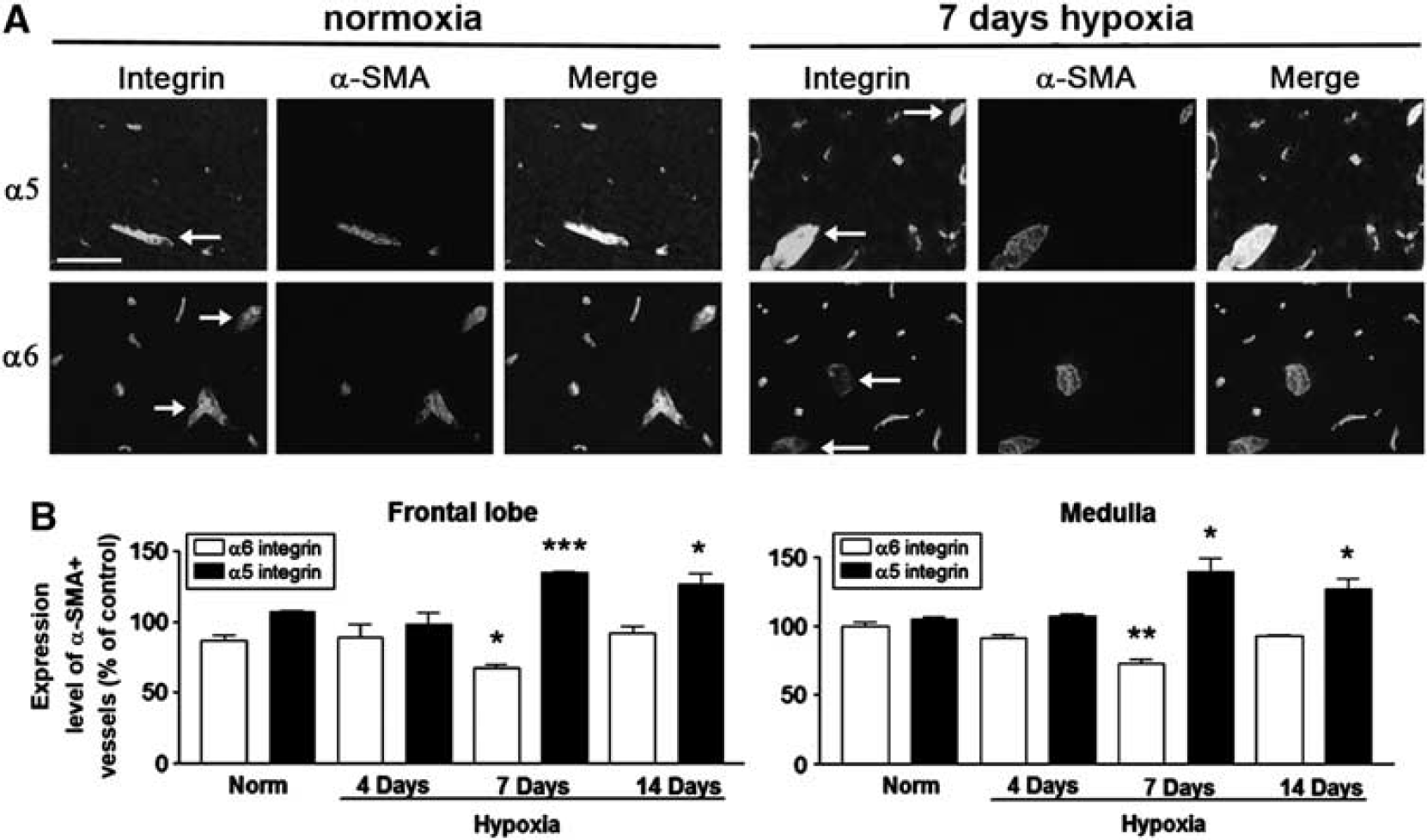
Cerebral hypoxia promotes a switch in endothelial β1 integrins within remodeling arterial vessels. (
Transforming Growth Factor-β1 Treatment In Vitro Promotes a Switch in Brain Endothelial Cell β1 Integrin Subunits from α6 to α5
Genetic knockout studies have shown that CD105 endothelial cell-specific conditional knockout mice display disrupted arterial integrity and AVMs, suggesting that CD105 is important for the maintenance of vessel stability (Mahmoud et al, 2010). By this reasoning it follows that when vessels remodel, they would transiently downregulate CD105 expression, and our observations support this idea. CD105 is a transmembrane cell surface protein that acts as an auxillary TGF-β1 receptor (Cheifetz et al, 1992), and alterations of CD105 expression trigger changes in TGF-β1 signaling pathways (Guo et al, 2004; Pece-Barbara et al, 2005). Linking these observations together, it is logical to speculate that during hypoxic remodeling, endothelial cells within cerebral arterial vessels downregulate CD105, which leads to alterations in TGF-β1 signaling and arterial vessel remodeling. As endothelial cells within remodeling arterial vessels display a switch in β1 integrins, it is logical to ask whether TGF-β1 signaling might mediate this switch in BEC β1 integrins. To address this question, we isolated and cultured BEC in vitro and treated them with TGF-β1 for 2 days, before examining