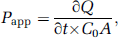Abstract
Ligand-mediated brain-targeting drug delivery is one of the focuses at present. Elucidation of exact targeting mechanisms serves to efficiently design these drug delivery systems. In our previous studies, lactoferrin (Lf) was successfully exploited as a brain-targeting ligand to modify cationic dendrimer-based nanoparticles (NPs). The mechanisms of Lf-modified NPs to the brain were systematically investigated in this study for the first time. The uptake of Lf-modified vectors and NPs by brain capillary endothelial cells (BCECs) was related to clathrin-dependent endocytosis, caveolae-mediated endocytosis, and macropinocytosis. The intracellular trafficking results showed that Lf-modified NPs could rapidly enter the acidic endolysosomal compartments within 5 mins and then partly escape within 30 mins. Both Lf-modified vectors and NPs showed higher blood–brain barrier-crossing efficiency than unmodified counterparts. All the results suggest that both receptor- and adsorptive-mediated mechanisms contribute to the cellular uptake of Lf-modified vectors and NPs. Enhanced brain-targeting delivery could be achieved through the synergistic effect of the macromolecular polymers and the ligand.
Keywords
Introduction
The brain capillary endothelium forms a formidable barrier to the entry of various physiologically active drugs, including gene medicines, into the central nervous system. Meanwhile, the tight junctions that seal brain capillary endothelial cells (BCECs) prevent circulating compounds, including these therapeutic drugs, from reaching the brain by passive transport or through the paracellular route (Abulrob et al, 2005). Transcytosis, which neither alters the cellular plasma membrane nor disrupts the cellular tight junctions, is considered to be the main active mechanism for the brain to take-up exogenous substances (Jones and Shusta, 2007). Many proteins, including lactoferrin (Lf) and transferrin, could effectively cross the blood–brain barrier (BBB) through the transcytosis mechanism of BCECs (Demeule et al, 2002; Fillebeen et al, 1999; Pan et al, 2004). The active mechanism of these proteins could be used advantageously to design nano-scaled brain-targeting drug delivery systems that might possess the similar intracellular trafficking process with the proteins (Huang et al, 2007b, 2008).
The first and most important step of the transcytosis process is endocytosis. There exist several endocytic mechanisms on the BBB (Smith and Gumbleton, 2006), including (1) receptor-mediated endocytosis in which a ligand-modified system will specifically bind to its cognate receptor to elicit cellular internalization; (2) adsorptive endocytosis in which the drug delivery system will bind to the cellular surface through nonspecific mechanisms, such as electrostatic interactions; and (3) macropinocytosis, which provides an efficient route for nonselective endocytosis of macromolecules (Walsh et al, 2006). Different mechanisms have different processes, e.g., receptor and adsorptive-mediated endocytosis could depend on clathrin and/or caveolae, respectively (Smith and Gumbleton, 2006). Until now, these endocytic mechanisms have been exploited to design brain-targeting gene delivery systems. For example, OX26, a monoclonal antibody to transferrin receptors, was attached to liposomes for targeting exogenous genes to the brain through receptor-mediated endocytosis (Shi et al, 2001; Xia et al, 2008). In addition, Lu et al (2006) prepared cationic albumin-conjugated nanoparticles (NPs), allowing exogenous genes delivering into the brain through adsorptive transcytosis.
In our previous studies, a novel efficient brain-targeting nonviral nano-scaled gene delivery system was constructed successfully (Huang et al, 2008). Polyamidoamine (PAMAM) dendrimer was used as the main macromolecular gene vector. In addition Lf, for the first time, was investigated as a brain-targeting ligand to modify PAMAM-based NPs to the brain, using hydrophilic polyethyleneglycol (PEG) as a spacer. There are two important attributes of Lf-modified NPs, namely (1) modified with Lf to target the corresponding receptors in the brain to enhance the brain-targeting ability, (2) using novel cationic macromolecular material, PAMAM, as the main gene vector, with high gene encapsulation ability.
Lactoferrin-modified NPs were first prepared in our laboratory, and their brain-targeting ability was fully shown (Huang et al, 2008). However, the brain-targeting mechanisms have not yet been elucidated. In this study, the uptake and transcytosis mechanisms of Lf-modified vectors and NPs were systematically investigated in vitro.
Materials and methods
Materials
Polyamidoamine dendrimer (generation=5, 21.43% w/w solution in methanol, containing 128 surface primary amino groups (molecular weight 28, 826 Da)) was purchased from Dentritech (Midland, MI, USA). α-Malemidyl-ω-N-hydroxysuccinimidyl PEG (NHS-PEG-MAL, molecular weight 3,400 Da) was obtained from Nektar Therapeutics (Huntsville, AL, USA). The BODIPY fluorophore (4,4-difluoro-5,7-dimethyl-4-bora-3a,4a-diaza-s-indacene-3-propionic acid, sulfosuccinimidyl ester, sodium salt), ethidium monoazide bromide (EMA), and LysoTracker Green were purchased from Molecular Probes (Eugene, OR, USA). Goat anti-bovine Lf antibody was obtained from Bethyl Laboratories (Montgomery, TX, USA). 14C-sucrose was purchased from Amersham Biosciences (Little Chalfont, UK). The plasmid DNA coding pGL2-Control Vector coding luciferase (Promega, Madison, WI, USA) was purified using the QIAGEN Plasmid Mega Kit (Qiagen GmbH, Hilden, Germany). Lactoferrin from bovine colostrum, SATA (N-succinimidyl S-acetylthioacetate), rabbit anti-goat IgG (immunoglobulin G) (whole molecule)-biotin antibody, streptavidin 10 nm colloidal gold labeled, phenylarsine oxide (PhAsO), filipin complex (from streptomyces filipinesis), colchicine,
Synthesis of Lactoferrin-Modified Vectors
A 1:2:1 (mol/mol/mol) conjugate of PAMAM, PEG, and Lf (designated as Lf-modified vectors) was synthesized successfully as described previously (Huang et al, 2008). Briefly, a 1:2 conjugate of PAMAM and NHS-PEG3400-MAL was prepared through specific reaction between the primary amino groups on the surface of PAMAM and the NHS groups of the bifunctional PEG derivative for 15 mins at room temperature. The resulting conjugate (designated as PEGylated vectors) was purified by ultrafiltration using a 5-kDa molecular weight cutoff membrane. At the same time, Lf was modified with thiol groups using SATA. The PEGylated vectors were then reacted with thiolated Lf, 1:1 (mol/mol), in phosphate-buffered saline (PBS, pH 7.0) for 24 h at room temperature to obtain Lf-modified vectors. The successful synthesis of Lf-modified vectors was shown previously (Huang et al, 2008).
