Abstract
Oxidative stress has been implicated in the cell death that occurs after ischemia–reperfusion of the brain, which causes the production of reactive oxygen species and a decrease in antioxidants, leading to mitochondrial dysfunction. However, the invasive methods used to collect much of this evidence are themselves stress inducing, which could skew the results. In this study, we aimed at demonstrating brain redox alterations after ischemia–reperfusion noninvasively, using Overhauser-enhanced magnetic resonance imaging. The reduction rate of 3-methoxycarbonyl-2,2,5,5-tetramethylpyrrolidine-
Keywords
Introduction
Oxidative stress has been implicated in the cell death that occurs after ischemia–reperfusion injury of the brain. The mitochondrial electron transport chain is one of the sources of the reactive oxygen species (ROS) that are generated after ischemia–reperfusion. These ROS, in turn, are believed to contribute to mitochondrial dysfunction. In the rat, transient middle cerebral artery occlusion (MCAO) induces ROS production and mitochondrial dysfunction, including the inactivity of electron transport chain enzymes. The mitochondrial dysfunction is attenuated by treatment with an antioxidant (Kuroda et al, 1996) or by the overexpression of mitochondrial Hsp70/Hsp75 (Xu et al, 2009), both of which decrease the ROS concentration.
Under normal physiologic conditions, oxidative damage is prevented by the regulation of ROS by the antioxidant network. However, ischemia–reperfusion induces ROS production, resulting in a decrease in antioxidants, such as ascorbic acid and reduced glutathione (GSH). This effect occurs in the rat striatum, in which the dialysate level of ascorbic acid is decreased after 4 h of global ischemia (Zhang et al, 2005). In addition, GSH breaks down to cysteine in the brain tissue after ischemia (Slivka and Cohen, 1993). This process may exacerbate the injury, particularly because cysteine is a more powerful inducer of neuronal cell death than even glutamate, a known excitotoxin (Olney et al, 1990). These findings suggest that the balance of oxidation versus reduction capacity, i.e., the redox balance, is lost by ischemia–reperfusion. However, although ischemia–reperfusion leads to oxidative stress when measured using invasive methods as mentioned above, there is little noninvasive experimental evidence for the increase in redox alterations in the brain after such an injury. The invasive methods used to collect much of this evidence are themselves stress inducing, which could skew the results.
Powerful imaging techniques for determining the redox status in vivo are available. The development of electron spin resonance (ESR) imaging (Lee et al, 2004; Sano et al, 2000; Sato-Akaba et al, 2008; Yokoyama et al, 1999), Overhauser-enhanced magnetic resonance imaging (OMRI) (Li et al, 2006; Liebgott et al, 2003; Utsumi et al, 2006), and MRI (Hyodo et al, 2008; Matsumoto et al, 2006) has permitted the noninvasive study of redox status in living animals. Especially, OMRI is a double-resonance technique that uses the presence of paramagnetic agents to enhance the signal intensity from nuclear spins by a process known as dynamic nuclear polarization, or the Overhauser effect (Krishna et al, 2002; Li et al, 2006; Lurie et al, 1988; Utsumi et al, 2006). Recent studies have successfully expanded this technique to obtain functional information, including noninvasive pO2 maps (Krishna et al, 2002) and simultaneous images of different redox reactions (Utsumi et al, 2006). These methods use nitroxyl radicals (i.e., nitroxides) as a redox-sensitive contrast agent for brain imaging. For example, nitroxyl radicals are directly reduced to their corresponding hydroxylamine by ascorbic acid; hence, their reduction indicates the antioxidant activity (Mehlhorn, 1991). Glutathione also affects the reduction of nitroxyl radicals indirectly (Bobko et al, 2007), through their interaction between ascorbic acid and GSH as a redox couple (Mehlhorn, 1991; Meister, 1994; Winkler et al, 1994). In addition, nitroxyl radicals can be reduced enzymatically by complexes I and II in the electron transport chain of the mitochondria (Chen et al, 1988; Quintanilha and Packer, 1977). In this study, we used the nitroxyl radicals to investigate whether the level of antioxidants or the activity of redox enzymes is altered in the brain after ischemia–reperfusion.
One nitroxyl radical, 3-methoxycarbonyl-2,2,5,5-tetramethylpyrrolidine-
Materials and methods
Chemicals
3-Carboxy-2,2,5,5-tetramethylpyrrolidine-
Methoxycarbonyl-PROXYL was synthesized from carboxy-PROXYL as previously described (Sano et al, 1997). Isotonic solutions containing methoxycarbonyl-PROXYL at a final concentration of 130 mmol/L were prepared by mixing the probe with 75 mmol/L of NaCl, sterilized by filtration (0.2 μm), and stored at −20°C before use.
Animals
Male Wistar rats (6 weeks of age) were purchased from Kyudou (Saga, Japan). The rats were housed in a temperature- and humidity-controlled room, and fed a commercial diet (MF; Oriental Yeast) and water ad libitum. Reversible focal cerebral ischemia was induced with an intraluminal suture to produce a model of MCAO. In brief, anesthesia was induced with 2% isoflurane (Dainippon Pharmaceutical, Osaka, Japan) in the air, and maintained with 1% isoflurane using a facemask. A 1-cm midline incision was made on the anterior neck, and the right common carotid artery, external carotid artery, and internal carotid artery were exposed. The common carotid artery and external carotid artery were ligated, and a suture was placed around the internal carotid artery for ligation. An embolus was made by inserting a 4-0 nylon surgical thread into the internal carotid artery through a small incision. The MCA was occluded by advancing the embolus into the internal carotid artery to block the origin of the MCA. After 1 h of MCAO, the MCA was reperfused by withdrawing the embolus. The rectal temperature was maintained by placing the rat on a heating pad under anesthesia. The blood pH, pO2, and pCO2 levels, which were analyzed using GASTAT-mini (Techno Medica, Yokohama, Japan), were found to be within the normal range (Table 1).
