Abstract
Therapeutic hypothermia promotes protection after traumatic brain injury (TBI). The mechanisms underlying hypothermic protection are multifactorial and may include the modulation of microRNA (miRNA) expression after trauma. We utilized microarrays to examine the effects of posttraumatic hypothermia on the expression of 388 rat miRNAs. Animals were subjected to sham or moderate fluid percussion brain injury, followed by 4 hours of hypothermia (33°C) or normothermia (37°C) and euthanized at 7 or 24 hours. At 7hours, 47 miRNAs were significantly different (P < 0.05) between TBI and sham (15 higher in TBI and 31 lower). After 24hours, 15 miRNAs differed by P < 0.05 (7 higher and 9 lower). The expression of miRNAs was altered by posttraumatic hypothermia. At 7hours, seven were higher in hypothermia than normothermia and five were lower. Some miRNAs (e.g., miR-874 and miR-451) showed the most difference with hypothermia, with changes verified by quantitative reverse transcriptase-PCR. Regionally specific miRNAs also showed responses to TBI and hypothermia treatments by in situ hybridization. In addition, in vitro neuronal stretch injury studies showed similar temperature-sensitive responses to specific miRNAs. These novel data indicate that the reported beneficial effects of early hypothermia on traumatic outcome may include temperature-sensitive miRNAs involved in basic cell-processing events.
Introduction
Therapeutic hypothermia has been reported to promote improved functional outcome and limit secondary damage in models of focal and diffuse traumatic brain injury (TBI) (Marion and Bullock, 2009). Moderate cooling initiated after the primary insult reduces contusion volume, protects selectively vulnerable neuronal populations, reduces diffuse axonal injury, and improves blood-brain barrier function (Dietrich et al, 2009). Indeed, hypothermia is one of the few cytoprotective treatments that has been successfully translated to specific patient populations including cardiac arrest and neonates experiencing hypoxic-ischemic insults (Bernard et al, 2002; Shankaran et al, 2005). In TBI, several single institutional studies have reported beneficial effects of moderate hypothermia on severely injured patients (Polderman, 2008). In contrast, multicenter trial data appear to indicate that patient-cooling strategies do not protect across the wide range of severe TBI populations (Clifton et al, 2011).
Over the last decade, various studies have identified temperature-sensitive pathomechanisms that may underlie the beneficial effects of early posttraumatic hypothermia. These include excitotoxicity, free radical generation, antiapoptotic mechanisms, as well as inflammatory mechanisms (Dietrich et al, 2009). The identification of multiple temperature-sensitive mechanisms supports the concept that moderate cooling may be protective by its ability to target multiple inflammatory cascades, and therefore act like a therapeutic cocktail. Although this conclusion makes sense from the treatment perspective, it remains unclear whether cooling strategies may also target more basic cellular processes known to be critical for cell survival or death.
The basic molecular and cellular processes that are affected by posttraumatic hypothermia remain poorly understood and could include posttranscriptional modifications. Recently, it has been shown that a class of small non-coding RNAs has wideranging and profound effects on the translation of many proteins and the function of cells in general. MicroRNAs (miRNAs) have been found in all species tested and many thousands have been identified. The miRNAs are ∼21 nucleotides long, which negatively regulate gene expression posttranscriptionally. They are initially transcribed as a longer primary miRNA transcript that is cleaved by the enzyme Drosha into a 70- to 100-nucleotide pre-miRNA. A second cleavage by Dicer produces the mature miRNAs, which incorporate into the RNA-induced silencing complex where they target the mRNA of protein-coding genes for either degradation or translational repression (Lee et al, 2002). The first miRNA was discovered in 1993 as a result of a genetic screen for mutations in Caenorhabditis elegans (Lee et al, 1993). Since then, miRNAs have been found to have an important regulatory role in many, if not all, cellular processes, including differentiation, growth and proliferation, cell cycle, lineage determination, and metabolism (Cao et al, 2006; Harris et al, 2010; Ivey and Srivastava, 2010; Lee and Dutta, 2006). Therefore, each miRNA can potentially target many genes and therefore may be able to affect entire pathways or cellular processes that involve multiple genes. Because miRNAs function at the posttranscriptional level to block translation of proteins, it is reasonable to hypothesize they may have a role in the mechanisms by which hypothermia may protect the cell from damage after TBI.
In the brain, the neuron-specific miRNA, miR-124, which targets over 100 non-neuronal transcripts, is sufficient to shift the development of a cell into a neuronal lineage (Lim et al, 2005). Another neuron-specific miRNA, miR-134, negatively regulates the size of dendritic spines (Schratt et al, 2006). Several neurodevelopmental disorders have been associated with miRNAs. For example, fragile X syndrome, one of the most common mental impairments, was the first to be associated with the involvement of miRNAs. Down's syndrome, caused by triplication of chromosome 21, is the most common cause of cognitive impairment. Five miRNAs are present on chromosome 21, miR-99a, let-7c, miR-125b-2, miR-155, and miR-802. Overexpression of these miRNAs is found in fetal brain and heart tissue of patients with Down's syndrome. MicroRNAs have recently been implicated in spontaneous Alzheimer cases, Huntington, and Parkinson's disease. The miR-133b is deficient in patients with Parkinson and is a miRNA specific to midbrain dopaminergic neurons (for a recent review, see Qurashi and Jin, 2010).
Recently, the role of miRNAs in CNS injury has become increasingly important (Madathil et al, 2010). Many of the predicted gene targets include proteins involved in inflammation, apoptosis, and oxidation, which participate in the pathogenesis of brain and spinal injury. In this regard, changes in miRNA expression have recently been noted after contusive spinal cord injury (Liu et al, 2009). Interestingly, circulating plasma miRNAs may potentially be used as biomarkers to indicate tissue damage. In one study, the presence of brain-specific miR-124 in plasma at 24hours after transient middle cerebral artery occlusion was found to be as much as 150 fold higher than sham animals (Laterza et al, 2009). Liu and colleagues profiled the expression of miRNAs in the brain and blood after experimental ischemic stroke, intracerebral hemorrhage, and kainite seizures (Liu et al, 2010a). This group reported several miRNAs up- or downregulated by more than 1.5 fold after each injury in both the brain and in the blood plasma. The miR-497 is upregulated after middle cerebral artery occlusion in rats, and proposed to promote ischemic neuronal death by negatively regulating antiapoptotic proteins, bcl-2 and bcl-w (Yin et al, 2010).
