Abstract
Obesity is an independent risk factor for stroke and is associated with poorer outcome after stroke. We investigated whether this poorer outcome is related to brain microvascular disruption. Focal cerebral ischaemia was induced in lean or obese (
Introduction
Obesity is an independent risk factor for stroke, and is associated with atherosclerosis, diabetes, and hypertension, conditions that predispose to stroke (Winter et al, 2008). The increased risk of stroke in obese individuals may also be accompanied by poorer prognosis after the ischaemic insult (Razinia et al, 2007). In support of this, recent studies have demonstrated greater brain damage in obese rodents after experimental stroke (Mayanagi et al, 2008; Terao et al, 2008). However, the mechanisms responsible for these detrimental effects of obesity on cerebrovascular injury are poorly delineated.
A crucial site of convergence for pathophysiological mechanisms involved in obesity and stroke is the brain microvascular endothelium. Obesity is a state of chronic systemic inflammation and is associated with vascular oxidative stress (Dandona et al, 2004). Inflammation and oxidative stress are important factors that contribute to disruption of the blood–brain barrier (BBB). Mechanisms include disruption of inter-endothelial tight-junction complexes (Schreibelt et al, 2007) and induction of proteases, in particular matrix metalloproteinases (MMPs), that degrade constituents of the vascular basement membrane (Zhao et al, 2007). We have shown previously that systemic inflammation exacerbates ischaemic brain damage through MMP-9-dependent alterations in BBB integrity (McColl et al, 2008). Clinically, loss of BBB integrity after stroke is associated with serious complications such as brain oedema and haemorrhagic transformation (HT) that correlate with poor prognosis (Simard et al, 2007). Accordingly, it is feasible that obesity may predispose to poorer outcome after stroke through aggravation of brain microvascular disruption.
In the present study, we sought to determine if obesity exacerbates brain microvascular disruption after experimental stroke.
Materials and methods
Focal Cerebral Ischaemia
Experiments were performed on 8- to 12-week-old male obese
In a separate experiment, lean and obese
Tissue Processing
Twenty-four hours after MCAo mice were perfused transcardially with 0.9% saline followed by 4% paraformaldehyde and brains post-fixed, cryoprotected, and frozen. Coronal brain sections (20 μm) were prepared on a cryostat.
Measurement of HT and Ischaemic Damage
To assess HT brain sections were stained with haematoxylin and eosin. Areas of HT were delineated at coronal levels (400 μm apart) and the total area of HT was calculated. For ischaemic damage, adjacent brain sections were stained with cresyl violet. Briefly, areas of damage were delineated at eight anatomically defined coronal levels and the total volume was calculated.
Immunohistochemistry
Primary antibodies were used as follows: goat anti-MMP-9 (1:400; R&D Systems, Abington, UK), and rabbit antilaminin (1:25; Sigma, Poole, UK). Endogenous peroxidise activity (except for double immunofluorescence) and nonspecific binding sites were blocked before incubating (4°C) sections in primary antibody solution. For peroxidise-based staining, sections were then incubated in biotinylated secondary antibody (1:200; Vector Laboratories, Peterborough, UK), immersed in avidin–biotin–peroxidase complex (ABC; Vector Laboratories) and colour-developed using a 0.05% diaminobenzidine solution (in 0.01% H2O2). For assessment of BBB disruption, primary antibody was omitted and a biotinylated anti-mouse IgG secondary antibody was used. For double labelling immunofluorescence, after primary antibody application sections were incubated in donkey anti-goat Alexa 488 and donkey anti-rabbit 594 (Molecular Probes, Paisley, UK), mounted, and coverslipped with ProLong Gold mounted medium (Invitrogen, Paisley, UK).