Arterial blood gases after transient MCAO
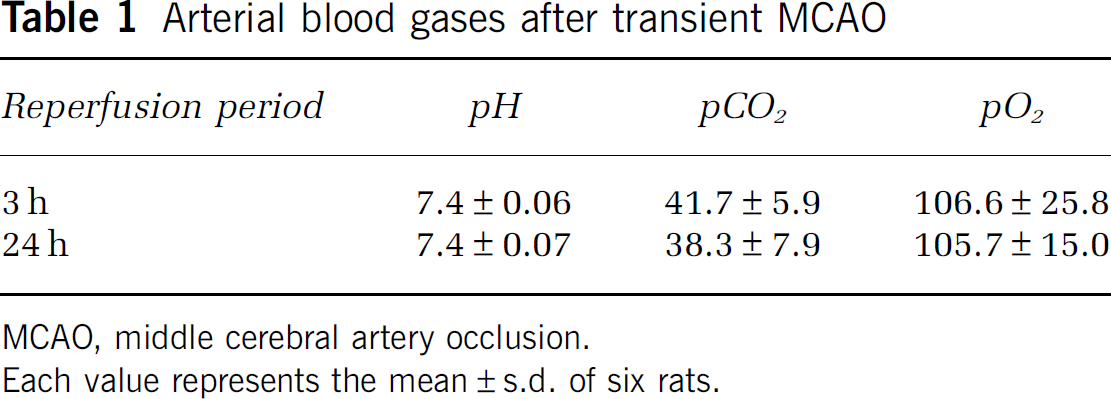
MCAO, middle cerebral artery occlusion.
Each value represents the mean±s.d. of six rats.
All the procedures and animal care were approved by the Animal Care and Use Committee, Kyushu University, and carried out in accordance with the Guidelines for Animal Experiments, Kyushu University.
Phantom Imaging
Overhauser-enhanced magnetic resonance imaging experiments were performed on a custom-built OMRI scanner (POEM; Philips Research Laboratories, Hamburg, Germany) with a surface-coil-type receiver coil for ESR irradiation. The resonant circuit consisted of a single-turn loop coil, a parallel coaxial line formed by 50-ohm coaxial cables, a half-wave line balun, and trimmer capacitors for matching and tuning, as reported previously (Matsumoto et al, 2007). The operating frequency of the surface coil was designed to have an ESR irradiation frequency of 222.6 MHz for OMRI. The OMRI images were obtained using the following conditions: FOV (field of view), 32 × 32 mm; matrix, 64 × 64; slice thickness, 20 mm; TR/TE/TESR (repetition time/echo time/the length of each period of ESR irradiation), 1,200/25/700 msecs. To validate the ability of the imaging method to provide spatially resolved redox status information, a phantom consisting of identical tubes containing methoxycarbonyl-PROXYL (0.5 mmol/L) was used. Methoxycarbonyl-PROXYL in the individual tubes was time dependently reduced by ascorbic acid or mitochondrial enzyme, as described using the terms and conditions given in the section ‘Kinetics of methoxycarbonyl-PROXYL reduction in the cytosolic or mitochondrial fraction,’ below.
Brain Imaging After Transient Middle Cerebral Artery Occlusion
The femoral vein was cannulated for the injection of methoxycarbonyl-PROXYL after the induction of anesthesia by pentobarbital (Dainabot, Tokyo, Japan). The rat was then placed in the resonator. Immediately after the administration of methoxycarbonyl-PROXYL (1.3 mmol/kg body weight), OMRI images were obtained (FOV, 32 × 32 mm; matrix, 32 × 32; slice thickness, 30 mm; TR/TE/TESR, 1,200/25/700 msecs). Conventional MRI experiments were performed on a 0.4 T MRI (Hitachi Medical Corporation, Tokyo, Japan) using a standard proton density-weighted spin echo sequence (TR/TE, 2,500/25 msecs). The OMRI images were superimposed on the MRI image by aligning the positions of markers (two capillaries containing methoxycarbonyl-PROXYL).
Semi-logarithmic plots of the time course of the OMRI signal change in the region of interest were used for calculating the reduction rate. A region of interest in each cerebral hemisphere was selected for this determination. The region of interest analysis was carried out using MATLAB (The MathWorks, MA, USA).
Quantification of the Methoxycarbonyl-PROXYL Level in the Brain
To examine the distribution of methoxycarbonyl-PROXYL in the brain, methoxycarbonyl-PROXYL was injected into the tail vein of rats at a dose of 1.3 mmol/kg body weight. Five minutes later, the rats were killed, their brains were removed, and brain samples were homogenized in a three-fold volume of saline. The homogenate solution was mixed with a ferricyanide solution (1 mmol/L final concentration). Ferricyanide quantitatively oxidizes hydroxylamine, which is produced by an in vivo reduction to its oxidized form (Krishna et al, 1992). X-band ESR spectra were recorded at room temperature using a JEOL JES-1X ESR spectrometer (microwave power: 5 mW; amplitude of 100-kHz field modulation: 0.063 mT; time constant: 0.03 secs; sweep rate: 5 mT/min), and were analyzed using an ESR Data Analyzer (JEOL, Akishima, Japan).
Kinetics of Methoxycarbonyl-PROXYL Reduction in the Cytosolic or Mitochondrial Fraction
The reduction of methoxycarbonyl-PROXYL was shown in the cytosolic or mitochondrial fraction obtained from the brain. The ESR spectra of methoxycarbonyl-PROXYL (10 μmol/L) were recorded in the cytosolic fraction without a substrate. The mitochondrial fractions were reacted with methoxycarbonyl-PROXYL (10 μmol/L) and a substrate as follows. Glutamate/malate (5 mmol/L) was used as a substrate for complex I, and succinic acid (20 mmol/L) was used for complex II. Antimycin A (0.05 mg/mL) was used as an inhibitor for complex III, and KCN (1 mmol/L) was used for complex IV. Immediately after starting the reaction, the ESR spectra were recorded.
Activity of Mitochondrial Electron Transport Chain Enzymes
Brain samples were homogenized on ice in seven volumes of buffer consisting of 10 mmol/L of HEPES (4-(2-hydroxyethyl)-1-piperazineethanesulfonic acid)–NaOH (pH 7.4) and 250 mmol/L of sucrose, and the suspensions were spun at 800 g at 4°C for 10 mins. The supernatants were then spun at 13,000 g at 4°C for 10 mins, and the pellets were washed with mitochondrial isolation buffer and spun again at 13,000 g at 4°C for 10 mins, to obtain the mitochondrial fraction.
The specific activity of the mitochondrial complex enzymes, including complexes I, II, III, and IV, was measured in the mitochondria isolated from the whole brains of each group, as previously described (Trounce et al, 1996). The specific activity of the rotenone-sensitive Nicotinamide adenine dinucleotide–ubiquinone oxidoreductase (complex I) was measured by the reduction in the ubiquinone analog, decylubiquinone. For the activity of succinate ubiquinone oxidoreductase (complex II), the reduction of 2,6-dichlorophenolindophenol when coupled to the complex II-catalyzed reduction in decylubiquinone was measured. For the specific activity of ubiquinol/cytochrome c oxidoreductase (complex III), the reduction of cytochrome c catalyzed by complex III in the presence of reduced decylubiquinone was monitored. The specific activity of cytochrome c oxidase (complex IV) was measured by after the oxidation of reduced cytochrome c, which was prepared in the presence of dithionite. The enzymatic activities were expressed in units per gram of tissue.
