Abstract
In experimental models, growth factors (GFs) such as vascular endothelial growth factor (VEGF), Angiopoietin 1 (Ang-1), or granulocyte-colony stimulating factor (G-CSF) mediate brain recovery after intracerebral hemorrhage (ICH). Our aim was to study the association between serum levels of GF and clinical outcome in patients with ICH. A total of 95 patients with primary ICH (male, 66.3%; mean age, 67.8±9.8 years) were prospectively included in the study within 12 h from symptoms onset. The main outcome variable was good functional outcome at 3 months (modified Rankin scale 2). Median serum levels of GF at 72 h from stroke onset were significantly higher in patients with good outcome (n=39) compared with those with poor outcome (all P<0.0001). Serum levels of VEGF 330 pg/mL, G-CSF 413 pg/mL, and Ang-1 35 ng/mL at 72 h were independently associated with good functional outcome (odds ratio (OR), 11.2; 95% confidence interval (CI): 2.9 to 43.0; OR, 19.6; 95% CI: 3.9 to 97.9; and OR, 14.7; 95% CI: 3.6 to 60.0, respectively), neurologic improvement (all P<0.0001) and reduced residual cavity at 3 months (all P<0.01). These results illustrate that high serum levels of GF are associated with good functional outcome and reduced lesion volume in ICH.
Introduction
Intracerebral hemorrhage (ICH) represents 10% to 15% of all strokes. Mortality ranges from 44% to 51% within the first month and only 20% of survivors can live independently at 6 months (Bamford et al, 1990; Broderick et al, 1993, 2007; Brown et al, 1996; Qureshi et al, 2001). There is no effective treatment for this disease so far, despite some clinical trials have been accomplished (Lyden et al, 2007; Mayer et al, 2008; Mendelow et al, 2005).
In animal models, ICH can induce cerebral angiogenesis and upregulation of vascular endothelial growth factor (VEGF); thus, the modulation of angiogenesis through altering the expression of VEGF and its receptors may be a potential strategy for promoting brain repair after ICH (Tang et al, 2007). Similarly, it has been shown that human neural stem cells overexpressing VEGF provide neuroprotection, angiogenesis, and functional recovery in a mouse ICH model (Lee et al, 2007). Other growth factors (GFs) have also been involved in neurorepair after ICH. Granulocyte-colony stimulating factor (G-CSF) induces long-term sensorimotor recovery after ICH with reduction of brain edema, inflammation, and perihematoma cell death (Park et al, 2005), whereas Angiopoietin 1 (Ang-1) and Tie-2 have shown a close relationship with angiogenesis in the rat brain after ICH (Zhou et al, 2008). According to this experimental evidence, we tested the hypothesis that high serum levels of GF might be associated with improved neurologic and functional outcome and reduced brain injury in patients with primary ICH.
Materials and methods
Study Population and Patients Characteristics
Between February 2005 and March 2007, 132 patients with a first-ever primary ICH of <12 h from symptoms onset, previously independent for their daily living activities and who were not on anticoagulant treatment, were prospectively evaluated to be included in the study. Patients with chronic inflammatory diseases (n=8), severe hepatic (n=4) or renal (n=2) diseases, hematological diseases (n=3), cancer (n=5) or infectious disease in the 15 days before inclusion (n=7) were excluded. Five patients did not accept their participation in the study and three patients were lost of follow-up; hence, a total of 95 patients (male, 66.3%; mean age, 67.8±9.8 years) were finally included in the study.
The protocol was approved by the ethics committee and informed consent was given by patients or their relatives.
Clinical Protocol
All patients were admitted to an acute stroke unit of 3 university hospitals and treated according to the Guidelines of the Cerebrovascular Diseases Study Group of the Spanish Society of Neurology (GEECV-SEN, 2004). Medical history recording potential vascular risk factors, blood and coagulation tests, 12-lead electrocardiography, and chest radiography were carried out at admission.
Stroke severity was assessed by a certified neurologist using the National Institute of Health Stroke Scale (NIHSS) at admission, 24, 48±6, and 72±24 h, and at 7±1 and 90±7 days. Functional outcome was evaluated at 3 months using the modified Rankin Scale (mRS).
