Abstract
Deep brain stimulation (DBS) represents a major advance in the treatment of various severe movement disorders or neuropsychiatric diseases. Our understanding of the mechanism of action of this surgical treatment has greatly benefited from functional imaging studies. Most of these studies have been conducted in patients with Parkinson's disease (PD) treated by bilateral subthalamic nucleus (STN) stimulation. These studies have notably underlined the fact that STN stimulation influences motor, limbic, or associative cortical-subcortical loops in various (sometimes contradictory) ways. We present an up-to-date review of the information provided by functional imaging studies in surgery for PD, dystonia, tremor, as well as in psychiatric disorders such as depression or obsessive-compulsive disorder. On the basis of this information, proposed mechanisms of action of DBS are discussed, as well as the need for additional approaches such as improved anatomical localization of the contact used for stimulation or a better understanding of the electrical distribution around the electrode.
Introduction
During the past two decades, deep brain stimulation (DBS) has been recognized as an efficient therapy for tremor (target=thalamus), Parkinson's disease (PD) (targets=subthalamic nucleus (STN), or internal segment of the globus pallidus (GPi), thalamus, pedunculopontine nucleus (PPN)), and dystonia (target=GPi) (Benabid et al, 1993; Coubes et al, 2000; Limousin et al, 1995). The indications are growing rapidly and recent reports suggest its efficacy in Gilles de la Tourette syndrome, depression, and obsessive-compulsive disorder (OCD) (Aouizerate et al, 2005; Houeto et al, 2005; Mallet et al, 2008; Mayberg et al, 2005). However, despite its effectiveness, its mechanisms of action remain a matter of debate (Benazzouz and Hallett, 2000; Kringelbach et al, 2007; McIntyre et al, 2004; Vitek, 2002). Functional imaging using positron emission tomography (PET) or single-photon emission computed tomography (SPECT) provides a direct in vivo approach to fill this gap in our knowledge. The aim of this review was to summarize the contributions that have been made by functional imaging in the understanding of DBS in different disorders and to consider the directions that future imaging studies of DBS may take.
Materials and methods
Data for this review were identified by searches on Medline using the terms ‘deep brain stimulation’, ‘thalamus’, ‘subthalamic nucleus’, ‘internal pallidum’, ‘Parkinson’, ‘tremor’, ‘dystonia’, ‘obsessive compulsive disorders’, ‘Tourette syndrome’, ‘depression’, ‘functional imaging’, ‘PET’, and ‘SPECT’. References were also identified from relevant articles and through searches of the author's files. Only papers published in English were reviewed.
Summary of the Different Radiotracers Used
Positron emission tomography and more rarely SPECT are classically used to better understand the mechanism of action and consequences of DBS. Functional magnetic resonance imaging (MRI) is not performed in DBS-implanted patients for safety reasons. Several PET radiotracers can be used. H215O explores the changes in cerebral blood flow induced by stimulation during a specific task. The dopaminergic function can be analyzed using 18F-Dopa, a presynaptic dopaminergic tracer, and [11C]-raclopride, a dopamine D2 receptor ligand, the binding of which is dependent on the amount of endogenous extracellular dopamine. In addition, several radiotracers have been developed for monitoring the integrity of the presynaptic dopaminergic system affected in PD based on DAT antagonists such as methylphenidate and cocaine (tropane derivatives) (Shih et al, 2006). By labeling glucose (18F-fluorodeoxyglucose, FDG), the radiotracer imaging technique can determine local metabolism.
Summary of the Model of Basal Ganglia Circuitry
Before considering functional imaging studies of DBS in different diseases, it is useful to give a brief and simplified overview of the organization of basal ganglia (BG) circuitry. Cortico-basal ganglia-cortical circuits are organized in parallel, largely segregated but interconnected, closed-loop projections (Alexander et al, 1986). These circuits can be divided into motor (putamen projecting mostly to the supplementary motor area (SMA), motor cortex, and the lateral premotor area), associative (dorsal caudate projecting to the dorsolateral prefrontal cortex (DLPFC)), and limbic loops (ventral striatum projecting mostly to the anterior cingulate cortex (ACC) and orbital frontal cortex; Figure 1). In each nucleus of the BG, motor, associative and limbic territories are represented (Alexander et al, 1986). This is crucial while analyzing the effect of DBS, notably STN DBS, during motor, cognitive, or emotional tasks.
