Abstract
Focal cerebral ischemia elicits strong inflammatory responses involving activation of resident microglia and recruitment of monocytes/macrophages. These cells express peripheral benzodiazepine receptors (PBRs) and can be visualized by positron emission tomography (PET) using [11C]PK11195 that selectively binds to PBRs. Earlier research suggests that transient ischemia in rats induces increased [11C]PK11195 binding within the infarct core. In this study, we investigated the expression of PBRs during permanent ischemia in rats. Permanent cerebral ischemia was induced by injection of macrospheres into the middle cerebral artery. Multimodal imaging 7 days after ischemia comprised (1) magnetic resonance imaging that assessed the extent of infarcts; (2) [18F]-2-fluoro-2-deoxy-d-glucose ([18F]FDG)-PET characterizing cerebral glucose transport and metabolism; and (3) [11C]PK11195-PET detecting neuroinflammation. Immunohistochemistry verified ischemic damage and neuroinflammatory processes. Contrasting with earlier data for transient ischemia, no [11C]PK11195 binding was found in the infarct core. Rather, permanent ischemia caused increased [11C]PK11195 binding in the normoperfused peri-infarct zone (mean standard uptake value (SUV): 1.93 ± 0.49), colocalizing with a 60% increase in the [18F]FDG metabolic rate constant with accumulated activated microglia and macrophages. These results suggest that after permanent focal ischemia, neuroinflammation occurring in the normoperfused peri-infarct zone goes along with increased energy demand, therefore extending the tissue at risk to areas adjacent to the infarct.
Introduction
Focal cerebral ischemia elicits strong inflammatory responses involving the rapid activation of glial cells (microglia, astrocytes) and recruitment of hematogenous cells (granulocytes, T cells, monocytes/macrophages) from the blood stream triggered by the upregulation of cell adhesion molecules, chemokines, and cytokines. These multifaceted responses follow specific temporospatial patterns (Hallenbeck et al, 1986; Schroeter et al, 1994; Schroeter et al, 1999; Mabuchi et al, 2000; Schroeter et al, 2001; Wang et al, 2008) and are termed ‘neuroinflammation.’ Although responses of inflammatory cells are only part of the complex process of neuroinflammation, in this study, we use them as a surrogate marker for the localization of neuroinflammatory processes overall. Different cell types involved in the neuroinflammatory response, namely activated microglia (Stephenson et al, 1995; Ji et al, 2008) and blood-borne cells of the monocyte-macrophage lineage (Zavala and Lenfant, 1987; Stephenson et al, 1995), express peripheral benzodiazepine receptors (PBRs). This expression pattern renders them detectable by the isoquinoline carboxamide PK11195 that selectively binds to PBRs (Benavides et al, 1983). In addition, reactive—but not resting—astrocytes demarcating the ischemic lesion from vital brain tissue have recently been shown to express PBRs (Chen et al, 2004; Rojas et al, 2007). Under physiologic conditions, PBR expression in the brain is undetectable by [11C]PK11195-PET. A recent study showed that an approximately four-fold increase in PBR mRNA levels leads to detectable [11C]PK11195 binding
11C-labeled PK11195 enables
We hypothesized that in permanent ischemia, increased [11C]PK11195 uptake marking an overexpression of PBRs would predominantly colocalize with activated microglia and macrophages in regions with preserved blood flow. We sought to characterize the metabolic situation in those areas that are subjected to inflammatory processes, to assess the risk of secondary damage of the respective tissue. To induce permanent ischemia, we used a macrosphere injection model of focal ischemia in rats that mimics arterio-arterial embolism, the leading etiology of human stroke (Mohr, 1993; Barnett et al, 2000; Grau et al, 2001; Prabhakaran et al, 2006).
Material and methods
Animals and Surgery
All animal procedures were carried out in accordance with the German Laws for Animal Protection and were approved by the local animal care committee and by the local governmental authorities. Spontaneously breathing male Wistar rats weighing 290 to 330 g (
Radiochemistry
Radiosynthesis of [11C]PK11195 was accomplished by
[18F]-2-fluoro-2-deoxy-d-glucose ([18F]FDG) was produced as described earlier (Hamacher et al, 1986).
