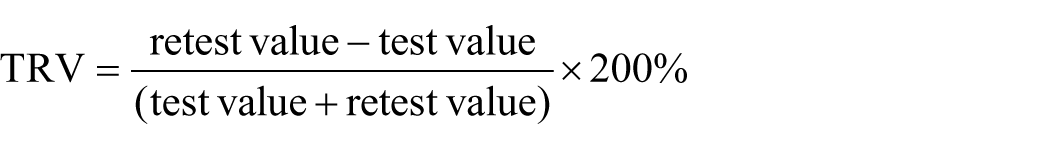Abstract
[11C]UCB-J is a radioligand targeting synaptic vesicle glycoprotein 2A, used to image synaptic density. For quantification, a small-volume centrum semiovale area was previously optimized as a [11C]UCB-J reference region (CS2mL); however, due to its small volume, its high variability resulted in reduced reliability. Herein, we evaluated an alternative reference region method to assess longitudinal test-retest reliability and detection of Parkinson’s disease (PD). For estimating distribution volume ratio (DVR), CS2mL and eleven white matter (WM) reference regions (range: 0.5–200 mL) were generated. Same-day and longitudinal test-retest variability (TRV) were assessed (24 healthy subjects (HS); n = 10 same-day and n = 20 longitudinal scans, range: 7–1028 days). Each reference region was used to evaluate the substantia nigra (SN) and caudate DVRs in HS (n = 25) and PD (n = 20); 10 mL was the optimal reference region volume, yielding [11C]UCB-J DVR measurements with reduced variability in TRV (same-day: 10 mL: 1.2 ± 5.7%, same-day: CS2mL: −0.9 ± 9.2% longitudinal: 10 mL: 1.5 ± 7.0%, CS2mL: 1.6 ± 11.9%), while maintaining <10% volume of distribution difference, compared to CS2mL. Further, a significant difference between PD and HS groups in SN and caudate DVRs was found using 10 mL, with greater effect size (Cohen’s d 0.61 for SN and 0.66 for caudate) compared to CS2mL (0.38 for SN and 0.43 for caudate).
Keywords
Introduction
Synaptic vesicle glycoprotein 2A (SV2A) is homogenously expressed on pre-synaptic vesicles of neurons throughout the central nervous system.1,2 Measurements of SV2A are useful as a surrogate marker for synaptic density, and recent advancements in positron emission tomography (PET) imaging have rendered in vivo synaptic density measurements possible.2–5
[11C]UCB-J, a radioligand targeting SV2A, has exhibited high specific binding, excellent brain uptake, and fast kinetic characteristics.5,6 As such, [11C]UCB-J PET has been applied extensively in humans to show differences in synaptic density in several neurological disorders including Alzheimer’s disease (AD)7,8 and Parkinson’s disease (PD).9,10
To validate the use of [11C]UCB-J in clinical studies, our group performed a kinetic evaluation of [11C]UCB-J in healthy subjects (HS) and we reported excellent same-day test-retest reproducibility of [11C]UCB-J. 6 Nevertheless, high longitudinal reliability is critical, particularly when studying neurodegenerative diseases that are progressive over several years, to evaluating pathological changes in synaptic density with time.11,12 Thus, optimizing quantification methodology can enhance statistical power to detect within-subject changes, allowing for clinical trials to have smaller population sizes or shorter duration.
Further, a reference region with minimal displaceable activity is ideally necessary to fully quantify and evaluate the specific binding of [11C]UCB-J. Binding potentials (BPND) have been derived using the centrum semiovale (CS)—a white matter (WM) region-of-interest (ROI) that has been optimized to have minimal displaceable activity and spill-in from gray matter (GM) regions5,7,13 as defined by the small-volume version of the CS by Rossano et al. 13 (referred to as CS2mL). ROIs derived from the WM can be appropriate reference regions due to the low uptake of SV2A compared to the GM. 5 However, due to its small volume (<2 mL), the CS2mL ROI introduces high variability into the measurements of specific binding, and longitudinal reliability was not considered in the initial selection of this CS ROI volume. Mecca et al. 7 proposed using the whole cerebellum as a reference region for evaluating differences in distribution volume ratios (DVRs) between AD patients and HS because the cerebellum exhibits a minimal difference in specific binding between these groups, and remains relatively intact in AD. Although the cerebellum provides a useful and appropriate reference ROI for studies of diseases wherein it is spared, cerebellar pathology is implicated in many neurodegenerative diseases.14,15 To this end, a WM ROI with less noise and minimal specific binding could be a useful reference region for many disorders.
Thus, to reevaluate reference regions for [11C]UCB-J, we created multiple sized WM ROIs for reference regions and performed same-day and longitudinal evaluations of test-retest reliability in HS. To optimize the WM reference ROI volume, we aimed to balance the high variability introduced using a small ROI with the bias induced by larger ROIs. In this context, we use “bias” to refer to the spill in of GM voxels at the boundaries of the WM into the computation of WM ROI values. As such, we compared the volume of distribution (VT) values among each reference ROI as well as the SD of the DVR test-retest variabilities (TRVs) computed using each reference region. Finally, we utilized the optimized reference ROI to reanalyze a subset of [11C]UCB-J PET images from a previous cross-sectional study of PD patients and HS, 10 to evaluate the reference region method when applied toward studying human pathophysiology.
Material and methods
Human participants
Twenty-four HS participated in the test-retest study (age: 40.3 ± 14.3 years, M:F = 13:11, 10 pairs of same-day scans, 20 pairs of longitudinal scans). These data are an extension of a previous same-day test-retest study. 6 A subset of subjects studied in Holmes et al. 10 who had arterial blood sampling were included in the PD cohort: 25 HSs (age: 60.6 ± 8.8 years, M:F = 11:14) and 20 PD subjects (age: 61.6 ± 8.6 years, M:F = 9:11, Movement Disorders Society-Unified Parkinson’s Disease Rating Scale III: 30.5 ± 9.0, Hoehn & Yahr: 2.0 ± 0.0, Montreal Cognitive Assessment: 26.4 ± 2.3, Disease duration: 4.7 ± 3.0). The data were collected from several protocols approved by the Yale University Human Investigation Committee and the Yale University Radiation Safety Committee, and in accordance with the United States federal policy for the protection of human research subjects contained in Title 45 Part 46 of the Code of Federal Regulations (45 CFR 46). Written informed consent was obtained from all participants after complete explanation of study procedures.
