Abstract
We hypothesized that magnetic resonance magnetization transfer (MT) imaging would be sensitive for detecting cerebral ischemic injury in white matter of neonatal brain. We compared the progression of changes in T2 and the MT ratio (MTR) after cerebral hypoxic-ischemic insults of differing severity in neonatal rats. Magnetization transfer imaging parameters were first optimized, and then MTR and T2 maps were acquired at various times after a mild (rather selective white matter) or substantial insult produced by unilateral cerebral hypoxia—ischemia. Depending on insult severity, time after insult, and region (e.g., subcortical white matter or cortex), cerebral hypoxia—ischemia produced reductions in MTR and an increase in T2. The exception was acutely at 1 to 5 h at which time points MTR was reduced ipsilaterally in white matter, whereas T2 was not affected significantly. Progression of imaging changes differed in rats grouped according to whether gross damage was present after chronic recovery. Behavioral changes were generally associated with chronic reductions in MTR and gross brain damage. Magnetization transfer imaging was capable of early detection of hypoxic-ischemic injury and particularly sensitive for identifying the progression of cerebral injury in white matter. Magnetization transfer ratio has potential for assisting with early diagnosis and treatment assessment for infants affected by perinatal hypoxia—ischemia.
Introduction
Standard proton magnetic resonance (MR) imaging detects primarily aspects of tissue water where contrast is obtained by using sequences sensitive to biophysical properties of water including its diffusibility, T1 or T2 relaxation within the tissue. Changes in contrast in standard MR images have assisted with diagnosis or detection of brain injury or disease such as stroke (Muir et al, 2006; Weber et al, 2005). However, these imaging methods are rather insensitive for detecting early injury or some of the irreversible cellular changes that can be detected histologically (Li et al, 2002; Meng et al, 2005). More sensitive may be an alternate MR imaging method—magnetization transfer (MT) imaging (Henkelman et al, 2001; Stanisz et al, 2005; Tofts, 2003). Magnetization transfer imaging sequences use an off-resonance radiofrequency pulse to saturate the restricted hydrogen pool, which in tissue consists predominantly of water that is bound to macromolecules and lipids. This results in an exchange of magnetization between the free and restricted hydrogen protons and produces a reduction in MR signal intensity depending on the rate and degree of this exchange. The most easily applied MT image for clinical use is the MT ratio (MTR), which is obtained from a ratio of the image or signal intensity acquired with and without the application of the off-resonance saturation pulse.
Changes in MTR in brain have been observed during normal development and with various disease processes (Tofts, 2003). For example, developmental increases of MTR in white matter are associated with myelination of axons (Engelbrecht et al, 1998; Wozniak and Lim, 2006; Xydis et al, 2006). Reductions in MTR in white matter occur in patients with multiple sclerosis within white matter lesions and regions of demyelination (Bagnato et al, 2006; Filippi and Agosta, 2007). Magnetization transfer imaging also detects tissue changes associated with stroke where, generally, reductions in MTR or increases in the MT transfer constant have been reported after cerebral ischemia or infarction and also in one study of periventricular leucomalacia (Ewing et al, 1999; Hanyu et al, 1998; Knight et al, 2005; Ordidge et al, 1991; Pui and Wang, 2005; Xydis et al, 2006). However, the results from these studies are limited, usually to measures in gray matter for a few times after insult, and little is known about the effects of cerebral ischemia on acute MT changes in white matter or its effect on injury severity or the progression of changes in immature brain over time.
We hypothesized that after cerebral hypoxia—ischemia in immature brain, MTR would vary depending on the severity of ischemic injury and be particularly sensitive for detecting white matter injury. Thus, we compared the progression of cerebral white and gray matter changes in T2 and MTR after an ischemic insult, where ischemic severity was varied in a neonatal rat model. In this model, the mild cerebral hypoxic—ischemic insult tends to produce relatively selective white matter injury and the more severe insult produces panne-crosis (Meng et al, 2006; Qiao et al, 2004). Magnetization transfer ratio changes were also compared with behavioral outcomes in the different group severities.
Materials and methods
Experiments were performed according to the guidelines provided by the Canadian Council on Animal Care and approved by the local and NRC and University of Calgary Animal Care Committees. Pregnant Wistar rats (Charles River Laboratories, Montreal, QC, Canada) arrived at least 1 week before delivery and were housed individually and fed ad libitum. Rats were used starting at 1 week of age.
Sequence Parameters and Magnetization Transfer Ratio Maps in Developing Brain
Initial experiments were performed to determine optimal MT sequence parameters for detecting gray and white matter contrast and standardize the imaging procedures for MTR imaging in rats at different stages of postnatal development. We examined the effect of varying the number of pulses, RF power, and frequency offset in eight rats. This was investigated in four different age groups: neonatal (1-week old), 2-week old, juvenile (5-week old), and adult.