Preparation of Nanoparticles
Lactoferrin-modified vectors were freshly synthesized and diluted to appropriate concentrations in distilled water. In 50 mmol/L of sodium sulfate solution, plasmid DNA solution was added to obtain specified weight ratios (10:1, PAMAM to DNA, w/w) and immediately vortexed for 30 secs at room temperature. Reporter genes encoding luciferase were used to complex with Lf-modified vectors, formulating Lf-modified NPs. PAMAM–DNA complexes were designated as unmodified NPs. Agarose gel electrophoresis was carried out to verify the complete complexation of vectors with DNA. Freshly prepared NPs were used in the following experiments.
Plasmid DNA was covalently labeled with fluorescent dye, EMA, when necessary. Fresh plasmid DNA solution (1 mg/mL of 0.05 mol/L Tris-HCl buffer, pH 8.0) was diluted with an aqueous solution of EMA (0.1 mg/mL) and incubated for 30 mins in the dark. The complex was then exposed to ultraviolet light for 1 h, and the resulting solution was precipitated by adding ethanol to a final concentration of 30% (v/v). The precipitate was collected by centrifugation and further dissolved in 50 mmol/L of sodium sulfate solution to obtain EMA-labeled plasmid DNA.
Characterization of Nanoparticles
The mean diameter and zeta potential of Lf-modified NPs were determined by dynamic light scattering using a Zeta Potential/Particle Sizer (PSS, Santa Barbara, CA, USA). To confirm the covalent conjugation of Lf in Lf-modified NPs, the immuno-gold staining technique was adopted. Goat anti-bovine Lf antibody was incubated for 1 h at 37°C with shaking at 80 r.p.m., to react with the Lf on Lf-modified NPs. Rabbit anti-goat IgG-biotin antibody was then added to react with goat anti-bovine Lf antibody for 1 h at 37°C with shaking at 80 r.p.m. Thereafter, the solution was further incubated for 1 h with streptavidin, 10 nm colloidal gold labeled, diluted 1:10 in 0.15 mol/L NaCl, 0.01 mol/L sodium phosphate, 5 mg/mL albumin, and 0.05% Tween 20, pH 7.0. The unreacted gold was removed through Sephacryl S-500 HP (GE Healthcare, Piscataway, NJ, USA), and the collected solution was centrifuged at 15,000 r.p.m. for 1 h at 4°C. The obtained NPs were resuspended with PBS (pH 7.0). Lactoferrin-modified NPs that were treated in the same way as mentioned above, except for the incubation with goat anti-bovine Lf antibody, were used as controls. Size and morphology of NPs were examined under a high-resolution transmission electron microscope (JEM-2010, JEOL, Tokyo, Japan).
Cellular Uptake at Different Temperatures
Lactoferrin-modified vectors were labeled with BODIPY according to Huang et al (2007a). Brain capillary endothelial cells were cultured in 33-mm dishes at a density of 1 × 105 cells per dish for 48 h, and examined under the microscope for confluency and morphology. The medium was removed, and the cells were incubated for 30 mins with BODIPY-labeled vectors (1 μmol/L in medium) at 4°C or with BODIPY-labeled vectors with and without the addition of 100 μmol/L free Lf at 37°C. After incubation, the wells were washed thrice with 2 mL PBS (pH 7.4, if not specified) and the cellular uptake was examined using a fluorescent microscope (IX2-RFACA, Olympus, Tokyo, Japan) and a confocal microscope (LeicalTCS SP2 AOBS, Heidelberg, Germany) for the X/Y and Z/Y sections, respectively.
Treatments with Inhibitors
Brain capillary endothelial cells were seeded at a density of 2 × 104 cells per well in a 24-well plate (Corning-Coaster, Tokyo, Japan), incubated for 72 h, and examined under the microscope for confluency and morphology. The medium was removed and the cells were pretreated with PhAsO (10 μmol/L), filipin (5 μg/mL), colchicine (10 μmol/L), excessive Lf (100 μmol/L), and polylysine (400 μg/mL) for 10 mins at 37°C. PhAsO is reported to inhibit clathrin-dependent endocytosis (Visser et al, 2004), and filipin is known to block the caveolae-mediated process (Kim et al, 2007). Colchicine is an inhibitor of macropinocytosis (Liu and Shapiro, 2003), whereas cationic polylysine could inhibit the uptake of cationic vectors. Excessive Lf might induce both competitive and cationic inhibition. Subsequently, BCECs were incubated with BODIPY-labeled PAMAM, BODIPY-labeled Lf, and BODIPY-labeled Lf-modified vectors in a final concentration of 1 μmol/L for 30 mins at 37°C. The cells were washed thrice with PBS and visualized under a fluorescent microscope. Brain capillary endothelial cells treated with BODIPY-labeled PAMAM, BODIPY-labeled Lf, and BODIPY-labeled Lf-modified vectors, without any inhibition, served as controls. For quantitative analysis, after being treated as described above, BCECs were incubated with Triton X-100 (1%, w/v) for cellular lysis and centrifuged at 3,000 r.p.m. for 1 min. The fluorescence intensity of BODIPY (excitation at 480 nm and emission at 510 nm) in the supernatant was assessed using a microplate fluorometer (LS 55, Perkin-Elmer, Waltham, MA, USA). Protein concentrations of the cell lysates were determined using the Bradford colorimetric assay with bovine serum albumin (Pierce, Rockford, IL, USA) as a protein standard. The relative uptake efficiency (RUE) was calculated as follows:
RUE=(experimental fluorescent intensity/mg protein)/(control fluorescent intensity/mg protein) × 100 (%)
The cellular inhibition of unmodified and Lf-modified NPs encapsulating EMA-labeled DNA was evaluated using similar methods as described above.