High Performance Liquid Chromatography Analysis of Ascorbic acid, Glutathione, and Cysteine
Brain tissue samples were homogenized in 5% metaphosphoric acid. After centrifugation at 14,000 g for 15 mins, the supernatant was filtered (0.45-μm filter, Minisart RC4; Sartorius Stedim Biotec GmbH, Goettingen, Germany) and analyzed for ascorbic acid, GSH, and cysteine by high performance liquid chromatography (HPLC) with electrochemical detection. Separation was achieved with a C18 reverse-phase column (MCM column 150 × 4.6 mm, MC Medical, Tokyo Japan). For the measurement of ascorbic acid, the mobile phase was 0.1 mol/L KH2PO4 buffer (pH 2.35). The HPLC flow rate was set at 0.5 mL/min. Electrochemical detection was performed using a Coulochem III detector (ESA Laboratories, MA, USA) equipped with a guard cell (M5020) and an analytical cell (M5011) set. The guard cell was set at +250 mV, electrode 1 at −50 mV, and electrode 2 at +200 mV. For the measurement of GSH and cysteine, the mobile phase consisted of 25 mmol/L sodium phosphate and 0.2 mg/mL sodium octyl sulfate in 2% HPLC grade acetonitrile, adjusted to pH 2.7 with phosphoric acid. The HPLC flow rate was set at 0.7 mL/min. The guard cell was set at +650 mV, electrode 1 at +100 mV, and electrode 2 at +600 mV.
Statistical Analysis
All the results are shown as the mean±s.d. Statistical significance was analyzed using the two-tailed t-test. A probability value of 0.05 was set as the minimum level of statistical significance.
Results
Figure 1A shows three tubes, containing the methoxycarbonyl-PROXYL radical solution (paramagnetic, enhanced), its hydroxylamine (diamagnetic, not enhanced), and water. The image obtained without ESR irradiation (i.e., the native 15 mT MR image) had a low signal–noise ratio. Electron spin resonance irradiation preceded each phase-encoding step of the MRI data acquisition, resulting in an increased nuclear magnetic resonance signal intensity of the tube containing methoxycarbonyl-PROXYL, but not hydroxylamine or water. Subtraction of the image obtained without ESR from that with ESR irradiation yielded a difference image that showed the pure distribution of the free radical (Figure 1A). The addition of ascorbic acid or mitochondrial enzyme reduced the nitroxyl radical to its corresponding hydroxylamine (Figure 1B). Thus, this technique should enable the visualization of these reduction reactions.
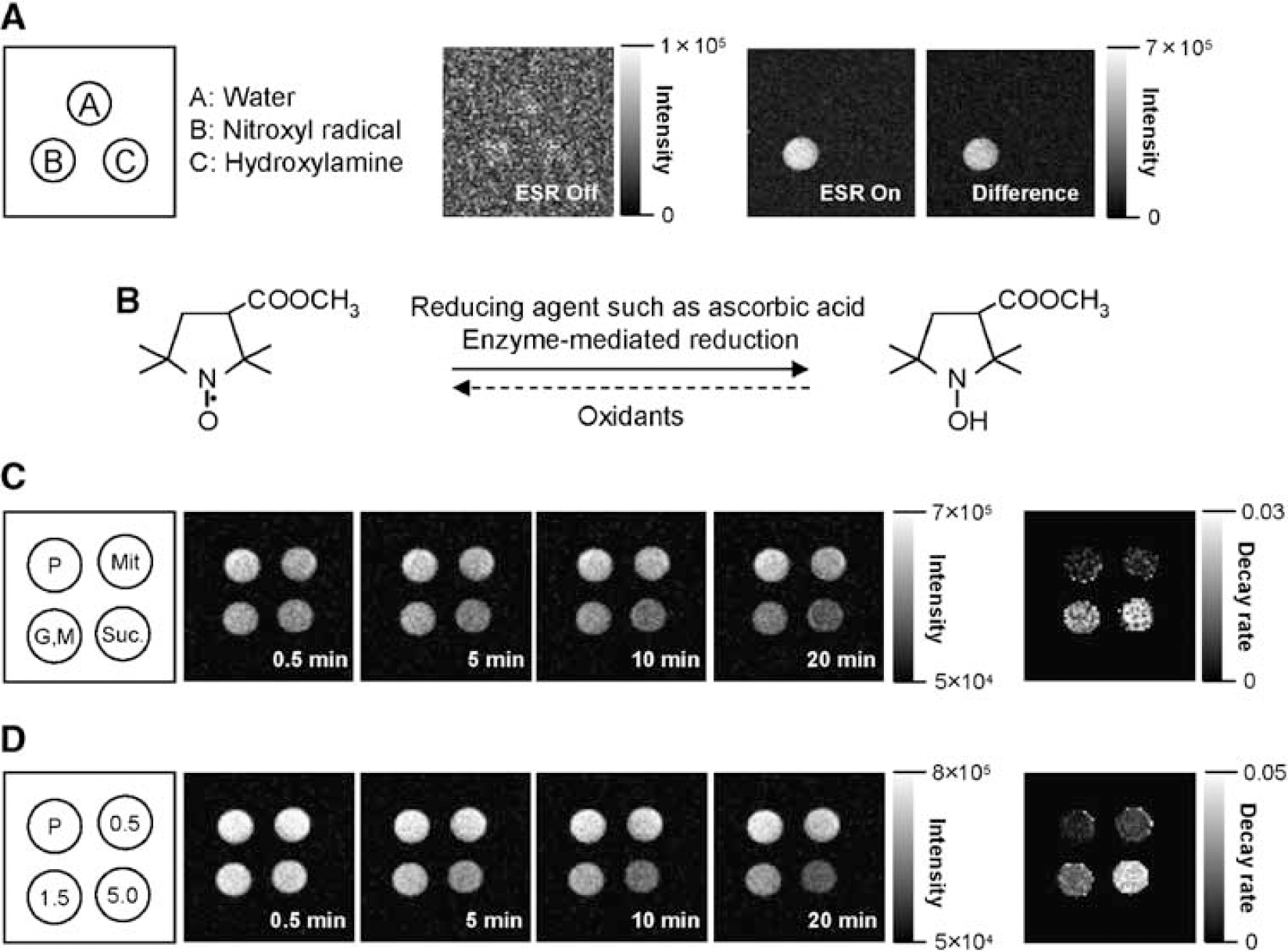
OMRI images of methoxycarbonyl-PROXYL phantoms. (
To assess the ability of our system to monitor redox status, we evaluated two redox phantoms, consisting of methoxycarbonyl-PROXYL and ascorbic acid or mitochondrial samples. Overhauser-enhanced magnetic resonance imaging scans were carried out as a function of time after the addition of methoxycarbonyl-PROXYL, as shown in Figures 1C and 1D. The image intensity of methoxycarbonyl-PROXYL in the mitochondrial fraction decreased gradually by mixing the sample with glutamate/malate and succinic acid, as substrates for complexes I and II, respectively. Figure 1C also shows the image for the rate constants in the mitochondrial fractions, which were calculated by assuming first-order kinetics for the time-dependent decrease in contrast. In the sample with endogenous mitochondria alone, the image intensity barely declined during the time of the experiment (Figure 1C). After mixing methoxycarbonyl-PROXYL with various concentrations of ascorbic acid, the image intensity decreased gradually (Figure 1D). The rate constant image showed clearly that ascorbic acid had a dose-dependent effect.