Magnetization transfer imaging was performed in isoflurane anesthetized rats using a 9.4-T/21-cm MR imaging system with a Bruker Avance console and a 3.5-cm diameter quadrature rf coil. Respiration was monitored and rectal temperature was controlled (37.5°C) while the animals were in the magnet. A distilled water phantom was used in initial studies to determine the extent of direct water saturation effects. For the MT sequence, proton density-weighted spin echo images with MT saturation on and off were acquired either as single or multiple (five) slices. Slice thickness was 0.8 mm, matrix size was 128 times 128, TR was usually 3 secs (5 secs for 100 pulses), TE was 15 ms, and the field of view was 2.0, 2.5, and 3.0 cm2 for neonatal, juvenile, and adult imaging, respectively. Testing frequency offsets of 500, 100, 1500, 2500, and 3000 Hz showed moderate saturation and good gray—white contrast at 1500 Hz. This frequency, also similar to that used by others previously, was then used for studies examining the effect of saturation pulse power and pulse number on MTR in developing brain (Gareau et al, 2001; Lemaire et al, 2000). Magnetization transfer ratio maps were acquired with different numbers of saturation pulses (20, 40, 60, 80, and 100) with a constant RF power (B1 of 6 μT). The RF power of the saturation pulses (60) was also varied using B1 values of 2, 4, 6, 8, and 10 μT.
For analysis of the images, MTR was calculated as MTR=((Mo-Ms)/Mo) × 100%, where Ms and Mo were the signal intensities obtained with and without MT saturation, respectively. Mean regions of interest from ipsilateral and contralateral regions of posterior subcortical white matter and gray matter of the parietal cortex were measured from the MTR maps. Differences in responses between gray and white matter were compared using Student's t-test.
Magnetization Transfer Imaging after Mild or Substantial Cerebral Hypoxia—Ischemia
Rat pups (53) were randomized to one of the three groups at 1 week of age: a sham control group or mild and substantial cerebral hypoxia—ischemia groups. Cerebral hypoxia—ischemia was produced as described in detail previously (Meng et al, 2006; Qiao et al, 2004). Briefly, the right carotid artery was ligated and severed under isoflurane (2% to 2.5% in oxygen) anesthesia. Local anesthetic was applied and the incision site was sutured closed. In the sham control group, the right carotid artery was isolated but not ligated. In the substantial group, animals were exposed to hypoxia (8% oxygen, 92% nitrogen) for 70 mins with a chamber temperature of 35.5°C. In the mild group, pups were exposed to hypoxia for 40 to 45 mins, with chamber temperature maintained at 34.5°C using a heating lamp. A relatively selective white matter injury with some gray matter involvement was produced consistently under such conditions. This models well the predominant white matter injury observed in premature and near-term infants where there can often also be some gray matter involvement (Back, 2006; Kinney, 2006).
The T2 and MT imaging within a maximum of three sequential scans per animal was performed at approximately 1 or 5 h or 1 day, 2 days, 1 week, or 4 weeks after the hypoxic-ischemic insult in the animals anesthetized with isoflurane. The MT imaging sequence parameters for 1- to 2-week-old rats were TR (repetition time) = 5000 ms for 60 pulses of 6 μT at 1500 Hz frequency offset, TE (echo time) = 15 ms, FOV (field of view) = 2.0 cm2 128 times 128 matrix, 5 slices 0.8 mm thick with an interslice gap of 0.2 mm. Juvenile rats (5-week old) had similar sequence parameters except for an FOV of 2.5 cm2, the use of 40 pulses of 4 μT for saturation, and an interslice gap of 1.6 mm, to adjust for the larger and older brain. Magnetization transfer ratio maps were analyzed as described above. The T2-values were obtained by a monoexponential fit of image intensities acquired using a multi-echo spin-echo sequence with a TE = 10 ms between echos for a total of 32 echos (TR = 5000 ms). Images were analyzed using locally developed software (Marevisi, Winnipeg, MB, Canada). A posterior slice through the midbrain was selected for measuring MTR and T2-values within parietal cortex and white matter in the right (ischemic) and left (contralateral) hemispheres. Differences between left and right hemispheres in hypoxic-ischemic animals were compared using Student's paired two-tailed t-tests.
Behavioral Testing
To examine the relationship between MR imaging changes and functional deficits, subgroups of animals surviving for 4 weeks after hypoxia—ischemia were administered neurological tests. The following three tests were performed:
Neurological score: At 2, 3, and 4 weeks after hypoxia—ischemia, animals were assigned a neurological score according to their circling and body torsion when lifted briefly by the base of the tail for three repeated tests. The score was assessed as follows: 0—no preferential circling when suspended or walking; 1—twists to same side 2/3 times when suspended and a normal walk or circles when walking; 2—twists to same side 3/3 times with or without circling or twists to same side 2/3 times with circling; 3—twists 3/3 times to same side when suspended and walks in a tight circle.
