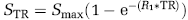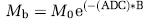Abstract
The well-vascularized nature of gliomas has generated a lot of interest in antiangiogenic therapies. However, the potential of vascular disrupting agents (VDAs) against gliomas has not been investigated extensively. In this study, we examined the in vivo efficacy of the tumor-VDA 5,6-dimethylxanthenone-4-acetic acid (DMXAA) against gliomas. Contrast-enhanced magnetic resonance imaging (MRI) and diffusion-weighted MRI were used to characterize the vascular and cellular responses of GL261 and U87 gliomas to DMXAA treatment. Therapeutic efficacy was assessed by Kaplan-Meier survival analysis. Before VDA treatment, minimal enhancement was detected within the tumor in both models. Longitudinal relaxation rate (R1 = 1/T1) maps acquired 24 h after treatment showed marked extravasation and accumulation of the contrast agent in the tumor indicative of treatment-induced vascular disruption. Normalized change in relaxation rate (ΔR1) values of the tumor showed a significant increase (P<0.01 GL261; P<0.05 U87) after therapy compared with baseline estimates. Mean apparent diffusion coefficient (ADC) values were significantly increased (P = 0.015) 72 h after therapy in GL261 but not in U87 gliomas. Vascular disrupting agent therapy resulted in a significant (P<0.01) increase in median survival in both models evaluated. The results highlight the potential of VDAs against gliomas and the utility of MRI in the assessment of glioma response to VDA therapy.
Introduction
Vascular proliferation is a critical component of glioma biology that strongly influences disease aggressiveness and patient survival (Brem, 1976; Abdulrauf et al, 1998; Cao et al, 2006). As a result, there has been considerable interest in therapies targeted toward tumor angiogenesis (Chamberlain, 2008). Several preclinical studies have reported the activity of antiangiogenic agents against gliomas (Sandstrom et al, 2004; Claes et al, 2008; Mathieu et al, 2008). Recent clinical studies have also investigated the activity of antiangiogenic agents in combination with chemotherapy with encouraging results (Narayana et al, 2009; Zuniga et al, 2009). Antiangiogenic agents, such as bevacizumab, are aimed at inhibiting new vessel formation by targeting specific angiogenic mediators or their receptors (Gerstner et al, 2007); in contrast, tumor-vascular disrupting agents (tumor-VDAs), such as combretastatin and 5,6-dimethylxanthenone-4-acetic acid (DMXAA), lead to disruption of the existing tumor vasculature (Tozer et al, 2005). Although the activity of VDAs against a variety of tumor types has been reported in preclinical model systems, only a few studies have examined the potential of VDA therapy against gliomas. Published reports of studies investigating the activity of VDAs against gliomas have also been carried out only in ectopic (subcutaneous) brain tumors (Eikesdal et al, 2002; Wachsberger et al, 2005). As tumor vascularization is an important characteristic of glioma biology, we hypothesized that selective disruption of tumor vasculature could be of potential therapeutic benefit in gliomas. To test this hypothesis, we examined the therapeutic activity of the small molecule tumor-VDA, DMXAA (Rehman and Rustin, 2008), against two experimental orthotopic models, murine GL261 gliomas and human U87 glioma xenografts. Using an imaging-based approach, we characterized the response of the two glioma models to DMXAA treatment.
Imaging techniques, such as magnetic resonance imaging (MRI) and PET (positron emission tomography), constitute an integral component of the diagnostic and therapeutic assessment of gliomas (Gerstner et al, 2008; Sorensen et al, 2008). Among the radiologic techniques currently available, MRI offers several advantages, including excellent soft tissue contrast, high temporal and spatial resolution without the use of ionizing radiation or radioactive tracers. Specifically, contrast-enhanced MRI (CE-MRI), a technique that provides information pertaining to tumor vascular physiology, is widely being used to evaluate the biologic activity of targeted therapies in preclinical models and in clinical trials (Akella et al, 2004; Jackson et al, 2007). In neuro-oncology, CE-MRI has been used to estimate parameters, such as cerebral blood volume and vascular permeability in gliomas (Cao et al, 2006; Claes et al, 2008; Akella et al, 2004). Therefore, in this study, using CE-MRI, we prospectively investigated the early vascular changes in murine GL261 gliomas and in human U87 glioma xenografts after treatment with the tumor-VDA DMXAA. This study included a baseline CE-MRI examination before DMXAA treatment and a follow-up study at 24 h after treatment.
Another MRI technique that is being widely investigated in preclinical and clinical studies for its utility as a biomarker of therapeutic response is diffusion-weighted MRI (DW-MRI) (Hamstra et al, 2008). Diffusion-weighted-MRI is a sensitive technique that allows detection of early cellular changes in tumors based on the Brownian motion of water (Koh and Collins, 2007). In experimental animal models, DW-MRI has been shown to provide tumor-specific information that strongly correlates with treatment response (Lee et al, 2006). Measurement of the apparent diffusion coefficient (ADC) from DW-MRI data sets has been correlated with disease progression and survival in patients with brain tumors (Hamstra et al, 2008). Therefore, in addition to CE-MRI, DW-MRI was performed 72 h after treatment and ADC maps were calculated to examine changes in water mobility as a measure of tumor response to DMXAA.
Finally, to determine the long-term therapeutic efficacy of DMXAA against the two glioma models, animals were monitored over a 40-day period and differences in survival between control and treatment groups were assessed by Kaplan-Meier analysis. The results of our studies show for the first time the potent tumor vascular disruption after DMXAA treatment in both glioma models evaluated. A statistically significant increase in median survival was also observed after VDA treatment compared with untreated controls.
Materials and methods
Cell Lines and Culture Conditions
GL261 murine glioma cells and U87 human glioma cells were grown on 100-mm tissue culture plates in complete DMEM (Dulbecco's modified Eagle's medium; Invitrogen, Grand Island, NY, USA) containing 10% fetal calf serum and 5,000 Units of penicillin/streptomycin at 37°C in 5% CO2 with media changes two to three times per week.