Injected Radiotracer
[11C]UCB-J was synthesized according to previously described procedures. 16 The injected radioactivity of [11C]UCB-J was 491 ± 185 MBq for the test-retest study cohort, and 538 ± 150 MBq for the PD cohort.
PET imaging experiments
All subjects participated in at least one 60-min [11C]UCB-J scans on the High Resolution Research Tomograph (HRRT, Siemens). 17 All participants in this study underwent arterial blood sampling. Radioactive concentration in plasma and parent fractions were measured to generate input function.
In the test-retest study, each subject participated in two [11C]UCB-J scans. There were 10 same-day and 20 longitudinal (1 week–1 month: 4 scan pairs, 1 month–1 year: 5 scan pairs, >1 year: 11 scan pairs, median 391 days, range: 7–1028 days) scan pairs. For the same-day group, the average time between injections was 5.3 h (range: 3–7 h) and they were allowed to consume a light lunch during the interval between the two PET measurements.
Blood analysis
Full details of the procedures of sampling and blood analysis were previously described. 6 For each arterial blood sample, plasma was obtained by centrifugation at 4℃ (2930 g for 5 min). For each arterial blood sample, plasma was obtained by centrifugation. Whole blood and plasma samples were counted in cross-calibrated gamma-counters (Perkin–Elmer, Waltham, MA, USA). Radiometabolite analyses were performed using an automatic column-switching HPLC system by measuring radioactivity in the plasma, plasma filter, plasma filtrate, and HPLC fractions.18,19 The arterial plasma input function was calculated as the total plasma activity multiplied by the parent fraction of [11C]UCB-J. 19
Image analysis
Processing of T1-weighted magnetic resonance (MR) images included skull- and muscle stripping procedures (FMRIB’s Brain Extraction Tool, http://fsl.fmrib.ox.ac.uk/fsl/fslwiki/BET).
The CS2mL and substantia nigra (SN) ROIs were defined in the automated anatomical labeling (AAL) template 20 in our previous work.10,13,21 For those ROIs, the individual MR image was spatially normalized to the AAL template in the Montreal Neurological Institute space using an affine linear plus nonlinear registration (BioImage Suite 2.5). 22 All other ROIs were defined based on FreeSurfer segmentation on the individual MR images. 23 The following FreeSurfer GM ROIs were created by merging original FreeSurfer ROIs. For cortical regions, parcellation was defined in Desikan–Killiany Atlas 24 : frontal cortex, parietal cortex, occipital cortex, temporal cortex, anterior cingulate, posterior cingulate, putamen, thalamus, caudate, hippocampus, entorhinal, amygdala, and brainstem. For the WM ROI segmentation, the cerebral WM ROI segmented by FreeSurfer was binarized and smoothed using a 10 mm full width half maximum (FWHM) gaussian filter to generate a WM map. 25 Ten differently sized ROIs were generated (0.5, 1, 2, 5, 10, 20, 45, 100, and 200 mL) in individual MR space by applying different thresholds on the smoothed WM map denoted as FreeSurfer-based WM (FBWM) ROIs (Supplemental Figure S1). Thresholds were selected to provide a wide range of WM ROI volumes beginning smaller than the CS2mL region and gradually increasing in volume to evaluate the impact of GM spill-in voxels from the boundaries of larger ROIs. The ROI volume did not vary between subjects. To extract ROI TACs from PET data, an average PET image (0–10 min) was aligned to each subject’s MR image via a rigid registration with mutual information; this transformation was used to align ROIs with PET space.
PET quantitative analysis
The one tissue compartment model was applied to estimate regional VT. DVR values were calculated using the CS2mL, and FBWM ROIs as reference regions. For the test-retest cohort, the reproducibility of VT and DVR values, calculated for each evaluated ROI using each reference region, were examined by calculation of the TRV as follows:
All data are presented as mean ± standard deviation (SD). To evaluate the TRV using methodology that is not reliant on arterial blood sampling, the simplified reference tissue model 2 (SRTM2) method was used to compute DVRs 26 (Supplemental methods).
For the PD cohort, the percent difference of DVR in the SN, caudate, and brainstem ROIs between HS and PD patient groups were calculated using the optimized FBWM and CS2mL as the reference ROIs. These regions were chosen because they exhibited a significant difference in BPND between HS and PD in Holmes et al. 23 Statistical analyses between groups were performed with two-tailed, unpaired t-tests, with significance defined as p < 0.05. The between-group percent difference, Cohen’s d, and intersubject %SD using each reference ROI were calculated.
Results
Injected radiotracer
There were no statistically significant differences between the test and retest condition or HS and PD patients in the injected amount of radioactivity, molar activity, or injected mass dose.
VT test-retest variabilities in each reference region
The average VT in the CS2mL across each subject’s first scan was 4.33 mL/cm3 ± 11.5%. VT in FBWM regions increased with increasing ROI volume (Figure 1). Only FBWM ROIs 10 mL and smaller exhibited less than 10% difference in mean VT compared to CS2mL, and %CoV was the second lowest in 10 mL (mean values, with %CoVs: 0.5 mL: 4.40 ± 13.1%, 10 mL: 4.68 ± 10.2%, 20 mL: 4.89 ± 10.1%, 100 mL: 6.44 ± 12.1%, Figure 1, Supplemental Table S2). While the mean TRV of VT in FBWM ROIs are close to 0% and consistent across different region volumes, the TRV SD decreases with increasing region volume for both the same-day and longitudinal scan pairs (same-day: 0.5 mL: −2.2 ± 14.5, 1 mL: −2.4 ± 12.0, 10 mL: −1.3 ± 8.5, 20 mL: −0.7 ± 8.2, 100 mL: 0.5 ± 7.9, and longitudinally: 0.5 mL: 0.1 ± 16.9, 1 mL: 0.1 ± 17.0, 10 mL: −0.5 ± 11.6, 20 mL: −0.3 ± 11.2, 100 mL: 0.3 ± 10.6, Supplemental Table S3). The VT TRV in CS2mL was −0.8 ± 9.9 and −0.6 ± 15.1 for same-day and longitudinal, respectively, demonstrating the higher TRV SD exhibited by CS2mL compared to larger regions such as the 10 mL FBWM ROI.
