Abstract
Neuregulin-1 (NRG1) signaling has multiple functions in neurons and glia. The data in this study show that NRG1 may also possess significant signaling and cytoprotective properties in human brain microvascular endothelial cells (BMECs). Neuregulin-1 mRNA and protein expression are present in these cells, and NRG1 receptors erbB2 and erbB3 are phosphorylated in response to NRG1. Neuregulin-1 triggers clear biologic responses in BMECs—elevated phospho-Akt levels, increased ring formation in a Matrigel assay, and decreased cell death after oxidative injury with H2O2. These data suggest that NRG1 signaling is functional and cytoprotective in BMECs.
Introduction
Neuregulin-1 (NRG1) isoforms are present in various tissues types, and have been described as oncogenes, glial growth factors, and a promoter of Schwann cell migration (Falls, 2003). In the nervous system, NRG1 and its erbB receptors perform multiple functions.
Neuregulin-1 was found to be neuroprotective in animal models of stroke (Xu et al, 2006). Although NRG1's actions on neurons and glia during ischemic brain injury has been studied (Xu et al, 2006), its role in brain endothelial cell function has not been characterized. It is likely that successful neuroprotective strategies must target the entire neurovascular unit consisting of the neuron, its associated glia, endothelium, and matrix (Iadecola, 2004; Lok et al, 2007; Park et al, 2003). Because of the importance of the endothelium in neurovascular signaling, we investigated NRG1 function in human brain microvascular endothelial cells (BMECs). Neuregulin-1 mRNA, protein expression, and activity were detected in BMECs. Additionally, NRG1 decreased endothelial cell death in an in-vitro model of oxidative stress. Our data suggest that NRG1 signaling is functional and cytoprotective in BMECs.
Materials and methods
Cell Culture
A human brain microvascular endothelial cell line was cultured in RPMI 1640 and characterized for brain endothelial phenotypes as previously described (Callahan et al, 2004).
Treatment with Neuregulin-1 for Signaling Studies
Recombinant human NRG1-β1 (carrier-free, R&D Systems, Minneapolis, MN, USA) was reconstituted in PBS and used for all experiments. Brain microvascular endothelial cells were grown on plates coated with 10 μg/mL Human Fibronectin (BD Biosciences, San Diego, CA, USA). When 80% confluent the next day, cells were serum-starved for 18 h. Vehicle or NRG1 ± PI3K inhibitor LY294002 was then added. At appropriate time points, cells were rinsed with cold phosphate-buffered saline and collected into lysis buffer. Cell lysates were centrifuged at 14,000 r.p.m. (10 mins, 4°C).
Evaluation of mRNA Expression
Total RNA extraction was accomplished using RNeasy mini kit (Qiagen Sciences, Germantown, MD, USA). The expressions of neurotrophins and their receptors were assessed by Oligo GEArray (OHS-031; SuperArray Bioscience Corporation, Frederick, MD, USA) and Oligo GEArray Trial Kit (SuperArray; GA-029), as suggested by the manufacturer. Image acquisition was performed using X-ray film and digital scanner.
Preparation of Brain Homogenates and Microvascular-Enriched Fraction
Brain samples were homogenized on ice in lysis buffer. After centrifugation, supernatant was collected. Microvascular-enriched fraction (MVE) was prepared using standard procedures (Galea and Estrada, 1991). Briefly, meninges, cerebellum, subcortical structures, and choroids plexus were removed and the brain placed in ice-cold phosphate-buffered saline. The volume of tissue and phosphate-buffered saline was measured and an equal volume of 26% dextran was added. This mixture was homogenized and centrifuged (13,500 g, 10 mins, 4°C). The supernatant was decanted and 3 mg of the pellet containing microvessels was homogenized in lysis buffer and centrifuged (14,000 r.p.m., 15 mins).