Positron Emission Tomography
Seven days after the induction of ischemia, PET imaging was performed on a microPET Focus 220 scanner (Concorde Microsystems, Inc., Knoxville, TN, USA; 63 image planes; 1.5-mm full width at the half maximum). Animals were anesthetized with 5% isoflurane, maintained with 2% isoflurane in 65%/ 35% nitrous oxide/oxygen, and placed in the scanner. The temperature was monitored using a rectal probe and maintained at 37 ± 0.5°C by a warm heating pad. After a 10-min transmission scan for attenuation correction, rats received an intravenous bolus injection of [11C]PK11195 (1.1 to 2.5 mCi per rat), and the emission data were acquired for 30 mins. After data acquisition, animals remained in the scanner under anesthesia. [18F]-2-fluoro-2-deoxy-D-glucose-PET was performed in the same session and without changing the position of the animal between scans, thus allowing for a precise coregistration of the resulting images. Independent measurements were ensured by using the 11C-labeled radiotracer first and waiting for five half-lives before the next tracer injection; i.e., 100 mins after [11C]PK11195 injection, animals received an intravenous bolus injection of [18F]FDG (1.8 to 2.3 mCi per rat), and the emission data were acquired for 60mins. After PET imaging, blood was collected from the tail vein to determine the blood glucose level. [11C]PK11195-PET data were reconstructed using a two-dimensional filtered back projection of the total uptake over the 30-min acquisition time. The [18F]FDG-PET data were reconstructed in time frames of 6 × 30, 3 × 60, 3 × 120, and 12 × 240 secs. An image-derived input function was extracted from the [18F]FDG-PET data, and parametric images from the kinetic constants were determined by applying a two-tissue-compartment kinetic model with four rate constants and the blood volume as free parameters. Therefore,
Magnetic Resonance Imaging (MRI)
On day 7 after induction of ischemia, and immediately before PET imaging, rats were reanesthetized with isoflurane, and experiments were carried out on a 4.7 T BioSpec system (Bruker BioSpin, Ettlingen, Germany) with a 30-cm bore horizontal magnet, equipped with a self-shielded gradient system (max gradient: 100 mT/m; rise time: <250 μsecs). Radio frequency transmission was achieved using a Helmholtz coil (12-cm diameter), and the signal was detected using a 22-mm diameter surface coil. The animals were positioned prone in a dedicated cradle using a stereotactic headholder with the surface coil placed directly over the head. Twelve contiguous 1-mm-thick T2-weighted coronal images of the brain were acquired (relaxation time/echo time = 3,000/ 14 msecs, 16 echoes, field of view = 3.0 cm, matrix 128 × 128) using a multislice multiecho Carr–Purcell–Meiboom–Gillsequence. During scanning, body temperature and respiration rate were monitored and maintained using a combination of in-house equipment and DASYLab (DasyLab, Moenchengladbach, Germany) version 9.0 software.
Image Analysis
For image coregistration, the image analysis software VINCI (MPI, Cologne, Germany) was used, allowing for a fast automated coregistration of all multimodal imaging data as described earlier (Cizek et al, 2004). Positron emission tomography images were additionally coregistered to anatomic data of a three-dimensional rat brain atlas constructed from the brain slices presented by Swanson (2003).
Quantitative data evaluation was based on a region of interest (ROI) analysis of PET images to determine local radiotracer uptake,
For the [11CjPKlligB-PET data, a standard uptake value (SUV) was calculated for each ROI, dividing the mean ROI activity by the injected radioactive dose per body weight. Standard uptake values were individually determined and then averaged between animals.
Immunohistochemistry
After PET imaging, rats were deeply anesthetized and decapitated. The brains were rapidly removed, frozen in isopentane, and stored at −80°C before further histologic and immunohistochemical processing. Adjacent serial coronal brain sections 10- μm-thick were cut at 500- μm intervals and stained using (1) H&E (hematoxylin and eosin) to depict histology; (2) mAb against the CD11b antigen to identify activated microglia/macrophages (clone OX42, dilution 1:1,000, AbD Serotec, Oxford, UK, cat-#MCA275R); or (3) mAB against the CD68 antigen to identify phagocytic microglia/macrophages (clone ED1, dilution 1:1,000, AbD Serotec, cat-#MCA341). The H&E staining was performed according to standard protocols. For visualization of anti-CD11b and anti-CD68, the ABC Elite kit (Vector Laboratories, Burlingame, CA, USA) with diaminobenzidine (Sigma, Munich, Germany) as the final reaction product was used.
Owing to a high cell density within the area of interest, and using cytoplasmatic stainings, a quantitative assessment of cell numbers was not feasible. To quantify cell accumulation in different regions, elliptic ROIs were placed in the infarct core, infarct rim, and in homotopic contralateral regions. The mean optical density of ROIs was assessed using the ImageJ software (National Institutes of Health, Bethesda, MD, USA, Version 1.38x, http://rsb.info.nih.gov/ij/).