To identify the redox status of the brain, the reduction of methoxycarbonyl-PROXYL was analyzed in live rats by OMRI. Figure 2 shows the OMRI data of the head region after 3 h of reperfusion after MCAO. For the in vivo experiments, two fiducial markers that contained methoxycarbonyl-PROXYL solution were placed to permit the superimposition of the image with that obtained using conventional proton-based MRI collected at 0.4 T. The anatomic images obtained using the proton density-weighted sequence at 0.4 T are shown in gray scale in Figure 2A. After the injection of methoxycarbonyl-PROXYL solution, the intensity in the rat's head region increased immediately (red area). The increased imaging intensity in the brain tissue was large compared with that in the facial muscle. The distribution of methoxycarbonyl-PROXYL in the left face was low, because the external carotid artery was ligated in this animal model (Figure 2A). The imaging intensity of methoxycarbonyl-PROXYL in the brain decreased gradually with time after its intravenous injection. Figure 2A also shows the image for the decay rates obtained from the methoxycarbonyl-PROXYL-enhanced OMRI images. The estimation and display of the rate of image-intensity loss from each pixel yields a parametric image of the rate of intensity loss. A similar decline in the decay rate of methoxycarbonyl-PROXYL was observed in the semi-logarithmic plot for the contralateral (open circles) and ischemic (closed circles) hemispheres of the rat brain after 3 h of reperfusion after MCAO (Figures 2B and 2C). The distribution of methoxycarbonyl-PROXYL in the brain was also investigated by determining the probe recovery in the brain. To determine the total concentration of methoxycarbonyl-PROXYL, which consisted of the nitroxyl radical and the corresponding hydroxylamine, potassium ferricyanide was added to the sample in order to oxidize the hydroxylamine to the nitroxyl radical. Figure 2D shows the concentration of methoxycarbonyl-PROXYL (nitroxyl radical and hydroxylamine form) in the brain. The total amount of methoxycarbonyl-PROXYL recovered from each hemisphere was the same.
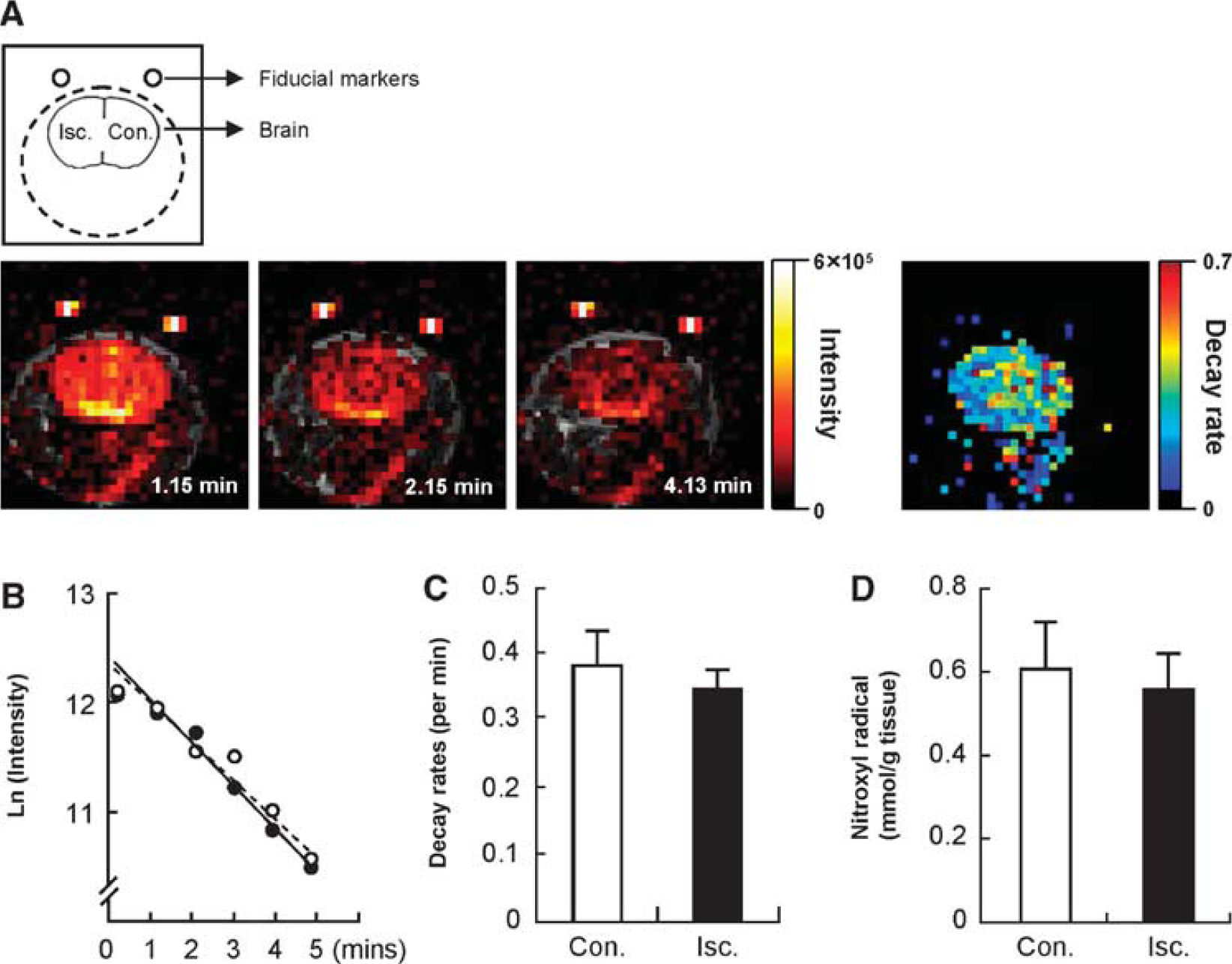
Signal decay rate of methoxycarbonyl-PROXYL in the brain after 3 h of reperfusion following 1 h of MCAO. (
The OMRI results of the rats after 24 h of reperfusion are shown in Figure 3. The increased imaging intensity in the brain tissue was similar to the level observed 3 h after reperfusion. However, at 24 h, the decrease in imaging intensity in the ischemic hemisphere was significantly slower than in the contralateral hemisphere. Figure 3A shows the image for the decay rate constants of the image intensities. In the decay rate images, the relatively rapid decay rates in the contralateral hemisphere are represented by aqua (0.255/min), yellow (0.423/min), or red dots (0.603/min), whereas the slower decay rates in the ischemic hemisphere are blue (0.109/min), iron blue (0.032/min), or black (0/min). The values in parenthesis are the signal decay rate. The signal decay rate calculated from continuous images in the ischemic hemisphere was significantly decreased compared with that in the contralateral hemisphere (Figures 3B and 3C). However, the distribution of methoxycarbonyl-PROXYL in the ischemic hemisphere after 24 h of reperfusion was the same as that to the contralateral hemisphere (Figure 3D). These results indicate that the difference of observed reduction was predominantly because of the conversion from methoxycarbonyl-PROXYL to its hydroxylamine form, rather than to the clearance of methoxycarbonyl-PROXYL from the brain tissue.