Immunoprecipitation/Western Blot Analysis
For western blot, equal amounts of protein were loaded per lane. For immunoprecipitation followed by western blot, the entire cell lysate from each plate was used and the procedure was performed as previously described (Sardi et al, 2006). Before gel electrophoresis, samples were heated in buffer with reducing conditions. After gel electrophoresis, proteins were transferred to polyvinylidene difluoride or nitrocellulose membranes, and treated with blocking buffer (Tris-buffered saline with 0.1% Tween 20, and 0.2% I-block (Tropix, Bedford, MA, USA) or 4% bovine serum albumin). Membranes were treated overnight (4°C) with primary antibody. After secondary antibody incubation, immune complexes were visualized by enhanced chemiluminescence. Primary antibodies: Neuregulin Ab-1 (7D5) from Lab Vision, Fremont, CA, USA; ErbB2 (Neu), phospho-erbB2 (Tyr1248), erbB3 (C-17), and erbB4 (C-18) from Santa Cruz Biotechnology, Santa Cruz, CA, USA, and pAkt (serine 473) from Cell Signaling Technology, Danvers, MA, USA. The mouse 4G10 anti-phospho-tyrosine antibody is a kind gift from Dr Gjoerup and Dr Roberts, Dana-Farber Cancer Institute, Boston, MA, USA. Dilutions: 1:1000 for NRG1, erbB2, erbB3, erbB4, and phospho-erbB2; and 1:5000 for 4G10 (anti-phospho-tyrosine).
Matrigel Tube Formation Assay for Angiogenesis
Brain microvascular endothelial cells were plated in 24-well plates in serum-free media in wells coated with Growth Factor-Reduced Matrigel Matrix (BD Biosciences). After 18h, photographs of five locations were taken (× 4 magnification). The numbers of complete rings in these fields were counted.
Induction of Oxidative Stress with H2O2
Cells were plated in wells coated with 10 μg/mL of human fibronectin factor (BD Biosciences). When 80% confluent, cells were placed in serum-free media with/without 100 μmol/L H2O2 for 18 h. Vehicle, NRG1, or NRG1 + AG825 (an inhibitor of the erbB2 tyrosine kinase activity) were added (AG825 5 to 10 mins before NRG1, and NRG1 5 to 10 mins before H2O2). After 18 h, cell viability was evaluated using MTT assay.
Assessment of Cytoxicity
Cell survival was measured by MTT assay, in which mitochondrial function was assessed by reduction of 3-(4.5-dimethylthiazol-2-yl)2,5-diphenyl-tetrazolium bromide (MTT; Sigma, St Louis, MO, USA) to a formazan product. After 18 h incubation with H2O2 or vehicle, cells were placed in media with 0.4% MTT. After 60 mins at 37°C, media was removed and cells were dissolved in dimethyl sulfoxide. Formazan formation was measured by reading absorbance at 570 nm with a reference setting of 630 nm on a microplate reader (FL600, Bio-Tek Instruments Inc.). Percent cell death was quantified as (100% – % cell survival).
Statistical Analysis
Matrigel ring formation data were analyzed using ANOVA and Tukey—Kramer tests. Statistical significance was set at P < 0.05. Data are expressed as mean + s.d. H2O2-induced cytotoxicity was analyzed as log-OD using a hierarchical linear mixed model with fixed cell means for all combinations of H2O2, NRG1, and AG825 concentrations and random intercepts among plates, among rows and columns within plate, and among replicate OD measurements for each well within each experiment. This is equivalent to a repeated measures strip-plot ANOVA with replicate OD measurements. Effects of experimental conditions relative to controls were estimated by linear contrasts. Point and interval estimates were back transformed to yield estimates of percent OD of each treatment relative to control OD. Percent cell death was calculated as 1% survival assuming a linear relationship between OD and density of live cells. Reported results are for two-tailed tests. Analyses were performed using SAS (version 9.1.3; SAS Institute, Cary, NC, USA). Statistical significance was set at P < 0.05. Data are expressed as mean ± confidence interval.
Results
Neuregulin-1 Signaling is Present in Human Brain Microvascular Endothelial Cells
Using a DNA microarray for neurotrophins/receptors, NRG1 mRNA was detected in BMECs, along with BDNF, NGF, FGF, and NT-5 (Figure 1A). Neuregulin-1 protein expression was found with multiple NRG1-immunoreactive bands corresponding to previously reported isoforms (Cote et al, 2005; Francis et al, 1999; Lebrasseur et al, 2003). Brain microvascular endothelial cells, MVE, and rat whole-brain homogenates all express a similar pattern of NRG-1 immunoreactive bands (Figure 1B). Although MVE contains other cell types, which express NRG1, enriching for endothelial cells did not decrease NRG1 immunoreactivity, further suggesting that BMECs express NRG1.