Statistical Analysis
Descriptive statistics and Student's
Results
Characterization of Macrosphere-Induced Focal Cerebral Ischemia by Multimodal Imaging In Vivo
In our macrosphere model of cerebral ischemia, four TiO2 spheres were injected into the ICA after ligation of the pterygopalatine artery (Figure 1, arrows), leading to a permanent occlusion within the ICA-MCA vessel arborization. Using multimodal imaging, the individual infarct was characterized with MRI depicting the exact anatomic location and extent of the infarct, and with the
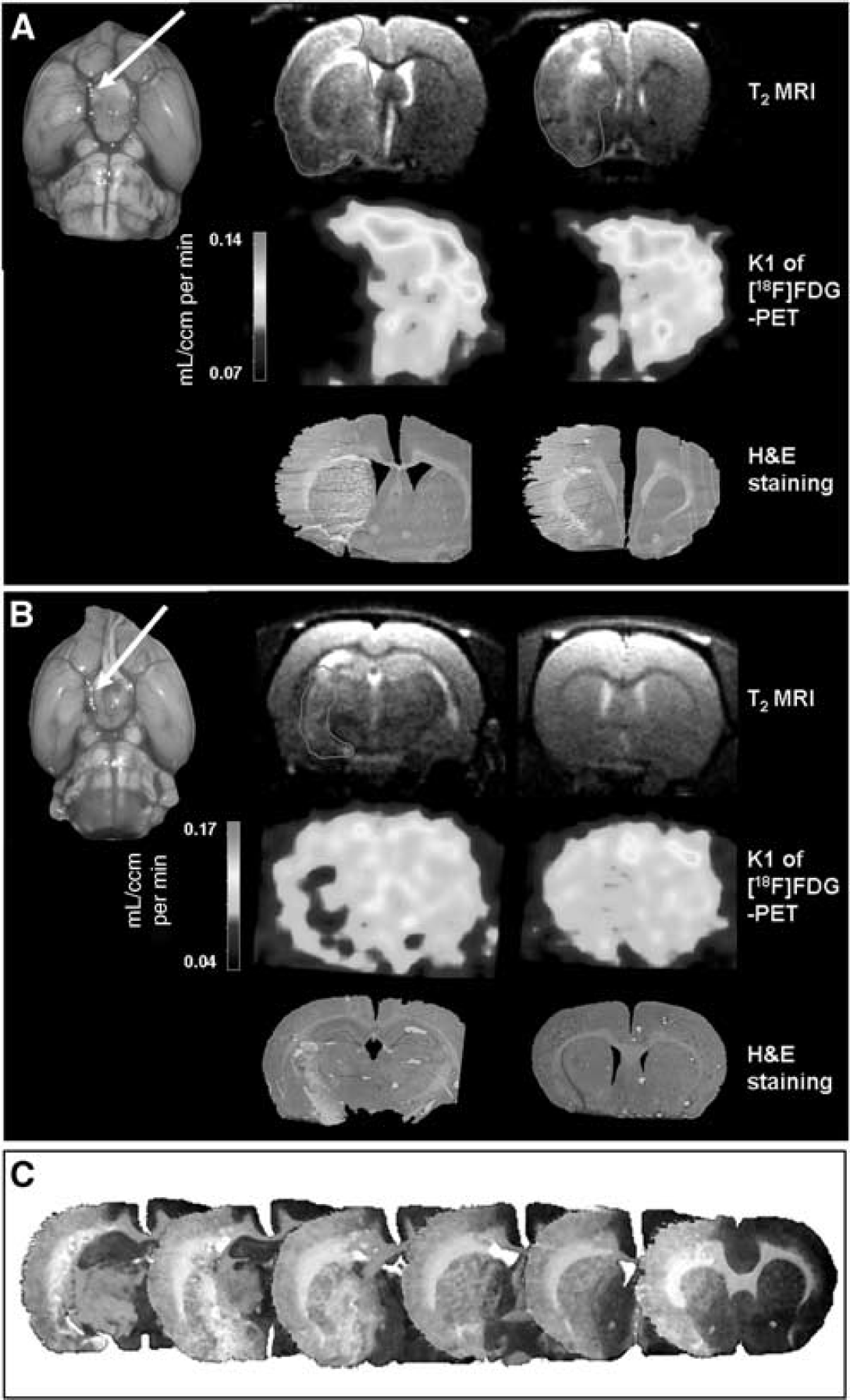
Multimodal imaging characterized the variable extent of macrosphere-induced cerebral ischemia
Neuroinflammatory Processes in the Macrosphere Model of Permanent Cerebral Ischemia
Neuroinflammatory processes were characterized immunohistochemically. All lesions showed pannecrosis within the infarct core. Some CD11b-positive