BEC expression of the integrin subunits α5, α6, and β1 by flow cytometry. As shown in Figure 6, TGF-β1 treatment of BEC produced marked changes in integrin expression. Both doses of TGF-β1 used (2 and 10 ng/mL) reduced α6 integrin expression to almost half that of control values, while concomitantly causing threefold increases in α5 integrin expression. Levels of the β1 integrin subunit were not significantly changed. This shows that TGF-β1 directly promotes an angiogenic switch in BEC expression of β1 integrins, from α6 to α5, consistent with the notion that a change in BEC CD105 levels in vivo leads to alterations in TGF-β1-mediated signaling, that may be instrumental in promoting ateriogenic remodeling events within the hypoxic CNS.
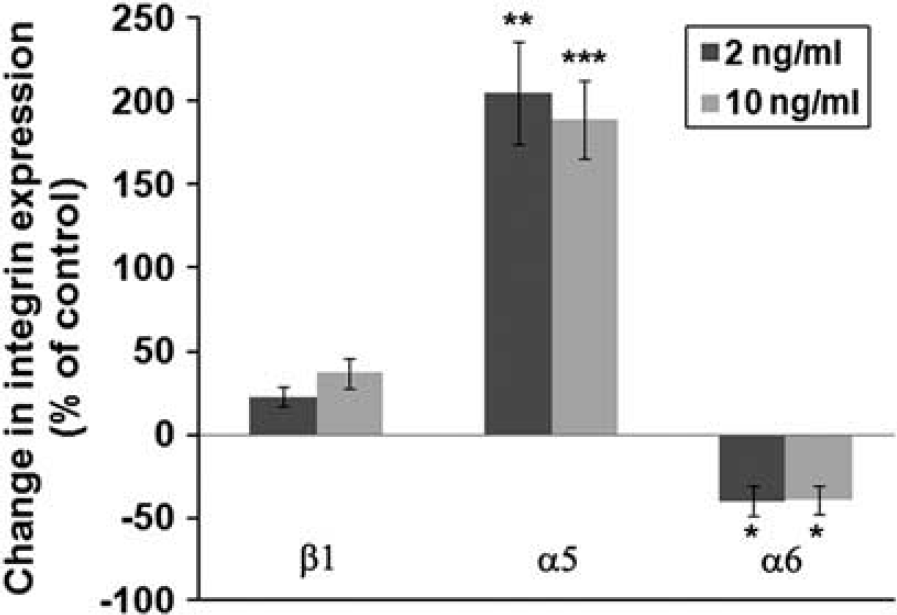
TGF-β1 promotes a switch in endothelial cell β1 integrins. Brain endothelial cells (BECs) were isolated and cultured as described in Materials and methods, and incubated in the absence or presence of 2 or 10 ng/mL TGF-β1 for 2 days and the expression level of the α5, α6, and β1 integrin subunits quantified by flow cytometry. All points represent the mean±s.e.m. of three experiments and are expressed as the percentage change relative to control conditions. Note that both doses of TGF-β1 reduced α6 integrin expression to almost half that of control values, while concomitantly causing a threefold increase in α integrin expression. ∗P<0.05, ∗∗P<0.02, ∗∗∗P<0.01.