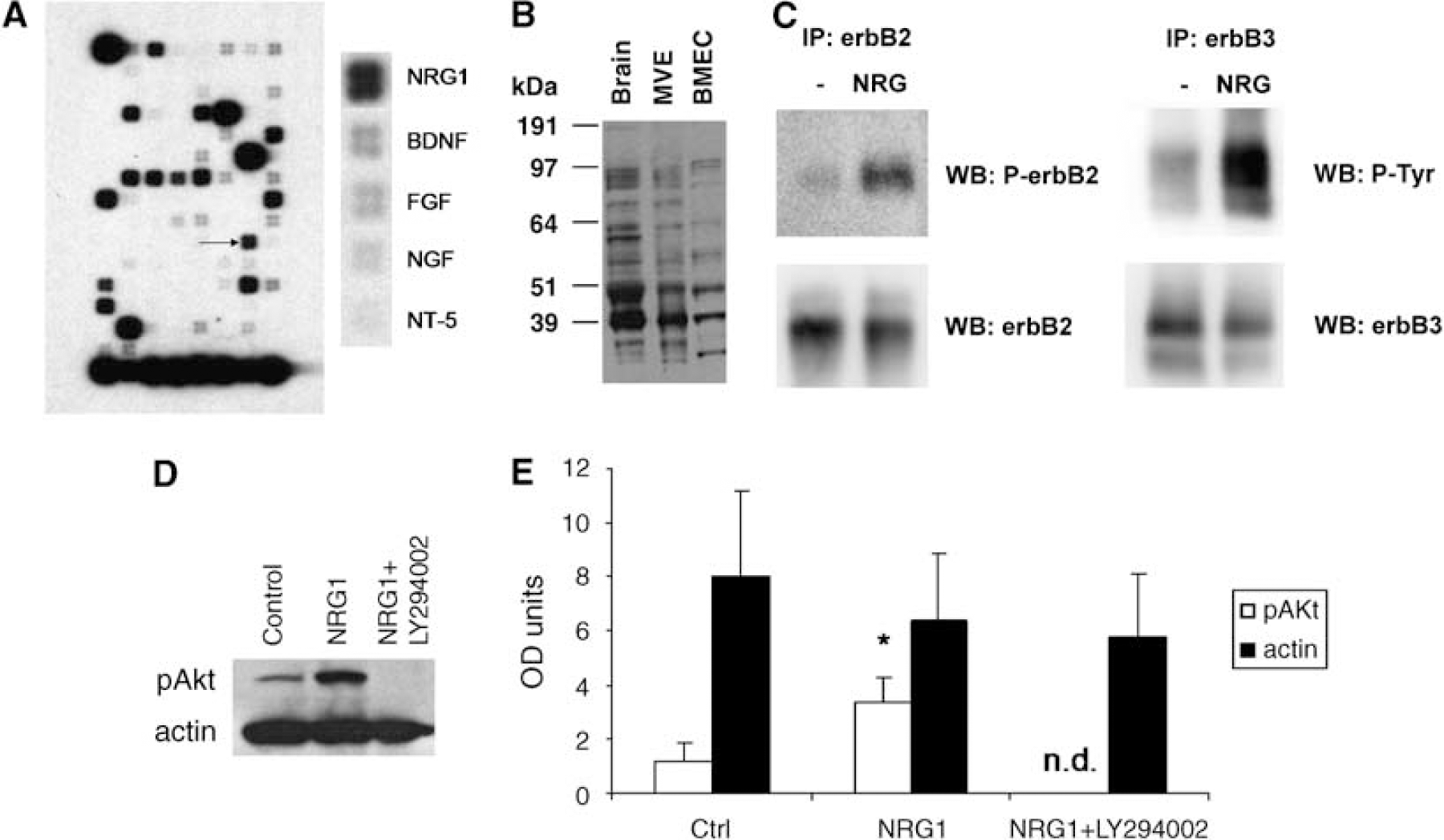
(
Immunoprecipitation/western blot showed the presence of erbB2 and erbB3 receptors that became phosphorylated after NRG1 incubation (Figure 1C). ErbB4 was not detected (data not shown). Neuregulin-1 increased Akt phosphorylation, which was abolished when the PI3-kinase inhibitor LY294002 (50 μmol/L) was added (Figure 1D and 1E).