Antihypertensive treatment with intravenous labetalol or captopril was administered in case of systolic blood pressure >185 mm Hg or diastolic blood pressure >105 mm Hg. Low-dose subcutaneous heparin treatment at admission was used for the prevention of deep vein thrombosis and pulmonary thromboembolism. A total of 25 (26.3%) patients were included in therapeutic clinical trials (3 patients in the Factor Seven for Acute Hemorrhagic Stroke (FAST) Trial (Mayer et al, 2008) and 22 patients in the Cerebral Hematoma and NXY Treatment (CHANT) Trial; Lyden et al, 2007).
Neuroimaging Studies
A computed tomography study was carried out at admission (median time from stroke onset, 5.4 (2.7, 8.2) h). Computed tomography follow-up studies were carried out in all patients who survived for 72 h (n=93), day 7±1 (n=91), and day 90±15 (n=68).
Lesion volumes were calculated by standard planimetric method using a semi-automated process by tracing the perimeter of appropriate high- and low-attenuation zones and calculating lesion areas for each slice multiplied by slice thickness to yield lesion volumes. The residual cavity volume at 3 months was determined using the same volumetric method described; each volume calculation was performed three times and the mean value was taken as definitive.
The ICH topography was classified as lobar when it affected predominantly the cortical or subcortical white matter of the cerebral lobes, or as deep when it was limited to the internal capsule, the basal ganglia, or the thalamus. The presence of intraventricular extension of the hematoma, and mass effect was also recorded.
All neuroimaging evaluations were carried out by the same neuroradiologist blinded to patients’ clinical and laboratory results.
Outcome Variables
The primary end point was good functional outcome defined as a mRS score 2 at 3 months.
The relative improvement of NIHSS score during the first 3 months and the relative change between the acute ICH volume and the volume of the residual cavity were evaluated as secondary end points. The relative improvement of the NIHSS score was calculated using the following formula: (NIHSS score on admission−NIHSS at 3 months/NIHSS admission) × 100. Dead patients (28.4%) were rated with a value of −100%. Good recovery was defined as a NIHSS improvement of 50%; this cut-off point was selected based on data obtained from a retrospective analysis of our patients database: In a total of 416 patients with primary ICH, excluding patients who were dead at 3 months, the median NIHSS improvement was 43.2% (25.1 to 74.9).
The relative change between the acute ICH volume and the volume of the residual cavity was calculated using the following formula: (ICH volume at admission−volume of residual cavity at 3 months/ICH volume at admission) × 100.
Laboratory Tests
Blood samples, drawn from all patients on admission, at 24±6 and 72±24 h, were collected in glass chemistry test tubes, centrifuged at 3,000 g for 10 mins, and immediately frozen and stored at −80°C. Serum VEGF and Ang-1 levels were measured with commercially available quantitative ELISA kits obtained from R&D Systems (Minneapolis, USA). Similarly, G-CSF levels were measured with commercially available ELISA kits obtained from Biosource Europe S.A. (Nivelles, Belgium). Determination of the levels was done in an independent laboratory, blinded to clinical and neuroimaging data.
Statistical Analysis
The results are expressed as percentages for categorical variables and as mean (s.d.) or median (quartiles) for the continuous variables depending on their normal distribution or not. Proportions were compared using the χ2 test, and Student's t-test or the Mann–Whitney tests were used to compare continuous variables between groups. The Spearman analysis was used for bivariate correlations.
The influence of GF serum levels on good functional outcome and on neurologic recovery (relative NIHSS improvement of 50%) was assessed by logistic regression analysis after adjusting for the main baseline variables related to good functional outcome and neurologic recovery in the univariate analyses (enter approach and probability of entry P<0.05). Owing to a lack of linearity of the odds ratios (ORs), GF serum levels were categorized using the best predictive value obtained by Receiver Operating Characteristic (ROC) analysis. Results were expressed as adjusted OR with the corresponding 95% confidence intervals (95% CIs). We carried out a sensitivity analysis based on patients who are not treated with investigational drugs. The statistical analysis was conducted using SPSS 16.0 (SPSS Inc. Chicago, IL, USA) for Windows XP.
Results
Primary Outcome
Table 1 shows the main characteristics of patients by functional outcome groups. Overall, 39 (41.1%) patients showed good functional outcome (mRS 2) at 3 months. A total of 29 patients were dependent and 27 died during the follow-up. Patients with good functional outcome showed milder stroke severity, lower rate of ventricular extension, and smaller ICH volume on admission. Similar results and P-values were obtained after the exclusion of patients included in clinical trials.