Currently, two studies have reported miRNA expression findings after experimental TBI. Lei et al (2009) assessed the expression of miRNAs in the ipsilateral cerebral cortex after fluid percussion (FP) injury, whereas Redell et al (2009) investigated altered expression in the hippocampus after controlled cortical injury. These studies demonstrated unique miRNA profiles in vulnerable brain regions after trauma. To date, no studies have determined the effects of posttraumatic hypothermia on patterns of miRNA expression. Because of the importance of posttraumatic temperature on neuronal survival, the goal of the present study was to determine miRNA profiles in the vulnerable cerebral cortex after FP injury in normothermia- and hypothermia-treated animals as compared with sham-operated controls. For this aim, we used a therapeutic hypothermia protocol that has been reported to reduce contusion volume and increase neuronal survival in the pericontusional cerebral cortex (Dietrich et al, 1994; Matsushita et al, 2001; Lotocki et al, 2009).
Materials and methods
Animal Groups
Male Sprague-Dawley rats weighing 280 to 340gm obtained from Charles River Breeders (Wilmington, MA, USA) were used for all experiments. Animal care was in accordance with the guidelines set by the University of Miami Animal Care and Use Committee. All animals were acclimated for at least 7 days before the study and exposed to a 12-hour light-dark cycle. Rats were allowed free access to water, but food was withheld overnight before brain injury. Animal groups included: sham-operated 7 and 24hours, TBI normothermia (37°C) 7 and 24hours, and TBI hypothermia (33°1C) 7 and 24hours, n = 5 per group.
Surgical Preparation and Fluid Percussion Injury
The basic surgical preparation for the moderate parasaggital FP brain injury was performed according to methods previously described (Lotocki et al, 2009). Rats were initially anesthetized with 3.0% isoflurane and a mixture of 70% nitrous oxide and 30% oxygen. The animals were placed in a stereotaxic frame, and a 4.8-mm craniotomy was made overlying the right parietal cortex (3.8 mm posterior to bregma and 2.5mm lateral to the midline; Zilles, 1985). A plastic injury tube was then placed over the exposed dura, was bonded by adhesive as well as dental acrylic and hardened, and the scalp was closed. Animals were returned to their home cage and fasted overnight. The next day, the animals were initially anesthetized with 3.0% isoflurane and a mixture of 70/30% nitrous oxide/oxygen. The femoral artery and vein were cannulated with PE-50 tubing (Becton Dickinson, Franklin Lakes, NJ, USA). Rats were then immobilized with pancuronium bromide (0.5 mg/kg intravenously). An endotracheal tube was inserted orally, and rats were mechanically ventilated and maintained on a mixture of 70% nitrous oxide and 0.5% to 1.0% isoflurane, with a balance of oxygen. In TBI animals, a FP (Dixon et al, 1987) device was used to induce moderate parasaggital FP injury (1.8 to 2.2 atmospheres). Physiological variables were maintained in normal ranges, and core and temporalis temperature was controlled at 37°C with a feedback heat lamp system.
Temperature Manipulations
Temperature manipulation was initiated 30minutes after TBI or sham surgery. Temporalis muscle and rectal (core) temperature probes were used to measure temperature. Selective brain hypothermia was induced 30minutes after injury, with target hypothermic temperature achieved 40 minutes after TBI and maintained using cooled air and heating lamps for a period of 4hours. Animals were allowed to rewarm slowly over a 1.5-hour period and returned to their cages. At 7 or 24hours after TBI, animals were deeply reanesthetized using isoflurane and euthanized. Animals used for miRNA arrays and reverse transcriptase-PCR (RT-PCR) were quickly decapitated, and the ipsilateral cortex was removed and flash frozen in liquid nitrogen. Animals used for in situ hybridization were perfusion-fixed transcardially with isotonic saline at a pressure of 100 to 120mm Hg for 2minutes and followed by fixative (4% paraformaldehyde in a 0.1 mol/L phosphate buffer, pH 7.4) for 28minutes. After perfusion, brains were removed and immersed in fixative at 4°C for 3 days. Brains were then frozen and cryosectioned at 12 or 20 μm.
MicroRNA Array
Total RNA was extracted from ipsilateral cerebral cortex using mirVana MiRNA isolation kit from Ambion (Austin, TX, USA), being careful to include isolation of small RNAs. Five microgram of each sample was sent to be analyzed by LC Sciences (Houston, TX, USA) using their μParaflo microfluidic chips containing 388 unique rat miRNA transcripts listed in Sanger miRBase Release 14.0. Samples from three animals per group included sham 7hours, sham 24hours, TBI normothermia 7hours, TBI hypothermia 7hours, TBI normothermia 24hours, and TBI hypothermia 24 hours. Each array was hybridized with two of the samples, randomly assigned. The samples were labeled with either Cy3 or Cy5. For the sample to be counted, the signal intensity had to exceed 3 × background standard deviation and then normalized using a LOWESS filter (locally weighted Regression). All samples with signals greater than 32 were measured. In-depth data analysis was provided by LC Sciences. The mean of each group was calculated, and one-way analysis of variance statistical analysis was performed. An miRNA was considered to be differentially expressed if the P-value was < 0.05 (Supplementary Table 1).
Quantitative RT-PCR of selected miRNAs

miRNA, microRNA; RT-PCR, reverse transcriptase-PCR.
Levels of expression of miR-874, miR-451, miR-497, miR-34a, miR-27b, and miR-9 compared with sham-operated animals are consistent with the trends shown in the array results. Means for each group are expressed as fold change compared with sham animals.
Fold-change values versus sham-operated animals are shown, mean ± s.e.m.
Reverse Transcriptase-PCR
The changes in the expression of some of the most interesting miRNAs were confirmed by quantitative real-time RT-PCR. In all, 20 ng of total RNA from each animal (n = 3 per group) was reverse transcribed using specific primers to the miRNA of interest (TaqMan MicroRNA Assay Reverse Transcription Primers; Applied Biosystems, Foster City, CA, USA), followed by real-time PCR with TaqMan MicroRNA assay primers. The quantitative PCR (qPCR) assays were performed in triplicate. The relative expression of miR-874, miR-497, miR-451, miR-34a, miR-27b, and miR-9 were analyzed using the standard 2−ΔΔCt method (Livak and Schmittgen, 2001) and one-way analysis of variance, followed by post hoc Holm-Sidak. Results are shown as fold change versus sham operated. U87 was used as the endogenous control for all samples. U87 (also known as SNORD87) is a small non-coding nucleolar RNA that is highly conserved and abundant, which functions in the modification of other small nuclear RNAs.