Confocal Microscopy
The EMA-labeled plasmid DNA solution was complexed with Lf-modified vectors to prepare EMA-labeled Lf-modified NPs as described above. Brain capillary endothelial cells were seeded in 33-mm dishes at a density of 5 × 104 cells per dish for 2 days. The medium was removed and the cells were treated with the fluorescent dye, LysoTracker Green, to label the acid compartments, including endosomes and lysosomes. The cells were then incubated with EMA-labeled Lf-modified NPs for 5 and 30 mins, respectively. After incubation, the wells were washed thrice with 2 mL PBS and examined under a confocal microscope (LeicalTCS SP2 AOBS) excited at 561 nm for EMA observation and at 488 nm for LysoTracker Green observation.
Transport Studies Across BCECs Monolayers
Brain capillary endothelial cells were seeded at a density of 5 × 104 cells per cm2 onto polycarbonate 24-well transwell filters of 1.0 μm mean pore size and 0.33 cm2 surface area (FALCON Cell Culture Insert, Becton Dickinson Labware, Franklin Lakes, NJ, USA). After culturing for ∼3 day, the cells were examined under the microscope for complete confluency. The cell monolayer integrity was monitored using an epithelial voltohmmeter (MILLICELL-ERS, Millipore, Billerica, MA, USA) to measure the transendothelial electrical resistance (TEER). Only cell monolayers with TEER exceeding 200 Ω cm2 were selected for this experiment (Xie et al, 2005).
PAMAM was iodinated as described previously with radioactivity of 1 μCi of 125I per μg PAMAM (Huang et al, 2007b). Radiolabeled PAMAM was applied to synthesize Lf-modified vectors. While BCECs were examined for complete confluency, 250 μL fetal bovine serum-free medium containing 0.6 μCi of 14C-sucrose was added to the donor chamber at time 0 to monitor the integrity of BCEC monolayers. At the same time, BCEC monolayers were treated separately with PAMAM, PAMAM plus excessive Lf (with a final concentration of 10 μg/μL), Lf-modified vectors, or Lf-modified vectors plus excessive Lf, with a final amount of 20 μg PAMAM per well. The incubation was performed at 37°C on a rocking platform at 50 r.p.m. At 5, 10, 15, 30, 45, and 60 mins, an aliquot of 20 μL from each acceptor chamber was removed and replaced with 20 μL of fresh fetal bovine serum-free medium. The radioactivity of 125I in each aliquot was assessed using a γ-counter (SN-695, Rihuan, Shanghai, China). Thereafter, 10 μL from the 20 μL aliquot was dissolved in 1 mL of scintillation cocktail and analyzed in a liquid scintillation counter (LS 6000SE, Beckman, Fullerton, CA, USA). The correction factor of 14C-sucrose was 25% for the equipment. The apparent permeability (Papp) was calculated according to Irvine et al (1999) as follows:
where ∂Q/∂t is the permeability rate (nmol/sec), C0 the initial concentration (nmol/mL) in the donor chamber, and A the surface area (cm2) of the membrane filter.
Both TEER and permeability of 14C-sucrose of BCEC monolayers were measured at time 0 and 60 mins, respectively, to monitor the integrity of monolayers. Brain capillary endothelial cell monolayers without any treatment served as controls.
The transport studies of Lf-modified NPs were similar to those of Lf-modified vectors described above. Brain capillary endothelial cell monolayers were treated separately with unmodified NPs, unmodified NPs plus excessive Lf, Lf-modified NPs, or Lf-modified NPs plus excessive Lf, with a final amount of 20 μg PAMAM per well. The results were expressed as Papp. Similarly, TEER and permeability of 14C-sucrose of BCEC monolayers were measured to monitor the integrity of monolayers.
Statistical Analysis
All quantitative measurements were carried out in quadruplicate. The data were expressed as mean±s.e. of the mean (s.e.m.). Statistical analysis was carried out by one-way ANOVA (analysis of variance) followed by Bonferroni's posttest using Stata software (version 7.0, StatSoft, Tulsa, OK, USA). Statistical significance was defined as P<0.05.
Results
Characterization of Nanoparticles
The mean diameter of Lf-modified NPs was 211±15.3 nm, whereas the zeta potential value was 25.17±2.12 mV. The result of transmission electron microscopy showed that Lf-modified NPs had a well-formed spherical shape and compacted structure. The size was ∼200 nm for the control NPs without treatment of the Lf antibody (Figure 1A) and 250 nm for Lf-modified NPs treated with the Lf antibody (Figure 1B). The size of Lf-modified NPs increased ∼50 nm after immuno-gold staining. It might be attributed to the conjugation of the Lf antibody and streptavidin on the surface of Lf-modified NPs. The increase in size influenced by the conjugated antibody and streptavidin is in good agreement with recent published work (Bhattacharya et al, 2008). The colloidal gold appeared as a black dot under the transmission electron microscope. Black dots were observed around Lf-modified NPs treated with the Lf antibody, indicating the presence of Lf on the surface of NPs (Figure 1B). In contrast, no black dots were observed in the control NPs without treatment of the Lf antibody (Figure 1A).