Baseline clinical characteristics, vascular risk factors, stroke subtype, biochemical parameters, and neuroimaging findings in patients with good or poor outcome
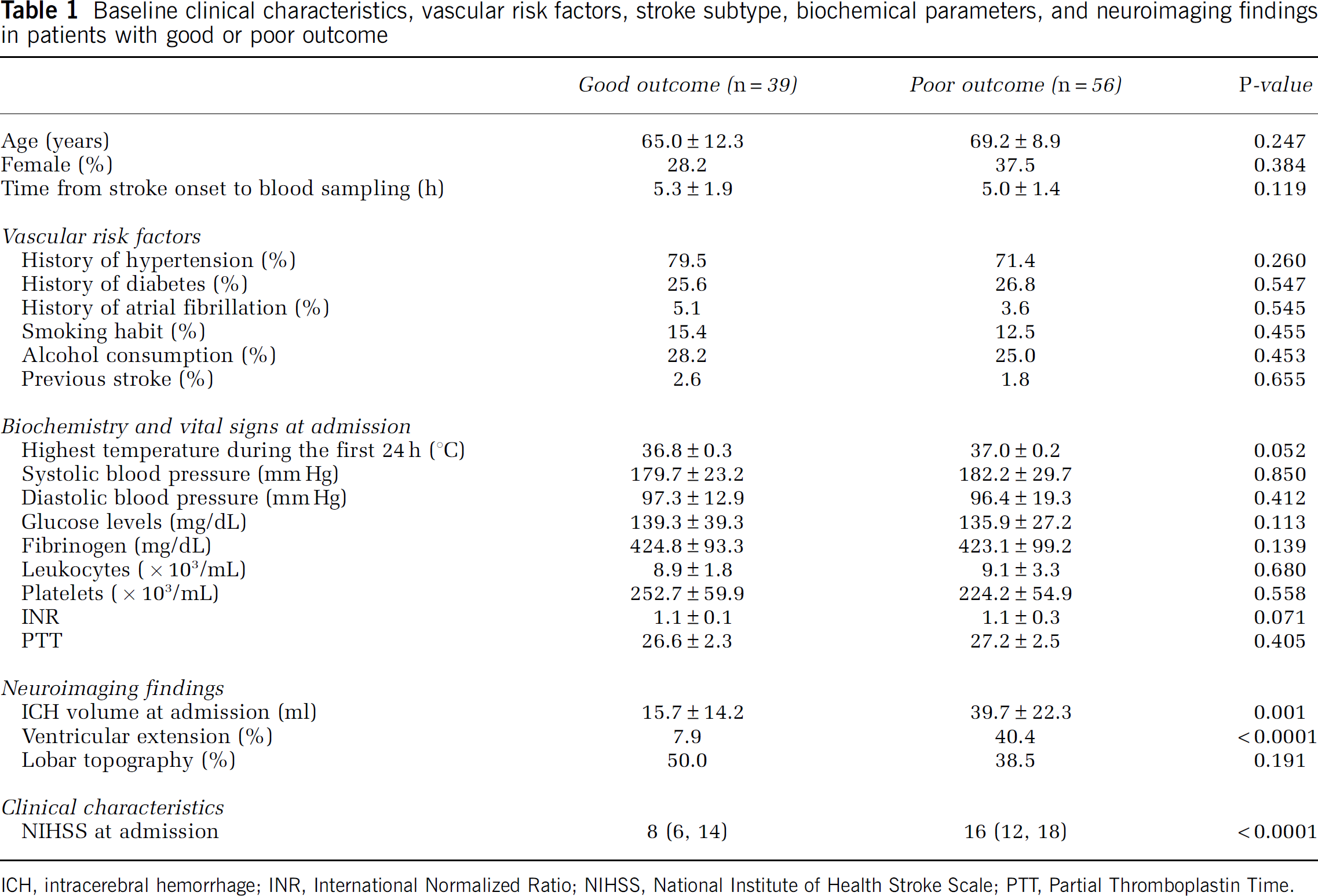
ICH, intracerebral hemorrhage; INR, International Normalized Ratio; NIHSS, National Institute of Health Stroke Scale; PTT, Partial Thromboplastin Time.
Growth factor levels were higher at 72 h than at baseline and at 24 h (Figure 1). Median (interquartile range) levels of GF at 72 h were significantly higher in patients with good functional outcome (Table 2). No significant correlations or associations were found between GF and baseline variables listed in Table 1 (r<0.2).