In situ Hybridization
To determine the regional patterns of expression for two of the miRNAs studied, we performed in situ hybridization experiments on miR-9 and miR-874. Sham, TBI normothermia, and TBI hypothermia animals were killed after 7 hours and brains were removed and flash frozen. Twelve-micrometer sections (bregma level −3.8 mm, two animals per group) were cryosectioned onto Superfrost Plus slides (VWR, West Chester, PA, USA). The miRNA signals were detected by using fluorescently labeled locked nucleic acid (LNA) probes and tyramide signal amplification, as previously described (Pena et al, 2009). Some slides were also processed without the LNA probe as a negative control. Slides were thawed and fixed in 4% formaldehyde in Tris-buffered saline (TBS: 137mol/L NaCl, 2.7mol/L KCl, 25mol/L Tris, pH 7.4) for 10minutes then washed several times in TBS. To remove residual phosphate, slides were processed with 1-ethyl-3-(3-dimethylaminopropyl) carbodiimide fixation. Slides were incubated in 0.13mol/L 1-methyimidazole, 300mol/L NaCl, pH 8.0 for 10minutes, followed by 1 hour in imidizole buffer plus 0.16M 1-ethyl-3-(3-dimethylaminopropyl) carbodiimide (Sigma-Aldrich, Atlanta, GA, USA). Slides were then washed in 2 × TBS/0.2% glycine and 2 × TBS. Slides were acetylated by incubating 30 minutes in 0.1 M triethanolamine and 0.5% acetic anhydride. After two washes in TBS, slides were prehybridized at room temperature in hybridization buffer containing 50% formamide, 5 × SSC, 5 × Denhardt's, 250 μg/mL yeast tRNA (Sigma-Aldrich), 500 μg/mL salmon sperm DNA (Sigma-Aldrich), 2% Blocking reagent (Roche, Indianapolis, IN, USA), 0.1% 3-((3-Cholamidopropyl) dimethylammonio)-1-propanesulfonate (Sigma-Aldrich), 0.5% Tween at 25°C for 2 hours in a humid chamber.
For hybridization, prehybridization buffer was removed and slides were incubated with 4 pmol of DIG-labeled LNA probe (mercury LNA Detection Probe, Exiqon, Vedbaek, Denmark) diluted in 100 μL of hybridization buffer for 16 hours at Tm–20°C. For miR-9, incubation was at 55°C and for miR-874, it was at 65°C. The following day, slides were washed in 50% formamide, 1 × SSC, 0.1% Tween 20 at the same temperature as hybridization for 1 hour. Slides were treated with 3% hydrogen peroxide in TBS/Tween 20 for 30 minutes. For probe detection, slides were incubated for 1 hour in 500 μL of blocking buffer containing 0.5% blocking reagent, 10% goat serum, 0.1% Tween 20 in TBS, followed by 1 hour in anti-DIG-FAB peroxidase (POD; Roche) diluted 1:500 in blocking solution. Slides were then washed in TBS/Tween and then 200 μL of TSA Plus Cy3 System working solution was applied for 10 minutes (Perkin Elmer Life Sciences, Waltham, MA, USA). After three washes in TBS, slides were coverslipped using Vectashield mounting medium with DAPI (Vector Labs, Burlingame, CA, USA) and analyzed by confocal microscopy. We specifically chose to evaluate expression patterns in vulnerable regions of the ipsilateral cerebral cortex.
Stretch-Induced Injury
To validate the temperature-sensitive response of specific miRNAs to trauma, cortical neurons were grown in Flex Plates (Flexcell International, Hillsborough, NC, USA) and injured using a model 94A Cell Injury Controller (Commonwealth Biotechnology, Richmond, VA, USA), as previously described (Ellis et al, 1995). Cortical neuronal cultures were prepared from embryonic E15-E16 rat cortices, as previously described (Keane et al, 1992). In brief, cortical tissue was disrupted into a cell suspension by gentle trituration and seeded in flex plates. Neurons were grown for 12 days until confluent in N5 medium containing 5% serum fraction. The cells were injured by the device delivering a 50-ms pressure pulse of compressed gas, which transiently deformed the silastic membrane of the Flex Plate equivalent to 6.5 mm deformation of the adherent cells. Cells were then incubated for 3 hours at either 33°C (hypothermia) or 37°C (normothermia). Separate plates were not injured but incubated as shams at both temperatures. Neurons were harvested and processed for miRNA isolation and quantitative miRNA RT-PCR, as described above for tissue samples. Three wells were pooled per group, n = 3.
Results
Samples from sham operated, TBI normothermia, and TBI hypothermia animals were hybridized to microarrays containing most of the known rat miRNAs to determine which were present in cerebral cortex samples. Samples were labeled with either Cy3 or Cy5, and representative pseudocolor arrays are shown in Figure 1. The mean intensity of each group was compared with the mean intensity of the other groups. Those that showed a difference with a P-value of < 0.05 were considered statistically significant and are listed in Supplementary Table 1 in order of significance.