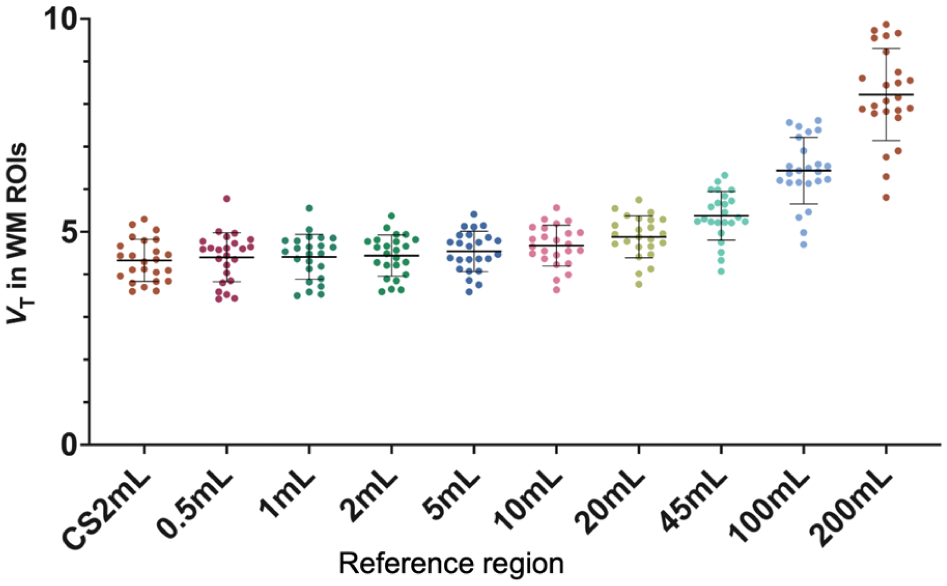
Volume of distribution (VT) calculated within each reference region for each subject’s first scan (n = 24). Regions of interest (ROIs) include the centrum semiovale (CS2mL) and Freesurfer-Based White Matter (FBWM) regions ranging from 0.5 to 200 mL.
Test-retest variability in gray matter regions for same-day and longitudinal groups
For each participant in the test-retest study, DVR was calculated for 12 GM regions by dividing the VT in each GM region by the VT in each reference region. VT TRV means and SDs were generally consistent across GM regions. The same-day group exhibited smaller variabilities of VT TRV in GM regions compared to the longitudinal group (Table 1). The mean TRV of the DVRs, averaged across all 12 GM regions, were small (p > 0.05 for t-test comparing test and retest value) and similar regardless of the reference region suggesting no systematic bias in both same-day and longitudinal group (Supplemental Table S3). The TRV SD of the DVRs, on the other hand, varied based on the reference region used, ranging from 3.1% in the 100 mL ROI to 9.8% in the 0.5 mL ROI for the same-day dataset, and ranging from 3.8% in the 100 mL ROI to 12.4% in the 0.5 mL ROI for the