Tumor Models
C57B16 mice and athymic NCr-nu/nu nude mice were purchased from the National Cancer Institute (Rockville, Maryland, USA) for establishing GL261 and U87 gliomas, respectively. Animals were provided food and water ad libitum and housed in micro isolator cages in a laminar flow unit under ambient light. The procedure for intracerebral implantation of tumor cells has been previously described (Ciesielski et al, 2006). Briefly, 8- to 12-week-old mice were anesthetized by an intraperitoneal injection of ketamine:xylazine anesthetic cocktail and fixed in a stereotactic head frame (David Kopf Instruments, Tujunga, CA, USA). A midline scalp incision was made and the bregma was identified. Stereotactic coordinates were measured (2.0 mm lateral and 1.0 mm anterior to the bregma) for implantation of cells into the deep frontal white matter. A burr hole was drilled at this point and 1 × 105 GL261 cells or 5 × 105 U87 cells suspended in 5 μL of DMEM were injected through a Hamilton syringe (Hamilton Company, Reno, NV, USA) with a fixed, 25-gauge needle at a depth of 3.0mm relative to the dura mater. Injections were administered at 1 μL/min. After the implantation of tumor cells, the needle was slowly withdrawn, the incision sutured, and the animal monitored for recovery. All experimental studies were carried out in accordance with protocols approved by the IACUC (Institutional Animal Care and Use Committee) at the Roswell Park Cancer Institute.
Experimental Design
The basic study design for investigating the antivascular and antitumor activity of DMXAA against gliomas is shown schematically in Figure 1A. Approximately 3 weeks after implantation, high-resolution T2-weighted (T2W) MR images were acquired to confirm the presence of tumor growth. Contrast-enhanced MRI examinations were performed using T1-weighted (T1W) fast spin-echo (SE) images over a 2-day period as described below. After baseline image acquisition, DMXAA powder (Sigma, St Louis, MO, USA) was dissolved in phosphate-buffered saline or D5W solution (VWR, Bridgeport, NJ, USA) before administration. C57B16 mice bearing GL261 gliomas were treated with a single dose of DMXAA (30 mg/kg, intraperitoneally). Although this is the documented maximum tolerated dose of DMXAA in mice, we have observed that some strains of nude and SCID (severe combined immunodeficiency) mice do not tolerate this dose. Therefore, on the basis of preliminary toxicity studies carried out in the laboratory, nude mice bearing intracranial U87 gliomas were treated with a single dose of 27.5 mg/kg DMXAA (intraperitoneally). Treatment was administered to mice used for imaging studies after baseline MRI acquisition and a second set of contrast-enhanced T1W images were acquired 24 h after treatment to visualize glioma vascular response to treatment. In addition, DW-MRI was performed 72 h after treatment to detect intratumoral changes in cellularity after treatment. Treatment efficacy was assessed by monitoring survival of control and DMXAA-treated mice over a 40-day period.
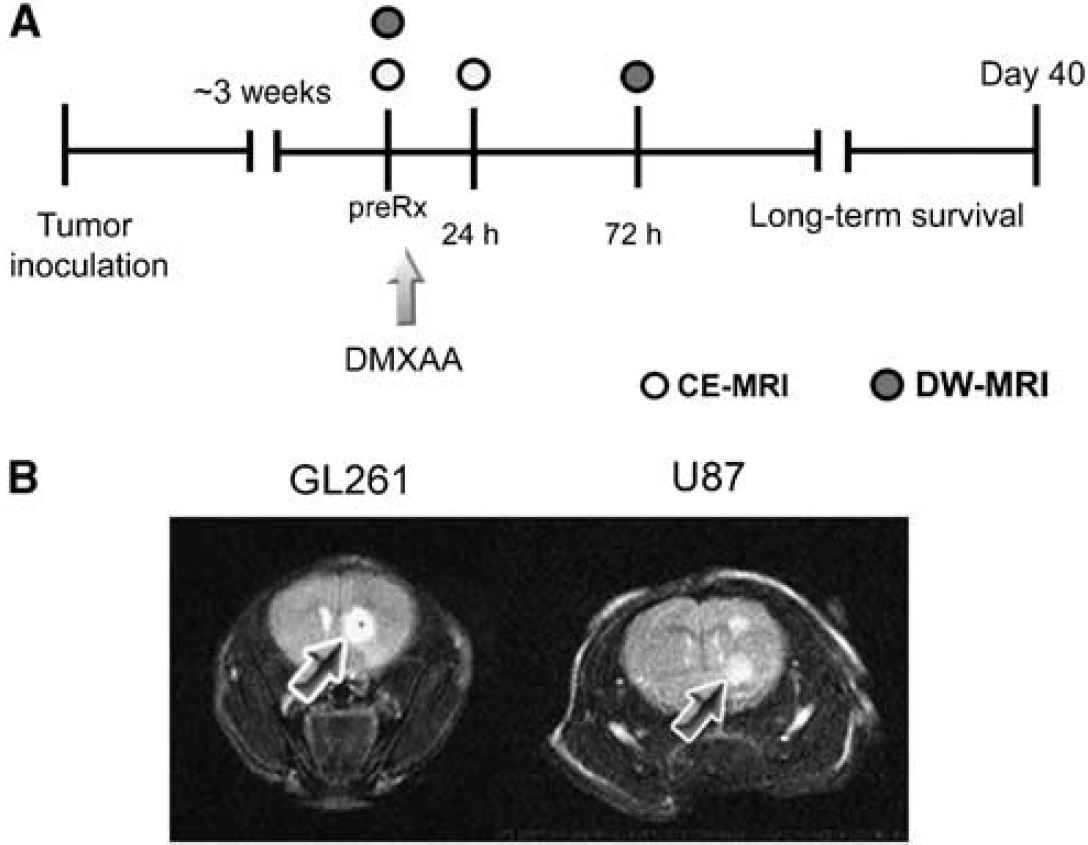
MRI of glioma response to therapy. (
Magnetic Resonance Imaging
Experimental imaging studies were carried out in a 4.7T/33-cm horizontal bore magnet (GE NMR Instruments, Fremont, CA, USA) incorporating AVANCE digital electronics (Bruker Biospec, ParaVision 3.1., Bruker Medical, Billerica, MA, USA), a removable gradient coil insert (G060, Bruker Medical) generating a maximum field strength of 950 mT/m, and a custom-designed 35 mm radiofrequency transmit/receive coil. Anesthesia was induced before image acquisition using 3% to 3.5% Isoflurane (Abbott Laboratories, Abbott Park, IL, USA) and maintained at ~2% to 2.5% during image acquisition. Animals were secured in a form-fitted MR-compatible mouse sled equipped with temperature and respiratory sensors (Dazai Research Instruments, Toronto, ON, Canada). An air heater system (SA Instruments Inc., Stony Brook, NY, USA, Model 1025) was used to maintain animal body temperature during image acquisition. A thermocouple embedded within the sled provided automated temperature control feedback. Care was taken to maintain animal body temperature and minimize motion during image acquisition.