Statistical Analysis
For all analyses, data are represented as mean ± s.d. Parametric data were analysed using Student's
Results
Increased Incidence of HT and Ischaemic Brain Damage in Obese Mice
Haemorrhagic transformation was observed in the ipsilateral ischaemic hemisphere of
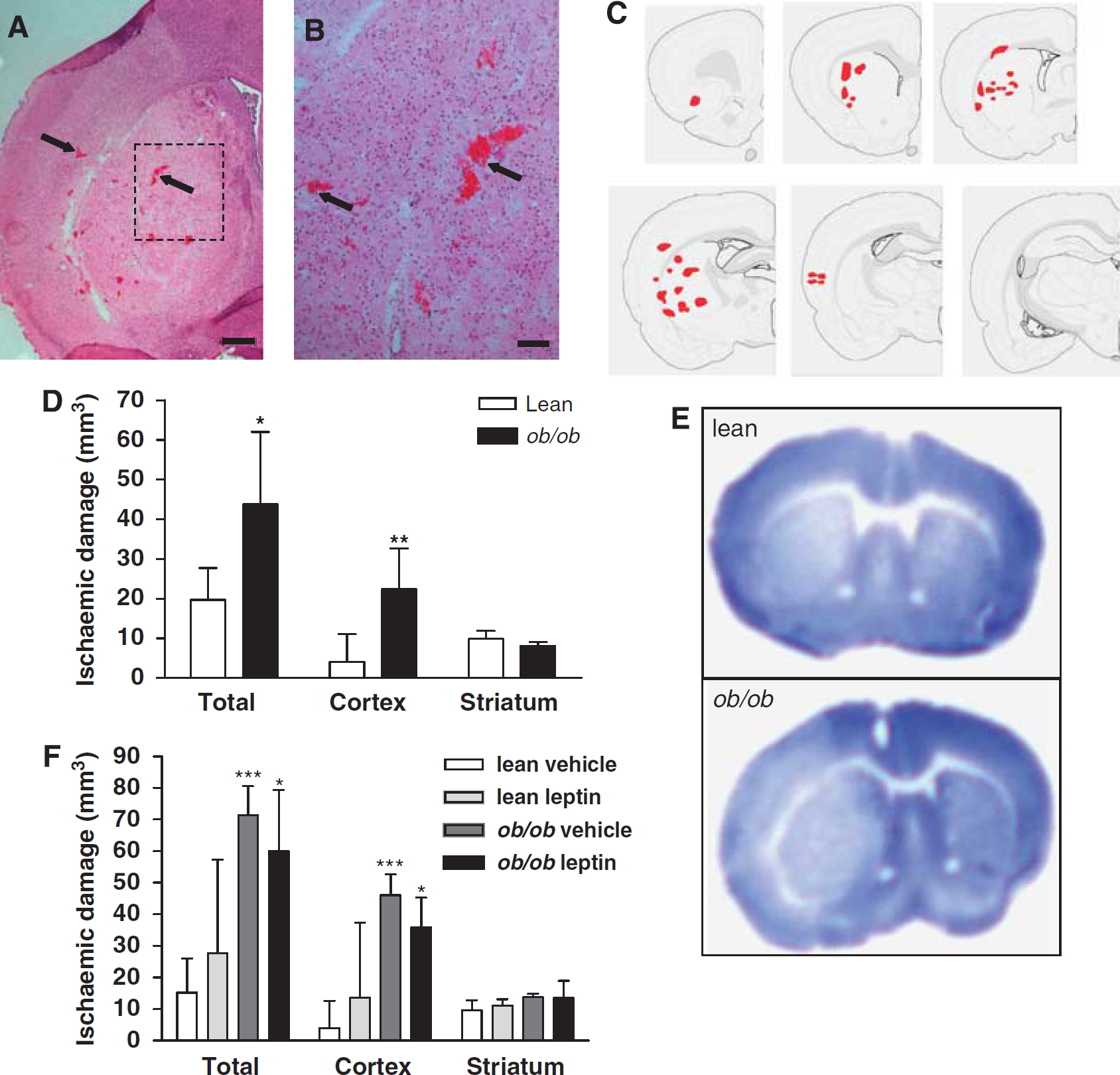
HT and ischaemic damage are increased in obese mice 24 h after MCAo. (
Increased HT and Ischaemic Brain Damage in Obese Mice are Independent of Leptin Deficiency
Neuroprotective actions of leptin have been demonstrated previously (Zhang et al, 2007); therefore, in a separate experiment, we assessed the effect of acute leptin replacement on ischaemic brain damage in lean and obese mice. Leptin administration did not affect the incidence of HT, as haemorrhages were present in the striatum and cortex of all
To verify that the absence of an effect of leptin administration on brain pathology was not due to lack of biological activity, we assessed the effect of leptin on food intake and body weight. Leptin (4 mg/kg, intraperitoneal.) significantly reduced food intake (vehicle, 4.0 ± 0.2 g versus leptin, 3.2 ± 0.3 g,
BBB Permeability and Brain Microvascular MMP-9 Expression are Increased after Focal Cerebral Ischaemia in Obese Mice
The marked increase in susceptibility to ischaemia-induced HT observed in
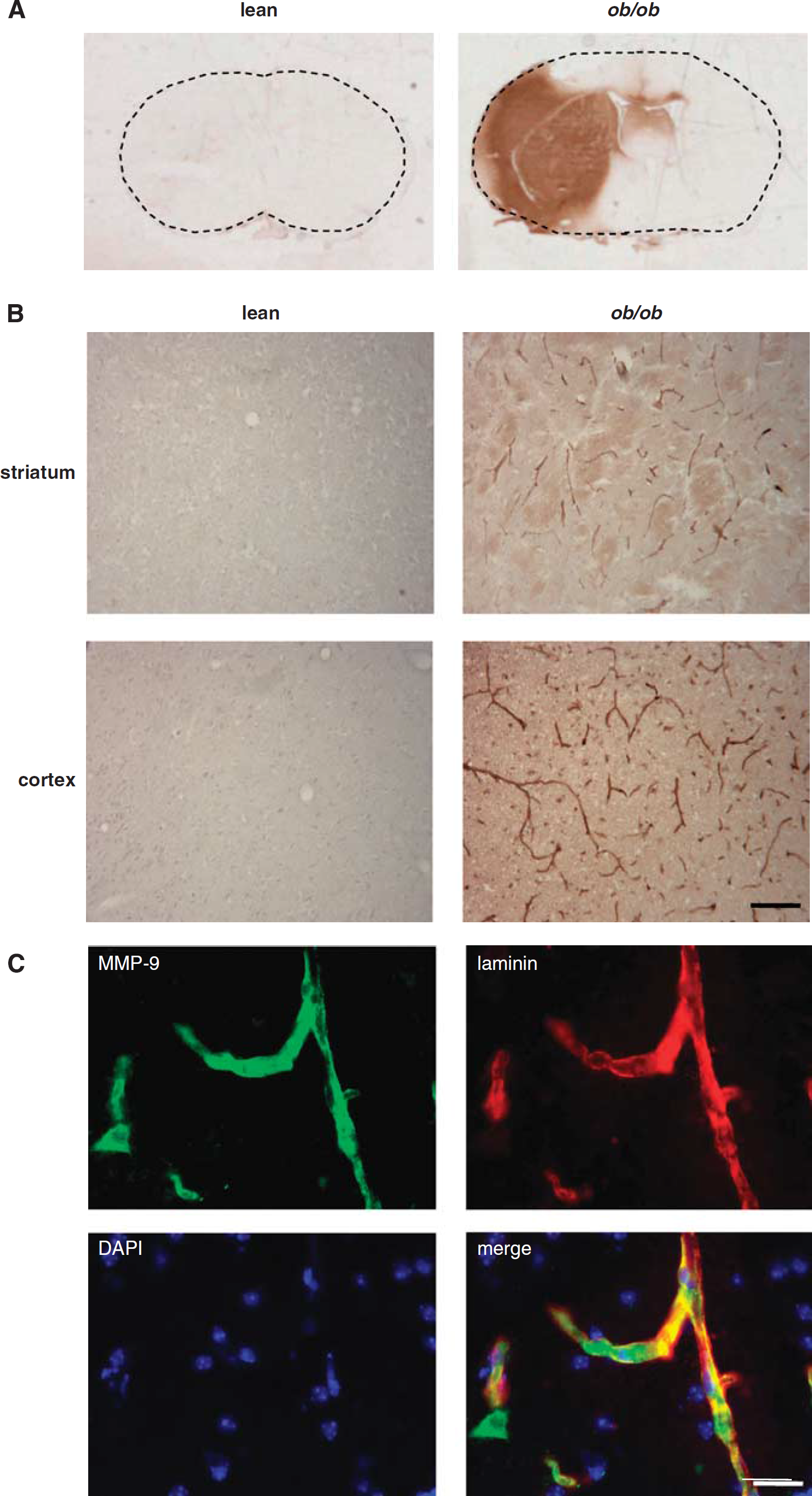
BBB permeability and MMP-9 expression are increased in obese mice after MCAo. BBB permeability was assessed by immunostaining for IgG. (
Experimental and clinical evidence implicates proteolytic disruption of BBB substrates by proteases such as MMP-9 in cerebrovascular disruption. We, therefore, assessed the expression of MMP-9 in the brain of obese and lean mice after MCAo. Extensive MMP-9 immunoreactivity was detected in the ipsilateral ischaemic striatum and cortex of
Discussion