longitudinal dataset (Figure 2, Supplemental Table S3). Lower variability in DVR TRV was exhibited by using larger reference regions (Same day: VT: −0.2 ± 7.6%, CS2mL: −0.9 ± 9.2%, 10 mL: 1.2 ± 5.7%, 45 mL: −0.3 ± 4.2% 100 mL: 0.7 ± 4.4% longitudinal: VT: 1.0 ± 10.4%, CS2mL: 1.6 ± 11.9%, 10 mL: 1.5 ± 7.0%, 45 mL: 1.0 ± 5.2% 100 mL: 0.7 ± 4.4%, Table 2, Table S3). Overall, TRV variability was lower for DVR2–200 mL compared to DVRCS2mL.
Same-day and longitudinal test-retest variability (TRV) of volume of distribution (VT) averaged across subjects for each region of interest.
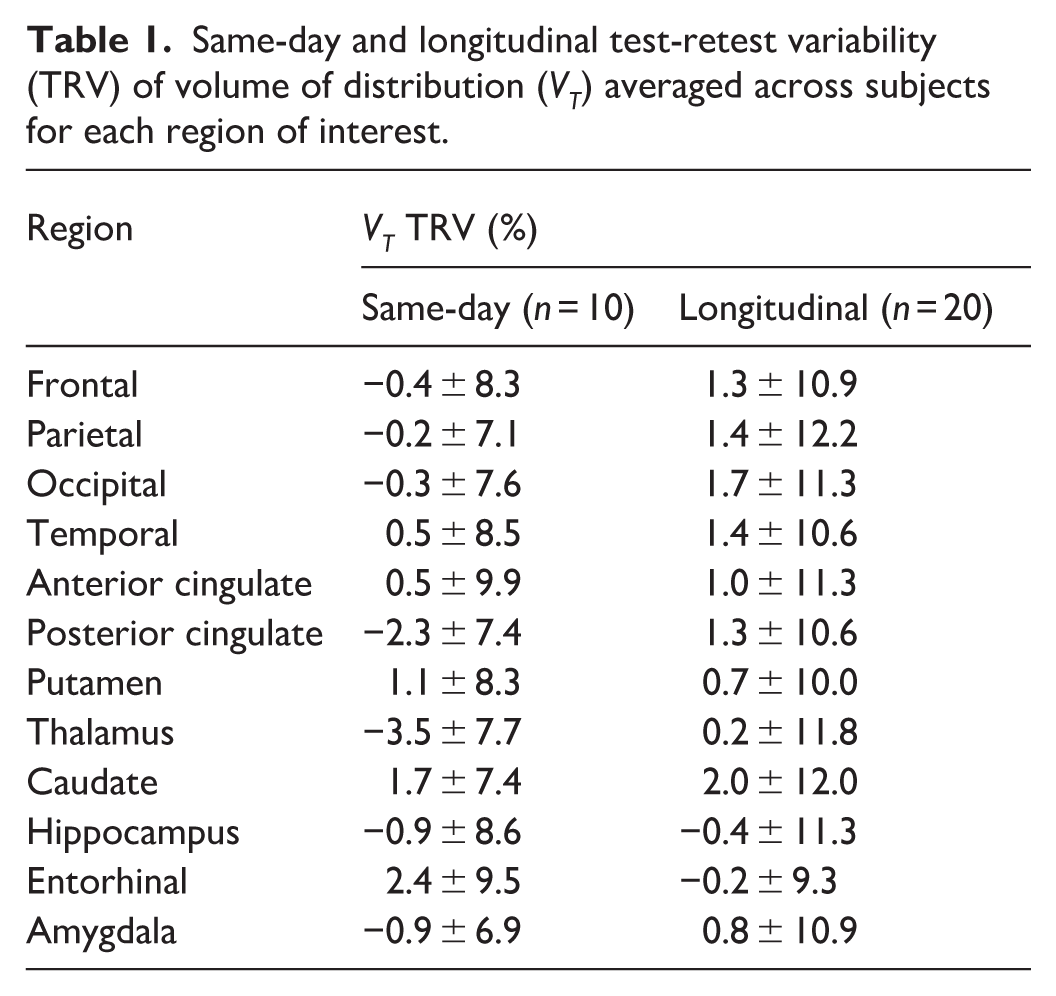
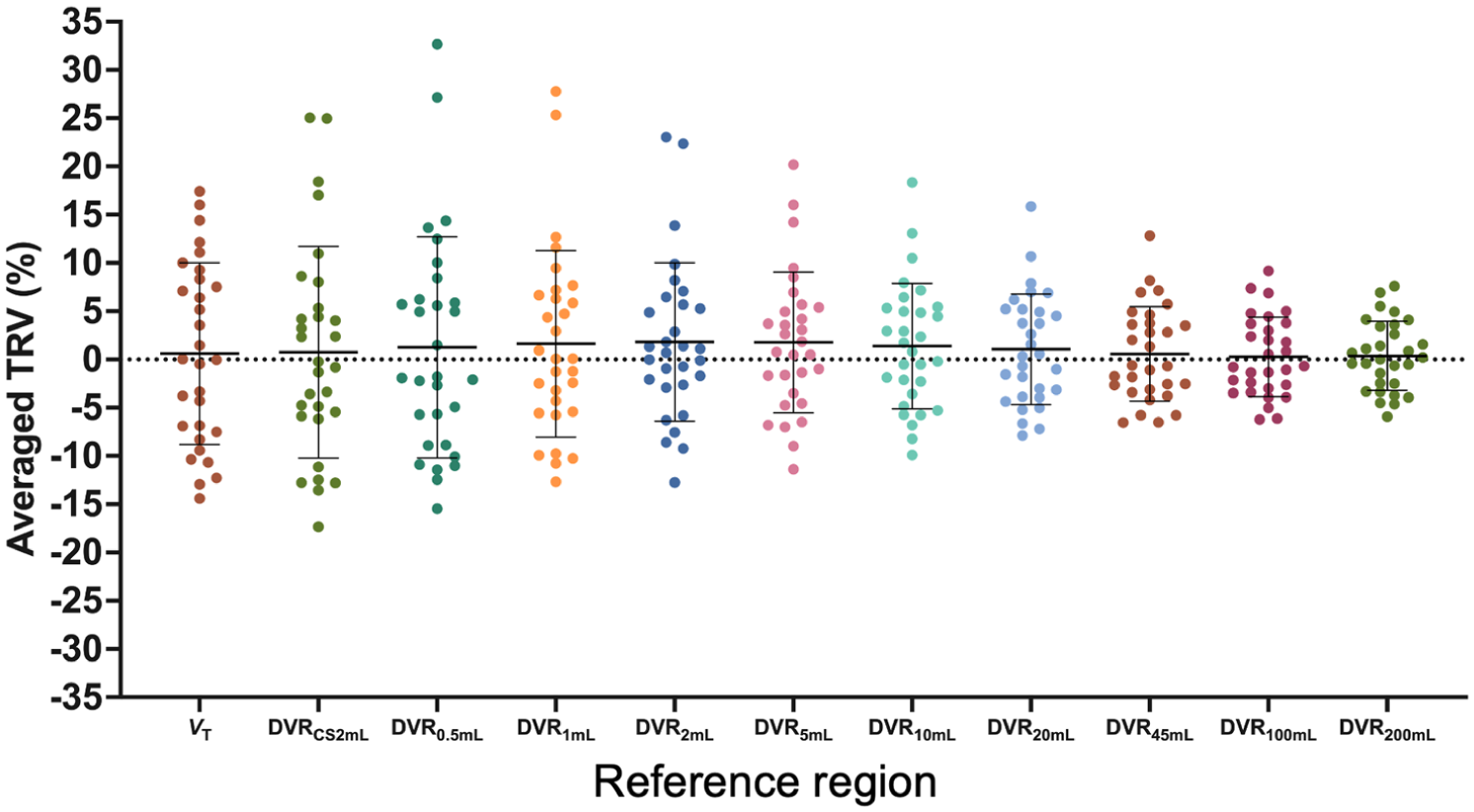
Test-retest variability (TRV) of distribution volume ratios (DVR) calculated using each reference region, averaged among 12 gray matter regions. Reference regions include the centrum semiovale (CS2mL) and Freesurfer-based white matter (FBWM) regions ranging from 0.5 to 200 mL.
Same-day and longitudinal test-retest variability (TRV) of distribution volume ratios (DVRs) calculated using the centrum semiovale (DVRCS2mL), 10 mL region (DVR10 mL), 45 mL region (DVR45 mL), and 100 mL region (DVR100 mL) as the reference region, averaged across subjects for each region of interest.