Growth factor levels at different timeframes by functional outcome
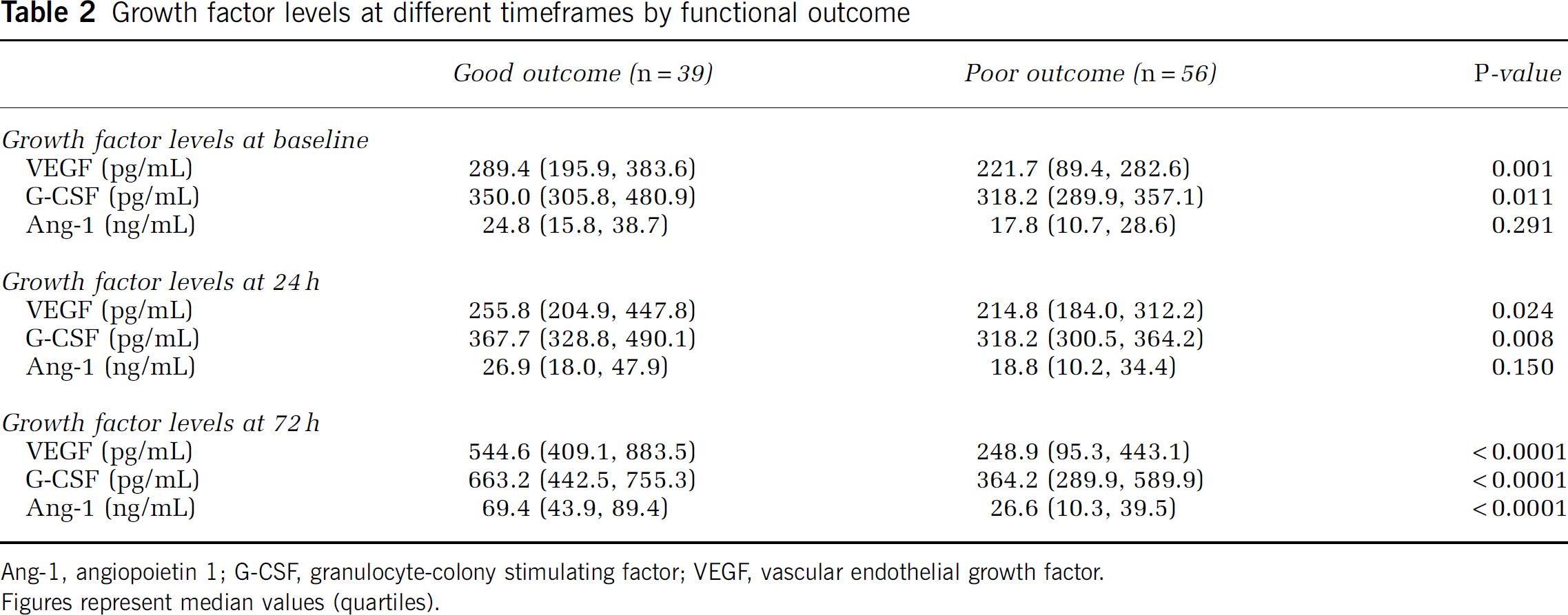
Ang-1, angiopoietin 1; G-CSF, granulocyte-colony stimulating factor; VEGF, vascular endothelial growth factor.
Figures represent median values (quartiles).
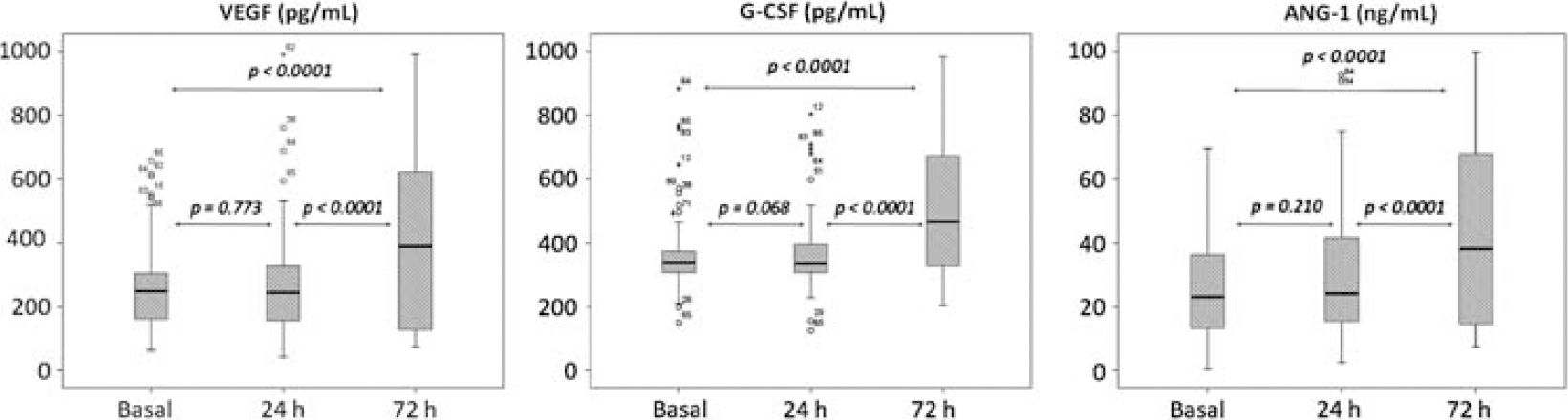
Temporal profile of GF serum levels during the first 72 h from stroke onset. Boxplots show median values (horizontal line inside the box), quartiles (box boundaries), and the largest and smallest observed values (lines drawn from the end of the box) of growth factors.