The first set of MRI examinations was performed 8 to 10 days after intracerebral inoculation of tumor cells to confirm the successful growth of tumors. Preliminary localizer images were acquired in the sagittal and axial planes before acquisition of T1W and T2W scans. T2-weighted fast SE images were acquired on coronal and axial planes to determine the presence and extent of tumors using the following parameters: TEeff (echo time) = 75 msecs, repetition time (TR) = 3,370 msecs, echo train length = 8, FOV (field of view) = 32 mm, matrix size = 256 × 256, 1-mm-thick slices, number of averages = 4, acquisition time = 7 mins 29 secs. Contrast enhanced-MRI was performed using the intravascular contrast agent albumin-(Gd-DTPA)35 (albumin-gadopentetate dimeglumine) according to methods previously described by us (Seshadri et al, 2006). At least two to three slices of the tumor were positioned for T1 measurements using the T2W coronal images as reference. Multislice relaxation rate (R1 = 1/T1) maps were obtained using a saturation recovery, FSE (fast SE) scan with variable TRs. The scan parameters were as follows: slice thickness = 1 mm, TEeff= 25 msecs, matrix size = 128 × 96, FOV = 32 mm, echo train length = 4, TR = 360 to 6,000 msecs, acquisition time = 4 mins 50 secs. Three precontrast T1W FSE images were acquired to obtain an average estimate of precontrast T1 values. Albumin-(Gd-DTPA)35 (courtesy of Dr Robert Brasch, University of California, San Francisco, USA) was then administrated at a dose of 0.1 mmol/kg as a bolus through tail-vein injection, and a second set of seven T1W FSE images were acquired. As each individual FSE scan was ~5 mins in duration, this allowed for the estimation of R1 for ~45 mins after contrast agent administration. The T1 relaxivity of the agent as determined at the Center for Pharmaceutical and Molecular Imaging, Department of Radiology, UCSF (University of California San Francisco) was 11.0 (mmol/L secs)−1 per Gd ion, at 25°C and 10 MHz.
Diffusion-weighted MRI was performed using a multislice diffusion-weighted SE sequence with the following acquisition parameters: TE/TR= 30/1,200msecs, 128 × 128 matrix, 3.2 × 3.2 cm, diffusion gradient strength (four variable gradient strengths per acquisition) = 8, 128, 256, 420 mT/m; diffusion B value (corresponding to four variable B values per acquisition) = 2.9, 512, 2,036.3, 5,470 secs/mm2; diffusion gradient duration = 6 msecs; diffusion gradients applied in X, Y, and Z directions; number of averages = 2, 1-mm slice thickness with a total data acquisition time of 20 mins 28 secs. Measurements were obtained at baseline (before DMXAA administration) and 72 h after treatment.
Image Processing and Analysis
After image acquisition, raw image sets were transferred to a processing workstation and converted into Analyze format (AnalyzeDirect, Version 8.0, Overland Park, KS, USA). Raw data were reformatted and object maps of regions-of-interest—tumor, muscle, contralateral brain tissue, and background noise were traced manually.
The relaxation rate R1 (R1 = 1/T1) and the maximal signal intensity Smax were calculated after subtraction of background noise using the following equation:
where STR is the signal intensity obtained at each TR time. R1 values obtained from the three precontrast scans and the seven postcontrast scans were averaged for tumor, brain, and muscle tissues, and the difference between the two values was reported as normalized change in relaxation rate (ΔR1) (tumor/muscle). ΔR1 after contrast agent administration was assumed to be proportional to the concentration of the agent in tissue. R1 maps were calculated on a pixel-by-pixel basis using MATLAB (Version 7.0, Mathworks Inc., Natick, MA, USA). A low-pass filter and a pseudo-color scale were applied in Analyze for visualization.
Apparent diffusion coefficient values were computed on a pixel-by-pixel basis by fitting the images from the DW-MRI sequence to the following equation:
where Mb and Mo are the MR signal intensities with and without diffusive attenuation, respectively, and B is the diffusion-weighting factor. Apparent diffusion coefficient maps were generated by fitting the raw data to the above equation using nonlinear regression analysis in MATLAB.
Survival Analysis
Animals were observed for clinical symptoms, including weight loss (> 20%), loss of movement, seizures, and ataxia, and killed according to institutional guidelines. The log-rank test was used to analyze statistical differences between Kaplan-Meier survival curves of animals in the control and treatment groups over a 40-day period.
Statistical Considerations
All statistical analyses were performed using GraphPad Prism version 5.00 for Windows (GraphPad Software, San Diego, CA, USA, http://www.graphpad.com). Measured values are reported as the mean ± s.e. of the mean. The two-tailed t-test was used for comparing CE-MRI data at baseline and posttreatment time points and P-values <0.05 were considered statistically significant. In total, 18 C57B16 mice (12 controls, 6 treated) and 23 nude mice (13 controls, 10 treated) were used for experimental studies. The presented CE-MRI results represent paired data sets obtained at baseline and 24 h posttreatment time points for a total of nine mice (four C57B16 mice bearing GL261 gliomas and five nu/nu mice bearing U87 glioma xenografts). Reported results of DW-MRI represent paired observations obtained at baseline and 72 h posttreatment time points in three animals with GL261 gliomas. Paired two-tailed t-test analysis with Bonferroni correction was used to examine differences between baseline and 72 h posttreatment ADC values for the whole tumor, contralateral brain, and muscle tissues.