Discussion
While chronic cerebral hypoxia leads to a robust vascular remodeling response throughout the CNS (LaManna et al, 2004), little is known about which part of the vascular tree is modified, or whether this response includes the formation of new arterial vessels. In this study, we examined these events in more detail, and defined the expression of endoglin (CD105), a marker of angiogenic endothelial cells in tumors (Minhajat et al, 2006; Yao et al, 2005). Interestingly, we found that cerebral hypoxia produced the biggest increase in the number of medium to large size vessels, with some slight reduction in the number of small vessels, consistent with the notion that hypoxia triggers active remodeling of small vessels into larger ones. These changes correlated with an increase in the number of arterial vessels. Surprisingly, hypoxia produced a marked reduction in CD105 expression on endothelial cells within actively remodeling arterioles, and these remodeling BEC displayed an angiogenic switch in β1 integrins (from α6 to α5), previously described during developmental angiogenesis (Milner and Campbell, 2002). In parallel in-vitro experiments, TGF-β1 promoted the angiogenic switch of BEC β1 integrins. These findings show that cerebral hypoxia promotes remodeling throughout the entire vascular network, including the formation of new arterial vessels. Furthermore, we have identified reduced CD105 expression levels as a novel marker of cerebral arteriogenesis. Taken together, our data suggest a mechanistic model in which BEC in remodeling arterial vessels downregulates CD105 expression, thereby altering TGF-β1 signaling, to promote an angiogenic switch in β1 integrins, facilitating vascular remodeling and the formation of new arterial vessels.
Arteriogenesis in the Hypoxic Central Nervous System
While angiogenesis, the formation of new capillaries from preexisting vessels, is regulated by hypoxia and a well-described network of downstream mediators (Folkman, 1995), arteriogenic remodeling is predominantly driven by hemodynamic factors, such as stretch or flow shear stress (Persson and Buschmann, 2011). In rat, chronic hypoxia (10% O2) promotes a twofold increase in cerebral blood flow during the first 2 days of hypoxia, which gradually returns to baseline by 4 days (Xu et al, 2004). Similar observations have also been made in mice (C Piantadosi, personal communication). Based on this, it seems likely that arteriogenesis in the cerebral hypoxia model is driven by an increased cerebral blood flow, as part of the physiological adaptive response to maintain oxygen delivery to the CNS. However, the elevated blood viscosity induced by hypoxia, which continues to increase throughout the hypoxic period, could also contribute to this process by increasing flow shear stress. While relatively little is known about cerebral arteriogenesis, a small number of studies in the last few years have begun to shed light on this process. Specifically, Buschmann and colleagues showed that occlusion of the vertebral arteries and left carotid artery triggered marked widening of the posterior cerebral artery. This effect was accelerated by delivery of granulocyte-macrophage colony stimulating factor, implicating a potential role for macrophages/microglia in cerebral arteriogenic remodeling (Buschmann et al, 2003). Additional studies have shown that arteriogenic remodeling of leptomeningeal collateral vessels on the surface of the brain is promoted by occlusion of the common carotid artery, and that this response is enhanced by delivery of granulocyte-macrophage colony stimulating factor (Todo et al, 2008) or adrenomedullin (Maki et al, 2011).
CD105 Expression and Function
In recent years, a number of studies have described elevated levels of CD105 on angiogenic endothelial cells in tumor-associated vessels in several organs, prompting the suggestion that CD105 may provide a useful diagnostic marker for angiogenic blood vessels in tumors (Minhajat et al, 2006; Yao et al, 2005). However, the specificity of CD105 for angiogenic tumor vessels was not observed in kidney or liver (Minhajat et al, 2006), and a separate study showed CD105 expression on all endothelial cells in normal brain (Matsubara et al, 2000). Our finding of decreased CD105 expression by BEC within remodeling arterial vessels is inconsistent with the idea of CD105 as a marker of angiogenic endothelial cells. Several reasons may account for this difference. First, the specificity of CD105 expression for tumorigenic angiogenic endothelial cells appears to be organ specific (Minhajat et al, 2006), and importantly is not observed in the brain (Matsubara et al, 2000). Second, all studies describing raised CD105 levels on angiogenic endothelial cells have been performed on tumor tissue, and it remains to be defined whether angiogenic vessels in other situations, for instance during development, also show this profile. Based on these findings, it seems likely that regulation of endothelial CD105 expression is organ specific. In the current study, we show that in the CNS, reduced CD105 expression can be used as a marker of arteriogenic remodeling. In future studies, it will be interesting to see if the same holds true for other organs.