ROC analysis showed that the best cut-off points of serum levels of GF at 72 h to predict good functional outcome were 330 pg/mL (area under curve 0.827, sensitivity 82%, specificity 79%, P<0.0001) for VEGF, 413 pg/mL (area under curve 0.774, sensitivity 83%, specificity 73%, P<0.0001) for G-CSF, and 35 ng/mL (area under curve 0.872, sensitivity 82%, specificity 81%, P<0.0001) for Ang-1.
Serum levels of VEGF 330 pg/mL, G-CSF 413 pg/mL, and Ang-1 35 ng/mL were independently associated with good functional outcome at 3 months (OR, 11.2; 95% CI: 2.9 to 43.0; OR, 19.6; 95% CI: 3.9 to 97.9; and OR, 14.7; 95% CI: 3.6 to 60.0, respectively) (Table 3). Only stroke severity remained significant after adjustment for ICH volume and ventricular extension. A sensitivity analysis carried out in the 70 patients who were not included in clinical trials showed similar significant results. In these patients, serum levels of VEGF 330 pg/mL, G-CSF 413 pg/mL, and Ang-1 35 ng/mL were independently associated with good functional outcome at 3 months (OR, 10.3; 95% CI: 2.0 to 53.5; OR, 26.4; 95% CI: 3.4 to 205.7; and OR, 21.8; 95% CI: 3.5 to 137.3, respectively) after adjustment for stroke severity, ICH volume, and ventricular extension.