Results
T2-Weighted Magnetic Resonance Imaging
To visualize glioma growth, T2W MR images were acquired at different times after intracranial implantation of tumor cells. Both GL261 and U87 gliomas appeared as well-defined hyperintense regions (arrows) at the site of injection on noncontrast-enhanced T2W SE images (Figure 1B). The presence of tumor was confirmed using T2W MRI on all the animals used for therapeutic evaluation.
T1-Weighted Contrast Enhanced-Magnetic Resonance Imaging
The vascular response of G1261 and U87 tumors to DMXAA treatment was evaluated using CE-MRI with albumin-(Gd-DTPA)35, a well-characterized intravascular MR contrast agent that has been used extensively in preclinical studies (Schmiedl et al, 1990). The study included a baseline MR examination before DMXAA treatment and a follow-up study at 24 h after treatment. R1 maps (R1 = 1/T1) were calculated on a pixel-by-pixel basis before and after DMXAA treatment to visualize treatment-induced changes in vascular integrity. Figure 2A shows colorized postcontrast R1 maps of a C57B16 mouse brain bearing an intracranial GL261 glioma before (pretreatment) and 24 h after DMXAA treatment (24 h post). Corresponding T2W images of the brain depicting the location of the tumor (arrows) are also shown. Minimal tumor enhancement was seen after administration of the contrast agent (postcontrast 1) with no visible increase over the 45 mins postcontrast imaging period (postcontrast 7) before DMXAA treatment (pretreatment). In sharp contrast, 24 h after treatment, marked extravasation and accumulation of the contrast agent was visible on the postcontrast R1 maps of the same animal indicative of significant vascular disruption after treatment (24h post). The longitudinal relaxation rate (R1 = 1/T1) of tissues is linearly related to contrast agent concentration. Therefore, the mean ΔR1 values of the tumor were calculated and normalized to ΔR1 muscle tissue to provide an indirect estimate of intratumoral contrast agent concentration at baseline and posttreatment time points. As shown in Figure 2B, a near five-fold increase (P < 0.001, n = 4) in normalized ΔR1 tumor/muscle value (1.7 ± 0.26) was observed at 24 h after treatment compared with baseline estimates (0.36 ± 0.05) indicative of DMXAA-induced vascular disruption.
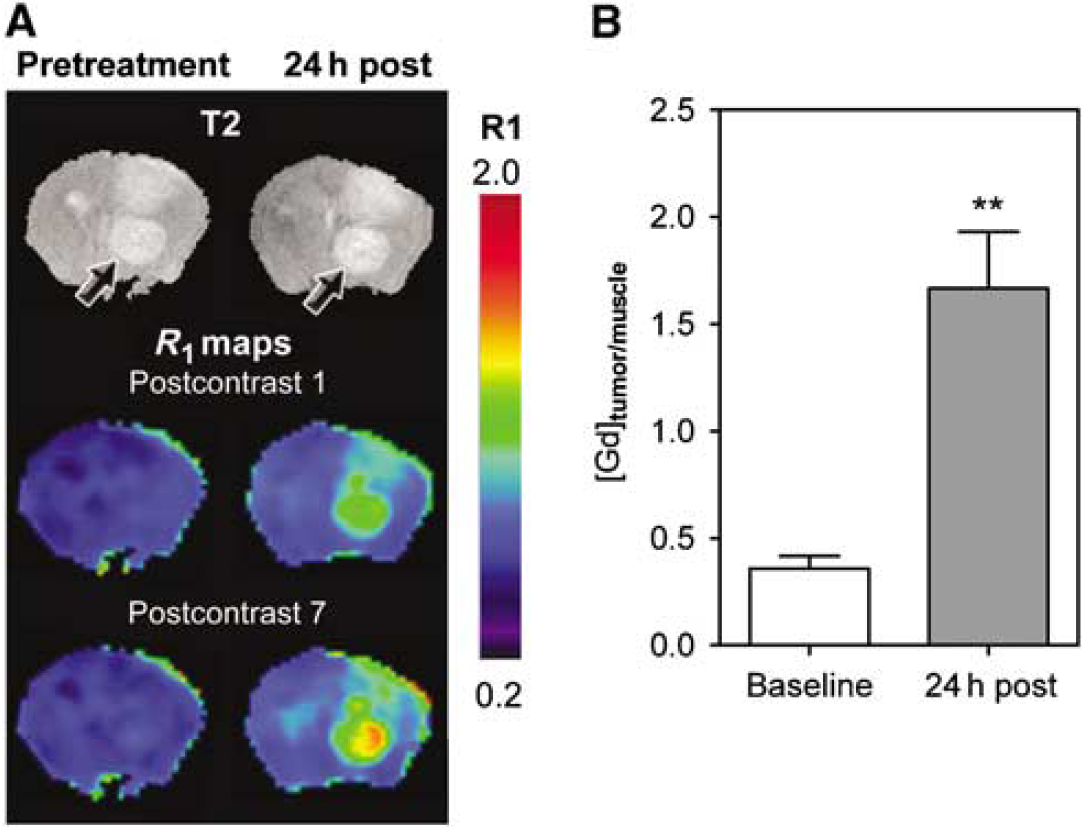
Contrast-enhanced MRI of response of GL261 gliomas to VDA therapy. (
Using the same study design, the vascular response of U87 gliomas was investigated. Baseline and posttreatment R1 maps (first and last postcontrast scans) of a nude mouse bearing a U87 glioma are shown in Figure 3A. Similar to GL261 tumors, minimal tumor enhancement was seen at baseline. Twenty-four hours after treatment, evidence of vascular disruption in the form of increased contrast agent accumulation within the tumor was observed on postcontrast R1 maps (Figure 3A). However, visible changes in R1 maps were much less pronounced in U87 xenografts compared with that in GL261 tumors. Normalized ΔR1tumor/muscle values of U87 gliomas (Figure 3B) also showed only a minimal increase in contrast agent concentration at the 24-h time point (0.85 ± 0.04) compared with baseline estimates (0.65 ± 0.07, P < 0.05, n = 5).