microglia were found within the necrotic core of the ischemic lesion as well, but those showed signs of disintegration (Figure 2A, arrow). As a constitutive expression, resting microglia showed a ramified shape and faint CD11b immunoreactivity in the contralateral hemisphere. Activated microglia and macrophages, as identified by CD11b immunoreactivity, were detected in the zone surrounding the infarct, constituting a thick rim bordering the lesion. In this area, intensely CD11b-positive microglial cells showed stellate morphology with stout processes (Figure 2A). This hypercellular rim of resident and infiltrating immune cells showed a significantly higher optical density (0.36 ± 0.025) than homotopic contralateral regions (0.25 ± 0.025,
Characterization of distinct local zones caused by cerebral ischemia
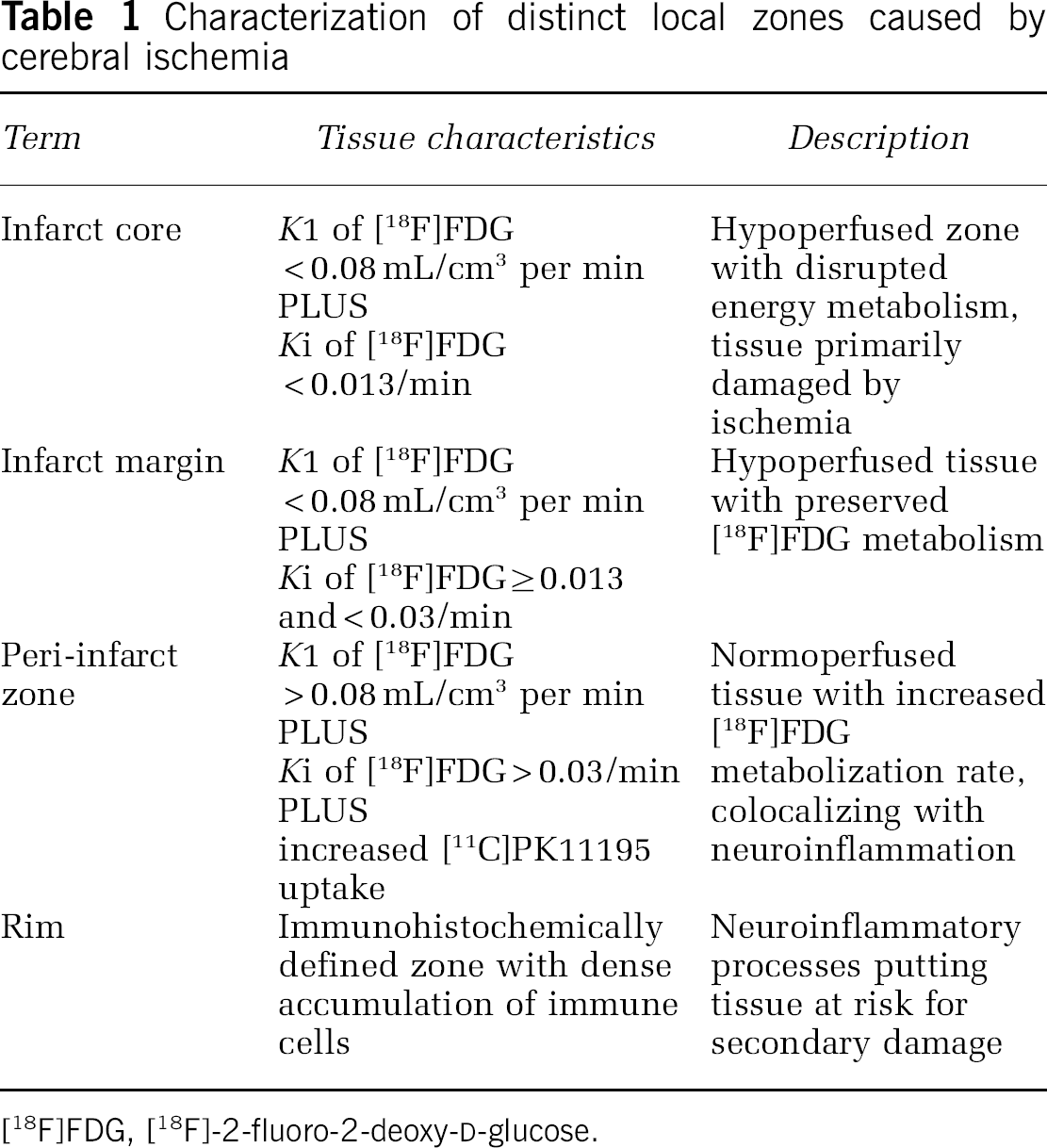
[18F]FDG, [18F]-2-fluoro-2-deoxy-d-glucose.