Adjusted OR of good outcome at 3 months for serum levels of GF at 72 h
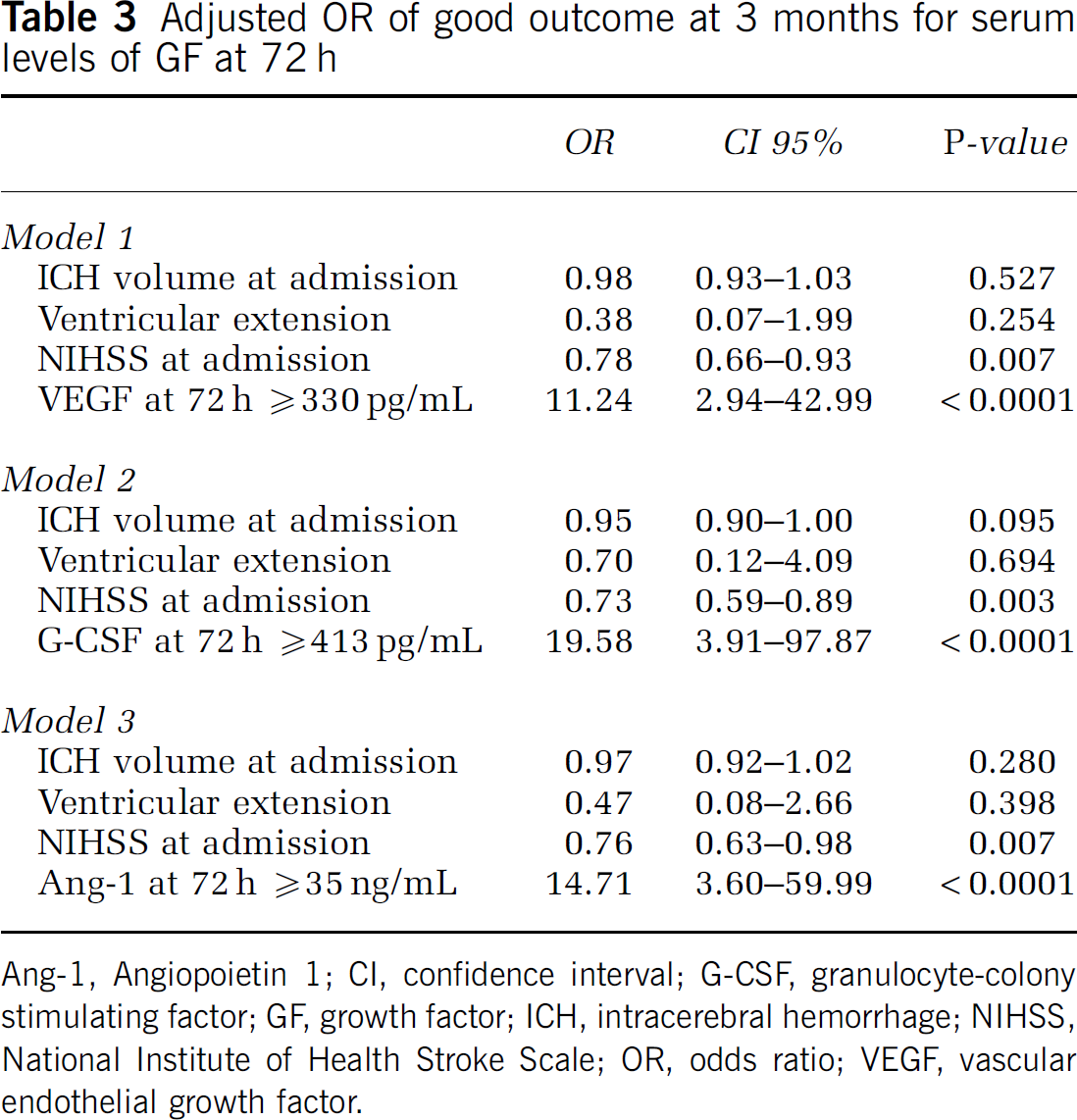
Ang-1, Angiopoietin 1; CI, confidence interval; G-CSF, granulocyte-colony stimulating factor; GF, growth factor; ICH, intracerebral hemorrhage; NIHSS, National Institute of Health Stroke Scale; OR, odds ratio; VEGF, vascular endothelial growth factor.
Secondary Outcomes
Serum levels of VEGF (r=0.831), G-CSF (r=0.762), and Ang-1 (r=0.887) at 72 h, but not at baseline and at 24 h, had a strong correlation with the relative improvement of the NIHSS score (Figure 2) (Supplementary Figure 1).
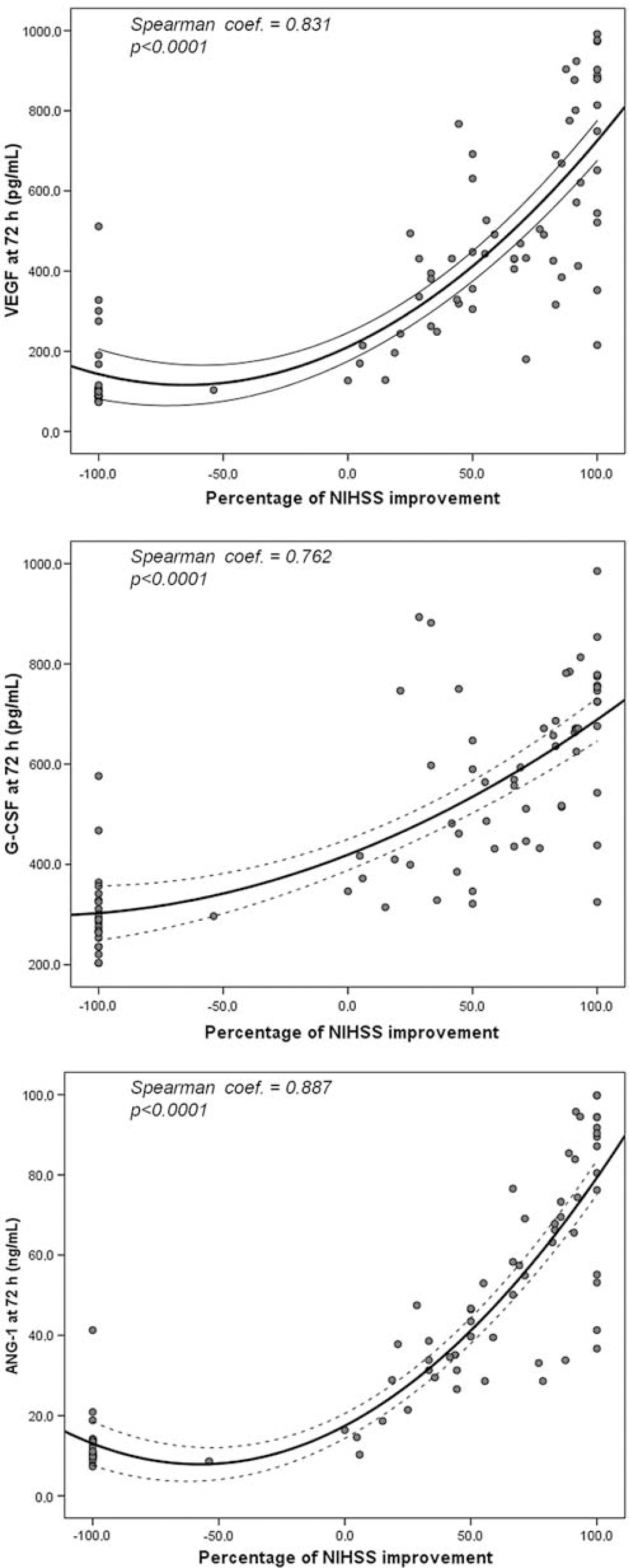
Scatterplots between GF serum levels at 72 h and the percentage of NIHSS improvement during the first 3 months (see text for explanations).