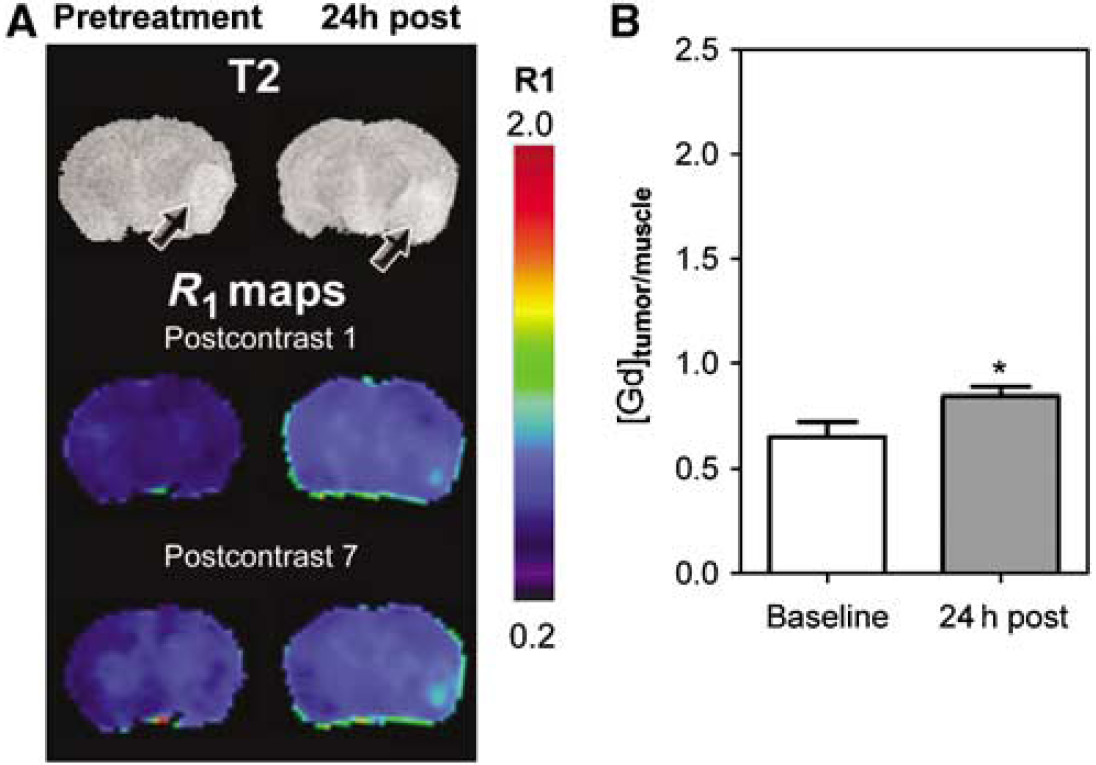
Vascular response of U87 gliomas to DMXAA. (
Diffusion-Weighted Magnetic Resonance Imaging
Diffusion weighted-MRI was performed 72 h after treatment and ADC maps were calculated to examine changes in water mobility as a measure of tumor response to DMXAA. Figure 4A shows pseudocolorized ADC maps of a GL261 glioma overlaid on the corresponding T2W images of a C57B16 mouse before and 72 h after therapy. Enlarged views of the tumor (regions-of-interest) are also shown. Regions of higher ADC were observed in GL261 gliomas at the 72-h time point compared with baseline measurements indicative of a response. Apparent diffusion coefficient values of all the three animals scanned at the 72 h posttreatment time point showed an increase compared with baseline estimates (Figure 4B). The mean ADC values of all the three animals at baseline was calculated to be 0.67 ± 0.06 (1 × 10−3mm2/sec. Seventy-two hours after treatment, an ~13% increase in the mean ADC values (0.75 ± 0.05, P = 0.015) was observed in GL261 gliomas. Diffusion weighted-MRI of nude mice bearing U87 gliomas showed no significant difference in ADC values (data not shown) 72 h after DMXAA treatment (n = 3) compared with baseline values or untreated controls (n = 4). Statistical analysis of ADC values of the contralateral normal brain tissue did not show any difference between the two time points.
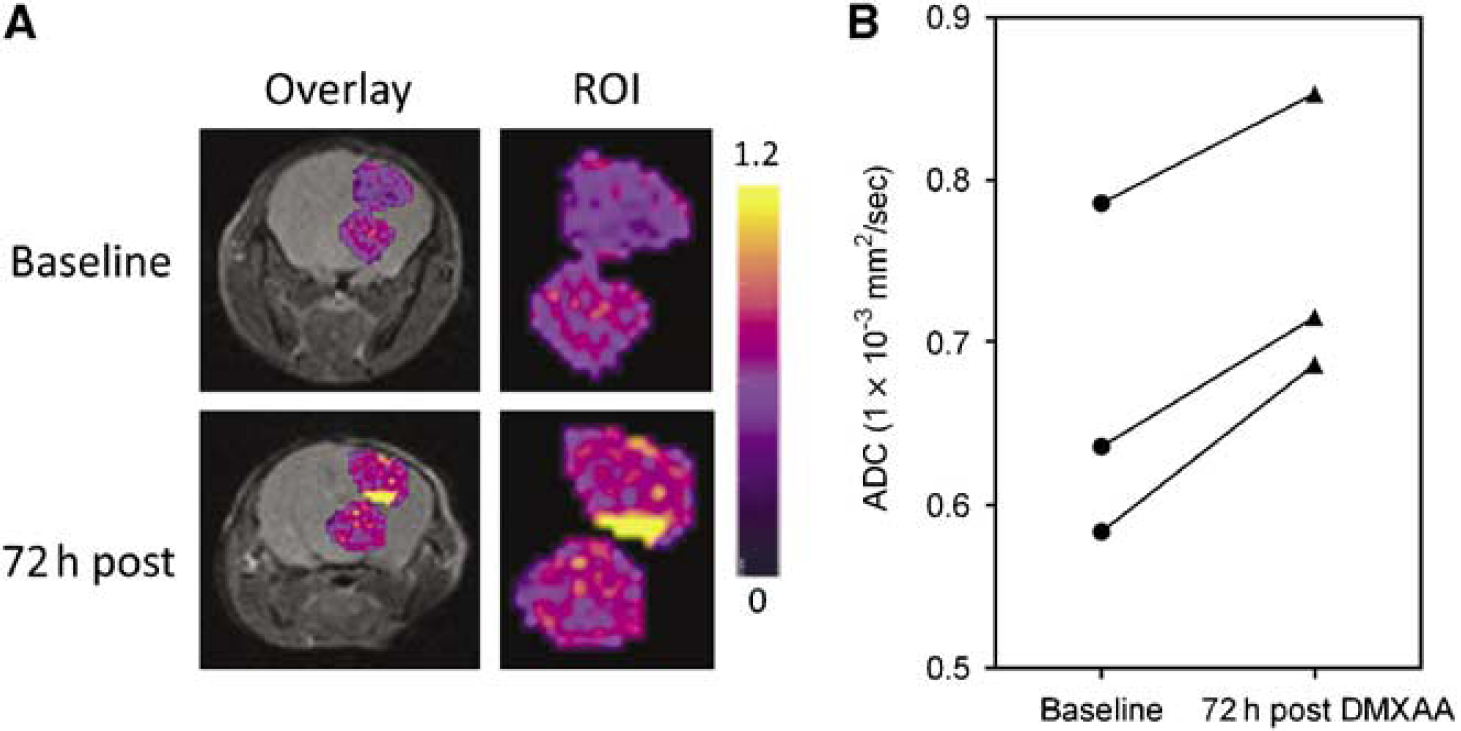
DW-MRI of GL261 response to 5,6-dimethylxanthenone-4-acetic acid (DMXAA). (
Long-Term Efficacy of Vascular Disrupting Agent Therapy