Cylinder test of asymmetrical forelimb use: The effect of ischemia on spontaneous forelimb use during upright postural support movements was tested at 4 weeks after cerebral hypoxia—ischemia by placing the animals into a clear plexiglas cylinder (20 cm in diameter) situated on a glass tabletop and videotaped from below through an angled mirror. The number of forelimb wall contacts (single-limb contacts and bilateral contacts) used for postural support were counted, and the percentage of bias or asymmetry of single-limb wall contacts was calculated as ((contralateral—ipsilateral)/(contralateral + ipsilateral) × 100).
Morris Water Maze test: Cognitive or spatial learning ability was tested in 5-week-old rats at 4 weeks after hypoxia—ischemia using the single-day Morris Water Maze test (Kraemer et al, 1996). A clear platform 1 cm below the water surface was placed in a 4-ft diameter pool containing water made opaque with milk powder. The rat was released into the water with its head facing the pool wall, and it was allowed to swim until it found the hidden platform or 60 secs had elapsed. It was placed on the hidden platform for 10 secs if initially unsuccessful. The latency to locate the hidden platform was recorded. The rats received four block sessions (four trials per block). Each block was separated by at least 1 h and each trial by a minimum of 5 mins to allow animals sufficient time to rest and warm under a heating lamp between trials.
Histology
After the cerebral hypoxia—ischemia and the last MR scan (at 1, 2, or 4 weeks) or behavioral testing (at 4 weeks), animals were injected with pentobarbital (120 mg/kg). After perfusion fixation with formalin or decapitation, brains were removed and paraffin embedded or stored frozen, respectively. Brain sections through anterior and posterior cerebrum were taken onto slides and stained with hematoxylin and eosin to determine the degree of permanent injury graded by extent of cerebral atrophy or cyst formation and white matter changes.