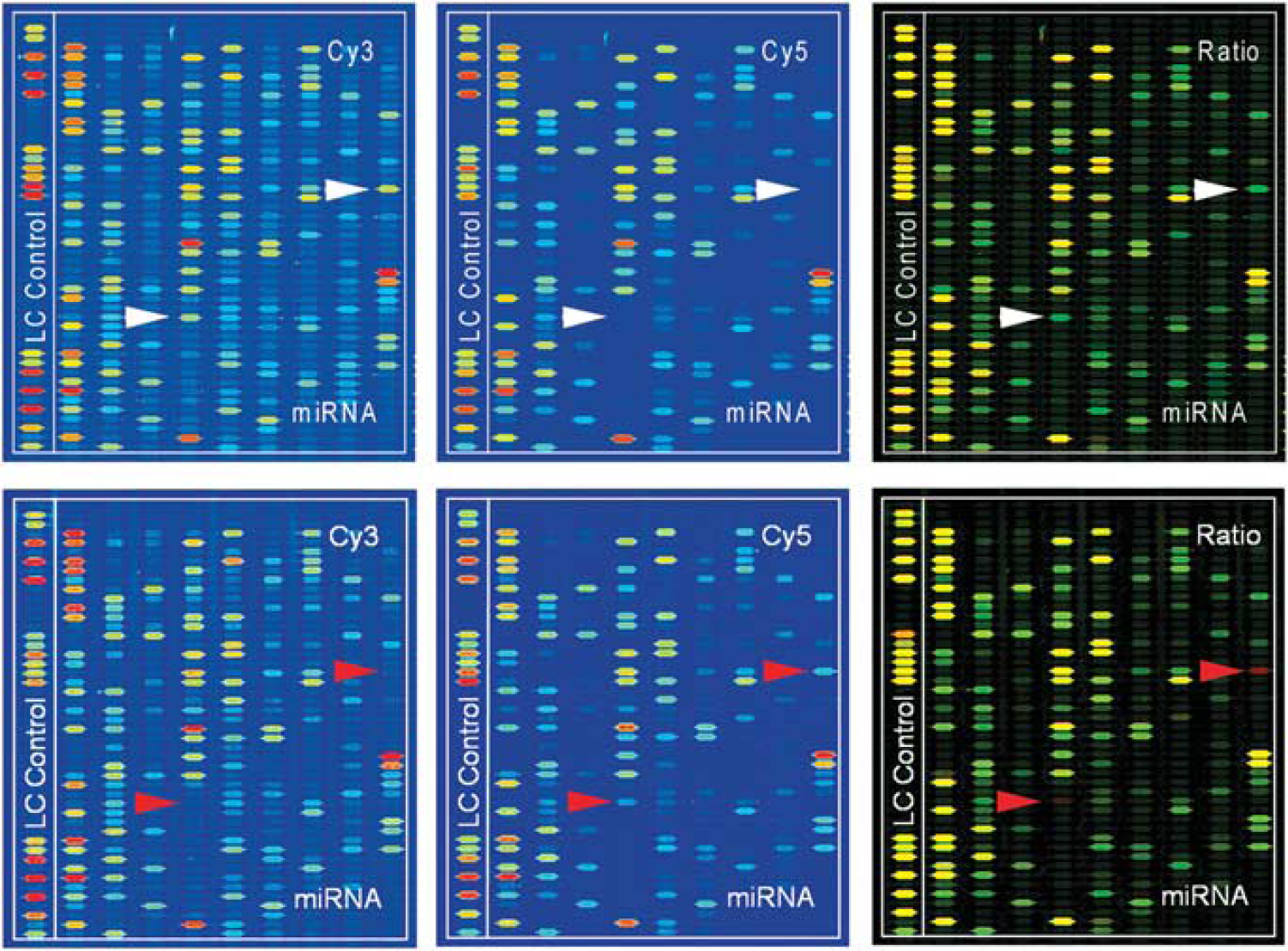
Pseudocolored Cy3/Cy5 images of representative microfluidic arrays of sham and traumatic brain injury (TBI) at 7hours. Merged image is Cy3/Cy5 overlay. Green bands represent lower expression after TBI versus sham (white arrowheads). Red bands in merged image represent higher expression after TBI (red arrow heads). Yellow bands show equivalent expression.
At 7hours after TBI, there were 49 miRNAs that differed by P < 0.05 from sham animals, 19 of which differed by P < 0.01. The heat maps (Figure 2) showed 16 miRNAs that were higher than sham (red) and 31 lower than sham (green). Some of the miRNAs that were the most elevated after TBI included the well-characterized miR-9 (P = 0.0004). The miR-9 is brain specific and involved in development (Sempere et al, 2004). Another, miR-290, the most highly enriched after TBI, was 142 fold higher than sham-operated rats. This miRNA is known to be involved in stem cell differentiation. The second most highly enriched miRNA after TBI was miR-874, which was 119 fold higher than sham (P = 0.02). This miRNA is known to have more than 100 possible gene targets.
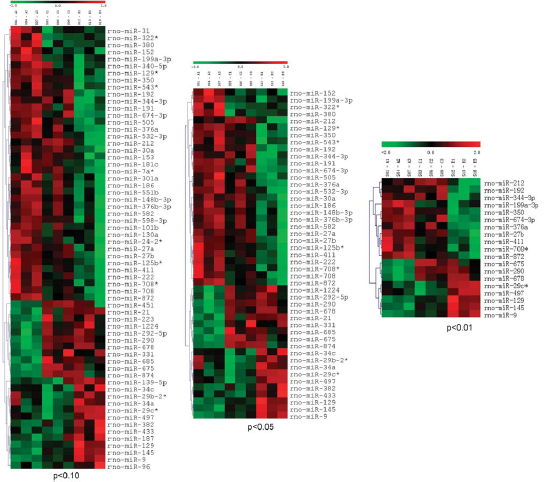
Alterations of microRNA (miRNA) expression at 7hours after traumatic brain injury (TBI). Heat maps of all miRNAs that were significantly different between TBI animals and sham-operated animals at 7 hours after injury. In all, 16 miRNAs were higher after TBI (red) and 31 were lower after TBI (green); P < 0.05.
At 24hours after TBI, there were 16 miRNAs that differed from sham (P < 0.05) of which only 5 differed by P < 0.01. Of these, seven miRNAs were higher than sham and eight were lower (Supplementary Figure 1). The major changes in expression of miRNAs after TBI therefore appear relatively transient, and occur predominately within the first day. For example, let-7d*, which decreased after TBI at 24 hours, was not detected to change at the earlier 7hour time point. This is a member of the let-7 family, which was one of the first miRNAs to be discovered in C. elegans (Reinhart et al, 2000) and is involved in cell differentiation and proliferation.
We next compared the expression of miRNAs after TBI in normothermia animals and hypothermia animals (Figure 3). At 7 hours after trauma, there were seven miRNAs that were elevated in the TBI hypothermia group compared with normothermia TBI, whereas five were lower. MiR-497 had a 2.4 fold increase in hypothermic versus normothermic animals. By 24hours after TBI, there was no difference between the two temperature TBI groups.