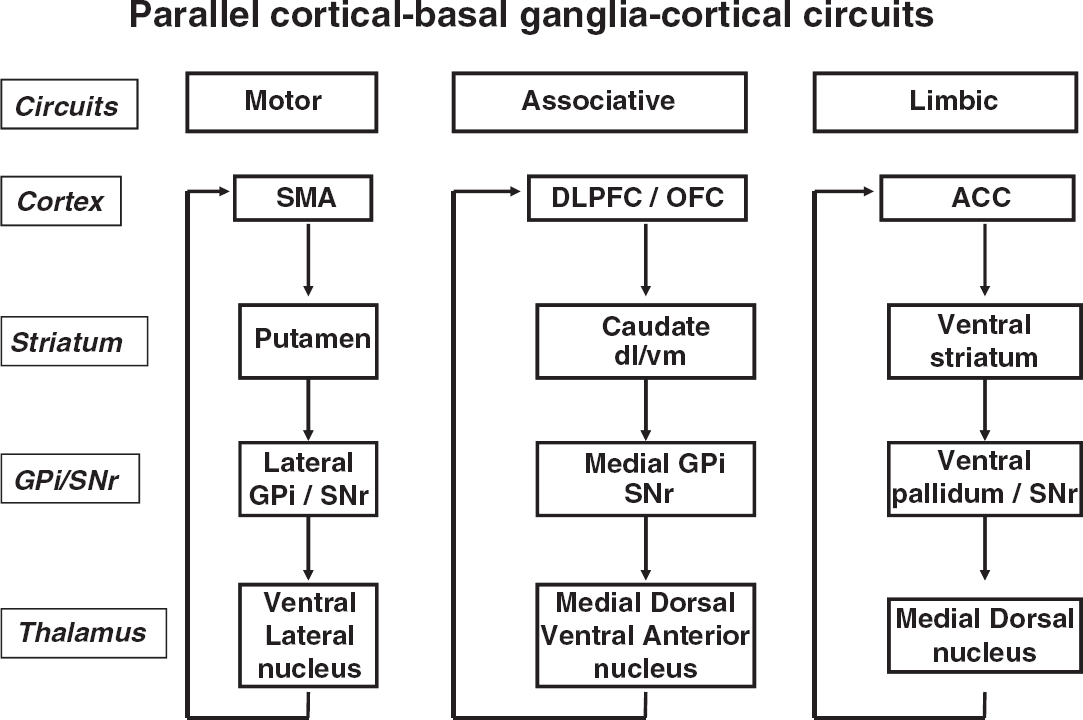
Parallel loop models of corticostriatal connectivity. SMA, supplementary motor area; DLPFC, dorsolateral prefrontal cortex; ACC, anterior cingulate cortex; GPi, internal globus pallidus; SNr, substantia nigra pars reticulata; VL, ventrolateral; VA, ventroanterior; DM, dorsomedial.
The PD model of bradykinesia has been developed from research conducted in a parkinsonian monkey in the late 1980s, and has then been progressively modified to become considerably more complex (DeLong, 1990; Yelnik, 2008). Although oversimplified, this model may help us understand or predict the changes of brain activation induced by DBS, especially in PD. Briefly, the cortex sends projections to the striatum. The striatum projects to the GPi through two different pathways: a direct striatal–pallidal pathway, which facilitates movement, and an indirect striatal–GPe–STN–GPi pathway, which inhibits movement. Both pathways receive dopaminergic innervation from the substantia nigra pars compacta (SNc), providing an inhibition of the excitatory indirect pathway and an excitation of the inhibitory direct pathway. The GPi, PPN, and substantia nigra pars reticulata (SNr) receive excitatory projections from STN. GPi, the main output area of BG, sends inhibitory projections to the thalamus, which in turn projects to the cortex (Figure 2A). In PD, the dopaminergic degeneration in SNc leads to a profound dysfunction of these pathways, with a hyperactivity of the indirect pathway and a reduction in activity of the direct pathway. In PD STN, neuronal activity is increased, which induces an inhibition of the thalamus through the Gpi and, in turn, a reduction in thalamocortical excitatory input (Figure 2B). According to this model, STN DBS, which is supposed to be inhibitory, or subthalamotomy should reduce the overactivity of STN and reverse the inhibitory output from the GPi to the thalamus and, in turn, improve cortical activation (Figure 2C). In dystonia, dysfunction is even more complex and both the indirect and direct pathways are hyperactive, leading to an abnormal pattern of GPi activity that reduces its inhibition of the thalamus and subsequently induces a release of thalamocortical activity (Vitek et al, 1999).
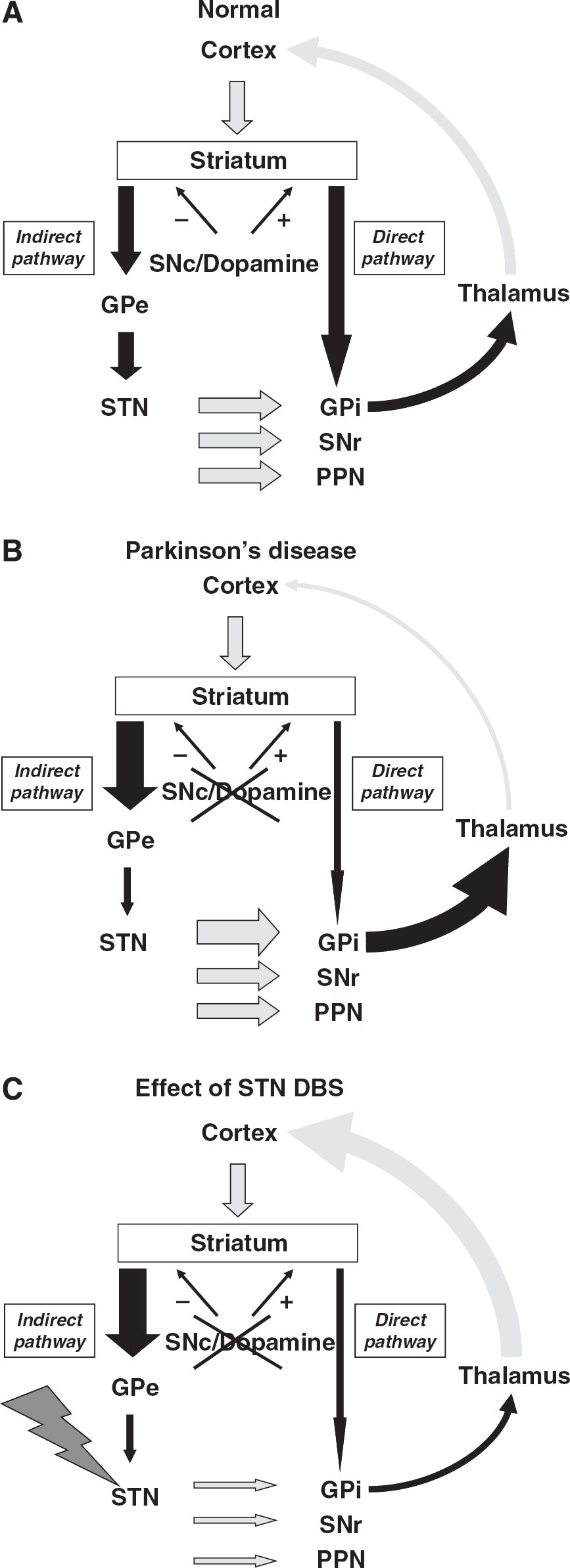
Cortical-subcortical loops in (
Functional Imaging Studies in Parkinson's Disease
Effect of DBS at rest
Subthalamic nucleus stimulation has been developed in the early 1990s to alleviate severe motor symptoms in PD (Limousin et al, 1995). This treatment improves all components of the parkinsonian triad, as well as levodopa-induced motor complications, and allows a major reduction in medication intake in patients with advanced PD (Kleiner-Fisman et al, 2006).