Results
Effect of Number of Saturation Pulses and Saturation Power on MTR
As the number of saturation pulses increased the MTR measured in either gray or white matter increased (Figure 1A). The MTR tended to reach a steady maximal level at 70% to 80% and this occurred with fewer saturation pulses at the older ages. Furthermore, during postnatal development, there were regional differences in MTR particularly between white and gray matter. Quantitatively, MTR was greater in gray matter than white matter in neonates but this reversed by 5 weeks of age (Figure 1B). Such a reversal in MTR with development has been reported previously and corresponds well with postnatal myelination in rat brain and the ontogenic decreases in water content and increases in lipid content (Matsumae et al, 2003; Wozniak and Lim, 2006).
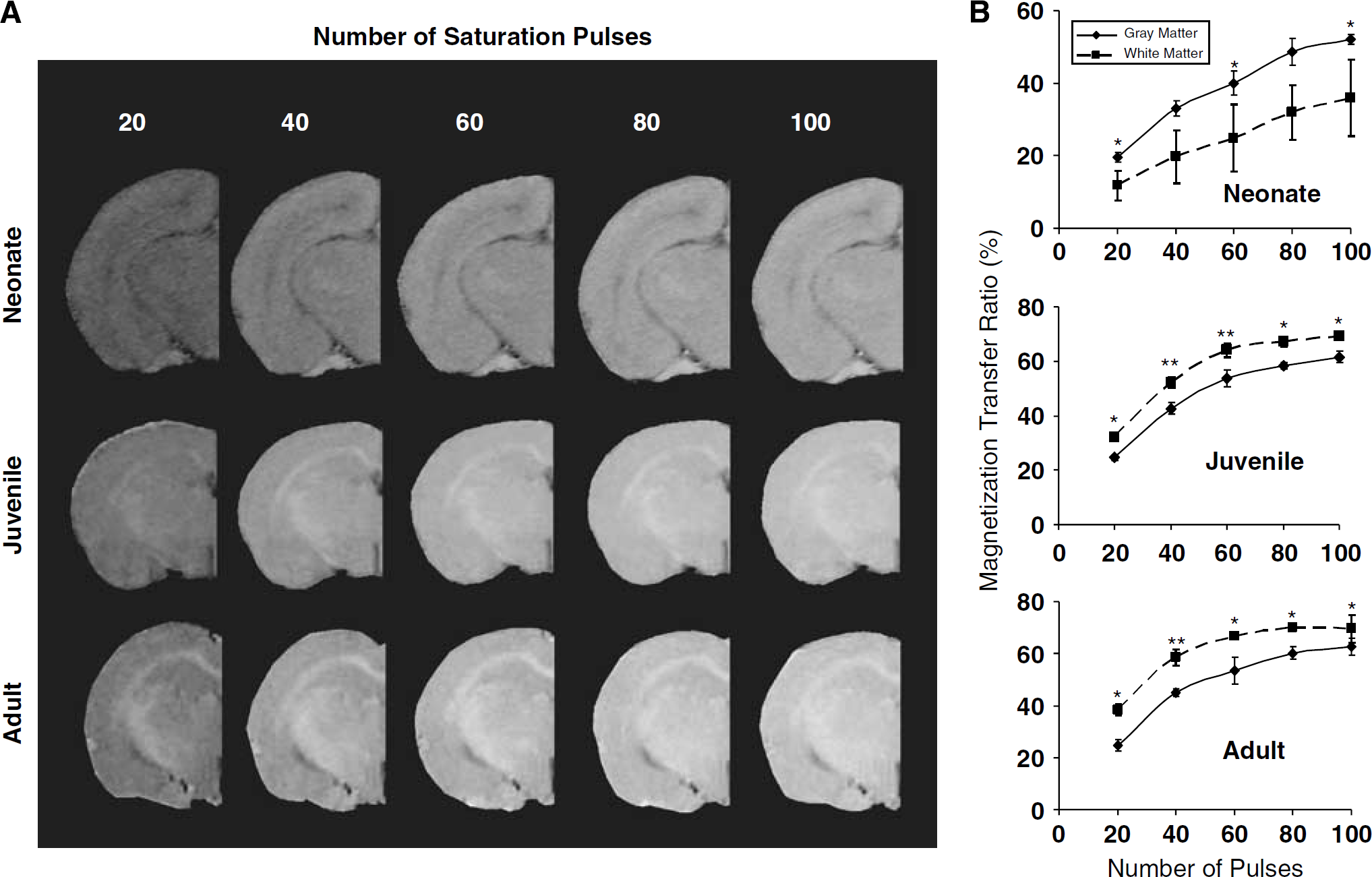
(
Similar to increasing the number of pulses, increasing the pulse power also increased the MTR (Figure 2). The MTR for gray matter exceeded that of white matter in neonates (Figure 2A). At 2 weeks of age, gray and white matter had similar MTR values, whereas in adults MTR of white matter exceeded that of gray (Figures 2B and 2C). Gray—white matter contrast in MTR maps for immature or neonatal brain tended to be highest at 6 to 8 μT and 60 to 80 pulses. Older brain tended to have best gray—white contrast at pulse powers of 4 to 6 μT.
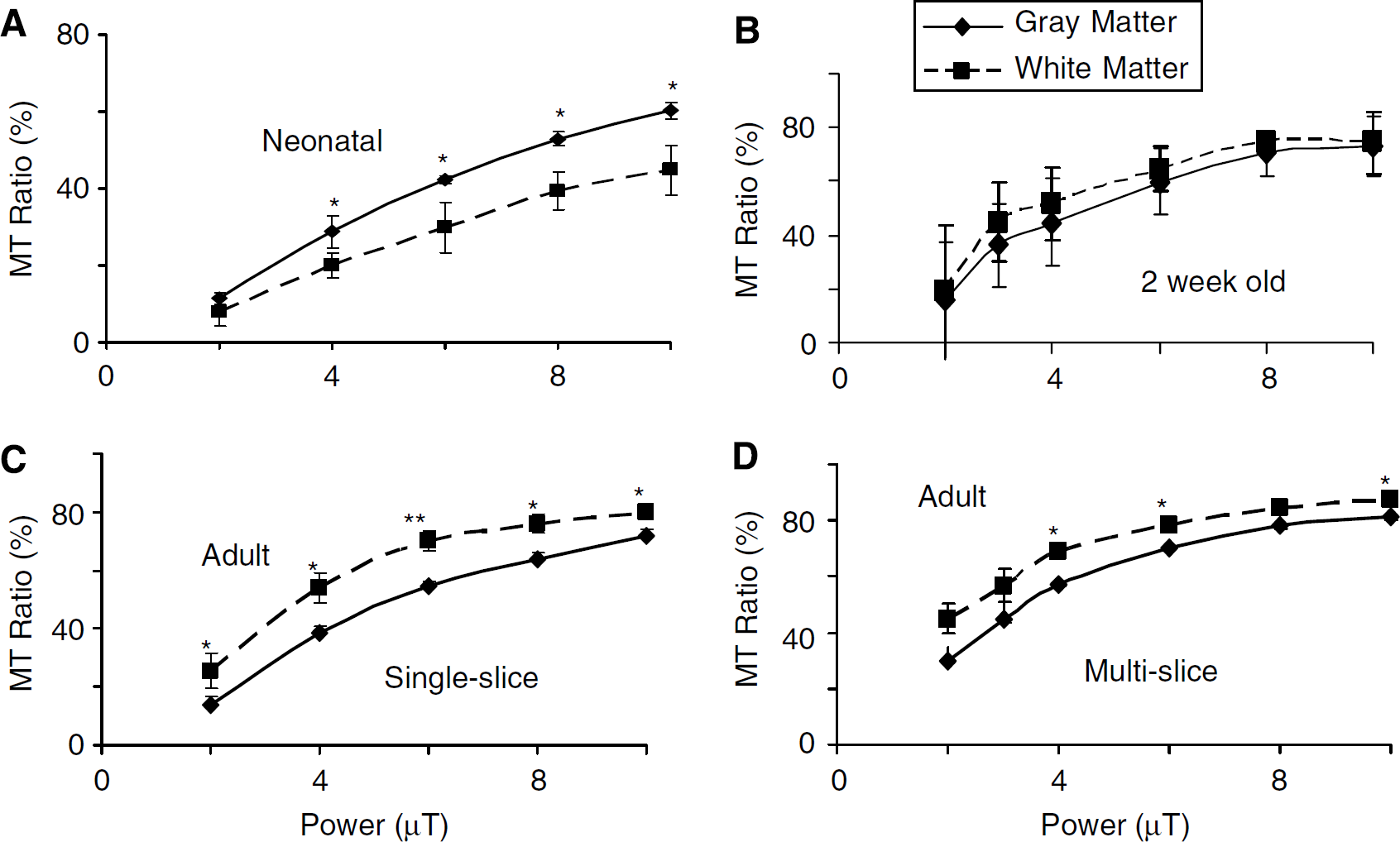
Effect on mean MT ratio in gray (parietal cortex) and subcortical white matter regions of changes in pulse power at different postnatal ages (
Gray—white contrast was also reduced in multi-slice MTR maps when compared with single-slice acquisitions (Figure 2D). Reduced power and number of saturation pulses improved contrast indicating that a lower RF power and fewer pulses are needed to approach an optimal MTR with multislice acquisitions, particularly in older brain. This resulted in a selection of 40 saturation pulses of 4 μT in our subsequent MTR studies in the more mature animals at 5 weeks of age.