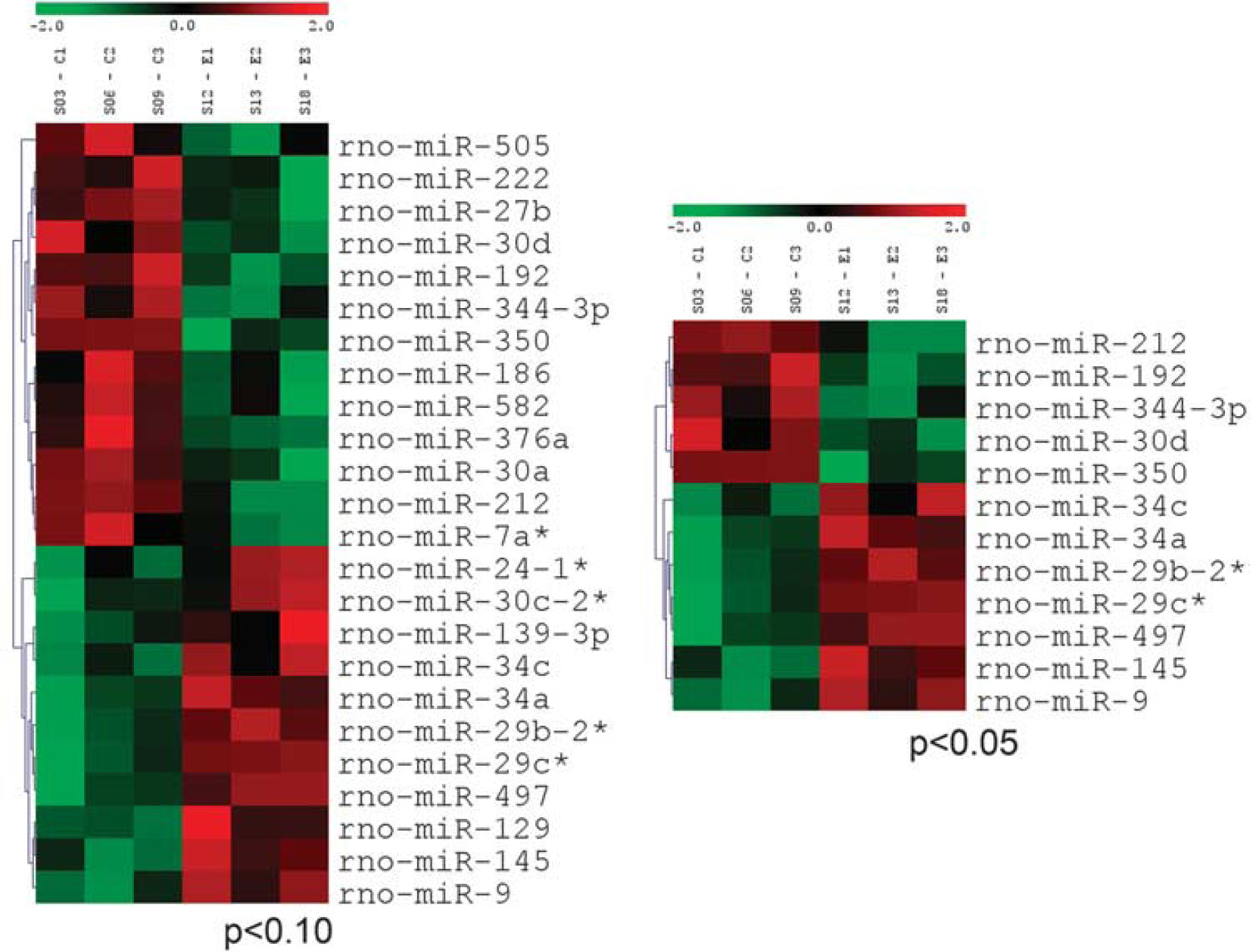
Effect of hypothermia treatment on microRNA (miRNA) expression after traumatic brain injury (TBI). TBI animals were subjected to 4 hours of normothermia (37°C) or hypothermia treatment (33°C), and killed at 7hours after TBI. The miRNA profiling revealed seven miRNAs that were higher in hypothermia than normothermia (red) and five that were lower (green); P < 0.05.
Quantitative Reverse Transcriptase-PCR
To validate the microarray results, we conducted quantitative real time RT-PCR on six of the miRNAs that demonstrated the most dramatic changes after TBI, including: miR-874, miR-451, miR-34a, miR-27b, miR-9, and miR-497. After subtracting the Ct for the small nuclear RNA U82 as an endogenous control, the relative amounts of these miRNAs after TBI with and without hypothermia treatment as compared with sham-operated animals was determined.
Real-time RT-PCR of miR-874 confirmed the results of the micro array, in that miR-874 was significantly higher after TBI in the normothermia animals at 7hours (2.56 fold higher, P < 0.05). Although still slightly elevated in normothermia TBI animals at 24hours (1.25 fold), the increase was not significant. In contrast, hypothermia TBI animals showed no significant difference in miR-874 levels from sham-operated controls (Table 1).
Real-time RT-PCR for miR-497 and miR-27b tended to corroborate the array results, but did not reach significance (Table 1). The miR-497 was 220% higher in the TBI animals at 7hours and 20% lower at 24 hours compared with controls. Hypothermia animals had a lower increase than the normothermia animals; however, differences did not reach significance. The miR-27b, which has been shown to regulate the transcription factor Mef2C, was decreased in the arrays in all TBI animals. In the RT-PCR results, after 7hours, miR-27b was decreased in both normothermia and hypothermia TBI animals (0.97 and 0.88 fold, respectively) and in the 24hours animals, below 0.64 and 0.95 fold, respectively, with some individual animals as low as 0.45 fold.
The miR-451 was increased ∼2.84 fold at 7hours after TBI in normothermia animals, but appeared at sham levels in the hypothermia group. The miR-34a, which is highly expressed in endothelial cells, showed a decrease in TBI animals at 7hours after injury. At 24hours after TBI, miR-34a was increased by threefold in normothermia animals, but not in hypothermia animals. The miR-34a is known to induce apoptosis. Although most of these qPCR results did not reach significance, they did validate the findings of the arrays.
In Situ Hybridization
We next evaluated the regional and cellular localization of both miR-9 and miR-874 by performing in situ hybridization in injured cerebral cortices using LNA probes. The miR-9 was constitutively expressed in the sham-operated animals in a perinuclear pattern (Figure 4). There was an apparent increase in miR-9 expression after TBI, as shown by increased red staining. miR-874 staining was not detected in sham animals (Figure 5A and 5D). However, after TBI, there was a strong increase in the staining pattern within regions of the cerebral cortex adjacent to the contusion site (Figure 5B and 5E). Interestingly, this staining pattern was markedly decreased with hypothermia treatment (Figure 5C and 5F). The location and appearance of labeled cells were consistent with the size and distribution of cortical neurons.