In the resting state, network analyses of 18F-FDG PET data have reported the presence of an abnormal spatial covariance pattern associated with the motoric features of PD (Eckert et al, 2007; Eidelberg et al, 1994; Moeller et al, 1999). This PD-related covariance pattern is characterized by increased pallidal, thalamic, and motor cortical metabolic activities associated with relative reductions in the lateral premotor cortex and the parieto-occipital regions (Eidelberg et al, 1994). A reduced expression of this abnormal PD-related covariance pattern has been observed during STN DBS (Trost et al, 2006), as well as during GPi DBS (Fukuda et al, 2001b). Interestingly, direct comparisons between STN DBS and dopaminergic medication have been carried out, which showed no difference in the degree of PD-related covariance pattern modulation (Asanuma et al, 2006; Hirano et al, 2008), supporting a common mechanism for these therapies (Krack et al, 2002; Vingerhoets et al, 2002).
Overall, a decrease in abnormal motor and cerebellar activities has been consistently reported with STN stimulation (Asanuma et al, 2006; Geday et al, 2009; Hershey et al, 2003; Trost et al, 2006). This decrease observed in the motor cortex was found to be proportional to the frequency of stimulation (Haslinger et al, 2005) and was associated with clinical improvements (Cilia et al, 2009). In addition, a significant correlation between improvement of rigidity and decreased regional cerebral blood flow (rCBF) in the SMA has recently been observed (Karimi et al, 2008). In contrast, STN DBS has been found to increase activity in precuneus and occipitotemporal cortices (Asanuma et al, 2006; Cilia et al, 2009; Geday et al, 2009; Goerendt et al, 2006; Grafton et al, 2006), consistent with the known preferential activation of these areas during resting states (Raichle et al, 2001). These results indicate that this rest-related activation is impaired in PD and restored by STN DBS.
With respect to subcortical structures, the literature is more equivocal. Indeed, metabolic studies using PET and 18F-FDG in resting PD patients have shown that STN DBS reduces glucose metabolism in the putamen and GPi (Asanuma et al, 2006; Trost et al, 2006). However, recent data suggested that STN DBS acts at rest through an excitatory mechanism with activation of the STN and the directly connected GPi (Geday et al, 2009; Hilker et al, 2008). Furthermore, although some studies reported increased activity in the thalamus during STN DBS (Goerendt et al, 2006; Hershey et al, 2003; Hilker et al, 2004; Karimi et al, 2008), others observed decreased activity (Geday et al, 2009). Altogether, these data are difficult to reconcile with a simple model of STN inhibition (which would only lead to an increase in cortical activation) or excitation (which would only, according to the BG model, lead to a decrease in cortical activation). Electrophysiological studies have also shown opposite findings, some showing that STN DBS induces an increase in GPi neuronal activity (in favor of an excitatory mechanism) and others finding a decrease in GPi and an increase in thalamic electrophysiological activity (in favor of an inhibitory mechanism) (Benazzouz and Hallett, 2000; Windels et al, 2000). It is noteworthy that methodological differences may account for some of the discrepancies (Geday et al, 2009). In keeping with this idea, it has recently been suggested, for instance, that increases in subcortical regions are an artifactual by-product of biased ratio normalization to the global mean used in studies comparing PD patients with healthy subjects (Borghammer et al, 2009).
Effect of DBS during motor execution
In PD, bradykinesia and rigidity have been shown to be related to a lack of activation of the DLPFC, SMA, and ACC because of an inhibition of the thalamocortical excitatory pathway (Jahanshahi et al, 1995; Jenkins et al, 1992; Playford et al, 1992). In parallel, the recruitment, even at an early stage of the disease, of ‘accessory’ pathways or regions has been recognized in numerous studies (Rascol et al, 1997; Sabatini et al, 2000; Samuel et al, 1997; Thobois et al, 2000). These overactivations could either correspond to a positive phenomenon (i.e., leading to the improvement of motor performance) or only reflect the inability of the central nervous system to select the appropriate program to perform the task (Hanakawa et al, 1999; Turner et al, 2003; Yu et al, 2007).