Acute or Subacute Effects of Cerebral Hypoxia—Ischemia on MTR and T2
Cerebral hypoxia—ischemia produced variable changes in MTR or T2 ipsilateral to the hypoxia—ischemia depending on insult severity and time after insult of the scans. Acute and subacute changes up to 24 h after hypoxia—ischemia were followed in animals with a mild or substantial hypoxic—ischemic insult (Figures 3 and 4). MTR percent unit values in contralateral white matter were similar at acute and subacute time points after hypoxia—ischemia (e.g., 53.3 ± 3 at 1 h and 54.9 ± 4 at 24 h in mild group; 52.8 ± 5 at 1 h and 57.6 ± 5 at 24 h in the substantial group) and not significantly different from those at 48 h for the mild group. One hour or 5 h after a mild insult, there was a trend for a left-right increase in T2 (NS) in cortical gray matter and no change in the subcortical white matter. In contrast, there was a modest decrease of MTR in white matter at 1 h, which became more pronounced at 5 h after the mild insult. In histological sections assessed at 2 days after insult, cell death was evident in white matter in the majority of animals. By 24 h, the changes were significant for both T2 and MTR in white matter. After a substantial hypoxic—ischemic insult, relatively pronounced increases in T2 and decreases in MTR were observed throughout much of the hypoxic—ischemic hemisphere with quantitative changes being significant in gray matter at 1 h after hypoxia—ischemia and in both gray and white matter at 5 or 24 h after hypoxia—ischemia. At 2 to 7 days after MR imaging, 1, 5, or 24 h after a mild insult, damage was detected histologically in the ipsilateral white matter with evidence for some cortical injury in the majority of animals. After a substantial insult, damage was apparent in ipsilateral white and gray matter in all animals.