In the present study, we show elevated microvascular MMP-9 expression and increased incidence of HT in obese, leptin-deficient
In addition to clinical data that have indicated increased risk of stroke in obese individuals (Winter et al, 2008), recent studies have shown that the severity of brain damage is increased after experimental stroke in obese rodents (Mayanagi et al, 2008; Terao et al, 2008). Our data expand on these previous findings to show that obese mice are highly susceptible to HT, which is associated with elevated microvascular MMP-9 immunoreactivity. HT is a serious complication in ischaemic stroke patients that correlates with poor prognosis (Paciaroni et al, 2008). Conventional risk factors for HT include hyperglycaemia and hypertension (Paciaroni et al, 2008), both of which are common in obese subjects. However, these factors are unlikely to account for the increased frequency of HT in the present study, because pre-ischaemic blood glucose levels were similar in lean and obese mice, and previous studies have not found significant differences in blood pressure (Vachharajani et al, 2005). Thrombolytic treatment also predisposes to HT (Zhao et al, 2007). Our data suggest that obese patients may be at further risk of HT if treated with thrombolytic agents.
The extravasation of all blood constituents, including erythrocytes, that occurs during HT is indicative of a catastrophic failure of microvascular integrity. This failure also underlies the increased permeability of the BBB and brain oedema that accompanies HT. In the present study, we found marked increase in BBB permeability to IgG in obese mice, which is consistent with their increased susceptibility to HT and confirms that obesity promotes severe disruption to the BBB. We also observed localisation of extensive MMP-9 immunoreactivity to the microvasculature in obese mice, suggesting that MMP-9 may be an important mediator underlying increased BBB disruption and HT in obese