Functional imaging studies using H215O PET have shown that, during motor execution, DBS of the STN induces an increased activation of the DLPFC, SMA, and ACC and, concomitantly, a decreased activation of the so-called ‘accessory’ pathways (Ceballos-Baumann et al, 1999; Grafton et al, 2006; Hershey et al, 2003; Limousin et al, 1997; Strafella et al, 2003a; Thobois et al, 2002). Interestingly, most of these studies are consistent with clear task-dependent changes of brain perfusion induced by STN DBS. In addition, it has been argued that during motor execution, STN DBS selectively improves the recruitment of appropriate motor areas and suppresses the activation of inappropriate or competing cortical regions, whereas in resting PD patients, the effect of STN stimulation is a reduction of an abnormally high activity in the motor cortex (Ceballos-Baumann et al, 1999; Geday et al, 2009; Payoux et al, 2004).
The neural correlates of GPi stimulation in PD have been less investigated. Two studies have found an increase in ACC and SMA activation during motor execution similar to what is observed after STN DBS (Fukuda et al, 2001a; Limousin et al, 1997). In contrast, no increase in DLPFC activation was observed, which constitutes a major difference from STN DBS and could be explained by differential pathways influenced by stimulating each target.
Effect of DBS of the PPN on gait-related symptoms of PD
The PPN region is involved in gait initiation and maintenance (Pahapill and Lozano, 2000). In advanced PD, gait and postural instability become a major issue for clinical management, as they are usually resistant to levodopa and STN DBS. In animal model of PD, the electrophysiological activity of PPN is reduced and stimulation or disinhibition of this structure can improve gait and akinesia (Jenkinson et al, 2004). Recently, some studies have shown that PPN DBS at a low frequency can improve gait disturbance and freezing in advanced PD (Stefani et al, 2007). At present, however, the mechanism of action of PPN DBS remains unclear. To date, only two PET activation studies have been reported, showing that (1) at rest, unilateral PPN DBS increases rCBF bilaterally in the thalamus and ipsilaterally to the stimulation in the putamen, cerebellum, and insular cortex in an advanced PD patient (Strafella et al, 2008); and (2) during a self-paced alternating motor task of the lower limbs, unilateral PPN DBS increases rCBF in the medial sensorimotor areas (including caudal SMA) in three advanced PD patients (Ballanger et al, 2009). Interestingly, this latter observation was associated with changes in the lower limb movement pattern represented by a lower frequency and an increase in amplitude. This indicates that PPN DBS may induce significant functional changes in the neural networks associated with the control of voluntary movements of the lower limbs. However, whether these findings account for the proposed clinical effects of DBS PPN remains to be proven in a larger sample of patients.
Effect of DBS during speech
Dysarthria is a disabling symptom frequently encountered in advanced PD. In healthy subjects, speech production is associated with a bilateral activation of the orolingual region of the primary motor cortex and of the cerebellum, thalamus, and caudate (Murphy et al, 1997). In PD, the profile of brain activation impairment during speech is different from the one reported during limb movements. Indeed, the primary motor cortex and cerebellum are underactivated, whereas SMA and DLPFC are overactivated during speech in PD patients relative to healthy controls (Pinto et al, 2004). These overactivations may correspond to compensatory mechanisms or may alternatively contribute directly to the pathophysiology of dysarthria. STN DBS does not usually improve speech intelligibility, but several studies have reported an improvement in some phonatory and articulatory speech parameters in patients with PD (Gentil et al, 2001). In PD patients with a clear improvement in the biomechanical components of speech induced by STN DBS, a PET activation study revealed that this improvement was associated with a correction of abnormalities of the brain activation profile described above (Pinto et al, 2004). Indeed, an increased activation of the primary motor cortex and cerebellum and a decreased activation of SMA were shown. This study also shows that the consequences of STN DBS are clearly task dependent and mostly consist of restoring a selective pattern of brain activation and suppressing inappropriate cortical recruitment.
Effect of DBS on cognition
Overall, STN stimulation does not lead to cognitive deterioration in carefully selected patients (Castelli et al, 2006; Witt et al, 2008). However, concerns have been raised by reports of changes in specific aspects of cognition (Krack et al, 2003). Of these, an impairment of specific cognitive tasks that require response selection through an inhibition of inappropriate or competing responses has repeatedly been reported (Ardouin et al, 1999; Jahanshahi et al, 2000a; Saint-Cyr et al, 2000). These response conflict tasks represented, for example, by Stroop and random number generation (RNG) are mostly mediated by ACC or the prefrontal cortex (Carter and van Veen, 2007; Jahanshahi et al, 2000b). Positron emission tomography studies comparing ‘on’ versus ‘off’ stimulation conditions in PD have shown that DBS of the STN is associated with decreased rCBF in the ACC and ventral striatum during Stroop and with decreased rCBF in the right orbitofrontal cortex, the left inferior frontal gyrus, and the left inferior temporal gyrus during a verbal fluency task (Schroeder et al, 2002, 2003). Random number generation is another example of a task, in which the participant has to generate numbers between 1 and 9 in a random manner that requires suppression/avoidance of habitual counting in series. During RNG, DLPFC and ACC are significantly activated relative to a control counting task, and rCBF in the left DLPFC is negatively associated with a measure of habitual counting during RNG and with rCBF in the temporal cortex (Jahanshahi et al, 2000b). It has been proposed that during RNG, strategic response selection and generation of numbers in a random manner is achieved through the inhibitory influence of the left DLPFC over the superior temporal cortex, the putative site of a number associative network, to keep activity in this area low and to prevent spreading activation in the network. With DBS of the STN, performance of PD patients on fast-paced RNG is worse on ‘on’ stimulation compared with ‘off’ stimulation. This is because of increased rCBF in the GPi and decreased rCBF in the left DLPFC and in the anterior and posterior cingulate cortex (Thobois et al, 2007). The impaired performance induced by STN DBS on verbal fluency, the Stroop, and fast-paced RNG has been related to a reduced activation of DLPFC and ACC, two areas involved in response selection through an inhibition of inappropriate or competing responses (Figure 3A; Thobois et al, 2007). In keeping with these observations, a significant linear relationship between glucose uptake and verbal fluency performance after STN DBS in advanced PD patients has recently been observed (Kalbe et al, 2009). The best correlations were found in the frontal lobe, including the left DLPFC and Broca area, as well as in the right dorsal ACC. Thus, in PD, DBS of the STN can improve the functioning of circuits involved in motor execution (Ceballos-Baumann et al, 1999; Grafton et al, 2006; Limousin et al, 1997; Strafella et al, 2003a; Thobois et al, 2002) and, conversely, disrupt circuits implicated in cognitive processing (Kalbe et al, 2009; Schroeder et al, 2002, 2003; Thobois et al, 2007), reducing notably ‘cognitive flexibility’ (Witt et al, 2004). Deficits have been reported in an abnormal metabolic cognitive network (PD-related cognitive pattern) associated with memory and executive dysfunction in nondemented PD patients, which is not influenced (i.e., not improved) by STN DBS (Huang et al, 2007a, 2007b) in contrast to the motor-related pattern (PD-related covariance pattern) (Asanuma et al, 2006; Hirano et al, 2008).