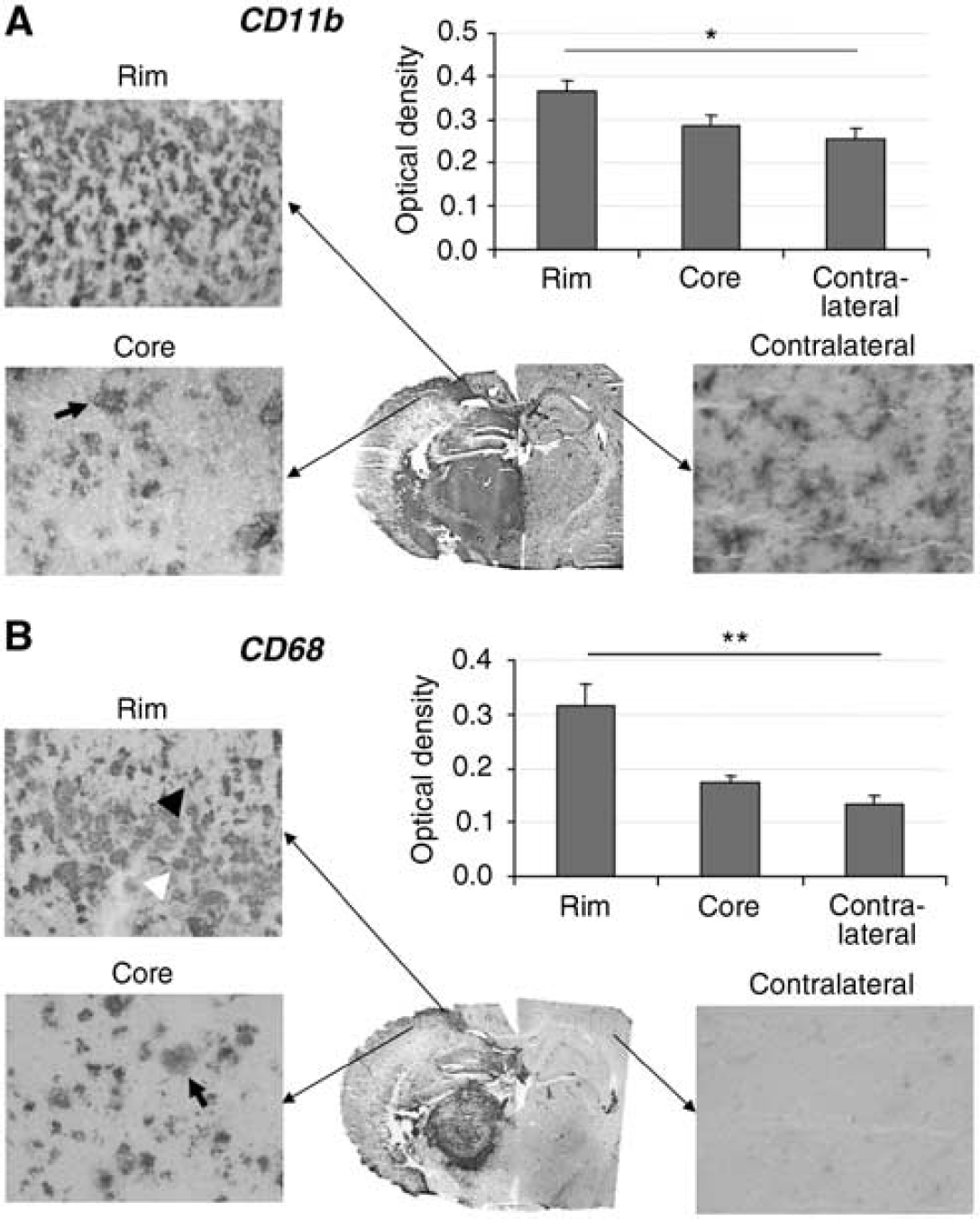
Characteristic neuroinflammation occurred in the macrosphere model of permanent cerebral ischemia. (
In Vivo Imaging of Neuroinflammation Using [11C]PK11195 and [18F]FDG
As detailed in the Material and Methods section, both [11C]PK11195- and [18F]FDG-PET were performed in the same session without changing the position of the animal between the scans, thus allowing a precise coregistration of both radiotracers. The [18F]FDG influx (
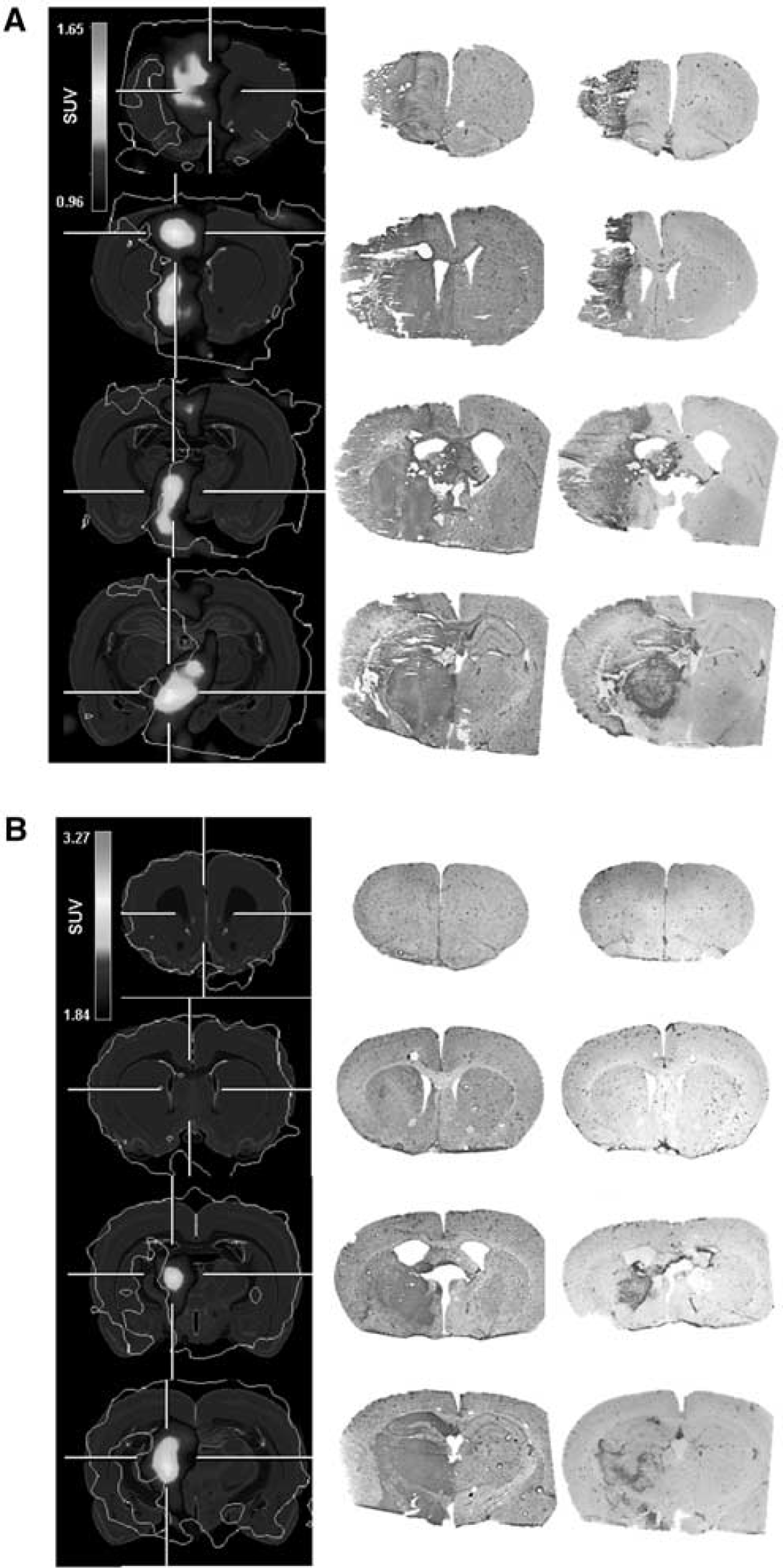
Increased [11C]PK11195 binding was observed only in areas of normoperfusion, but never within the ischemic core itself (Figure 3, left column). More specifically, [11C]PK11195 uptake was restricted to the perfused tissue of the peri-infarct zone in all experimental animals independent of the variable location and size of the infarction. The localization of the [11C]PK11195 signal corresponded well to the massive accumulation of activated microglia and macrophages as depicted by CD11b immunohistochemistry (Figure 3, middle column), and to CD68 + phagocytosing cells as far as they were located in perfused areas (Figure 3, right column).