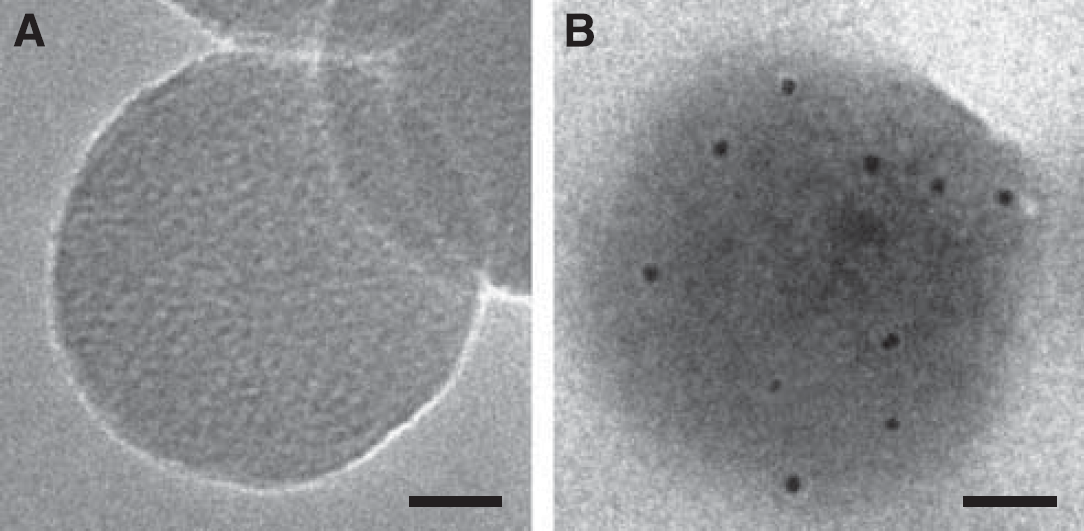
Transmission electron micrographs of Lf-modified NPs without any treatment (
Cellular Uptake at Different Temperatures
The results of cellular uptake at different temperatures are shown in Figure 2. The cellular uptake of Lf-modified vectors at 37°C was much higher than that at 4°C, and was significantly inhibited by excessive free Lf (Figures 2A–2C). Similar results were observed using a confocal microscope. The vectors were weakly internalized at 4°C and predominantly associated with the plasma membranes (Figures 2D, 2G, and 2J). On the same incubation at 37°C, the vectors were internalized into intracellular structures and the amount of cellular internalization increased significantly (Figures 2E, 2H, and 2K). When coincubated with excessive Lf, the cellular uptake of the vectors and intracellular fluorescence were decreased markedly (Figures 2F, 2I, and 2L).
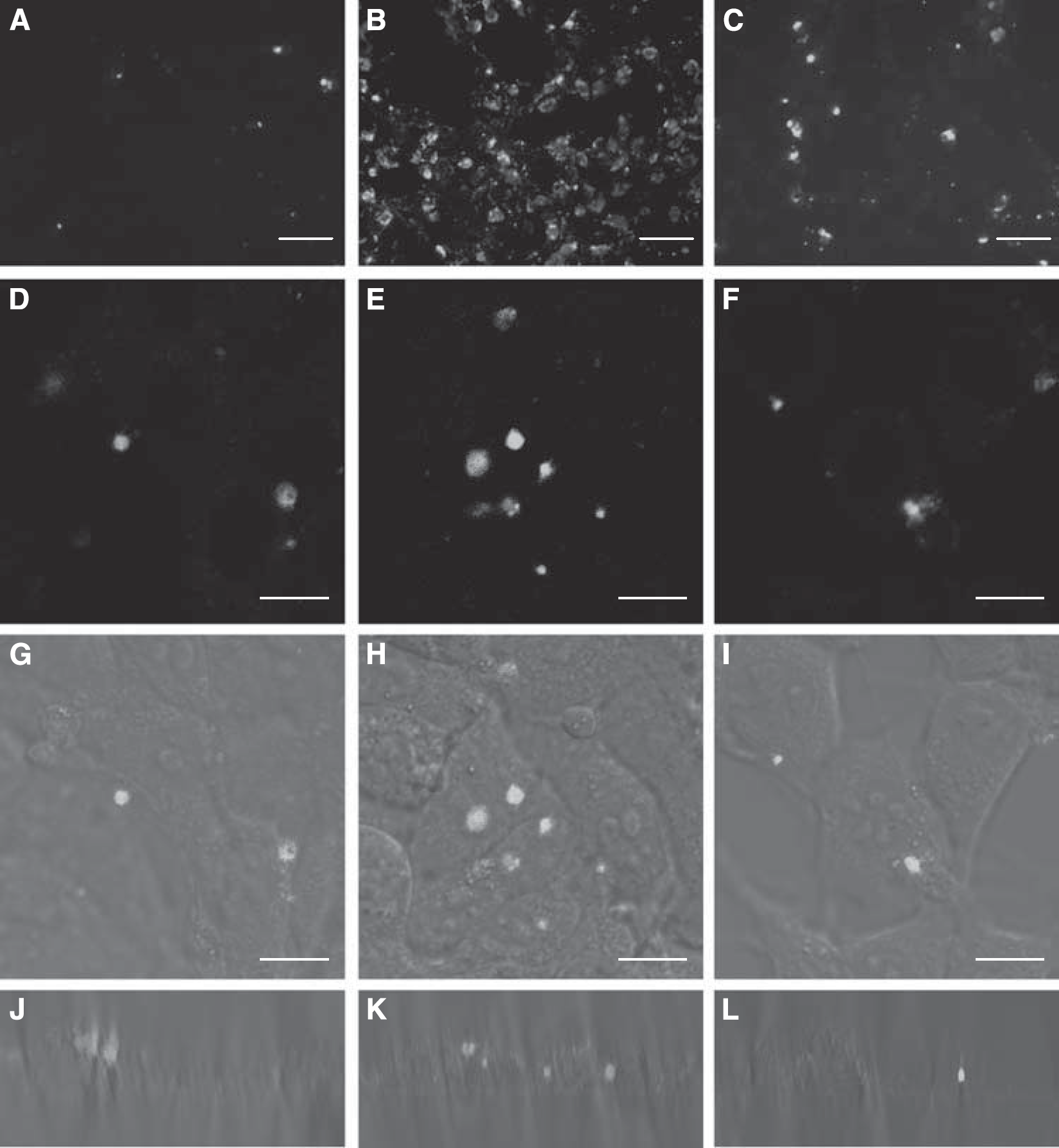
Cellular uptake of BODIPY-labeled Lf-modified vectors. BCECs were incubated with Lf-modified vectors for 30 mins. (
Cellular Uptake with Inhibitors
The impacts of different inhibitors are shown in Figures 3 and 4. The results of fluorescent microscopy showed that the cellular uptake of PAMAM was inhibited by all the used inhibitors, except PhAsO (the inhibitor of clathrin-dependent endocytosis) (Figures 3A–3F). Similarly, all the inhibitors except macropinocytosis-inhibited colchicine could reduce the cellular uptake of Lf (Figures 3G–3L). Furthermore, the cellular uptake of Lf-modified vectors was inhibited by all the used inhibitors (Figures 3M–3R). The result of quantitative analysis was consistent with that of fluorescent microscopy. The RUE of filipin, colchicine, excessive Lf, and cationic polylysine on the cellular uptake of PAMAM was 41.67, 51.73, 70.22, and 32.61%, respectively (Figure 3S). The RUE on the cellular uptake of Lf was 42.71, 54.52, 36.85, and 73.53%, in the presence of PhAsO, filipin, excessive Lf, and polylysine (Figure 3S), respectively. Moreover, the RUE of PhAsO, filipin, colchicine, excessive Lf, and polylysine on the cellular uptake of Lf-modified vectors was 34.14, 32.85, 57.37, 40.76, and 68.36%, respectively (Figure 3S).