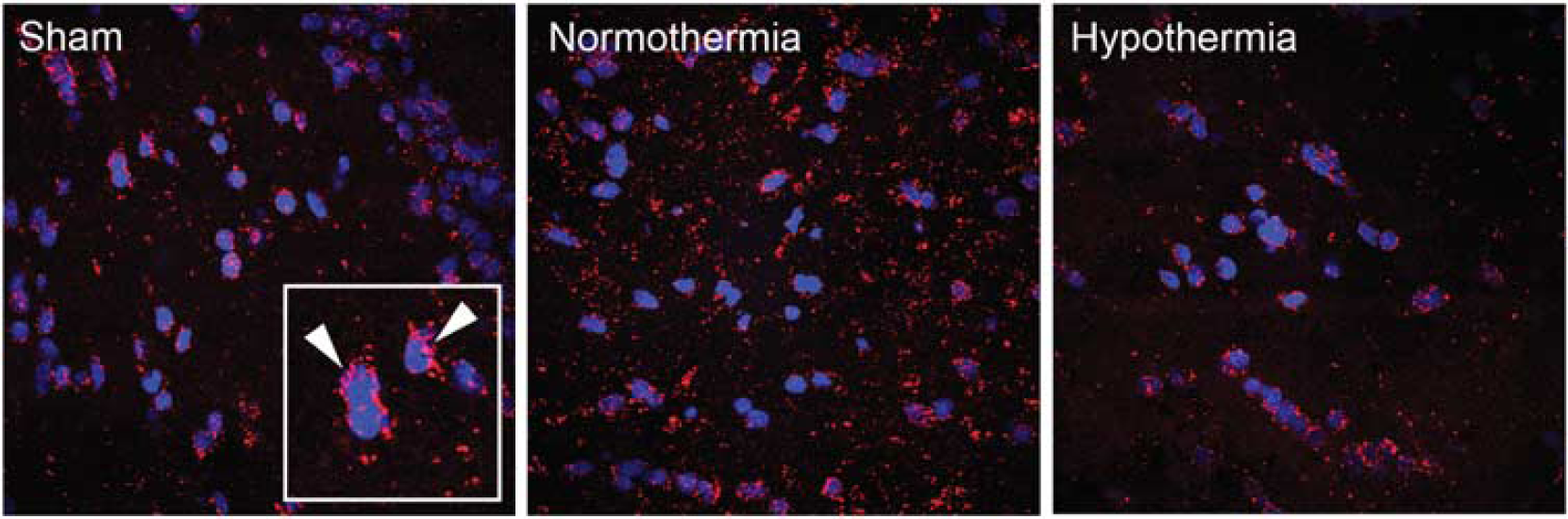
In situ hybridization of miR-9. Confocal image of ipsilateral cortex in sham, 7-hour traumatic brain injury (TBI) normothermia, and 7-hour TBI hypothermia show miR-9 constitutively expressed in sham-operated animals, with an apparent increase after TBI. Cy3-labeled miR-9 (red) is shown to localize in a perinuclear fashion (inset, arrowheads). Staining with 4',6-diamidino-2-phenylindole delineates the nuclei (blue). Magnification: × 60.
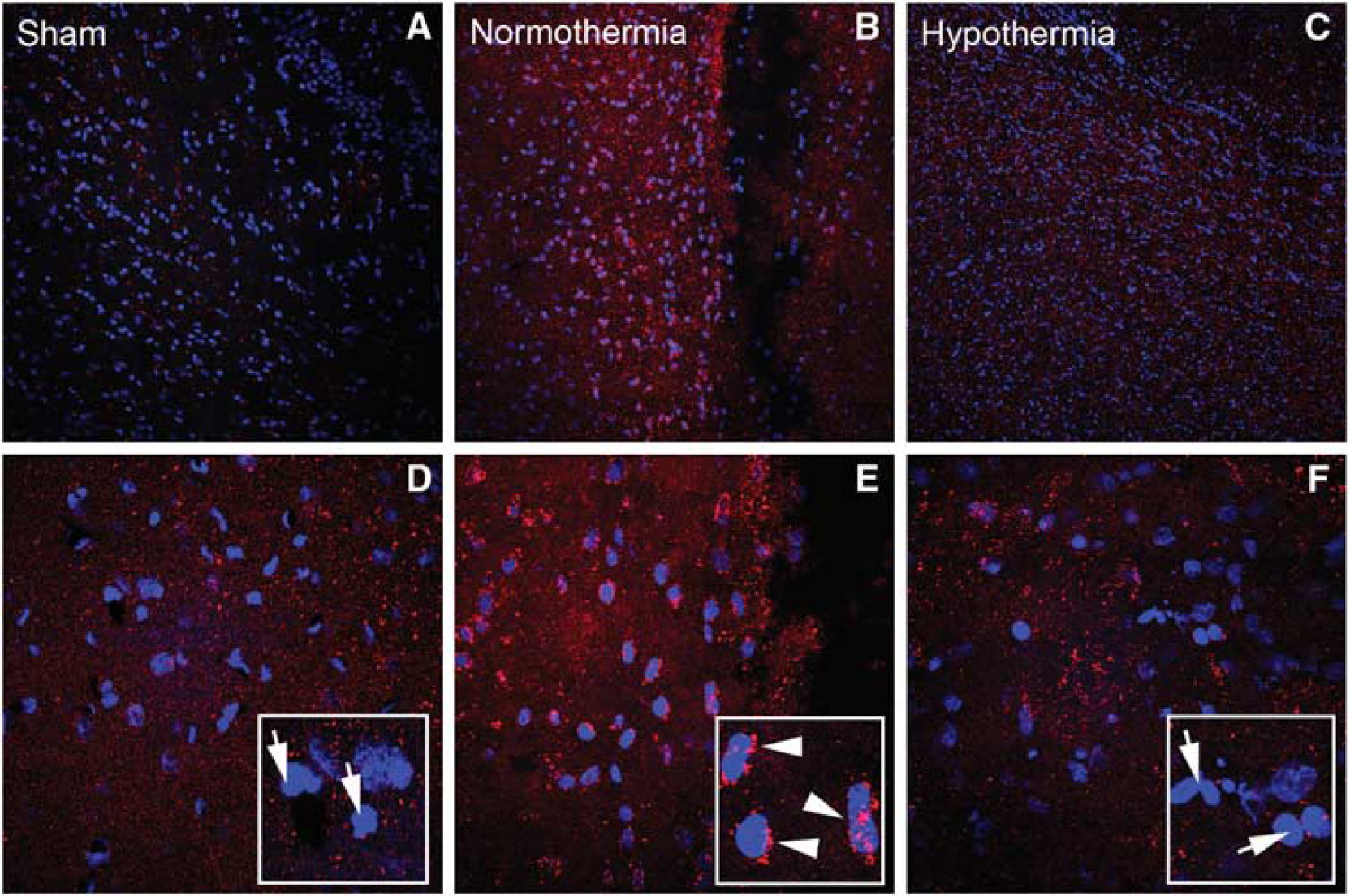
In situ hybridization of miR-874. Confocal images of ipsilateral cortex in sham (
Neuronal Stretch Injury Model
To determine whether we could measure a similar change in miR-874 expression in cultured neurons, we used a stretch-induced model of neuronal injury. Quantitative RT-PCR results on non-injured neurons under 3hours of normothermic or hypothermic conditions showed no difference between the sham groups. However, 3hours after stretch injury, miR-874 expression was increased in the normothermic cells (Figure 6). In contrast, injured cells treated with 3 hours of hypothermia showed no increase in miR-874 over sham levels.