Effect of DBS on emotional processing and behavior
Deep brain stimulation of STN can induce a wide range of behavioral and mood changes in PD (Appleby et al, 2007). Among these behavioral and mood changes, manic episodes have been reported (Mallet et al, 2007; Ulla et al, 2006). One recent single case study showed that mania induced by STN DBS was associated with an increased activation of the dorsal ACC, which is consistent with what is classically found in bipolar disorders (Figure 3B; Blumberg et al, 2000; Ulla et al, 2006). This STN-DBS-induced ACC dysfunction could explain the difficulties in performing Go/NoGo reaction time tasks (Hershey et al, 2004) and the emotional liability of these patients. However, another study did not confirm this finding and found, in contrast, a decreased ACC activation (Mallet et al, 2007). These discrepancies may be partly because of differences in electrode placement but these results need confirmation on a larger group of patients. Interestingly, acute depression induced by DBS of the STN has also been shown to be associated with ACC and prefrontal overactivation; which could mean that the kind of mood changes induced by DBS may vary on the basis of the precise location of the electrode and the presurgical psychiatric state of the patient (Bejjani et al, 1999). Another single report using functional MRI has also shown that depression induced by DBS of the STN was correlated with changes in mesolimbic cortical structures, especially the superior and medial (Brodmann's area (BA) 9/10) prefrontal and anterior cingulate cortices (BA 24), anterior thalamus, caudate, and brainstem (Stefurak et al, 2003).
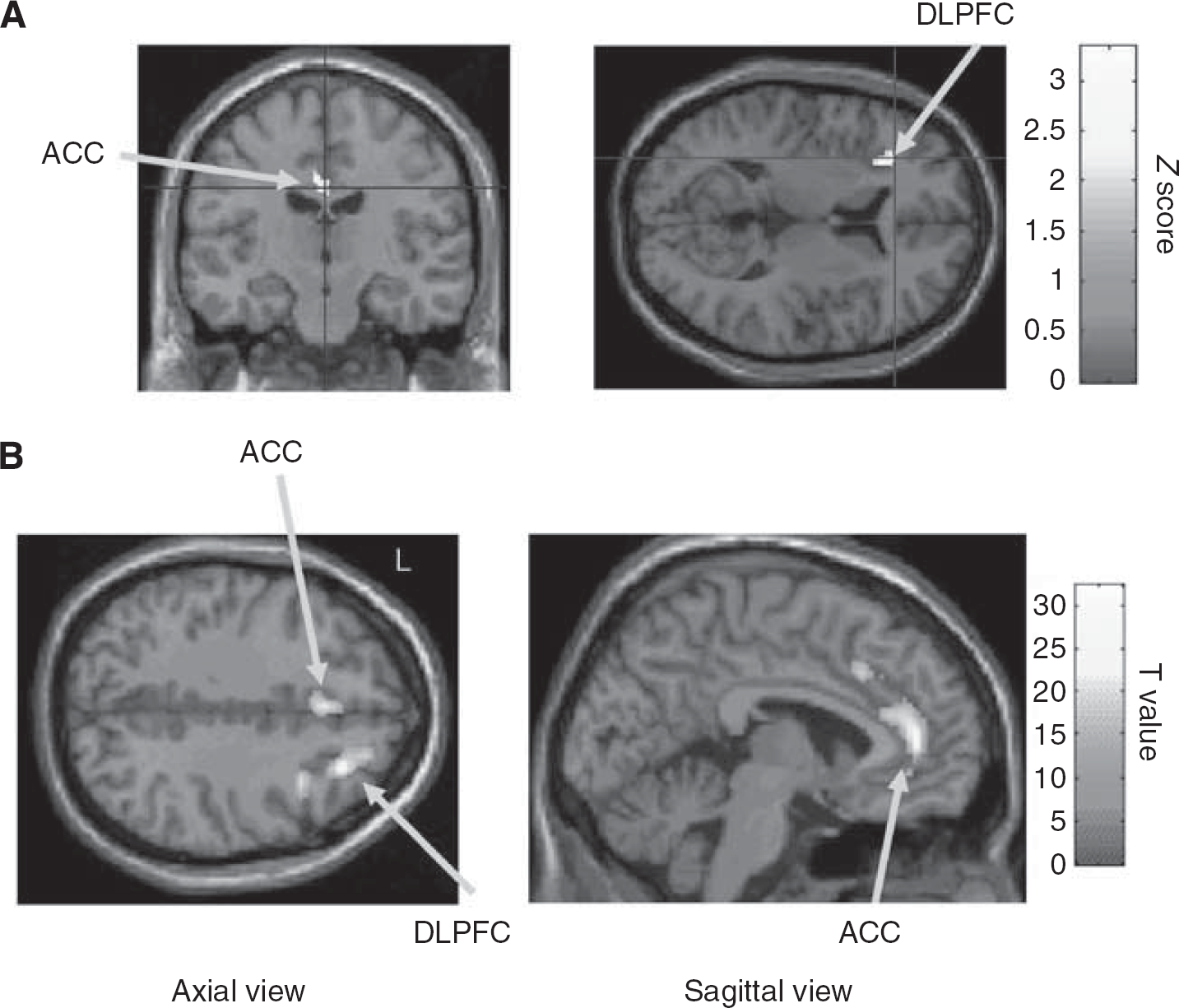
(
In addition, several studies have shown that bilateral STN stimulation disturbs negative emotional information processing with an impairment of the recognition of fear or anger in facial expressions (Dujardin et al, 2004). These difficulties have been related to a deficit of activation of the right fusiform gyrus, which is involved in the processing of emotion in visual stimuli, when PD patients were viewing emotionally expressive faces in the on-stimulation condition (Geday et al, 2006; Phan et al, 2002). Another recent study using PET and FDG showed that the deficit induced by DBS of the STN in recognition of emotion from facial expressions, especially fear, was related to a dysfunction of the orbitofrontal cortex, a region classically involved in emotion processing (Le Jeune et al, 2008).
Another important issue is the induction of apathy after STN DBS in some patients (Drapier et al, 2006). The improvement in apathy by dopaminergic drugs suggests that the worsening of apathy in some cases after surgery may be related to a reduction of these treatments after surgery (Czernecki et al, 2008). However, the reason why some patients develop apathy whereas others do not remains unknown. Therefore, further functional imaging investigations of the mechanisms of development or worsening of apathy after surgery for DBS of the STN are required.