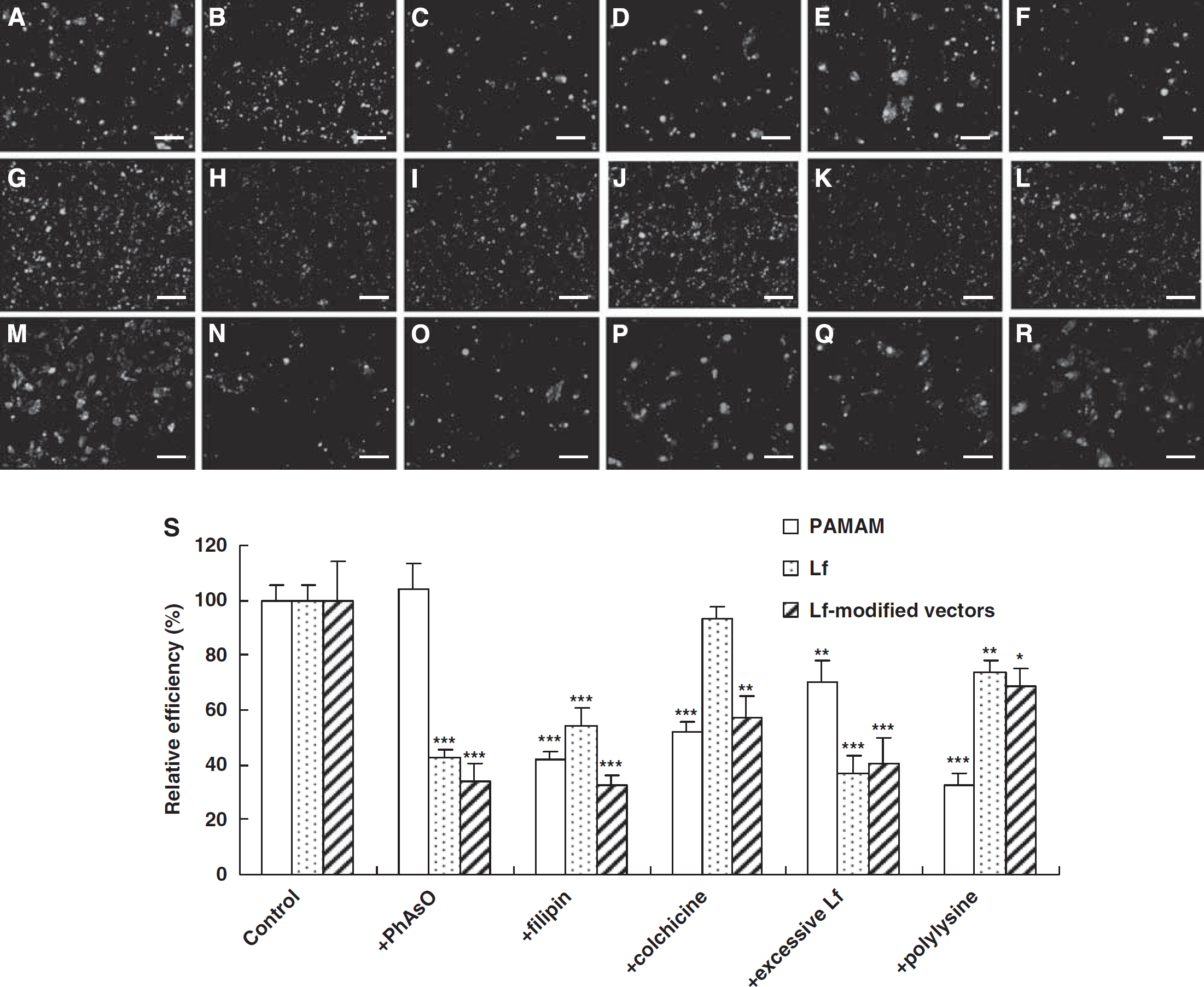
Qualitative and quantitative evaluation of the cellular uptake of different vectors is shown. The cellular uptake of BODIPY-labeled PAMAM (
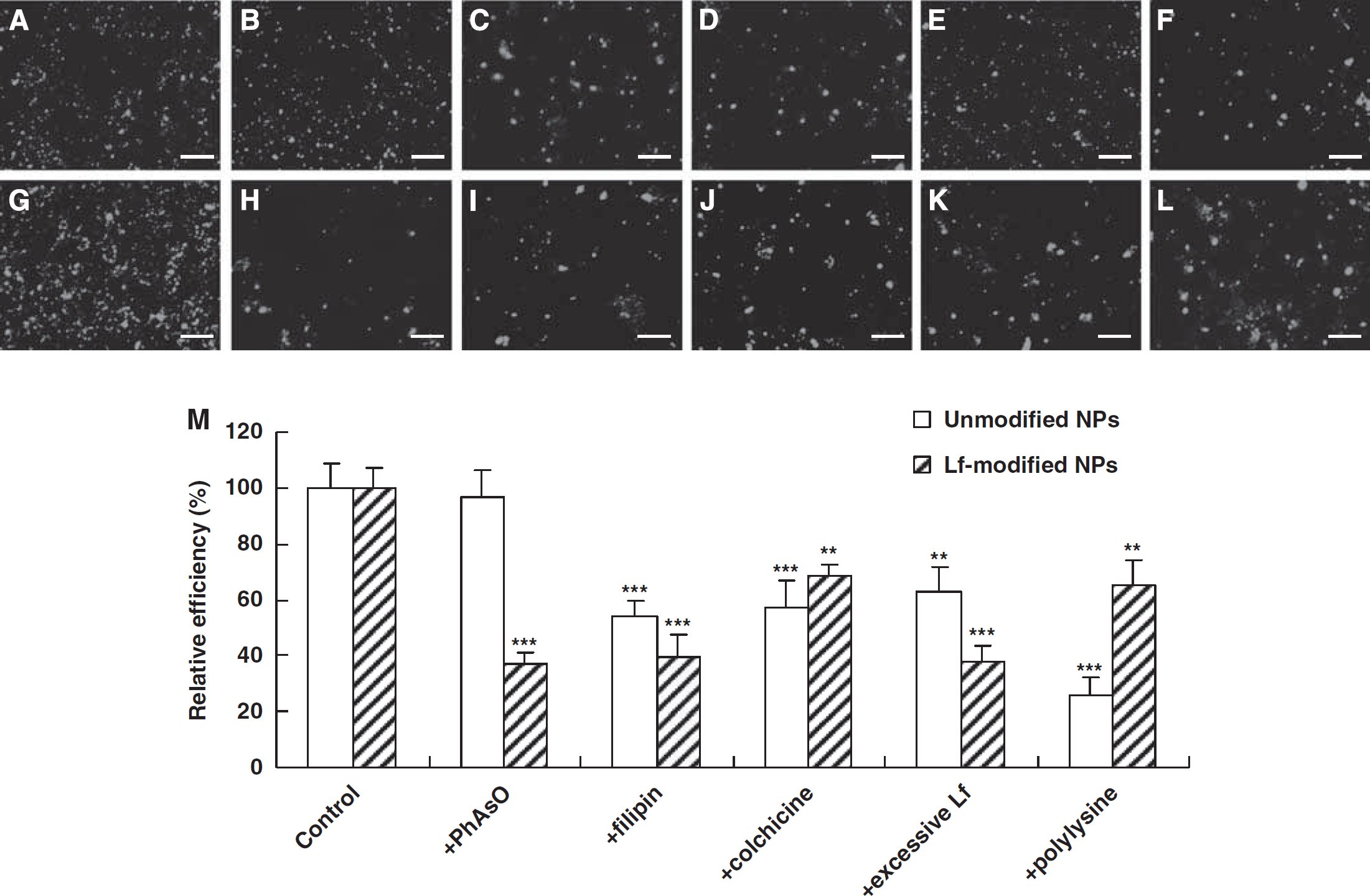
Qualitative and quantitative evaluation of the cellular uptake of unmodified and Lf-modified NPs encapsulating EMA-labeled DNA is shown. The cellular uptake of unmodified NPs (
Similar results were observed using different NPs encapsulating EMA-labeled DNA. As shown in Figure 4, the cellular uptake of Lf-modified NPs was inhibited by all the used inhibitors (Figures 4G–4L), whereas the cellular uptake of unmodified NPs was not inhibited by PhAsO (Figures 4A–4F). In addition, the RUE of filipin, colchicine, excessive Lf, and cationic polylysine on the cellular uptake of unmodified NPs was 53.56, 57.45, 63.12, and 25.98%, respectively (Figure 4M). The RUE of PhAsO, filipin, colchicine, excessive Lf, and polylysine on the cellular uptake of Lf-modified NPs was 36.89, 39.35, 68.45, 38.23, and 65.48%, respectively (Figure 4M). The extent of cationic inhibition of NPs was a little higher than that of corresponding vectors. This might be attributed to the decreasing charges of NPs when encapsulating to the anionic DNA.
Confocal Microscopy Studies
The plasmid DNA was labeled with EMA to construct fluorescent NPs for localization in BCECs. The acid compartments including endosomes and lysosomes were stained green with LysoTracker Green. As shown in Figures 5A–5C, almost all of the red fluorescence colocalized with the green fluorescence, suggesting that most of the Lf-modified NPs entered the acid compartments within 5 mins. After 30 mins of incubation, some Lf-modified NPs escaped from the acid compartments, whereas some remained in these compartments (Figures 5D–5F).