We then examined the long-term consequence of tumor vascular disruption induced by DMXAA in both glioma models by monitoring long-term survival after treatment. The median survival of control and DMXAA-treated animals was calculated using the method of Kaplan and Meier, and differences were analyzed for statistical significance using the log-rank test. As shown in Figure 5, a significant but differential increase in median survival was observed after DMXAA treatment in GL261 and U87 models. The median overall survival of control C57B16 (n = 12) mice bearing GL261 gliomas was 19.5 days (Figure 5A). In comparison, GL261 tumor-bearing animals treated with DMXAA (n = 6) showed a median survival of 29 days (P=0.003, log-rank test). In the U87 xenograft model, DMXAA-treated animals (n = 10) showed a median survival of 34 days (P = 0.0005, log-rank test) compared with untreated control animals (n = 13) that showed a median survival of 26 days from the day of implantation (Figure 5B). Overall, animals treated with DMXAA showed significantly prolonged survival (~50% for GL261 and ~30% for U87 gliomas) compared with untreated controls.
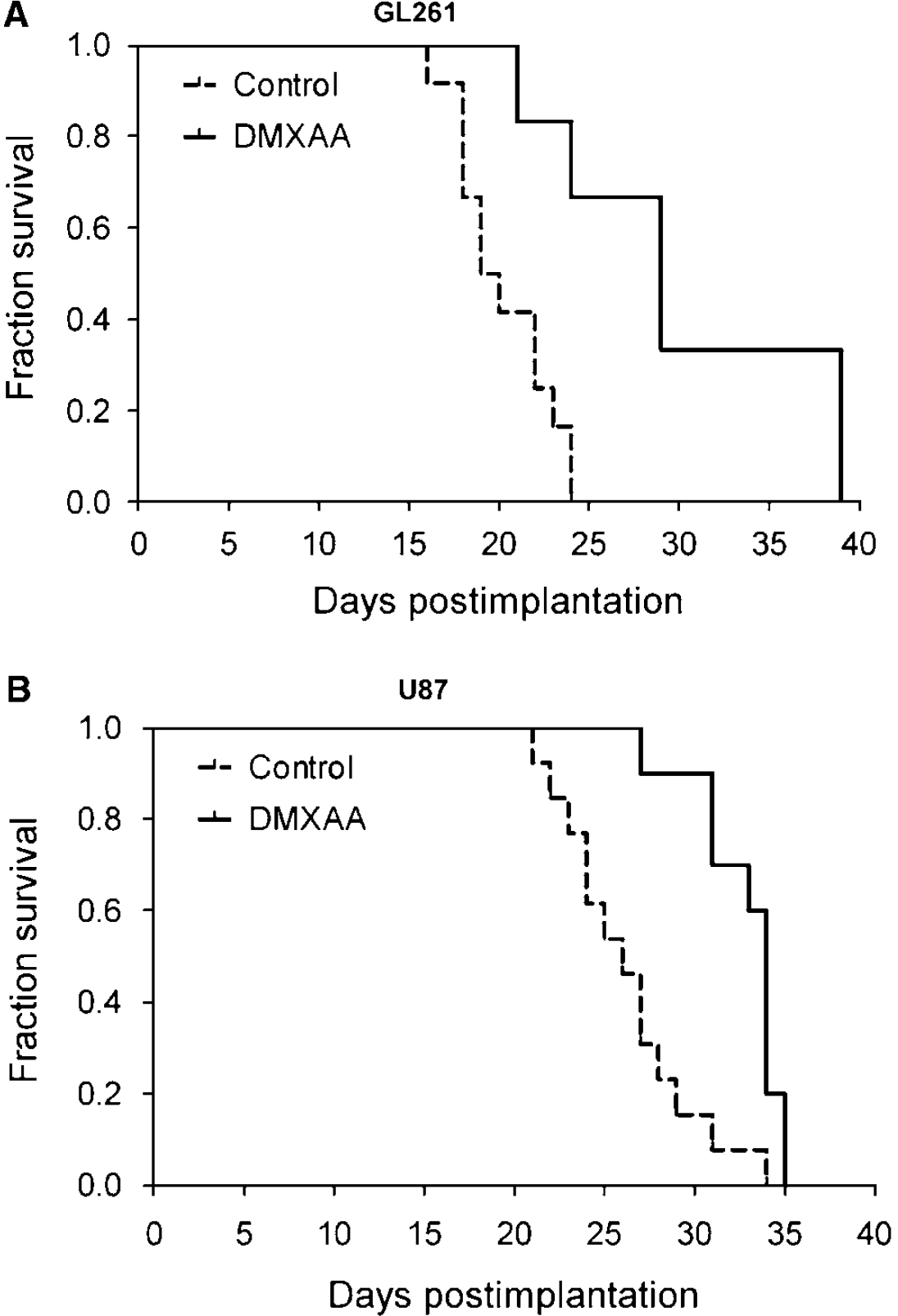
Therapeutic efficacy of 5,6-dimethylxanthenone-4-acetic acid (DMXAA) against gliomas. Kaplan-Meier survival analysis of GL261 (
Discussion
The aggressive clinical course of gliomas often limits treatment options and contributes to poor long-term survival in patients. Therefore, the need to investigate and develop novel and effective therapies in gliomas is clearly evident.