mice. Quantitative assays of MMP-9 activity will be required to verify changes on a functional level. Previous studies have shown that MMP-9 can disrupt multiple components of the BBB, including the tight-junction proteins claudin-5 and occludin, and the basement membrane protein collagen-IV (Candelario-Jalil et al, 2009). Furthermore, inhibition of MMPs attenuates BBB disruption and reduces thrombolysis-induced HT after experimental stroke (Candelario-Jalil et al, 2009).
A chronically elevated inflammatory profile is a feature of obesity and inflammation is implicated in microvascular dysfunction associated with obesity (Singer and Granger, 2007). Increased leukocyte–endothelium interactions and intracellular adhesion molecule-1 (ICAM-1) expression after experimental stroke in obese mice have recently been reported (Mayanagi et al, 2008; Terao et al, 2008). Leukocytes, in particular neutrophils, contain abundant proteases, including MMP-9; therefore, increased leukocyte adhesion could be a mechanism underlying the increased microvascular MMP-9 expression in the present study. In support of this, brain neutrophil accumulation was significantly greater after MCAo in obese mice (data not shown). More generally, growing evidence suggests that systemic inflammatory conditions, such as obesity, are important modifiers of stroke outcome (McColl et al, 2009).
One important caveat of the present study is that we cannot exclude that the effects of obesity are mediated through alterations in cerebral perfusion (e.g., through collateral vessels) and, indeed, given the pro-coagulatory state that is associated with obesity, it will be important to consider this potential mechanism in future studies. In summary, we have shown that obese
Footnotes
Acknowledgements
We acknowledge financial support by the Research Councils UK and the Medical Research Council.
The authors declare no conflict of interest.