Altogether, these imaging studies clearly show that the limbic changes associated with DBS of the STN are related to modifications of the functioning of limbic networks. They again reveal a differential action of DBS of the STN on the motor, associative, and limbic BG-frontal circuits.
Effect of DBS on vegetative/autonomic functions
Little data are available with regard to the effect of DBS of the STN on autonomic function, but some studies have shown that this therapy improves autonomic symptoms and fluctuations, such as drenching sweats (Witjas et al, 2007). In addition, weight gain has consistently been reported after DBS of the STN (Barichella et al, 2003). This weight gain has been related to a restoration of normal resting energy expenditure (Montaurier et al, 2007). Another aspect of the nonmotor signs that may be influenced by DBS of the STN is sleep. Several studies have indeed shown that DBS of the STN improves sleep architecture (Arnulf et al, 2000). Some data have also shown in PD that DBS of the STN positively influences urodynamic parameters during storage phase (Finazzi-Agro et al, 2003). A recent study has found that this improvement in urinary bladder function is related to a decreased activation of the ACC and lateral PFC during bladder filling in on-stimulation condition (Herzog et al, 2006). It has been suggested that DBS of the STN improves continence, which in turn leads to a reduction in the activation of areas involved in the maintenance of continence.
Does DBS have a neuroprotective effect?
Some animal experiments have suggested that DBS of the STN could be neuroprotective (Piallat et al, 1996). However, functional imaging studies using PET and 18F-Dopa, which allow the analysis of the rate of dopaminergic degeneration, failed to show any neuroprotective effect of DBS of the STN in PD (Hilker et al, 2005). Nevertheless, to draw definitive conclusions with regard to this question, it would be of interest in the future to include a control group.
Does STN DBS increase striatal dopamine release?
Some animal studies have suggested that STN DBS could increase the extracellular content of striatal dopamine release (Bruet et al, 2001). However this was not confirmed using PET studies and [11C]-raclopride, a D2 dopamine antagonist ligand (Strafella et al, 2003b; Thobois et al, 2003). Nevertheless, this does not rule out the idea of increased striatal DA release with STN DBS. Indeed, [11C]-raclopride has relatively poor sensitivity for detecting low levels of DA release. In fact, as the test–retest variability of [11C]-raclopride binding in normal subjects under baseline conditions is between 7% and 13%, we can assume that small variations of DA release, leading to a smaller modification of [11C]-raclopride binding, may be within the range of this baseline variability and thus difficult to detect when using such a radioligand (Volkow et al, 1993). Therefore, if STN DBS increases striatal DA release, this phenomenon will remain of small magnitude.