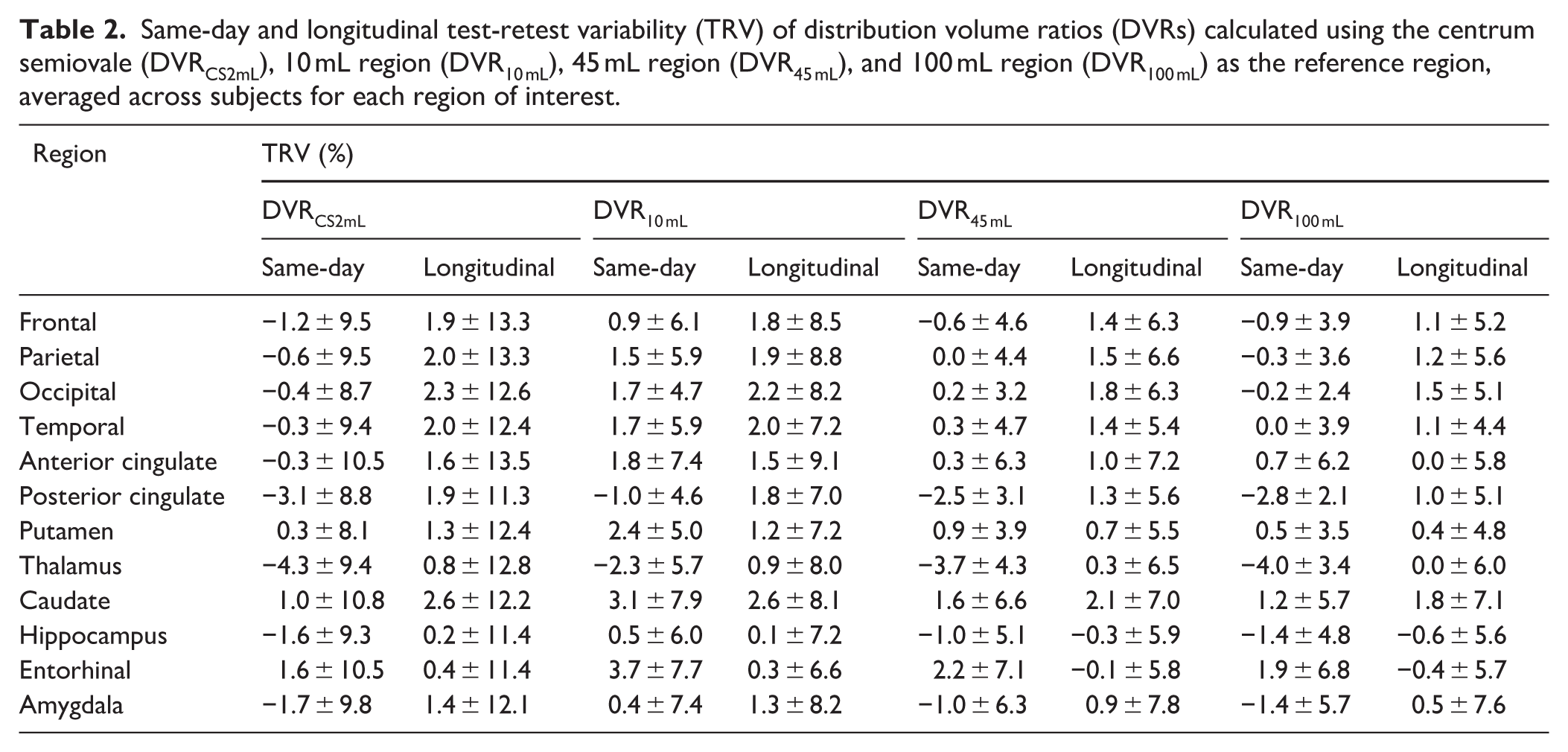
For the longitudinal group, the averaged TRV among 12 GM ROIs were plotted versus the number of days between the first and second scans. For each reference region, the TRV did not significantly change within our study duration, indicating that the inter-scan interval had no consistent effect on TRV up to 1028 days (Figure 3 for VT, DVRCS2mL, DVR10 mL, and DVR45 mL). The residual sum of squares was computed for each interscan interval plot using each reference region, demonstrating the reduction in SD induced using larger reference regions (DVRCS2mL: 2910.4, DVR10 mL: 1022.6, DVR45 mL: 535.8). Balancing the smaller variability achieved using larger ROIs against the small VT difference from the previously validated CS2mL region (<10%), and thus small GM spill-in bias, observed in the smaller reference regions, we propose the 10 mL FBWM as the optimal reference region.
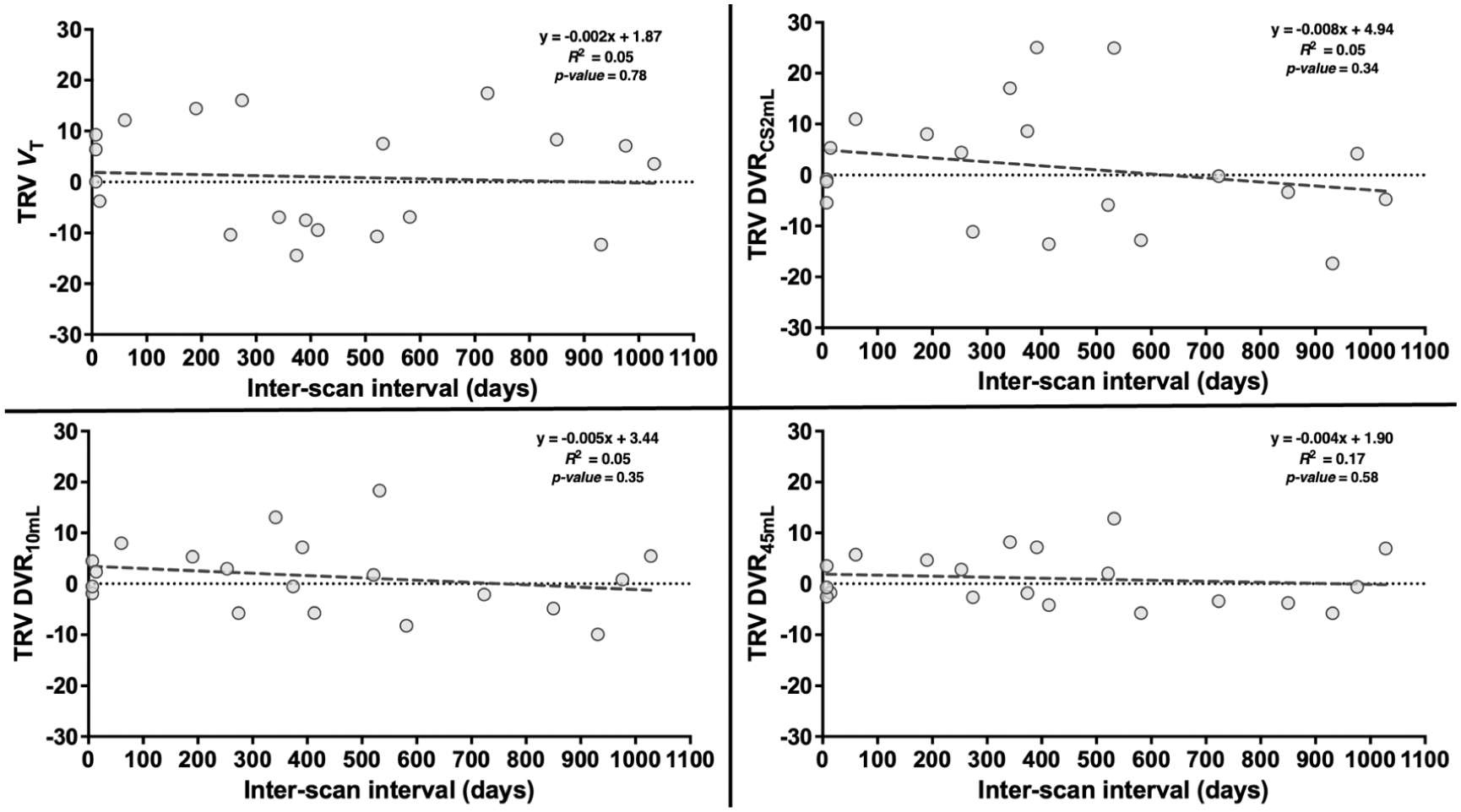
Test-retest variability (TRV) of volume of distribution (VT) and distribution volume ratios (DVR) calculated using each reference region, averaged among gray matter regions, plotted against interscan interval. Reference regions include the centrum semiovale (CS2mL) and the 10 and 45 mL Freesurfer-Based White Matter (FBWM) regions. The equation of the line of best fit, R2, and p-values are also displayed for each method.
To confirm the reduced TRV variability exhibited by DVRs calculated using the 10 mL FBWM compared to the CS2mL region using a reference tissue method without arterial blood sampling, SRTM2-based DVR values were computed (Supplemental Table S4). For the evaluated GM ROIs, the TRV was low regardless of the reference region used; however, the variability in TRV was consistently lower using DVRs computed with 10 mL FBWM compared to CS2mL.
FBWM reference region method applied to Parkinson’s disease dataset
For the 10 mL reference region, DVRs in the SN and caudate were significantly different between HS and PD groups (p < 0.05) but were not significant with the CS2mL reference region (Figure 4). The DVR in the brainstem was not significantly different between groups for either reference region. The intragroup SD for the SN was 16.1% and 14.7% using DVRCS2mL and DVR10 mL, respectively, for the HS group, while the SD was 18.6% and 16.0% using DVRCS2mL and DVR10 mL respectively, for the PD group. The intragroup SD for the caudate was 12.0% and 10.6% using DVRCS2mL and DVR10 mL, respectively, for the HS group, while the SD was 15.3% and 14.8% using DVRCS2mL and DVR10 mL respectively, for the PD group. Further, DVR10 mL exhibited a higher group percent difference and Cohen’s d for each ROI, compared to