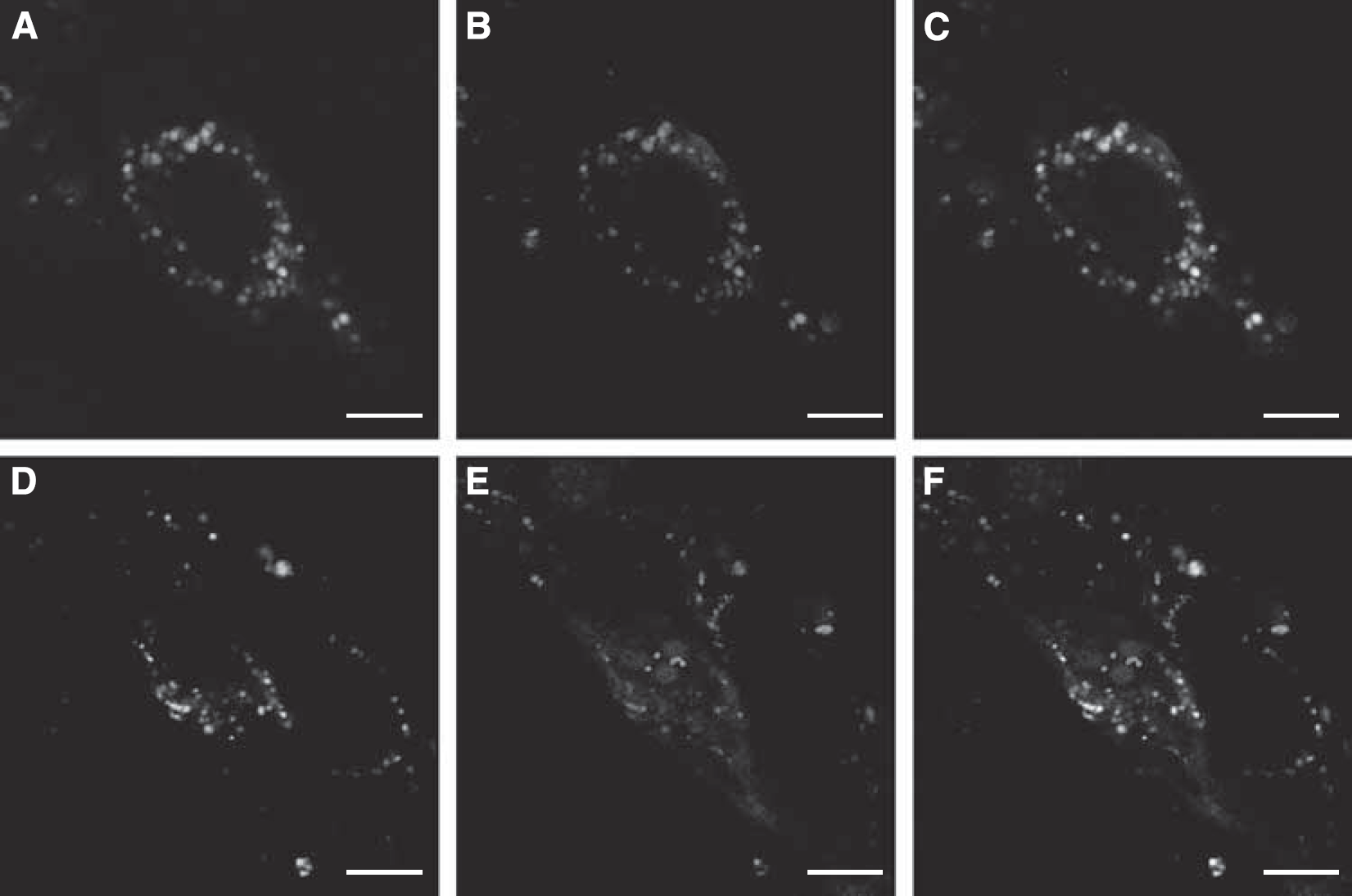
Confocal images of BCECs incubated with Lf-modified NPs for 5 mins (
Transport Studies Across BCECs Monolayers
The results of transport studies of vectors are shown in Figure 6A. Papp of PAMAM across BCEC monolayers reached a platform after 10 mins. Papp of Lf-modified vectors was significantly higher than that of PAMAM at each time point and reduced markedly in the presence of excessive free Lf. Furthermore, Papp of unmodified vectors was not apparently influenced by excessive Lf. Similarly, the results of transport studies of NPs are shown in Figure 6B. Papp of Lf-modified NPs was significantly higher than that of unmodified NPs at each time point and reduced markedly in the presence of excessive free Lf. Papp of both Lf-modified NPs and unmodified NPs increased with incubation time. Furthermore, Papp of unmodified NPs was not apparently influenced by excessive Lf. TEER and permeability of 14C-sucrose of BCEC monolayers showed no significant difference from that of controls (data not shown), indicating the nontoxic effects of different vectors on BCECs in the used dosage and verifying the integrity of BCEC monolayers during the experiments.
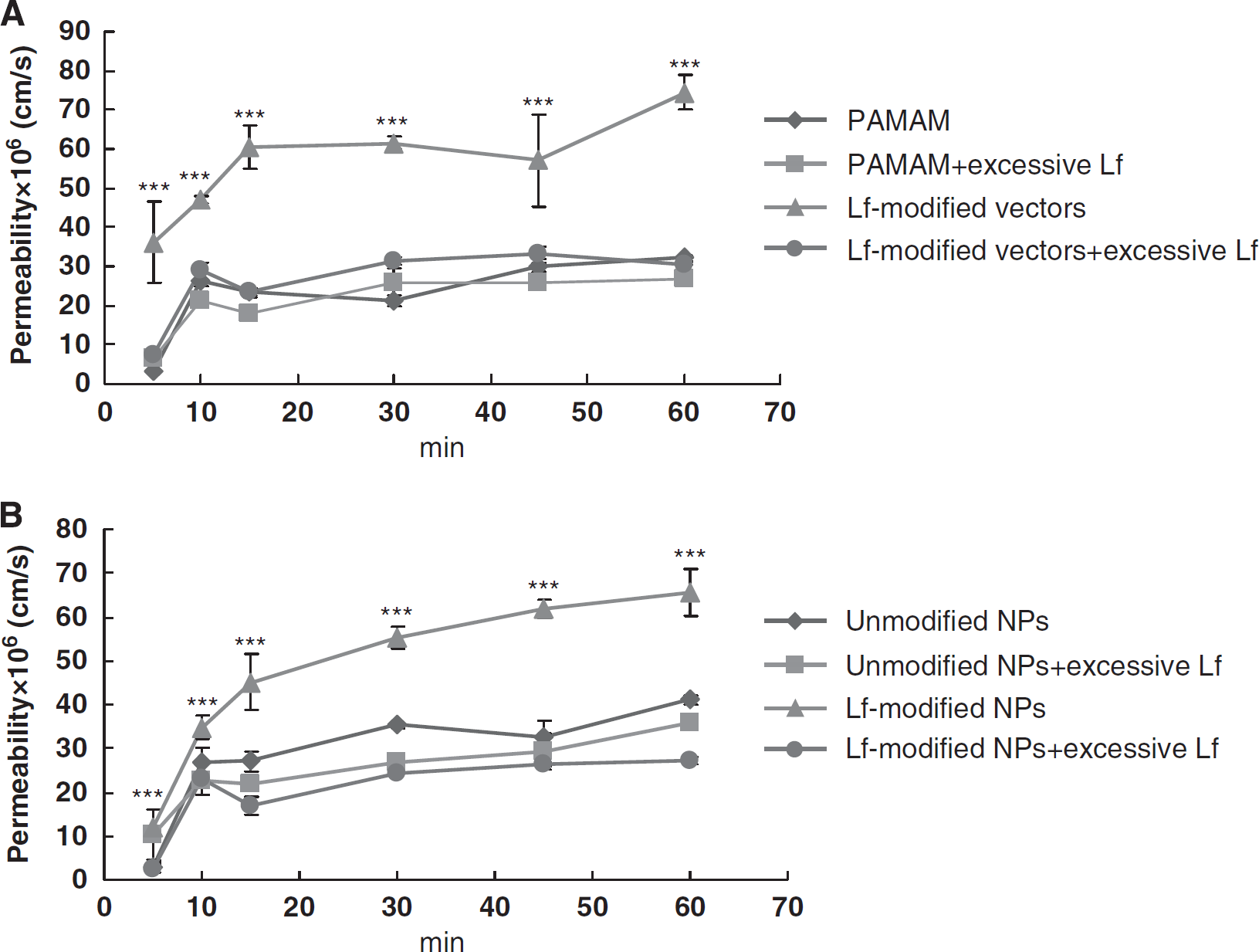
The permeability of 125I-labeled vectors (
Discussion
Nano-scaled brain-targeting drug delivery systems have been used for small molecules, proteins, and genes (Huang et al, 2007b, 2008; Juillerat-Jeanneret, 2008; Pardridge, 2007; Visser et al, 2005). Efficiently designing these systems and regulating their fate in vivo are supported by the elucidation of their exact targeting mechanisms. Ligand-mediated brain-targeting drug delivery to the brain is one of the focuses at present (Juillerat-Jeanneret, 2008; Pardridge, 2007). In our previous studies, Lf was exploited as a novel brain-targeting ligand and successful gene delivery targeting to the brain was achieved (Huang et al, 2008). In this paper, the brain-targeting mechanisms of Lf-modified vectors and NPs were systematically investigated for the first time.
Lactoferrin-modified vectors designed in this study mainly consist of the ligand, Lf, and the dendritic PAMAM. Several lines of evidence have indicated the presence of specific Lf receptors in the brain (Suzuki and Lonnerdal, 2004; Talukder et al, 2003). Lactoferrin is reported to be weakly cationic (Chasteen and Woodworth, 1990) and target Lf receptors in both the BBB and the neural cells (Huang et al, 2007a; Ji et al, 2006). In addition, PAMAM is cationic itself with primary amino groups on the surface (Kang et al, 2005; Luo et al, 2002; Patil et al, 2008). Thus, the synthesized Lf-modified vectors were also cationic. The cellular uptake of Lf-modified vectors was energy dependent and inhibited by excessive Lf (Figure 2). Furthermore, the uptake of Lf-modified vectors and NPs by BCECs was related to the endocytic processes including clathrin-dependent endocytosis, caveolae-mediated endocytosis, and macropinocytosis, with regard to the Lf and cationic properties of vectors (Figures 3 and 4). These results suggested that both receptor- and adsorptive-mediated mechanisms might contribute to the cellular uptake of Lf-modified vectors and NPs. In fact, the