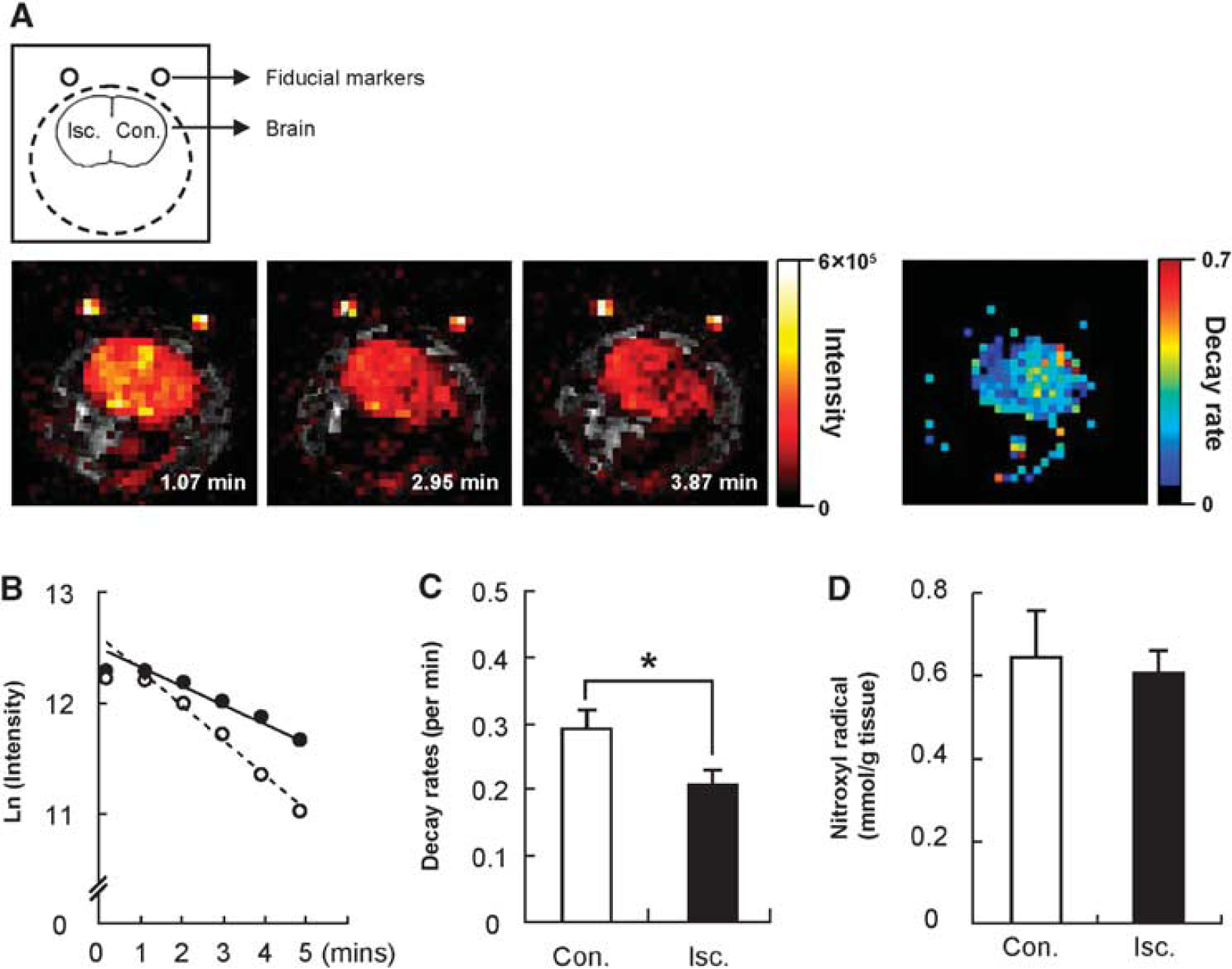
Signal decay rate of methoxycarbonyl-PROXYL in the brain after 24 h of reperfusion following 1 h of MCAO. (
To identify the mechanisms responsible for the reduction of methoxycarbonyl-PROXYL in the brain redox image, the signal decay rates in cellular fractions obtained from the brain were estimated by X-band ESR (Table 2) and calculated by assuming first-order kinetics for the time-dependent decrease. To examine its reduction at the mitochondria, methoxycarbonyl-PROXYL was mixed with the mitochondrial fraction. Two segments of the electron transport chain are primarily responsible for the reduction of nitroxyl radicals, complex I and complex II (Chen et al, 1988; Quintanilha and Packer, 1977). Only a small signal decay appeared when glutamate/malate was used, as a substrate for complex I, in both the hemispheres, whereas the addition of succinic acid, as a substrate for complex II, resulted in the clear decay of the methoxycarbonyl-PROXYL ESR signal. The signal decay rates were the same between the hemispheres after 3 h of reperfusion. Antimycin A and KCN are well-known inhibitors of electron transport in the respiratory chain of the mitochondria. Neither Antimycin A nor KCN inhibited the methoxycarbonyl-PROXYL reduction in the mitochondria with succinic acid as a substrate. The signal decay rates in the cytosolic fraction of the contralateral and ischemic hemispheres were also comparable at 3 h of reperfusion after MCAO.