Functional Characterization of a Distinct Neuroinflammatory Peri-infarct Zone
We used the rate constant
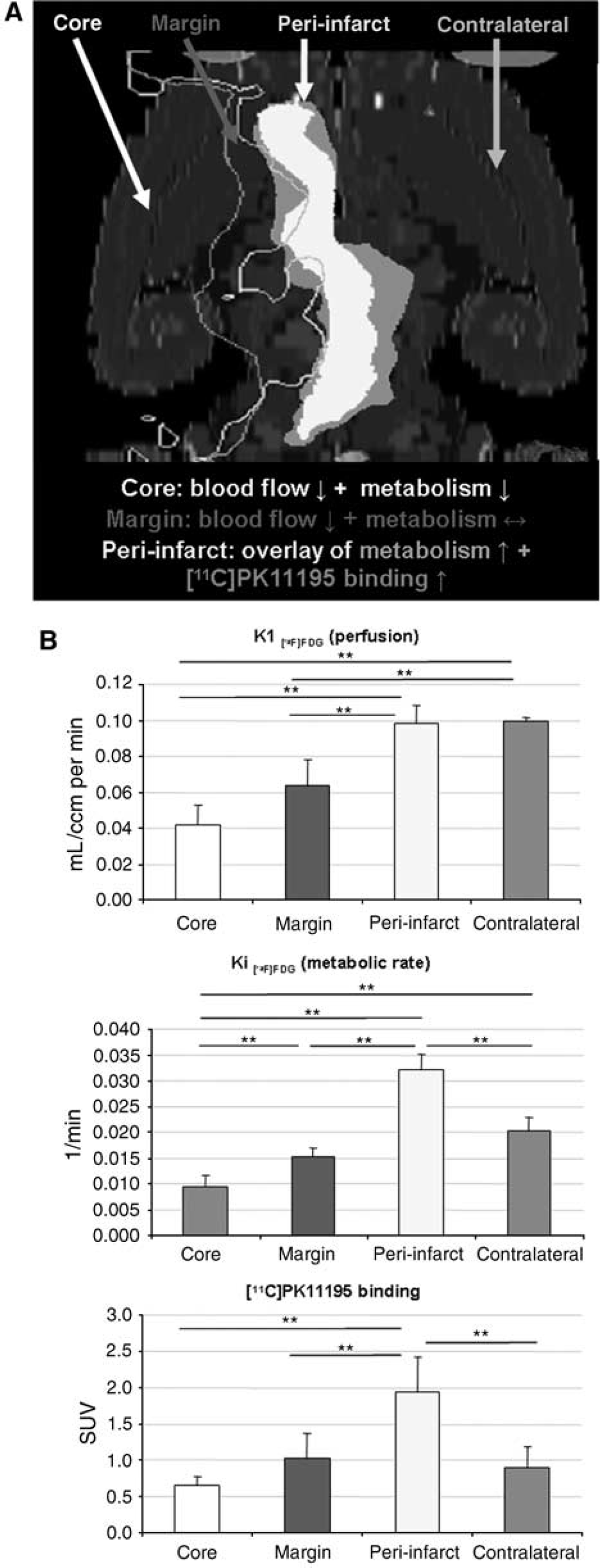
Neuroinflammation occurring in a distinct peri-infarct zone puts the vital brain tissue at risk for secondary damage. [18F]FDG transport rate constant (
Standard uptake values for [11C]PK11195 were determined as a quantitative assessment of neuroinflammatory processes (Figure 4B, lower diagram). No relevant [11C]PK11195 binding was detected in the infarct core (SUV: 0.65 ± 0.13) or in the infarct margin (1.02 ± 0.35) compared with that in homotopic contralateral regions (0.90 ± 0.28). Exclusively in the peri-infarct region, we observed significantly increased [11C]PK11195 binding (1.93 ± 0.49) compared with that in homotopic contralateral regions (0.90 ± 0.28;
Supplementary Table 1 provides the neuropathological extent of individual ischemic lesions and the respective functional data collected from distinct regions.
Discussion
Focal ischemia induces a neuroinflammatory response with an essential effect on infarct demarcation and tissue regeneration (Stoll et al, 1998; Dirnagl et al, 1999). This process is mediated by resident microglia and by hematogenous monocytes. The histologic differentiation between resident and invading cells is difficult, as there is no immunocytochemical marker to distinguish between the two populations (Flaris et al, 1993). Earlier studies have approached this problem using morphologic criteria along with a detailed histopathological time course to identify and track invading cells (Mabuchi et al, 2000). Others have used bone marrow chimeric mice to label hematogenous cells (Schilling et al, 2003), or depleted peripheral macrophages to identify the role of residential cells (Schroeter et al, 1997). All these studies agree that resident microglia predominate over invading monocytes in the dense cellular rim of the peri-infarct zone (Schroeter et al, 1997; Mabuchi et al, 2000; Schilling et al, 2003). Both cell populations play the same functional role in neuroinflammation, i.e., phagocytosing debris and producing pro-inflammatory cytokines, and therefore represent a target for therapeutic interventions. Novel therapeutic strategies aiming at neuroinflammatory processes hold promise for treating stroke patients in the future. However, the development and clinical implementation of such treatment critically depends on reliable tools to noninvasively monitor neuroinflammation
In the macrosphere model, permanent focal ischemia is induced by an MCA occlusion with a defined number of TiO2 spheres (Gerriets et al, 2003). From a clinical point of view, the macrosphere MCA occlusion model mimics arterio-arterial embolism of ‘hard’ arteriosclerotic plaque material more closely than the classic occlusion method with an intraluminal thread. As arterio-arterial embolism is one of the most frequent causes of stroke, the macrosphere model seems to be a most relevant model for studying the pathophysiology of stroke (Mohr, 1993; Barnett et al, 2000; Grau et al, 2001; Prabhakaran et al, 2006).