Forty-two (44.2%) patients showed a NIHSS score improvement >50% at 3 months. Serum levels at 72 h of VEGF 330 pg/mL, G-CSF 413 pg/mL, and Ang-1 35 ng/mL were independently associated with a NIHSS relative improvement of 50% during the first 3 months (OR, 41.1; 95% CI: 9.1 to 185.8; OR, 227.9; 95% CI: 20.5 to 2535.7; and OR, 312.9; 95% CI: 34.5 to 2838.9, respectively). Diastolic blood pressure, stroke severity, ICH volume, and ventricular extension were not remained significant after adjustment. A sensitivity analysis carried out in the 70 patients who were not included in clinical trials showed similar significant results (data not shown).
Serum levels at 72 h of VEGF 330 pg/mL, Ang-1 35 ng/mL, and, in particular, of G-CSF 413 pg/mL were associated with a higher percentage of lesion volume reduction (Figure 3).
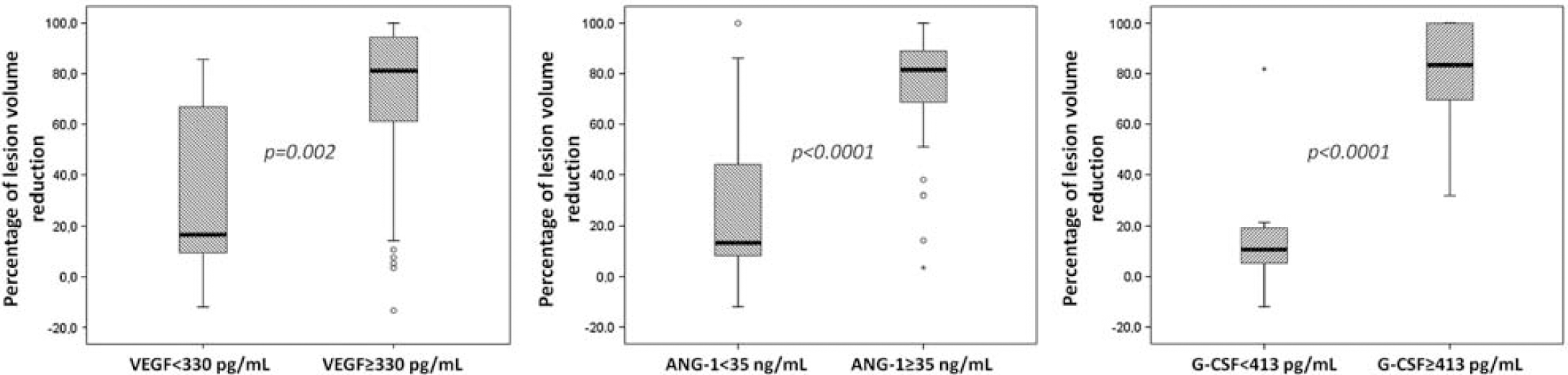
Relative lesion volume reduction for the categorized VEGF, G-CSF, and Ang-1 serum levels according to the best predictive capacity of recovery. Boxplots show median values (horizontal line inside the box), quartiles (box boundaries), and the largest and smallest observed values (lines drawn from the end of the box) of growth factors.