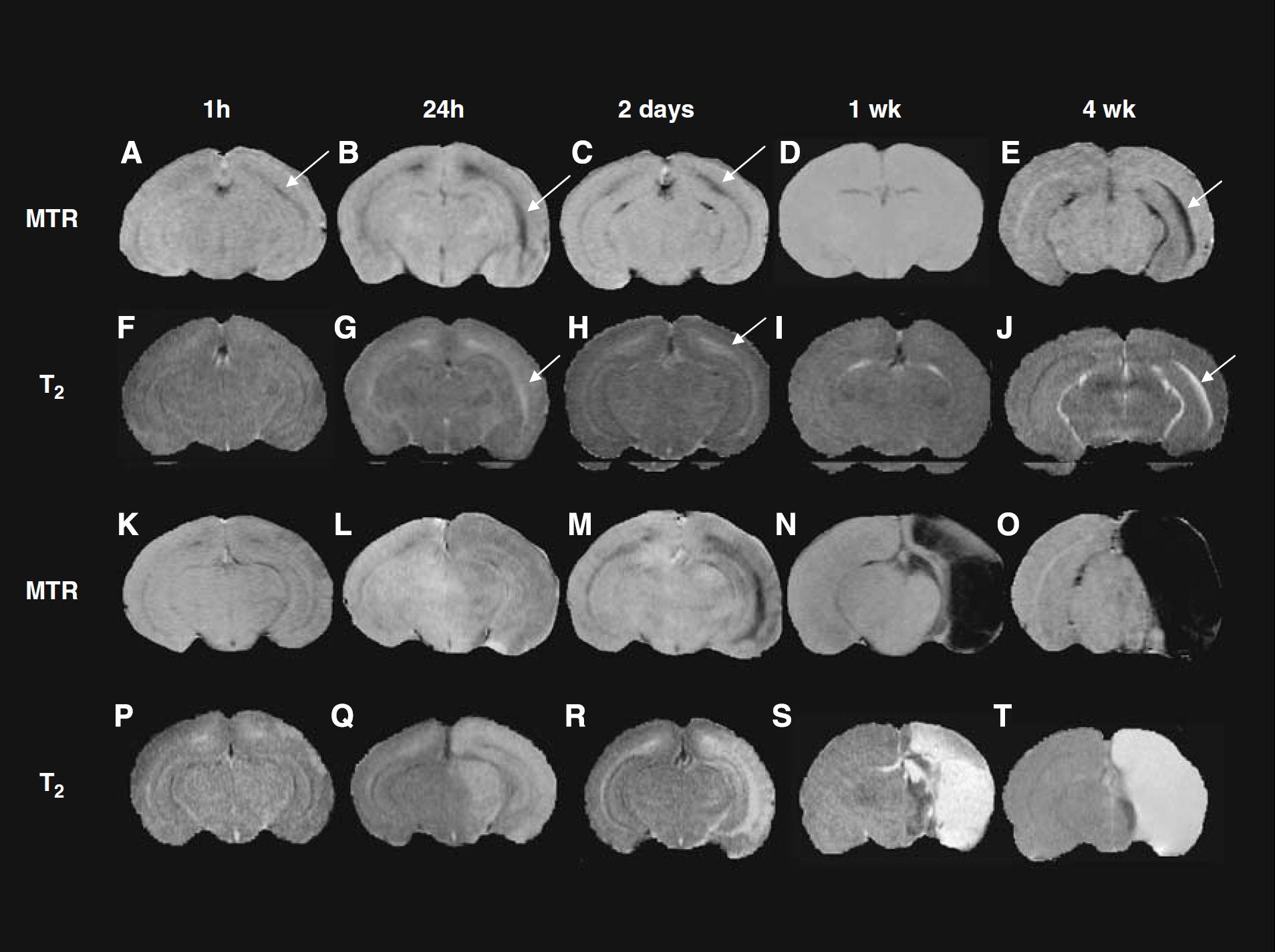
Representative MTR maps (

Early acute changes in MTRs (
Chronic Effects of Mild Cerebral Hypoxia—Ischemia
Changes in MTR and T2 continued to evolve at more prolonged times of 2 days, 1 week, and 4 weeks after cerebral hypoxia—ischemia (Figures 3C to 3E and 3H to 3J). In these animals, a mild insult produced either grossly visible tissue damage or no gross tissue damage in brains assessed histologically 1 to 4 weeks after insult (Figure 5). The gross damage included loss of periventricular white matter and lateral ventricular enlargement, presence of several small periventricular cysts or glial scars, and mild cerebral atrophy including cortical thinning and atrophy of subcortical structures such as the basal ganglia. To define the different MR imaging changes associated with obvious permanent injury, the MR data in the mild group were divided into animals with and without gross damage and analyzed separately (Figure 6). Permanent damage in the contralateral hemisphere was observed very infrequently (e.g., Figure 5B).