Neuregulin-1 is Proangiogenic and Cytoprotective in Brain Microvascular Endothelial Cells
Neuregulin-1 increased ring formation in a Matrigel assay for angiogenesis (Figures 2A and 2B). At baseline there was an average of 300 rings per well. In NRG1-treated wells, there was an increase of 30 to 40 rings per well>(P < 0.05).
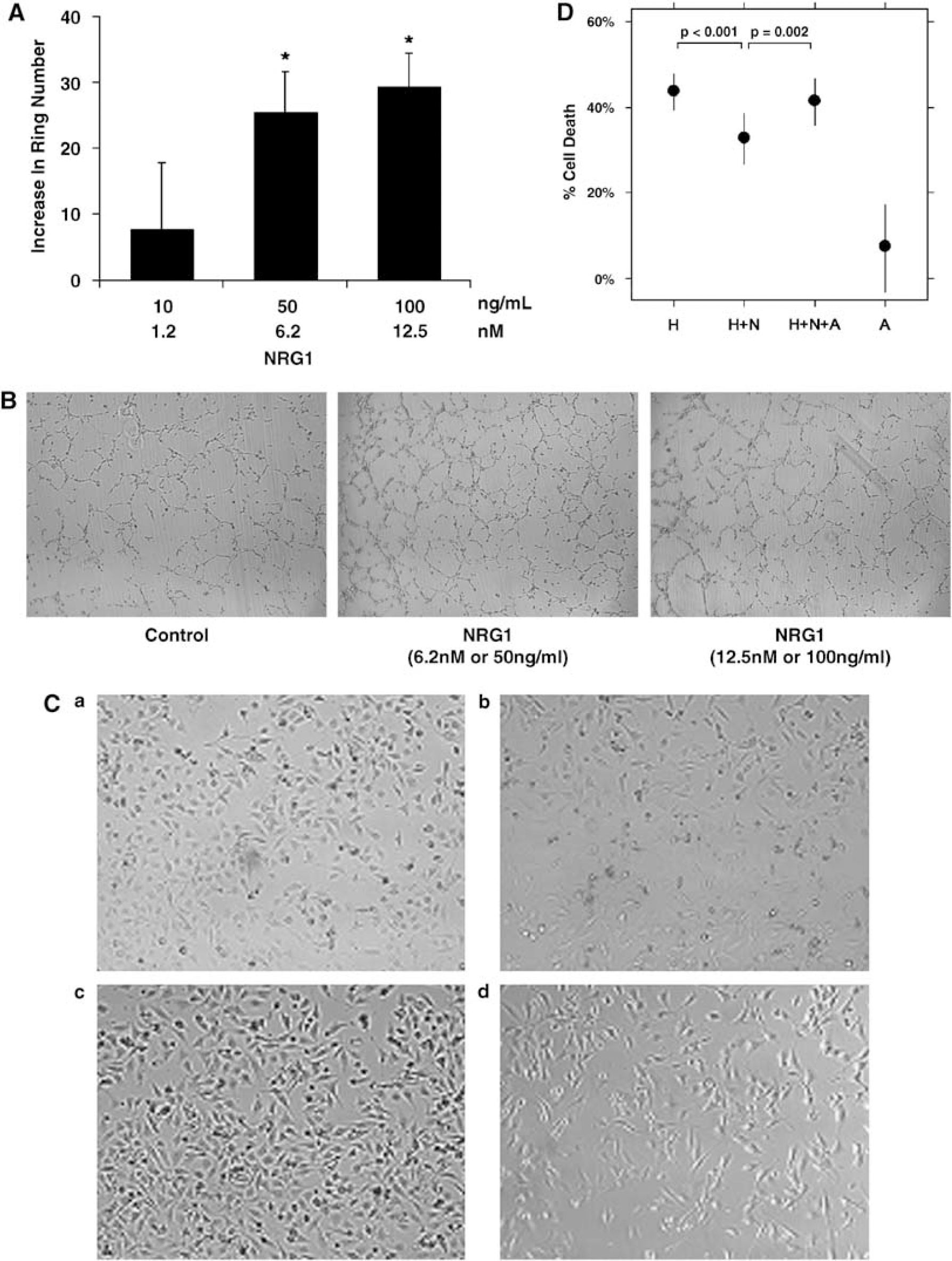
(
Oxidative stress was induced using H2O2, and cell viability assessed with MTT assay (Figures 2C and 2D). Eighteen hours exposure to 100 μmol/L H2O2 resulted in 44% cell death compared with untreated cells (P < 0.001). Addition of NRG1 reduced cell death to 33% (P < 0.001). Further addition of the erbB2 tyrosine kinase inhibitor AG825 negated the cytoprotective effect of NRG1 and cell death increased back to 42% (P = 0.002). AG825 alone did not affect cell survival.