Discussion
This prospective study shows a relationship between serum GF and brain recovery in patients with ICH. The increase in GF levels during the first 72 h after ICH was independently associated with a good functional outcome at 3 months. This favorable effect on the primary variable was supported by positive effects on the reduction of lesion volume and in the relative neurologic improvement of 50% at day 90. Importantly, these effects remained significant after adjustment for the well-recognized prognostic factors such as ICH volume, stroke severity, and ventricular extension.
Serum levels of GF increased during the next 3 days after ICH. The greater the magnitude of this increase, the better the neurologic improvement and the larger the reduction of lesion volume at 90 days. These findings are in line with experimental studies, which indicate that GFs, such as VEGF, Ang-1, or G-CSF, might mediate endothelial cell regeneration, angiogenesis, and sensorimotor recovery (Lee et al, 2007; Park et al, 2005; Tang et al, 2007; Zhou et al, 2008).
A critical part of the deleterious effects of ICH is caused by the early enlargement of the hematoma, which starts within 3 h after ICH onset, but also by perihematoma edema and inflammation that increase gradually for at least 72 h (Castillo et al, 2002; Montaner et al, 2005; Silva et al, 2005). Consequently, it is tempting to hypothesize that GFs might contribute in restoring endothelium integrity and decreasing brain edema, inflammation, and perihematoma cell death (Krizanac-Bengez et al, 2004; Park et al, 2005). However, GFs, especially VEGF, could also induce recovery through the relationships between angiogenesis and macrophage inflammation. Increased microvessel density induced by GF is detected mainly in regions also containing macrophages, suggesting a role in ‘clean-up’ of necrotic debris (Manoonkitiwongsa et al, 2001, 2006). Accordingly, we postulate that the recovery mediated by GFs could be due to the angiogenesis and more robust inflammatory responses that allow faster removal of the hematoma necessary for the subsequent neurorepair processes.
Growth factors may also have an important role in brain repair after ICH as a result of the stimulation of the endothelial progenitor cell recruitment (Engelmann et al, 2006; Honold et al, 2006; Nan et al, 2005). Several studies have shown that angiogenic GFs such as VEGF, Ang-1, and G-CSF promote the mobilization and incorporation of endothelial progenitor cells to the neovascularization areas (Asahara et al, 1999; Hattori et al, 2001; Kalka et al, 2000), which results in a reduction of neurologic deficit in rats after hemorrhagic brain injury (Kalka et al, 2000).
As we have mentioned above, GFs can mediate recovery through endothelial cell regeneration, angiogenesis, decreasing brain edema, and perihematoma cell death, regarding the relationships between angiogenesis and macrophage inflammation, and stimulating the recruitment of endothelial progenitor cells. However, most of these mechanisms must occur in the brain tissue to promote recovery. Then, why circulating levels of GFs should reflect brain recovery after ICH? The fact that injury provoked by ICH induces blood–brain barrier disruption would allow cerebral substances to reach the blood torrent. According to this hypothesis, we suggest that the blood–brain barrier disruption caused by ICH may lead to an important increase in GF concentration in the bloodstream, reflecting the overexpression of these GFs in the brain tissue where they mediate the beneficial effects stated above.
In our opinion this study has some strengths. First, ICH volume and residual cavity were semiautomatically measured reducing the probability of intrarater variability. Second, GF were measured three times during the acute phase showing a consistent increase at 72 h. We recognize also some limitations. First, we did not determine GF at longer intervals after the acute phase of ICH; hence, we cannot conclude if good recovery was associated with an acute and transient increase in GF levels or it was the result of a sustained overexpression of these factors during the follow-up. Second, we cannot rule out a GF increase as an epiphenomenon of milder ICH. However, the favorable effect of the acute increase in serum levels of GF was independent of other important prognostic variables at baseline. Finally, 3 patients were treated with the recombinant activated factor VII vs placebo in the FAST Trial, which showed a reduction in the hematoma growth, and 22 patients in a failed neuroprotectant trial. However, a sensitivity analysis excluding these patients showed a similar effect.
In conclusion, an increase in serum levels of VEGF, G-CSF, and Ang-1 after ICH is independently associated with a better clinical outcome. Whether GF may promote brain repair and regenerative angiogenesis in humans remains to be clarified.