receptor- or adsorptive-mediated mechanism has been applied for designing brain-targeting drug delivery systems(Huang et al, 2008; Jones and Shusta, 2007; Lu et al, 2006). The results in this paper indicated that enhanced brain-targeting delivery could be achieved through the synergistic effects of the macromolecular polymers and the ligand. In addition, Lf was reported to transport across the BBB through a unidirectional receptor-mediated process (Fillebeen et al, 1999; Huang et al, 2007a). Excessive Lf could significantly inhibit the transcytosis of both Lf-modified vectors and NPs across the BCEC monolayers (Figure 6), suggesting that the Lf receptor-mediated mechanism has an important role in the transcytosis process of Lf-modified delivery systems across the BBB.
Lf-modified NPs prepared from the complexation of cationic Lf-modified vectors and anionic genes were still cationic, with a final positive zeta potential. In our previous studies, the modification of Lf could significantly enhance the brain-targeting efficiency of Lf-modified vectors and expression products of Lf-modified NPs, ∼4.6- and 4.9-fold, respectively, compared with that of unmodified counterparts (Huang et al, 2007b, 2008). In this study, intracellular localization and BBB-crossing mechanisms of Lf-modified NPs were investigated. The intracellular trafficking results showed that Lf-modified NPs could rapidly enter the acidic endolysosomal compartments within 5 mins and then partly escape after 30 mins of incubation (Figure 5), suggesting that the endocytic process should be the main pathway for the uptake of Lf-modified NPs. Moreover, some Lf-modified NPs remained in the acid compartments, showing the possibility to transport across the BBB. This was shown in vitro wherein Lf-modified NPs were observed to be transported across the BBB monolayer (Figure 6B), and in vivo wherein the exogenous gene expression using Lf-modified NPs was observed in both the BBB and the parenchyma (data not shown).
Otherwise, NPs with different sizes were reported to penetrate the BBB through different mechanisms, such as receptor-mediated transcytosis (Bharali et al, 2005; Jain, 2007; Liu et al, 2008; Reimold et al, 2008). The result of dynamic light scattering showed that the size of Lf-modified NPs was ∼200 nm, in harmony with that observed using the transmission electron microscope (Figure 1).
Conclusions
This is the first study to systematically investigate the uptake and transcytosis mechanisms of Lf-modified vectors and NPs in vitro. Collectively, the results showed that Lf-modified NPs could efficiently cross the BBB. Furthermore, both receptor- and adsorptive-mediated mechanisms contribute to the cellular uptake of Lf-modified vectors and NPs. Lactoferrin could be used as a brain-targeting ligand to construct drug delivery systems efficiently targeting the brain and crossing the BBB. Enhanced brain-targeting delivery could be achieved through the synergistic effect of the macromolecular polymers and the ligand.
Footnotes
Acknowledgements
The authors thank Professor Jianhua Zhu (School of Pharmacy, Fudan University) for his kind technical assistance in radiolabeling-related experiments.
The authors declare no conflict of interest.