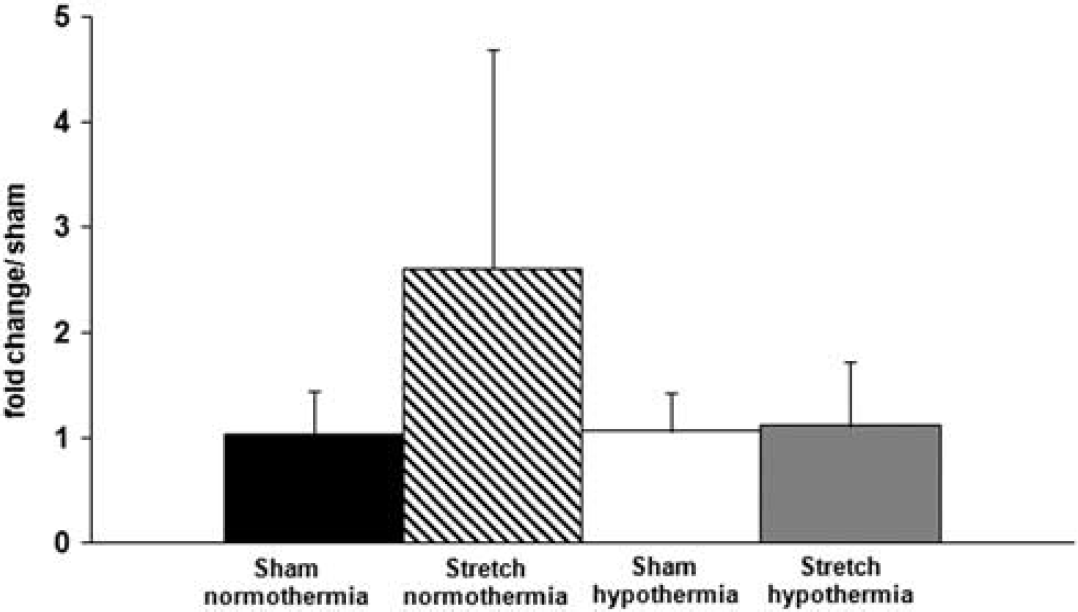
Quantitative reverse transcriptase-PCR of cortical neuron culture stretch-induced injury. Amounts of miR-874 relative to sham, uninjured, cells show an increase of 3.2 fold in normothermic-injured cells (diagonal stripes bar). Injured hypothermic neurons show no increase in miR-874 expression over sham levels (gray bar).
Discussion
The major findings of this study are that moderate TBI differentially alters the expression of specific miRNAs in the vulnerable cerebral cortex and that a therapy that has been reported to improve traumatic outcome significantly alters their regulation. We found specific miRNAs that were either up- or downregulated after TBI. In addition, some of these responses to trauma were significantly affected by posttraumatic hypothermia. Although most of the known functions of miRNAs are associated with cell differentiation, development, or oncogenesis, this is the first study to report temperature-sensitive changes in miRNAs after TBI with moderate hypothermia treatment. On the basis of the predicted gene targets with potential binding sites for the miRNAs that we found to be differentially expressed, it appears that relatively mild temperature modulations after trauma may influence essential cellular events, including protein and vesicle transport, as well as mitochondria and other organelle membrane functions.
As already discussed, effects of TBI are complex and involve complex pathological processes (Raghupathi, 2004; Stoica and Faden, 2010). Inflammation, neurodegeneration, apoptosis, synaptic plasticity, axonal damage, and other secondary injury mechanisms all contribute to the ongoing damage that occurs after TBI. Hypothermic treatment after TBI has been shown to attenuate some of these deleterious mechanisms and improve behavioral and histopathological outcomes (Dietrich et al, 2009).
Systematic and integrative analysis of possible gene targets as analyzed by DAVID (the database for annotation, visualization, and integrated discovery) bioinformatics resource (http://david.abcc.ncifcrf.gov; Dennis et al, 2003; Huang da et al, 2009) allows one to extract biological features/meaning associated with large gene lists. It systematically maps a large number of interesting genes in a list to the associated biological annotation (ontology terms) and then statistically highlights the most overrepresented biological annotation. In our study, several miRNAs were chosen to analyze based on whether they were the most highly enriched after TBI or whether they showed differences between normothermic and hypothermic groups. The miRNAs were analyzed using the miRDB search tool (http://mirdb.org/miRDB) and microrna.org (http://www.microrna.org/microrna) to identify possible gene targets of each miRNA. These gene lists were then loaded into DAVID for analysis.
The miR-874 yielded 102 possible gene targets in 3 functional categories made up of 24 clusters. Five clusters had an enrichment score of > 1.3, meaning they were statistically overrepresented in the sample. The genes represented in these clusters were involved in endomembranes, vesicle transport, Golgi and trans Golgi, membrane organization, endocytosis, mitochondrial and organelle membranes, and intracellular protein transport, to name a few (Supplementary Table 2).
The miR-290 had 313 possible gene targets that fell into 72 clusters. The most enriched cluster had 31 genes involved in zinc and other metal-binding annotations. Cluster 4 included genes that are associated with protein transport and intracellular localization (Supplementary Table 3). The miR-497, which was 2.4 fold higher in normothermia TBI, had 398 possible gene targets. These fell into 127 clusters, with 16 having significant enrichment. The most significant cluster included genes associated with protein localization and intracellular protein transport (cluster 1). The most highly significant genes are listed in Supplementary Table 4. Cluster 2 involved nuclear and organelle lumen proteins. Clusters 3 and 4 were related to axonal projection and neuronal development and migration. Cluster 5 was involved in brain development.