The molecular and phenotypic differences between normal tissue vasculature and tumor-associated vasculature offer a unique opportunity that has been exploited for selective therapeutic targeting (Eberhard et al, 2000; Ruoslahti, 2002). This has been pursued primarily using two approaches, namely (1) antiangiogenic agents, such as bevacizumab and DC101, that are aimed at preventing or inhibiting new vessel formation typically by targeting a specific angiogenic molecule or its membrane receptor (Jain, 2001; Tong et al, 2004) and (2) VDAs that selectively destroy existing tumor vessels (Kelland, 2005). Examples of VDAs include combretastatin, ZD6126, and the small molecule DMXAA (ASA404). It is believed that VDAs differ from antiangiogenic agents both in their mode of action and in their potential clinical application. Vascular disrupting agents are targeted toward larger solid tumors with established vasculature in contrast to antiangiogenic agents targeted toward smaller tumors with associated neovasculature (Kelland, 2005).
Gliomas are highly angiogenic, aggressive brain tumors that are often nonresponsive to therapy (Brem, 1976; Abdulrauf et al, 1998; Cao et al, 2006). Changes associated with angiogenesis in gliomas have been correlated with an aggressive disease phenotype and poor clinical outcome (Abdulrauf et al, 1998; Cao et al, 2006). These observations have led to the investigation of the potential of antiangiogenic agents in gliomas in preclinical and clinical settings (Claes et al, 2008; Narayana et al, 2009). However, the potential of VDAs against gliomas has not been reported extensively. Therefore, in this study, we investigated the antivascular activity and efficacy of the tumor-VDA DMXAA against gliomas. The agent has been shown to be well-tolerated in Phase I clinical trials (McKeage et al, 2006). Results of a randomized Phase II clinical trial in patients with non-small-cell lung cancer has also shown improvement efficacy with DMXAA in combination with carboplatin and paclitaxel (McKeage et al, 2008). Using MRI, we examined the response of intracranial GL261 murine gliomas and U87 human glioma xenografts to VDA therapy along with long-term survival analysis. Our results show potent antivascular activity of DMXAA that translated into a survival benefit in both models evaluated.
Radiologic techniques are important components of the diagnostic and prognostic armamentarium in neuro-oncology (Gerstner et al, 2008). A number of noninvasive imaging methods, including PET, perfusion computed tomography, and MRI are currently being used to evaluate the activity of targeted therapies in clinical trials. Contrast enhancement within tissue detected by MRI or computed tomography is commonly used as an indicator of malignant progression in gliomas (Gerstner et al, 2008; Sorensen et al, 2008). Clinical trials of antiangiogenic agents have used CE-MRI for the assessment of biologic activity with encouraging results (Akella et al, 2004). Most CE-MRI studies are typically carried out using a paramagnetic contrast agent that results in the shortening of the longitudinal (TJ relaxation time of tissues. Tissue blood volume is extracted from changes in signal intensity through the application of a pharmacokinetic model with associated assumptions. However, the use of freely diffusible tracers has led to difficulties in interpretation, particularly after treatment with antiangiogenic agents.
The amount and rate of contrast agent uptake within a tissue after intravenous administration is related to the extent of tissue blood supply (perfusion) and transendothelial transport (diffusion) of the agent. With small molecular weight agents that freely diffuse across the endothelium, the intravascular concentration of the contrast agent after administration of a bolus injection decreases with time during the course of a single MR examination. As large molecular weight contrast agents show minimal transendothelial diffusion and remain intravascular for longer periods of time, these agents are considered to be more suited as probes for assessing tumor vascular permeability compared with small molecular weight agents (Brasch et al, 1997; Barrett et al, 2006). Therefore, in this study, CE-MRI was performed with an intravascular contrast agent to characterize the vascular response of gliomas to VDA therapy. The agent used in our study has been well-characterized and widely used in preclinical studies to estimate tumor vascular permeability (Schmiedl et al, 1990; Raatschen et al, 2008). As the relaxation rate of tissues and not signal intensity is linearly related to contrast agent concentration, the change in tissue longitudinal relaxation rate (ΔR1) after intravenous administration of the contrast agent was used as an indirect estimate of its tissue concentration. Dynamic R1 mapping was used to visualize the effect of VDA therapy on glioma vasculature.
Antiangiogenic agents have been shown to decrease tumor vascular permeability and interstitial fluid pressure while inhibiting new vessel formation (Tong et al, 2004). These ‘normalizing’ effects are believed to contribute to a functionally efficient vascular network thereby, improving drug delivery and penetration (Jain, 2001). In contrast, VDAs, such as combretastatin-A4-phosphate and DMXAA, affect the structure and integrity of the tumor endothelial lining resulting in alterations in vascular permeability, eventually leading to blood flow stasis and shutdown (Tozer et al, 2005; Kelland, 2005). Previous reports by us and others have shown increased vascular permeability as the major mechanism of action of the VDA DMXAA (Zhao et al, 2005; Seshadri et al, 2005). Consistent with these known, previously observed biologic effects of VDAs, the results of our CE-MRI studies provided evidence of marked alterations in vascular permeability in both models 24 h after treatment (Figure 2 and Figure 3). Treatment with DMXAA led to considerable extravasation of the contrast agent as shown by the significant increase in R1 after treatment compared with baseline levels.
In addition to CE-MRI, we used DW-MRI to assess changes in the cellularity of gliomas after treatment. The technique is widely being investigated in preclinical and clinical systems for its utility as a biomarker of disease and therapeutic response (Lee et al, 2006; Hamstra et al, 2008). The principles and the biologic basis of DW-MRI have been described extensively (Koh and Collins, 2007; Hamstra et al, 2008). The technique measures the random Brownian motion of water molecules within biologic tissues (free diffusion) as an indirect measure of tissue cellularity and membrane integrity (Hamstra et al, 2008). In DW-MRI, a quantitative estimate of the mobility of water protons is obtained by calculating the ADC from the detected attenuation in signal intensity within tissue (Koh and Collins, 2007; Lee et al, 2006). Parametric mapping of ADC values provides a visual estimate of changes in cellularity within a given tissue of interest. Highly cellular regions in tissue with restricted water diffusion are associated with low ADC values and correspondingly, regions with low cellularity show higher ADC values (Lee et al, 2006; Hamstra et al, 2008; Koh and Collins, 2007).