DVRCS2mL (SN: DVRCS2mL: 6.5% difference, Cohen’s d: 0.38, DVR10 mL: 9.0% difference, Cohen’s d: 0.61. caudate: DVRCS2mL: 5.7% difference, Cohen’s d: 0.43, DVR10 mL: 8.0% difference, Cohen’s d: 0.66. brainstem: DVRCS2mL: 2.4% difference, Cohen’s d: 0.23, DVR10 mL: 5.0% difference, Cohen’s d: 0.56. Figure 4, Supplemental Table S5). Of note, the %CoV for VT computed in the CS2mL region was 15% and 12% for HS and PD, respectively; for the 10 mL ROI VT CoV was 11% for both groups (Supplemental Figure S3).
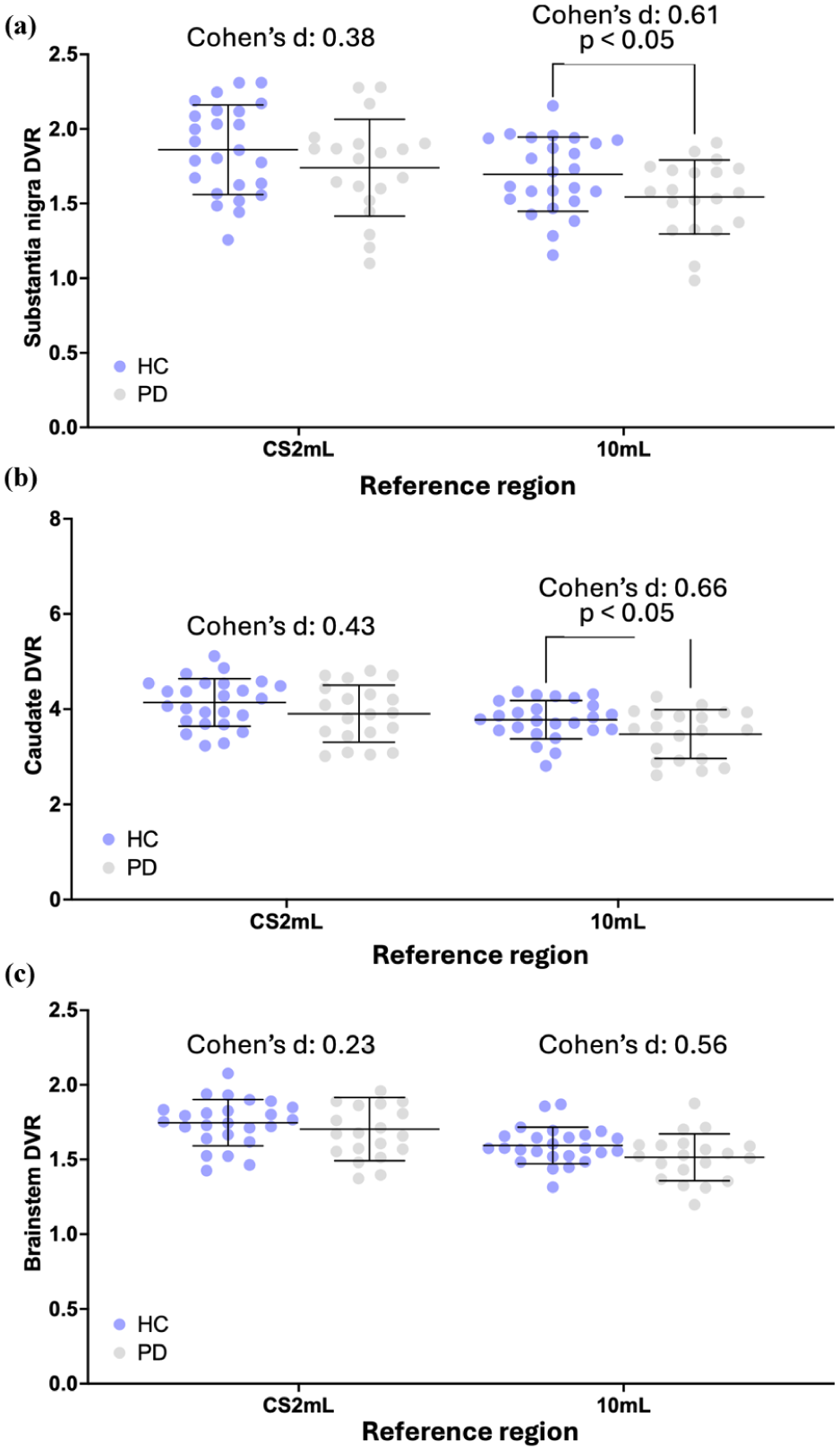
(a) Substantia nigra, (b) caudate, and (c) brainstem distribution volume ratios (DVR) calculated for Parkinson’s disease (PD) and healthy subjects (HS). Reference regions include the centrum semiovale (CS2mL) and 10 mL Freesurfer-based white matter (FBWM) region. p-values are displayed demonstrating significant difference between HS and PD groups (p-value < 0.05). Effect size is displayed using Cohen’s d.
Discussion
In this study, the longitudinal and same-day test–retest reproducibility using [11C]UCB-J was examined, and the development of WM-based reference regions was proposed and applied toward the test-retest reproducibility outcomes and a dataset comparing [11C]UCB-J uptake in PD and HS. Overall, we demonstrated that [11C]UCB-J exhibits excellent longitudinal test-retest reliability regardless of the reference region used; however, an optimized 10 mL WM region can yield [11C]UCB-J DVR measurements with reduced inter-individual variation compared to the CS2mL because of its larger volume, without introducing significant specific activity (i.e., the 10 mL FBWM ROI was the largest region to exhibit a <10% difference in VT compared to the CS2mL, while maintaining the second lowest %CoV across different region volumess). Indeed, a significant group difference between PD and HS groups was elicited from SN and caudate DVR values using the 10 mL FBWM region instead of CS2mL. Consequentially, we demonstrated the utility of improved reference regions for [11C]UCB-J PET for assessing synaptic density in disorders that necessitate longitudinally reliable measurements.
Same day and longitudinal test-retest variability
In agreement with our previous study, the same-day TRV in VT and DVR for several regions was low, regardless of reference region. 6 Similarly, the longitudinal TRVs of regional VT and DVR values demonstrated reasonable test-retest reliability, but with higher variability than same-day TRV, sometimes greater than 10%. This is in agreement with a study by Tuncel et al., 27 in which the 28-day test-retest repeatability of [11C]UCB-J PET was evaluated, demonstrating a TRV variability of VT ranging from 4.2% to 6.8%. Interestingly, using a larger CS region (mean volume 5.4 mL), they did not demonstrate a drastic improvement in TRV variability.