Reduction rates of methoxycarbonyl-PROXYL in cell fractions (× 10−3/min)
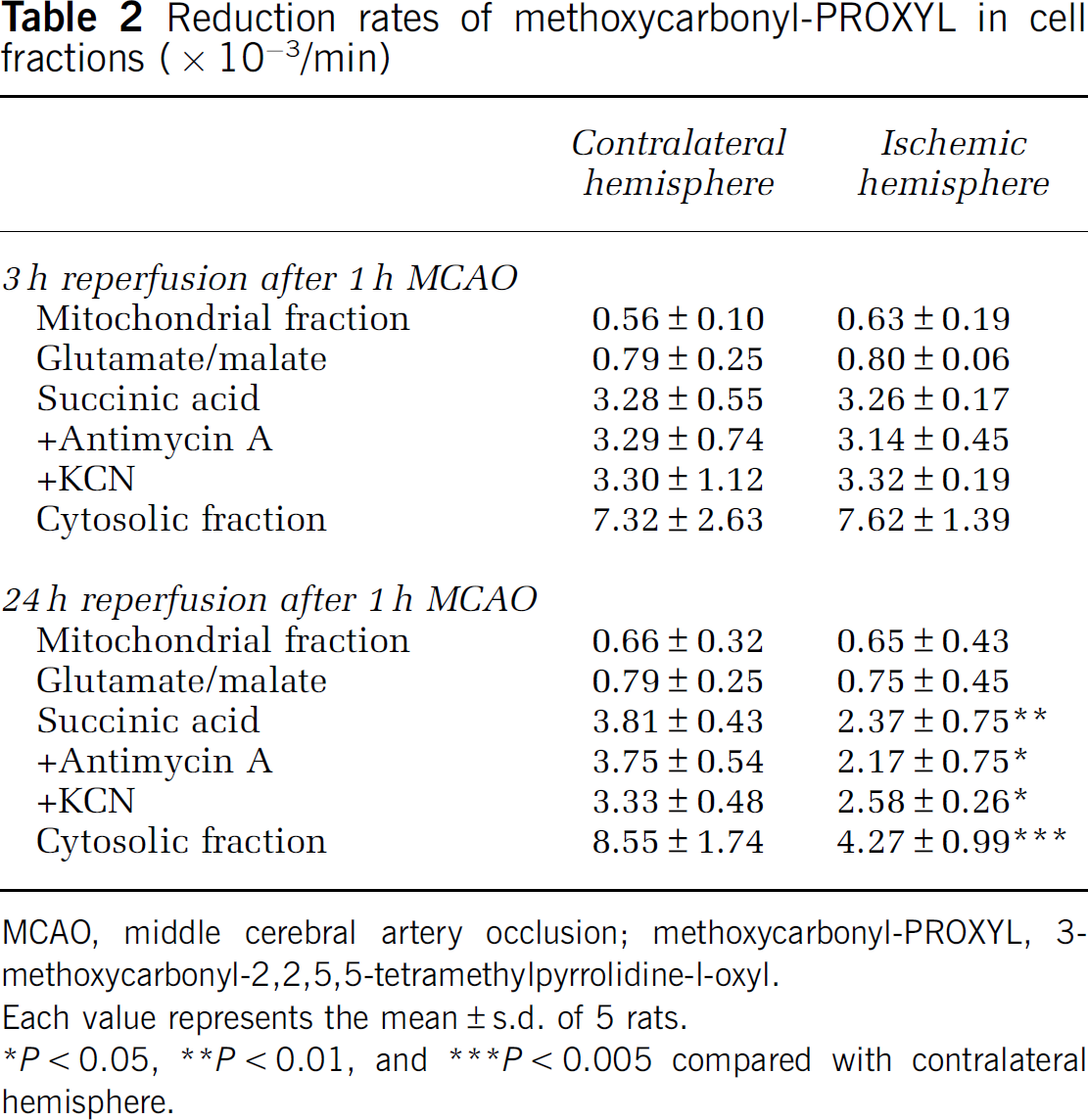
MCAO, middle cerebral artery occlusion; methoxycarbonyl-PROXYL, 3-methoxycarbonyl-2,2,5,5-tetramethylpyrrolidine-l-oxyl.
Each value represents the mean±s.d. of 5 rats.
∗P<0.05, ∗∗P<0.01, and ∗∗∗P<0.005 compared with contralateral hemisphere.
In the group examined after 24 h of reperfusion after MCAO, the reduction rate of methoxycarbonyl-PROXYL in the mitochondria obtained from the ischemic hemisphere was decreased with succinic acid as the substrate. These signal decay rates were not affected by the addition of Antimycin A or KCN. The ESR signal of methoxycarbonyl-PROXYL in the cytosolic fraction of the ischemic hemisphere also decayed more slowly than that of the contralateral hemisphere. These results suggested that the redox status determined from the brain image might reflect the reduction of methoxycarbonyl-PROXYL in the cytosolic fraction and the electron transfer function at complex II.
As methoxycarbonyl-PROXYL is believed to be reduced by mitochondrial enzymes or ascorbic acid, we measured the activity of the mitochondrial complex enzymes and the antioxidant levels in the brain tissue after transient MCAO. Figure 4 shows the activity of complexes I and II in the contralateral and ischemic hemispheres of the brain 3 and 24 h after transient MCAO. The activity of complex II in the ischemic hemisphere was clearly decreased after 24 h of reperfusion (Figure 4B), whereas no discernible change was observed after 3 h of reperfusion (Figure 4A). The activity of complex I in the ischemic hemisphere was not significantly different from that of the contralateral hemisphere at both reperfusion periods. The activities of complexes III and IV were also unchanged at both of the reperfusion periods (data not shown).