Functional imaging studies in dystonia and tremor
Dystonia is characterized by an excess of brain activation leading to sustained muscle contractions inducing abnormal postures and movements. The causes of dystonia can be divided into two broad groups: primary and secondary dystonias. Although the latter refers to dystonia that is primarily a result of specific factors (such as exposure to certain medications, lesions of BG, or trauma), the precise cause of primary dystonias is unknown, although it has become clearer that they are likely to have a genetic basis. In addition, in patients with primary dystonia, brain MRI is normal in contrast to secondary dystonias. In primary dystonia, overactivations were found in the DLPFC, premotor cortex, ACC, cerebellum, and putamen, whereas a decrease in rCBF is usually observed in the primary motor cortex (Ceballos-Baumann et al, 1995b; Detante et al, 2004; Ibanez et al, 1999; Kumar et al, 1999; Playford et al, 1998). Conversely, in secondary dystonia, rCBF is often increased in the primary motor cortex (Ceballos-Baumann et al, 1995a). Other studies on writer's cramp and Meige's syndrome have shown an altered somatotopic representation, which contributes to the loss of functional selectivity of muscle activity (Delmaire et al, 2005).
Few studies have analyzed the impact of GPi DBS on the abnormalities of brain activation in dystonia. Two studies have been conducted in generalized primary dystonia, and both showed during motor execution a major reduction in brain overactivity, including, notably, in the prefrontal cortex (Detante et al, 2004; Kumar et al, 1999). In tardive dystonia, similar results were obtained, but the stimulation-induced decrease mostly involved the cerebellum and primary motor cortex (Figure 4; Thobois et al, 2008). These convergent data clearly show that GPi DBS in dystonia acts through the restoration of a selective brain activation pattern and the suppression of the recruitment of inappropriate cortical regions.
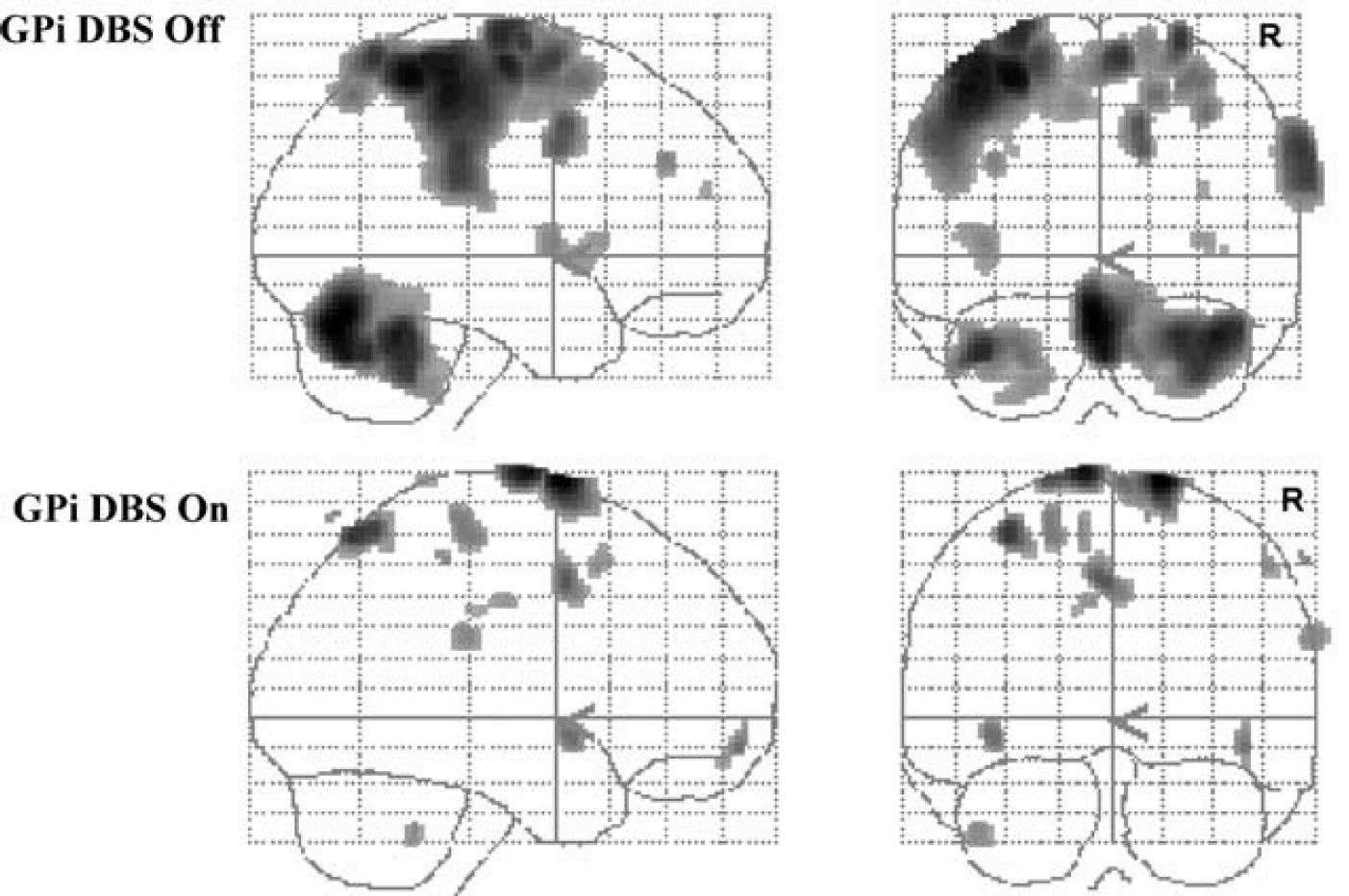
Decreased activation induced by GPi DBS during motor execution in a group of patients with tardive dystonia. PET H215O study. Data are superimposed on coronal and sagittal sections of a glass brain.