There are several potential sources of the higher variability in TRV exhibited in the longitudinal study. First, not all subjects underwent MR imaging on the same scanner, introducing potential variability because MR images are essential for accurate ROI segmentation. 28 Further, physiological variability could be introduced by a difference in synaptic density exhibited by some participants between their first and second scan. To this point, only HS were evaluated, with an average age of 39.8 years and longest interval between scans of <3 years; thus, any change in synaptic density would be unexpected. 29 Age-related reductions in synaptic density have been demonstrated in HS, 30 and studies have suggested that the caudate exhibits the largest synaptic density reduction per year.31,32 This pattern is not replicated in our data. To assess longitudinal age-related reductions in synaptic density, an increased sample size and longer interscan interval would be necessary. Nevertheless, an undiagnosed neurological condition in any of the evaluated subjects could result in a change of synaptic density between the scans, introducing inter-individual variability in TRV values. Finally, human- and instrument-induced variability can be associated with several daily processes including radiosynthesis, tracer injection, blood sampling, and scanner calibration.33,34 These sources of variability likely create bias spatially all-throughout each image, providing motivation for using the DVR as the primary measure of specifically bound tracer.
The CS2mL has been optimized and validated as an appropriate reference region with minimum specific binding for calculating [11C]UCB-J DVR as well as BPND for some populations.5,13,35 Indeed, the CS2mL has been used as a reference region for several studies evaluating regional synaptic density in health 30 and disease.9,27,36–38 Although the small volume (2 mL in template space, <2 mL in subject space) and deep location renders the previously proposed CS2mL ROI relatively invulnerable to GM spill-in effects that would bias DVR values, due to the smaller proportion of voxels at the boundary of WM and GM, it also results in VT measurements that are computed from a small number of voxels, increasing DVR variability. Figure 1 visually demonstrates the high variability of the CS2mL VT, which is propagated into the average DVRCS2mL displayed in Figure 3. This phenomenon results in the higher variability in DVRCS2mL TRVs (Table 2) compared to those calculated using any other evaluated reference region.