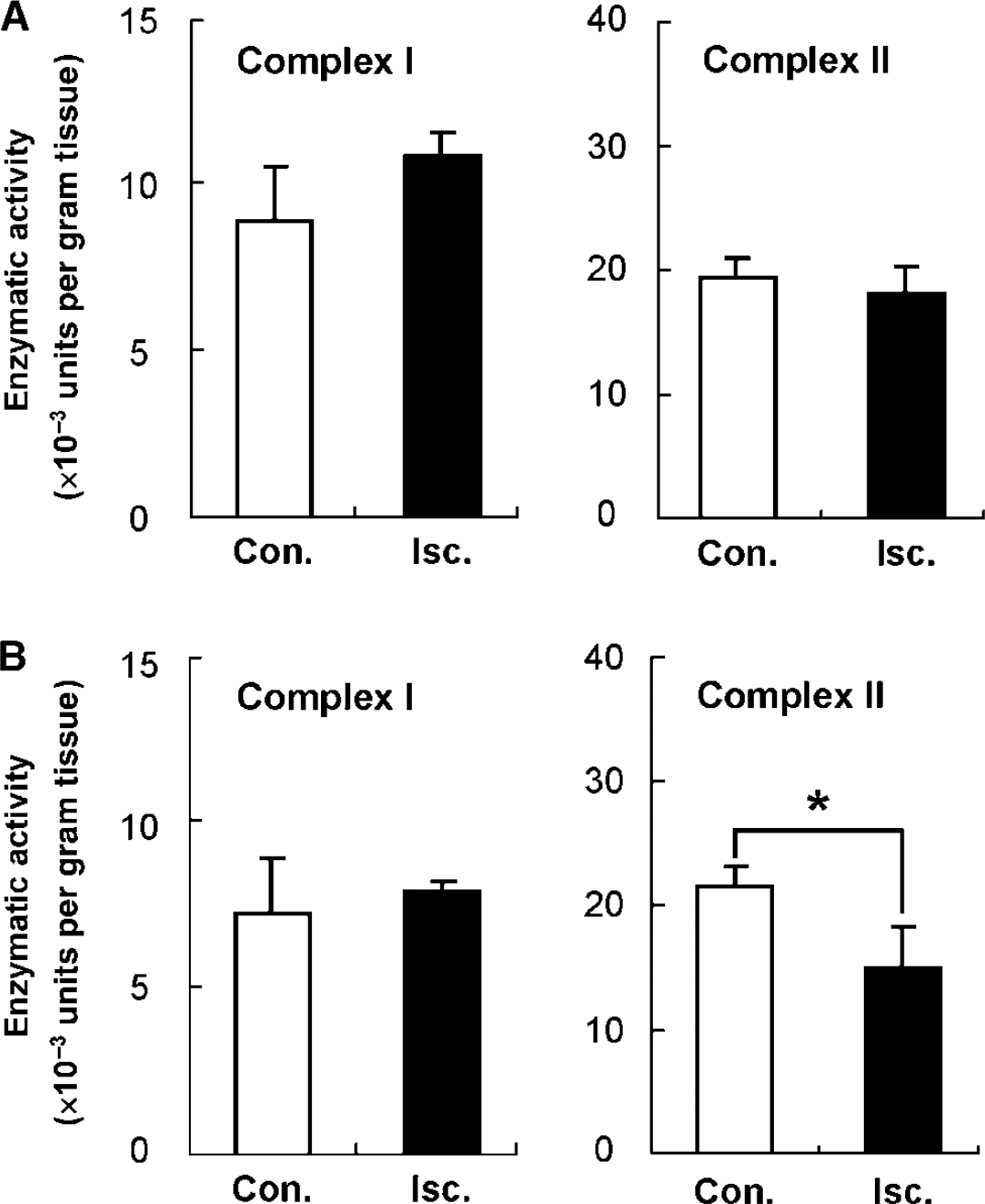
Estimation of the mitochondrial complexes I and II activities after 3 h (
The levels of low-molecular-weight antioxidants measured in the rat brain tissue after MCAO are shown in Figure 5. The concentration of GSH in the ischemic hemisphere was significantly lower than in the contralateral hemisphere after 3 h of reperfusion (Figure 5A), whereas the ascorbic acid and cysteine levels had not yet changed. However, the concentrations of ascorbic acid and GSH in the ischemic hemisphere were both significantly decreased after 24 h of reperfusion (Figure 5B). Conversely, the concentration of cysteine in the ischemic hemisphere increased to more than thrice the level in the contralateral hemisphere.
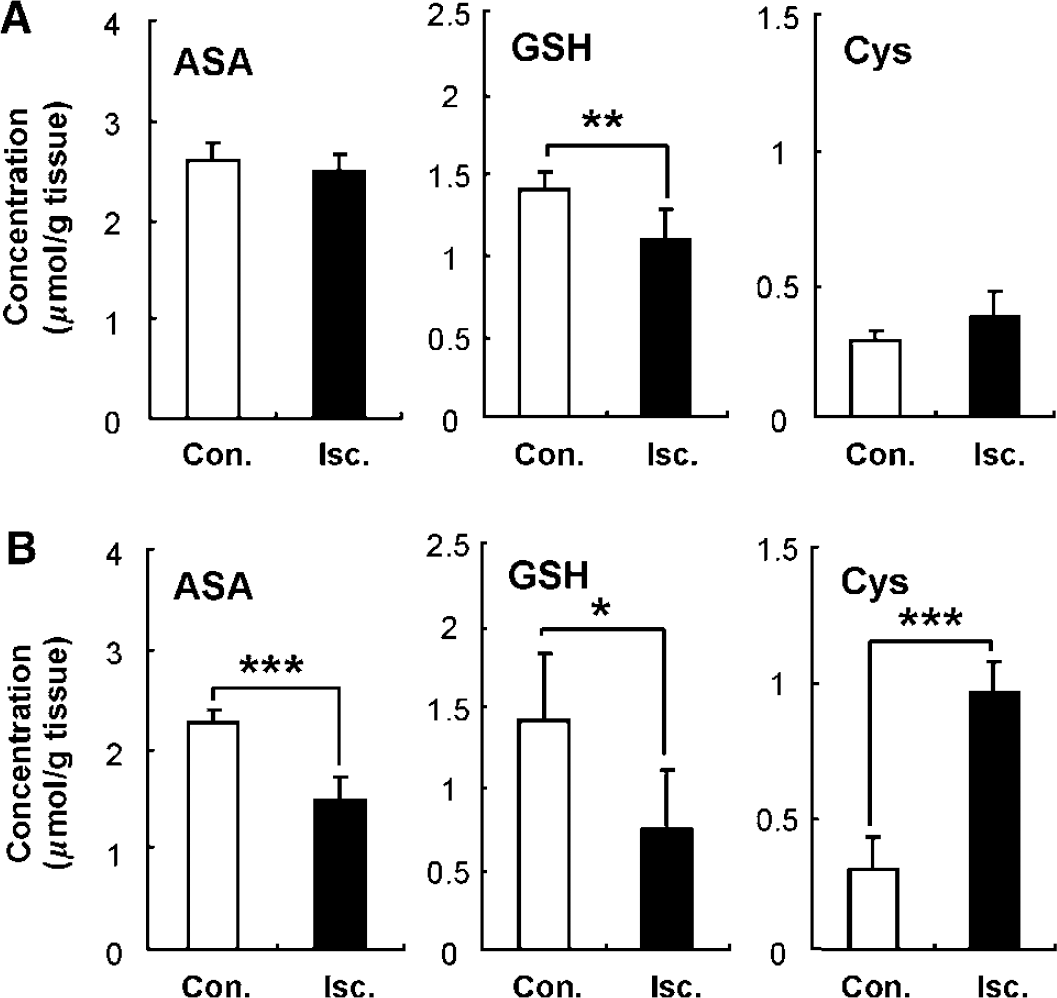
Estimation of the antioxidant levels after 3 h (
Discussion
The results of this study showed that the reduction of methoxycarbonyl-PROXYL was influenced by redox alterations after transient MCAO. The reduction of nitroxyl radicals is a useful index for estimating the ‘net’ redox status noninvasively, using MRI or OMRI for detection, whereas the details of the in vivo redox status have remained largely unknown. In this study, we were able to show the reduction mechanisms of methoxycarbonyl-PROXYL in the ischemic brain by a comprehensive investigation of the redox status. Our findings suggested that the redox imaging of the brain reflected alterations in the antioxidant level and the complex II activity in the mitochondria after ischemia–reperfusion.