In the macrosphere stroke model, depending on the exact macrosphere trapping within the MCA arborization, infarcts affect between 20% and 60% of the hemisphere (Gerriets et al, 2003). We used T2-weighted MRI to morphologically assess the extent of individual lesions
Characterizing metabolically distinct regions of the ischemic brain, we identified an ‘infarct margin’ directly adjacent to the (necrotic) infarct core that showed reduced blood flow—indicated by a reduction in the [18F]FDG transport rate constant (
However, although there might be additional neuroinflammatory processes going on within the infarct core, as is also suggested by our immunohistochemical data, those events in the infarct core are of minor functional relevance with regard to secondary tissue damage and recovery of function, as the infarct core is likely to turn into necrosis in permanent ischemia. In contrast, with regard to recovery, the viability of the peri-infarct zone is of major importance, and secondary tissue damage occurring in this region might be detrimental to regeneration. In this study, we show for the first time that neuroinflammatory processes in this peri-infarct zone are accompanied by an increased metabolic rate constant of [18F]FDG that was 60% higher than that of the remaining brain tissue, indicating enhanced energy metabolism. At the same time, tissue perfusion was not increased to compensate for this increased metabolism, thus putting this tissue at risk for metabolic disturbance.
Several earlier studies in animals and in humans analyzed the localization and time course of stroke-induced neuroinflammation, and found inflammation predominantly within the ischemic core (Cremer et al, 1992; Garcia et al, 1994) or in peri-infarct regions (Gerhard et al, 2000; Pappata et al, 2000; Mabuchi et al, 2000; Price et al, 2006). As discussed, we attribute differences in localization to different experimental ischemia models or to the individual clinical situation, respectively. However, to the best of our knowledge, our study is the first to correlate the individual site of neuroinflammation to the functional, i.e., metabolic state of the respective tissue. Furthermore, using a surrogate parameter for cerebral blood flow, we show that the lack of reperfusion in the ischemic core excludes [11C]PK11195 binding in permanent focal ischemia.
The complete functional repertoire of the inflammatory cells that are activated and/or recruited after stroke is still unknown. Besides the harmful effects, such as the production of cytotoxic agents and free radicals, beneficial functions include the demarcation of necrotic tissue and the promotion of tissue repair and regeneration. However, despite the potential beneficial effects that neuroinflammatory processes may promote, we propose that the focal metabolic disturbance we observe in the peri-infarct zone in combination with the concurrent destructive potential of neuroinflammation, e.g., the production of reactive oxygen species by inflammatory cells, primarily put the affected tissue at risk of secondary damage. Novel treatment strategies aimed at reducing these neuroinflammatory processes may, at least to some extent, aid in balancing this delicate equation between beneficial and detrimental effects of neuroinflammation.
Multiple attempts have been made in recent years to develop a radiotracer with higher affinity to PBRs than PK11195 (Zhang et al, 2003; Fujimura et al, 2006; Brown et al, 2007). Higher affinity suggested by the differences in dissociation constants may address some of the issues related to nonspecific binding reported with PK11195. However, in our model of permanent focal ischemia, nonspecific binding of [11C]PK11195 did not pose a significant problem because of the intense and circumscribed signal of specific [11C]PK11195 accumulation, with a mean SUV of 1.93 within the ROI and a good signal-to-noise ratio. We propose that although novel PBR ligands with higher binding affinity may be helpful in studying disease models with more subtle microglial activation like that associated with neurodegenerative disorders, the robust neuroinflammatory processes occurring with permanent focal cerebral ischemia can be detected unequivocally with the well-established [11C]PK11195.
In brief, the exact colocalization of neuroinflammatory processes with an increased [18F]FDG metabolic rate constant as shown in this study for the first time identifies the peri-infarct zone to be at risk of secondary tissue damage by neuroinflammation and metabolic disturbances. Therefore, the peri-infarct zone may represent the ‘tissue-to-treat’ with any novel therapeutic strategies aiming at neuroinflammatory processes, and the evaluation of the differential effects of such treatments should include specifically the peri-infarct zone. The possibility of analyzing neuroinflammation noninvasively and repeatedly over time in the same experimental subject makes [11C]PK11195 a promising radiotracer in the preclinical and clinical development of such treatments (reviewed by del Zoppo et al, 2000;Wang et al, 2007). Further studies using these imaging techniques might help in uncovering the functional role of inflammatory cells, the relationship between infarct core and peri-infarct zone, and factors that determine peri-infarct tissue to survive or to succumb to death. In addition, using [11CjPKlligB-PET to stratify patients according to their individual neuroinflammatory patterns may help to select individuals who would benefit most from immunomodulatory treatments.