In this study, DW-MRI showed a significant increase in the mean ADC values of GL261 gliomas 72 h after treatment compared with baseline estimates (Figure 4). Apparent diffusion coefficient maps showed a heterogeneous pattern of response to DMXAA at the 72-h time point. This could be reflective of the spatial variation in tumor vascular damage induced by DMXAA, because VDAs are believed to be more effective in the central regions of the tumor containing established vasculature (Tozer et al, 2005). The classic pattern of tumor response to VDAs reported in preclinical studies involves induction of central necrosis with a fraction of viable cells found in the periphery that survive treatment (Tozer et al, 2005). Although spatial correlation between the vascular damage (R1 change) and cell death (ADC change) would have yielded useful results, this was not performed because of the difference in time points between CE-MRI and ADC data acquisitions.
Consistent with the less-pronounced vascular response observed with CE-MRI, DW-MRI of U87 xenografts did not show a significant change in ADC after treatment. This is not surprising when considering the difference in DMXAA dose used between the two models. 5,6-Dimethylxanthenone-4-acetic acid has been observed to show a rather steep dose—response curve in preclinical model systems with significant species and strain differences in pharmacokinetics. As the aim of our study was to evaluate the response of murine gliomas and human glioma xenografts to DMXAA rather than to compare differences in their response, we used two different but well-tolerated doses of DMXAA (30 mg/kg for GL261 tumors and 27.5 mg/kg for U87 tumors). This could at least partly explain the differences in the degree of response between the two models as detected by DW-MRI and the survival benefit observed (10 days for GL261; 8 days for U87). In addition, the vascular disruptive effects of DMXAA are a consequence of both direct drug effects on the endothelium and indirect effects through induction of cytokines, such as tumor necrosis factor-α (Baguley and Wilson, 2002). In a recent study, we have shown differences in cytokine induction and the vascular response of ectopic and orthotopic murine fibrosarcomas established in C57B16 mice treated with the same dose of DMXAA (Seshadri et al, 2008). Therefore, it is plausible that variable degrees of cytokine induction between GL261 and U87 gliomas after DMXAA treatment could have also contributed to the observed differences in vascular response and survival. However, given differences in the underlying tumor biology (disease aggression, angiogenic characteristics) between the two models, it is difficult to obtain valid conclusions on the differential response observed between U87 and GL261 models. One potential alternative that could be considered for future studies would be to examine the possibility of implanting GL261 tumors in nude mice. Such a design would eliminate one variable (mouse strain) and enable utilization of the same dose of the agent against both tumors.
Finally, a discussion of the implications and the limitations of the study are warranted. Although only a single dose of DMXAA was evaluated, treatment with a single injection of the tumor-VDA resulted in a statistically significant survival benefit in both glioma models evaluated (Figure 5). However, we observed no evidence of ‘cure’ with VDA treatment. Given the data obtained from several preclinical reports (Baguley and Wilson, 2002), suggesting that the true value of VDAs lies in their use in combination with chemotherapy or radiotherapy, this is not an entirely surprising observation. Second, the blood-brain barrier is a critical factor that influences delivery of chemotherapeutic agents to brain tumors (Gerstner and Fine, 2007). Studies in preclinical models have shown that treatment with antiangiogenic agents reduces the permeability of the blood-brain barrier through stabilization of the vasculature (Claes et al, 2008; Mathieu et al, 2008). In contrast, treatment with hyper osmotic agents, such as mannitol, have also been shown to result in the disruption of the blood-brain barrier and contribute to enhanced efficacy of boron neutron capture therapy (Barth et al, 2000). In our study, CE-MRI provided evidence of disruption of the blood-brain barrier after VDA treatment in both glioma models. Therefore, it could be hypothesized that optimization of dose and schedule of VDAs, such as DMXAA, in combination with chemotherapy, would potentially enable increased drug delivery to gliomas. We are currently planning to evaluate the combination of DMXAA with chemotherapeutic agents, such as temozolomide, against gliomas to test this hypothesis. Finally, there has been an increased interest in the identification of early biomarkers that can reliably predict disease aggression or therapeutic response. With the increased development of targeted therapies, it is believed that imaging methods are likely to provide indirect evidence of the early biologic activity of these agents. Specific MRI parameters of tumor vascularity and cellularity are currently being investigated for their potential value in predicting tumor response to conventional and novel anticancer therapies. In our study, CE-MRI allowed measurement of the early changes in vascular permeability after DMXAA treatment. This observation is particularly important as enhancement of vascular permeability is the major mechanism of action of the agent consistently observed in preclinical models (Zhao et al, 2005; Seshadri et al, 2005). Therefore, MRI measurements of permeability using an intravascular contrast agent could potentially serve as a surrogate marker of efficacy that is mechanistically related to the pharmacologic activity of DMXAA. However, in our study, because of the relatively small sample size used in the imaging study, we did not perform a direct correlation between the early vascular response and treatment outcome. Further examination with a larger cohort of animals is required to determine the true predictive ability of such MRI-based biomarkers. Recent clinical approval of the intravascular MR contrast agent, MS-325 (Vasovist) for MR angiography at least offers the potential of incorporating the methodology for the assessment of biologic activity of VDAs in patients in the future.
In conclusion, our findings show proof of principle of the potential of antivascular therapy in gliomas. To the best of our knowledge, this is the first report on the antivascular and antitumor activity of DMXAA in two orthotopic glioma models.
Footnotes
Acknowledgements
The authors would like to thank Drs Robert Brasch and Yanjun Fu (University of California, San Francisco) for providing albumin-(Gd-DTPA); Dr Joseph Spernyak (RPCI) for useful discussions; and Steve Turowski and Alison Arter for their technical assistance.