Transient MCAO causes the production of ROS, such as superoxide anion radicals (Ohtaki et al, 2007), hydrogen peroxide (Lei et al, 1997), and hydroxyl radicals (Liu et al, 2003). Superoxide dismutase regulates the level of superoxide anion radicals and generates hydrogen peroxide, which, in turn, is converted to water and oxygen by catalase and GSH peroxidase. Moreover, the management of hydroxyl radicals depends on ascorbic acid and GSH (Cohen, 1994). In this study, we found that the level of GSH in the ischemic hemisphere had already significantly decreased after 3 h of reperfusion following MCAO, and that the ascorbic acid had also decreased after 24 h of reperfusion. As the levels of these antioxidants were decreased by the ischemia–reperfusion insult, their ability to defend the cells from oxidative stress was impaired. To counteract the decrease in antioxidants, transient MCAO mice can be treated with dehydroascorbic acid, which is transported to the blood–brain barrier and retained in the brain as ascorbic acid (Agus et al, 1997; Huang et al, 2001). Huang et al showed that the administration of dehydroascorbic acid significantly improves the cerebral blood flow and functional outcome, and significantly decreases the volume of infarcted brain tissue after ischemia–reperfusion. In addition, we observed not only a decrease in GSH but also an increase in cysteine in the ischemic hemisphere after 24 h of reperfusion. The breakdown of GSH by γ-glutamyltransferase generates cysteinylglycine, which can be taken up by a transporter or hydrolyzed to cysteine and glycine by an extracellular dipeptidase (Hanes et al, 1952). Therefore, the changes that we observed in both the GSH and the cysteine levels in the rat model may have arisen from the breakdown of GSH, which could further enhance the damage after transient MCAO, because the generated glutamate and cysteine can cause neuronal cell death (Meister and Tate, 1976).
Ischemia–reperfusion causes ROS overproduction by the mitochondria, leading to an increase in oxidative stress, an impairment of complex II, and mitochondrial dysfunction (Chen et al, 2007; Piantadosi and Zhang, 1996). In this study, we also observed a decrease in complex II activity after transient MCAO. Mitochondrial ATP (adenosine triphosphate) production is the major energy source for brain cells, because many cell functions, including neurotransmitter turnover, require ATP. Hence, mitochondrial dysfunction leads to severe ATP depletion, thus compromising neuronal signaling.
In this study, we used a noninvasive technique to observe redox alterations associated with the above-mentioned disease status. The fact that methoxycarbonyl-PROXYL has good permeability through the blood–brain barrier was confirmed not only by MRI study (Hyodo et al, 2008) but also by autoradiography (Anzai et al, 2003) and microdialysis (Shiba et al, 2008) studies. Moreover, methoxycarbonyl-PROXYL is a sensitive indicator of redox reactions (Figure 1). Therefore, we used methoxycarbonyl-PROXYL as a blood–brain barrier-permeable imaging probe to examine redox states in the brain. The 3-h reperfusion period after MCAO was not associated with changes in ascorbic acid concentration, mitochondrial electron transport chain activity, or decay of the methoxycarbonyl-PROXYL imaging intensity. Conversely, the reduction rate obtained from the continuous images of the ischemic hemisphere showed significant decrease compared with the rate in the contralateral hemisphere after 24 h of reperfusion. The total methoxycarbonyl-PROXYL levels were the same in both hemispheres, suggesting that the difference in intensity decay represented the reduction of methoxycarbonyl-PROXYL to its hydroxylamine.
We also estimated the reduction rates of methoxycarbonyl-PROXYL in the organelles of brain cells ex vivo to clarify the factors responsible for the methoxycarbonyl-PROXYL reduction. Similar to previous reports, we found that the reduction of methoxycarbonyl-PROXYL occurred in both the cytosolic and mitochondrial fractions. In particular, the reduction rates were slower when these fractions were obtained from the ischemic hemisphere than from the contralateral hemisphere after 24 h of reperfusion. At this time point, we also observed a decreased concentration of ascorbic acid and reduced complex II activity in the ischemic hemisphere, suggesting that the ischemia–reperfusion-induced changes affected the reduction of methoxycarbonyl-PROXYL in the brain.
Nitroxyl radicals react with superoxide anion radicals or hydroxyl radicals in the presence of reducing agents (Krishna et al, 1992; Kudo et al, 2008). These reactions form the chemical basis for using nitroxyl radicals as spin probes for in vivo ESR spectroscopy to examine the generation of free radicals noninvasively. We previously observed the enhanced ESR signal decay of a spin probe with intermediate permeability in vivo after 30 mins of reperfusion following MCAO (Yamato et al, 2003). However, in this study, the signal decay of methoxycarbonyl-PROXYL was not enhanced but rather was decreased after 24 h of reperfusion. Hence, the signal decay of methoxycarbonyl-PROXYL in the brain of transient MCAO rats might be altered by different factors at different time points after the injury.
In conclusion, we used OMRI to show the redox status of the live rat brain after ischemia–reperfusion injury. Decreased antioxidant levels and increased mitochondrial dysfunction were reflected in the brain redox image and were closely associated with the ischemia–reperfusion injury. Hence, redox imaging of the brain may be useful for noninvasively evaluating the disease state after ischemia–reperfusion injury.