What function does endoglin (CD105) have in regulating endothelial cell behavior? CD105 mutations in humans result in hereditary hemorrhagic telangiectasia, a disease characterized by disordered vascular remodeling and the appearance of AVMs (McAllister et al, 1994). Consistent with this, CD105 global knockout mice die at mid gestation due to a major failure of vascular development (Bourdeau et al, 1999), and endothelial cell-specific CD105 knockout mice display defective vascular integrity and vascular remodeling, increased endothelial proliferation and AVMs (Mahmoud et al, 2010). These collective findings suggest that CD105 is important for the maintenance of vessel stability, and that in its absence, vessels fail to form stable structures, resulting in excessive endothelial cell proliferation and vascular malformations. This reasoning suggests that endothelial cells within remodeling vessels will transiently downregulate CD105 expression, which is consistent with our observations in the hypoxia model. CD105 is a transmembrane protein that is an auxiliary TGF-β receptor (Cheifetz et al, 1992). In endothelial cells, TGF-β1 has positive or negative effects, stimulating endothelial cell proliferation and migration through the activin receptor-like kinase 1 (ALK1) pathway or inhibiting these responses via the ALK5 pathway (ten Dijke et al, 2008). Interestingly, CD105 strongly influences TGF-β1 signaling pathways (Guo et al, 2004), best illustrated by the finding that CD105 null endothelial cells proliferate faster and are more responsive to TGF-β1 (Pece-Barbara et al, 2005). Taken with evidence that TGF-β1 stimulates arteriogenesis in the peripheral circulation (van Royen et al, 2002), our data suggest that BEC within remodeling arterial vessels downregulates CD105 expression, which triggers alterations in TGF-β1 signaling, leading to arteriogenic remodeling.
Angiogenic/Arteriogenic Switch of Endothelial β1 Integrins
Integrins have critical roles in regulating vascular remodeling both during development and in the adult (Silva et al, 2008). The α5β1 integrin is expressed at high levels by angiogenic blood vessels in development, downregulated during vessel maturation (Milner and Campbell, 2002) and then reinduced by angiogenic vessels in the adult CNS (Milner et al, 2008a). Significantly, upregulation of endothelial α5β1 integrin expression has also been reported on arteriogenic vessels (Cai et al, 2009). An angiogenic role for the α5β1 integrin has been shown both in vivo (Francis et al, 2002) and in vitro (Wang and Milner, 2006). Conversely, the α6β1 integrin mediates endothelial differentiation and stability of blood vessels (Germain et al, 2010). During developmental angiogenesis in the CNS, BECs switch integrins from α5 to α6 dominant (Milner and Campbell, 2002). In the current study, we found that in the actively remodeling arteriogenic foci of the hypoxic CNS, BEC also displayed the α6 to α5 integrin switch. As the factor/s responsible for triggering this angiogenic switch have yet to be identified, we were motivated by the observation that vessels displaying this switch also expressed low levels of CD105, suggesting a potential role for TGF-β1. Our in-vitro studies confirmed that TGF-β1 promoted the angiogenic switch of BEC integrins, consistent with a role for TGF-β1 in cerebrovascular remodeling: (1) TGF-β1 promotes upregulation of α5 integrins on several different cell types (Roberts et al, 1988), (2) TGF-β1 is an important regulator of vascular remodeling (Madri et al, 1992), and (3) TGF-β1 levels in the ischemic brain are increased at the same time that vascular remodeling is ongoing (Krupinski et al, 1996; Lehrman et al, 1998). In summary, our findings show that cerebral hypoxia promotes arteriogenic remodeling, and that these events can be identified by reduced CD105 expression. These data suggest a mechanistic model in which BEC within remodeling arterial vessels downregulate CD105 expression, triggering alterations in TGF-β1 signaling, to promote the angiogenic switch in β1 integrins and formation of new arterial vessels. Manipulation of this mechanism might provide a novel approach for enhancing brain arteriogenesis, which could have important clinical implications for the treatment of ischemic stroke.
Footnotes
Disclosure/conflict of interest
The authors declare no conflict of interest.