Thus, a FBWM ROI is likely to be the appropriate choice—that is, if the ROI volume is large enough to overcome the variability seen in smaller reference regions but small enough to avoid GM spill-in. Figure 1 and Supplemental Table 1 demonstrate the higher VT in larger regions, compared to the CS2mL, likely due to the spill in of GM voxels containing variable concentrations of [11C]UCB-J. On the other hand, the variability in DVR TRVs exhibited by using ⩽5 mL FBWM ROIs (Figure 2 DVR0.5–5 mL) was higher compared to FBWM ROIs ⩾ 10 mL in volume. Tables 1 and 2 demonstrate that longitudinal and same-day DVR10 mL TRVs show reduced variability compared to DVRCS2mL. These results are in agreement with results published by Tuncel et al. 27 they computed DVRs using a larger CS2mL reference region with a mean volume of 5.4 mL and did not demonstrate a resulting increase in TRV variability. Based on these factors, the 10 mL FBWM region was selected for further evaluation using the PD cohort.
Clinical application: Comparing SV2A binding in PD and healthy subjects
Variability in outcome measurements imposes a limit on the detection of a group difference in [11C]UCB-J binding. Studies have demonstrated reduced regional synaptic density in PD subjects compared to HS using the CS as a reference region.9,10,39–41 We investigated the impact of using the 10 mL FBWM reference region, in place of the CS2mL, to evaluate and compare the variability and group difference in synaptic density measurements within the SN, caudate, and brainstem of PD and HS; these regions were chosen due to their significance, prior to multiple comparisons correction, in another study by Holmes et al.9,10
The group percent difference for reference region VT values increased with larger ROI volume, with percent differences greater than or equal to 8% only exhibited by ROIs greater than 45 mL in volume (Supplemental Figure 1). Compared to the CS2mL, VT calculated within the 10 mL FBWM ROI exhibited a smaller difference between PD patients and HS, suggesting that the 10 mL ROI does not introduce displaceable binding into the denominator of DVR calculations. Compared to values calculated using the CS2mL, DVR10 mL exhibited lower variability, as well as a large percent difference and effect size between the PD and HC groups for each evaluated region. These results are not in agreement with a study by Holmes et al., 10 in which they found a significant difference in SN, caudate, and brainstem using the CS as a reference region for computing a binding potential (BPND) with the simplified reference tissue model 2 method; however, this is likely due to differences in methodology and cohort, the latter of which was larger, including subjects without arterial blood sampling. Of note, several PD subjects included in Holmes et al. 10 with extensive disease burden (>6 years since diagnosis) did not have arterial blood sampling. Nevertheless, the results herein suggest that the 10 mL FBWM ROI is a better and possibly optimal reference region for studying synaptic density changes in neurodegenerative diseases in which the cerebellum is implicated in disease pathogenesis.
In fact, knowledge of the variability induced by using a particular reference region may inform future study designs. For example, based on a simple power analysis, about 10 subjects are needed to measure a difference, or change, of 10% in synaptic density in the SN, based on the variability measured using the 10 mL reference region to calculate DVR in the PD and HS cohorts. This is in comparison to about 15 subjects needed using the CS2mL.
Although the low specific binding of [11C]UCB-J and relatively low variability within the 10 mL FBWM ROI encourages its use as a reference region for longitudinal studies, this method is not suitable for use in patients with WM pathology. WM lesions or atrophy, often induced by inflammation such as in multiple sclerosis and HIV encephalopathy, stroke sequelae, and other conditions, can render reference region measurements inaccurate. 42 Thus, future optimization of this method should be aimed toward avoiding the inclusion of abnormal WM in reference regions.
It should be considered that all data were acquired using the HRRT, and corrected for head motion using event-by-event motion correction with a head tracking system 43 as well as frame-based image registration. 44 Partial volume correction may be necessary to produce similar results using data acquired with poorer spatial resolution. 25 Future studies repeating the current analysis using other scanners and motion correction strategies would further elucidate the generalizability these results.
In all, in this study we demonstrated the long-term reliability of [11C]UCB-J PET in conjunction with a new optimized WM reference region for assessing regional synaptic density in age-related neurogenerative disorders. As such, future [11C]UCB-J PET studies incorporating the 10 mL reference region may benefit from the reduced variability introduced by this region.
Supplemental Material
sj-docx-1-jcb-10.1177_0271678X261441071 – Supplemental material for Optimized reference region and the effect on test-retest reliability and sensitivity to differences between Parkinson’s disease and control groups with [11C]UCB-J
Supplemental material, sj-docx-1-jcb-10.1177_0271678X261441071 for Optimized reference region and the effect on test-retest reliability and sensitivity to differences between Parkinson’s disease and control groups with [11C]UCB-J by Nikkita Khattar, David Matuskey, Jean-Dominique Gallezot, Mika Naganawa, Sophie E Holmes, Faranak Ebrahimian Sadabad, Mahsa Mayeli, Irina Esterlis, Christopher H van Dyck, Adam P Mecca, Deepak Cyril D’Souza, Nabeel B Nabulsi, Sjoerd J Finnema, Yiyun Huang, Richard E Carson and Takuya Toyonaga in Journal of Cerebral Blood Flow & Metabolism
Footnotes
Acknowledgements
The authors appreciate the excellent technical assistance of the staff at the Yale University PET Center.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by NIH Medical Scientist Training Program Training Grant (T32GM136651), NINDS (R01NS124819; R01NS094253), NIA (R01AG052560), Nancy Taylor Foundation, AbbVie Inc.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: SJF is a full-time employee of AbbVie but their employment does not constitute any competing interest for the content presented in this article. All other authors have no relevant financial or non-financial interests to disclose.
Author contributions
TT, REC, and NK collectively contributed to the conception of the study and its design. J-DG, MN, REC, and TT contributed to image processing algorithm development. DM, SEH, FES, MM, IE, CHVD, APM, DCD, REC, SJF, and TT were responsible for human data acquisition, recruitment, evaluation, and care. YH and NBN were responsible for tracer synthesis and quality control tests. TT and NK performed data analysis. TT, REC, NK, SJF, and DM contributed to data interpretation. The first draft of the manuscript was written by NK and TT. All authors participated in the editing the manuscript and approved the manuscript and this submission.
Ethical considerations
All procedures performed in studies involving human participants were conducted in accordance with the ethical standards of the institutional and/or national research committee, as well as the 1964 Helsinki declaration and its subsequent amendments or comparable ethical standards.
Consent to participate
Informed consent was obtained from all individual participants included in the study.
ORCID iDs
Data availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.*
Supplemental material
Supplemental material for this article is available online.