Discussion
Our data show that NRG1 and its erbB2 and erbB3 receptors may mediate important biologic functions in BMECs. Neuregulin-1 increases pAkt, which may enhance survival of endothelial cells. Alternatively, NRG1 may protect BMECs by upregulating other trophic factors. These pro-angiogenic and cytoprotective actions may also contribute to neuronal survival.
The presence of erbB receptors in BMECs parallels the finding of these receptors in other types of endothelial cells (Russell et al, 1999). Although erbB2 lacks ligand-binding activity and erbB3 lacks efficient kinase activity, together the erbB2-erbB3 pair is the most active erbB heterodimer in terms of cell growth and differentiation. Downstream pathways include the Ras-Erk pathway for proliferation, the PI3 kinase pathway for survival, as well as mitogen-activated protein kinase, phospholipase-Cγ, protein kinase C, and the Janus kinase (Jak-STAT; Citri et al, 2003). The ability to activate multiple pathways suggests that NRG1 signaling may be active in endothelial homeostasis.
There are several caveats in this study. First, our cell-culture data are obtained in a cell line rather than primary cells. This cell line was previously shown (Callahan et al, 2004) to possess cerebral endothelial phenotypes. Furthermore, the presence of NRG1 in MVE of rat brain supports the cell-culture data. Second, many NRG-1 immunoreactive bands are detected by western blotting. The finding of multiple isoforms is a well-known property of NRG1 (Falls, 2003). The reader is referred to a recent manuscript (Cote et al, 2005) for a study of the specificity of NRG1-immunoreactive bands in cardiac endothelial cells. Third, our biologic assays are limited to two endpoints, angiogenesis and cytoprotection. Perhaps NRG1 might have other effects on endothelium. Additionally, our cytoprotection is relatively modest. Larger effects were seen in PC-12 cells transfected with erbB4 receptors (Goldshmit et al, 2001), possibly because of the higher number of erbB4 receptors in these cells. Alternatively, the modest effects of exogenous NRG1 in our study could be because of high levels of endogenous NRG1, although the lack of significant baseline erbB and Akt phosphorylation argues against this hypothesis. It is possible that endogenous NRG1 is subjected to posttranslational processing resulting in isoforms that do not act within the endothelial cell. Lastly, although AG825 is relatively specific for erbB2 at the concentration (5 μmol/L) used, it may inhibit other types of tyrosine kinases. However, its IC-50 is 0.35 for erbB2, 19 for EGFR, 40 for PDGFR, and > 100 for InsR (Levitzki et al, 1995). So at 5 μmol/L, its specificity for erbB2 is 4 times more than that for EGFR, and 100 times more than that for PDGFR and for insulin receptor.
Neuregulin-1 is shown here to protect BMECs against oxidative stress. Further study is required to determine whether NRG1 is more broadly cytoprotective against other insults, and whether it mediates both acute neuroprotection and postinjury repair. It is likely that endothelial function and neuronal health are linked, and cytoprotection of the brain endothelium may increase neuronal survival during both processes.
In summary, our data suggest that the NRG1 signaling is active in BMEC biology, including the promotion of angiogenesis and protection against oxidative stress. Neuregulin-1's potential role in endothelial signaling merits further study, as neurovascular homeostasis is essential for salvaging brain function in stroke, brain trauma, and neurodegeneration.
Footnotes
Acknowledgements
The authors thank Dr Eric Macklin for statistical analysis of the cytotoxicity data, and Dr Klaus van Leyen, Dr Douglas Sawyer, Dr Xiaoying Wang, and Ms Rui-Ying Zhu for many helpful discussions.
None.