Deep brain stimulation in movement disorders was first applied to treat severe tremor (essential or parkinsonian tremor) (Benabid et al, 1993). The pathophysiology of essential tremor is still a matter of debate, but the implication of the cerebellum is widely accepted (Lorenz and Deuschl, 2007). In parkinsonian tremor, hyperactivation of the cerebellum has been shown (Deiber et al, 1993; Fukuda et al, 2004). This hyperactivation is reversed by thalamic DBS (Deiber et al, 1993; Fukuda et al, 2004). In addition, thalamic DBS induces a reduction in the sensorimotor cortex activation ispilateral to stimulation (Fukuda et al, 2004).
Functional imaging studies in psychiatry
As noted in the Introduction, the field of DBS has expanded and is now under investigation in psychiatric disorders such as depression or OCD (Mallet et al, 2008; Mayberg et al, 2005). For instance, stimulation of the ventral ACC (BA 25) has been applied to severely depressed patients with promising results (Mayberg et al, 2005). This surgical approach to treat medically refractory depression has been based on convergent data, suggesting that this region is involved in sadness and in the modulation of negative effect (Mayberg et al, 1999). In addition, it has been shown that successful antidepressant treatment by medication or other approaches led to a reduction in the activity of this region (Mayberg et al, 2000). In accordance with these data, before surgery, a hyperactivation of the ventral ACC (BA 25) and a hypoactivation of the dorsal ACC were observed in depressed patients. Several months after surgery, concomitantly with an improvement in depression, an increased activation of the dorsal ACC and a decreased activation of the ventral ACC were observed (Mayberg et al, 2005). The latter result fits well with the conclusion of the meta-analysis of emotion activation studies conducted by Phan et al (2002), showing that sadness was significantly associated with activity in ventral ACC. This result is also in line with many theories of the functions of the medial prefrontal cortex suggesting functional divisions within the ACC, the ventral section being more implicated in limbic processing and the dorsal section in cognitive tasks (Amodio and Frith, 2006; Koski and Paus, 2000; Margulies et al, 2007).
In OCD, a dysfunction in the orbitofrontal, ACC, striatum, and thalamus has been found (Rauch et al, 2006). Stimulation of a ventral striatum/ventral capsule target led to a significant activation of orbitofrontal and anterior cingulate cortices, striatum, Gpi, and thalamus (Rauch et al, 2006). Furthermore, metabolic studies showed that stimulation of the anterior capsule induced a decrease in prefrontal metabolic activity, especially in the subgenual ACC, which is considered to reflect an interruption of the cortico-thalamo-striato-cortical circuit (Van Laere et al, 2006). In addition, the degree of improvement in OCD inversely correlated with the metabolism of the left ventral striatum, amygdala, and hippocampus (Van Laere et al, 2006). All these findings support a major role for the dysfunction of the limbic circuits in OCD pathophysiology and show that DBS modulates these pathways.
Conclusion
Functional imaging with PET has provided a unique window for analyzing in vivo the consequences of DBS on brain activation patterns and has been applied to various disease that benefit from this surgical approach. These studies provide crucial information for a better understanding of the disease itself and, conversely, for clarifying the mechanism of action of DBS. All these imaging studies show that DBS finely tunes specific circuits depending on the task and the underlying disease. As previously suggested, the results of imaging studies also show that the effects of DBS of the STN are task specific and depend on the particular networks engaged by specific tasks (Thobois et al, 2007). This fits well with animal studies, which suggest that subcortico-cortical connectivity depends on brain state, such as whether the animal is at rest or active (Magill et al, 2004, 2006; Sharott et al, 2005). Furthermore, it has been proposed that the mechanism of action of STN stimulation is not merely inhibitory or excitatory but could at the same time inhibit the soma of STN neurons and excite the projections from STN neurons or fibers passing near STN or other nuclei (McIntyre et al, 2004). It is possible that the balance between the inhibition of soma and the excitation of axons could be different for specific neural networks engaged by particular tasks. This could explain, for example, the differential effect of STN stimulation on the different frontostriatal circuits during motor versus cognitive tasks.
In addition, the functional imaging approach clearly shows that stimulating a very small structure such as STN induces complex modifications of the pattern of brain activation involving entire networks and not only isolated regions. Therefore, the findings of these studies have to be interpreted in terms of functional connectivity between different brain areas, leading to a better knowledge on how one region interacts with others (Thobois et al, 2007). In addition, independently of the therapeutic action of DBS, these surgical procedures when combined with imaging represent a unique in vivo opportunity in humans to directly ‘manipulate’ the functioning of specific structures and to understand the functions of these structures.
One of the critical points in relation to the interpretation of the results of functional imaging studies is the precise location of electrodes, to determine which of the motor, associative, or limbic territories are affected by stimulation depending on the topography of stimulation. In the same manner, a better understanding of the diffusion of the electrical field around the electrode is crucial and would enhance our understanding and interpretation of the results from imaging studies (Maks et al, 2009; Vasques et al, 2009). In addition, as most functional imaging studies have investigated modifications of brain perfusion, the study on the consequences of DBS in terms of changes of neurotransmitter release remains largely unknown and would be of great interest. Therefore, in future, this imaging methodology will continue to offer an incomparable window into the mechanism of action of DBS, which, 20 years after the introduction of this surgical approach, is still incompletely understood (Kringelbach et al, 2007).
Footnotes
Acknowledgements
Individual contributions to the paper: B Ballanger and S Thobois conducted the bibliographical search and wrote the first draft of the paper. M Jahanshahi and E Broussolle made corrections to the paper for important intellectual content.
The authors declare no conflict of interest.