The miR-27b, which decreased after TBI, had 367 potential targets in 96 clusters. The most highly significant clusters involved genes associated with regulation of transcription and amino-acid metabolic process (cluster 1), neuron differentiation and development (cluster 2), and macromolecular complex assembly (cluster 3). The miR-451 had 20 potential gene targets, but none of them are related functionally. The miR-34a had 164 target genes in two clusters. Cluster 1 included E2F transcription factor genes, and cluster 2 was zinc finger proteins. The miR-9, which went up slightly after TBI at 7hours, had 260 target genes in 68 clusters. The most significant were (1) actin-binding proteins, (2) basal plasma membrane, and (3) negative regulators of transcription.
In our screen of the miRNA transcriptome, we found that at 7hours, there were 16 miRNAs that significantly increased and 31 that decreased. Two other studies in TBI found similar findings. Redell et al (2009) reported that 35 miRNAs increased and 50 decreased after controlled cortical impact in the hippocampus. Lei et al (2009) using a FP model, reported that 13 miRNAs increased and 14 decreased. Our current findings along with the two published TBI studies have reported that more miRNAs are downregulated after injury than upregulated. In the FP study by Lei and colleagues (2009), miR-21 was found to increase from 1.5 to 2 fold. In our study, this miRNA also was seen to go up by ∼2 fold at 7hours after TBI. Finally, Wang et al (2010) reported a decrease in miR-107 after controlled cortical impact injury in the hippocampus. This miRNA is associated with wound repair and regulates granulin. We detected no change in levels of miR-107 after TBI.
In a screen of hippocampus miRNAs after controlled cortical impact (Redell et al, 2009), several miRNAs that were shown to increase after injury, miR-223 and miR-451, were also seen to increase in our study. However, others such as miR-107, miR-130a, miR-433-3p, and miR-541 remained unchanged in our experimental model (Redell et al, 2009). Two of the most highly induced miRNAs in our study, miR-874 and miR-290, were not reported in the other TBI studies, although miR-290 has been seen to be increased after experimental brain ischemia (Madathil et al, 2010). It is important to note that the cellular and molecular response to brain injury can vary greatly depending on the type of injury. It is common to find different histological, biochemical, or molecular changes as a result of cerebral ischemia, FP, or cortical impact injury. Interestingly, in a study of plasma miRNAs in severely injured TBI patients, several miRNAs detected were also seen in our rat model of TBI. The miR-16 and miR-451 were upregulated in both human plasma and rat cortex. Importantly, the miRNAs that showed the most increase in human plasma; miR-574-3p, miR-574-5p, and miR-1246 are not yet identified in the rat genome, as miR-874 is not found in the human genome.
The changes measured on the microarrays of some of the miRNAs that were affected the most by post-TBI hypothermia were verified by qRT-PCR. The miRs-874, −451, −497, and −9 were all seen to increase after injury when measured by qPCR. Additionally, miR-34a and miR-27b both decreased, as was shown by the arrays as well. Localization of miR-9 and miR-874 to perinuclear locations in injured cerebral cortex cells by in situ hybridization confirmed the cytoplasmic nature of miRNA-mediated posttranscriptional activity. As expected, these miRNAs are transported out of the nucleus and into the cytoplasm where they can associate with the RNA-induced silencing complex and block protein translation. In our cell culture studies where neurons were damaged by a stretch injury model, elevations in miR-874 were observed under normothermic but not under hypothermic conditions. In contrast, no differences in baseline expression were observed between normothermic and hypothermic sham-treated neurons. These data emphasize the potential of temperature modifications after TBI, influencing miRNA expression in neurons.
An important finding of the present study was that some of the miRNAs that were increased by TBI were attenuated by hypothermia. Previous studies have shown miRNAs to be sensitive to severe temperature changes in both plants and animals (Zhou et al, 2008; Liu et al, 2010b). However, these studies only investigated the normal functioning of miRNAs in response to environmental factors including profound temperature fluctuations. In our study of TBI, miR-874 was highly upregulated after TBI under normothermic conditions but the levels were lower in the hypothermic animals. This miRNA is predicted to target genes involved in intracellular transport of proteins and vesicles, potassium-chloride transporters, and membrane organization. Upregulation of this miRNA would be predicted to lead to the decreased production of several key proteins involved in normal cellular function and influence cell vulnerability after TBI. Thus, if moderate levels of hypothermia decreased this negative effect on a critical cell function, then a vulnerable cell could be protected from irreversible injury. Other miRNAs investigated in this study that increased after TBI and target genes involved in protein localization and intracellular transport include miR-497 and miR-290.
The miR-9, which negatively regulates transcription and downregulates proteins associated with actin-binding proteins and basal plasma membranes, was increased after TBI. This response could potentially cause damage to the cytoskeleton and cellular integrity. The levels of miR-9 in the hypothermia animals were similarly decreased compared with normothermia animals. The miR-27b, which decreased after TBI, is a positive regulator of nucleic-acid metabolic process and macromolecule complex assembly. This decrease in miR-27b seen after TBI may likewise be contributing to the disruption of normal cell function. In our study, the majority of miRNA expression changes were observed at the early postinjury time point. By 24hours, differences in miRNA expression patterns between normothermic and hypothermic TBI rats were not apparent. This transient nature of the temperature sensitivity of miRNA expression is consistent with previous studies where early postinjury alterations in mRNA levels are reported. It is possible that longer cooling and/or survival periods after TBI may demonstrate different patterns of miRNA expression than reported in the present study.
In summary, our results indicate a significant role for miRNAs in the pathophysiology of TBI. Because TBI regulates whole classes of genes vital to cellular function and health, these molecular responses to trauma make a strong potential target for therapy. The fact that therapeutic hypothermia modifies specific miRNA responses to trauma indicates that this mechanism may underlie some of the benefits of hypothermia on traumatic outcome. By knocking down or overexpressing specific miRNAs with hypothermia or alternate strategies, downstream injurious cascades may be eliminated or reduced to promote protection of vulnerable cells.
Footnotes
Acknowledgements
The authors thank George Lotocki for assistance in graphic artwork design, and Doris Nonner and Juan Pablo de Rivera Vaccari for assistance in the neuronal stretch injury.
The authors declare no conflict of interest.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