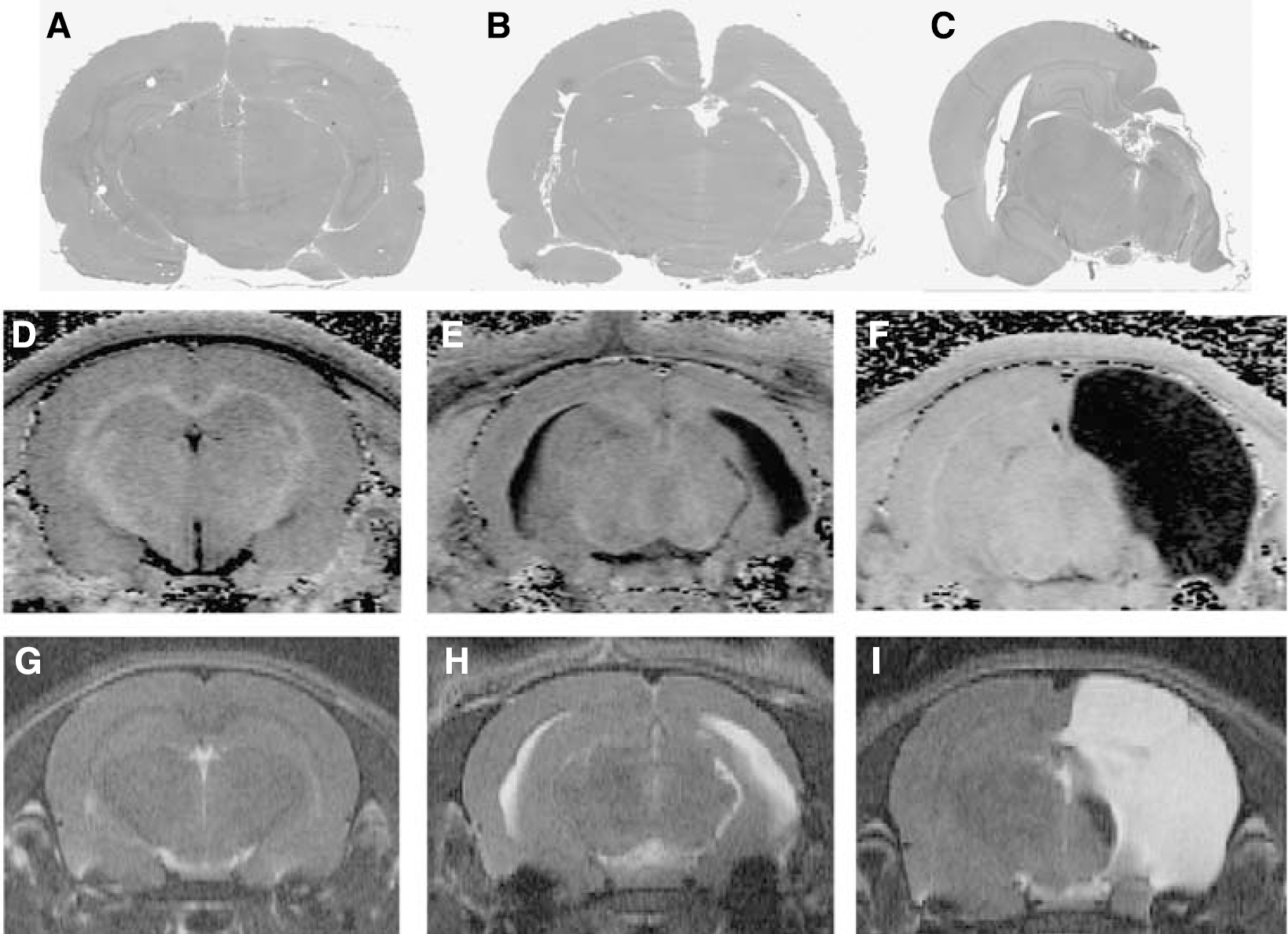
Representative histological sections from rats corresponding to the group subjected to mild hypoxia—ischemia without gross damage (Mild I) (
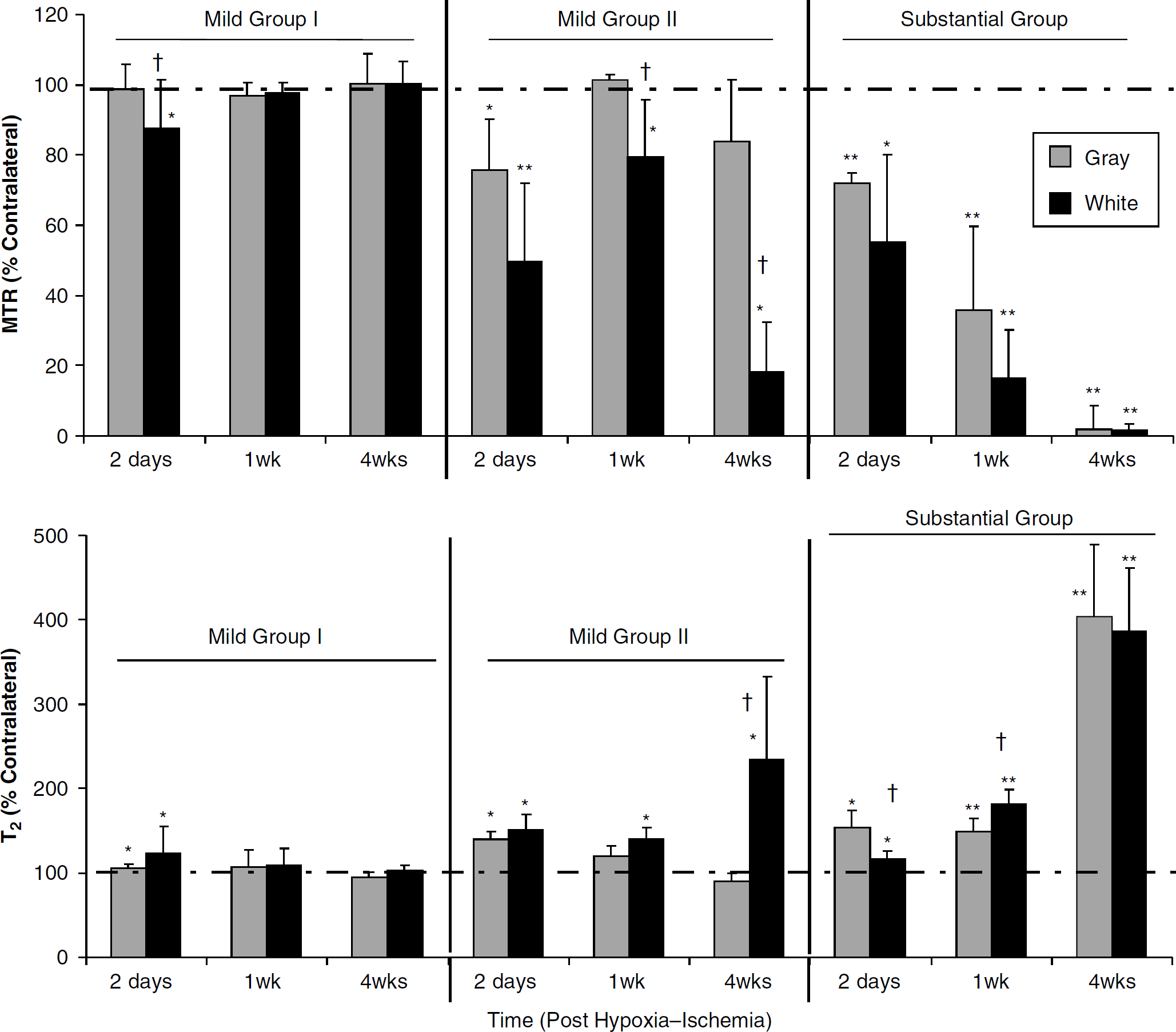
Subacute and prolonged (> 24 h) changes in mean MTRs (
After a mild insult without gross damage (Mild I), the changes in MTR and T2 followed a similar pattern (Figure 6). Significant decreases in MTR and increases in T2 were observed within white matter at 2 days after insult. These changes were not observed at 1 and 4 weeks after insult that may suggest a recovery from edematous changes. Indeed, microscopic examination of hematoxylin and eosin-stained sections in this subgroup at 1 to 4 weeks after insult detected subtle changes consisting of slight thinning of the white matter and cortex ipsilateral to the hypoxia—ischemia.
Two days after a mild insult with gross damage (Mild II), there were reductions in MTR in both gray matter of parietal cortex and white matter (Figure 6). Magnetization transfer ratio and T2 in cortex returned toward baseline at 1 and 4 weeks, whereas white matter changes persisted or increased. The normalization of imaging changes in parietal cortex was associated with a resolution of edema and loss of necrotic cells resulting in cortical atrophy, whereas white matter changes were adjacent to cyst formation.
Chronic Effects of Substantial Cerebral Hypoxia—Ischemia
A substantial insult resulted in progressive increases in T2 and decreases in MTR in much of the hypoxic—ischemic hemisphere (Figure 6). Responses were similar in gray and white matter. The large increases in T2 and decreases in MTR at 4 weeks after insult reflect the presence a cystic infarct.
Behavioral Changes with Differing Injury Severity
There were differences in the behavioral responses between groups for all three tests examined (Figures 7A to 7C). The rat pups subjected to substantial hypoxia—ischemia had significant impairments in function compared with sham controls according to their neurological score and their performances on the water maze and cylinder test. This is generally consistent with the behavioral differences reported by others using a similar model of neonatal cerebral hypoxia—ischemia (Bona et al, 1997; Ikeda et al, 2006). It also corresponds with the extensive damage and cavitation detected in these animals using T2 and MT imaging.
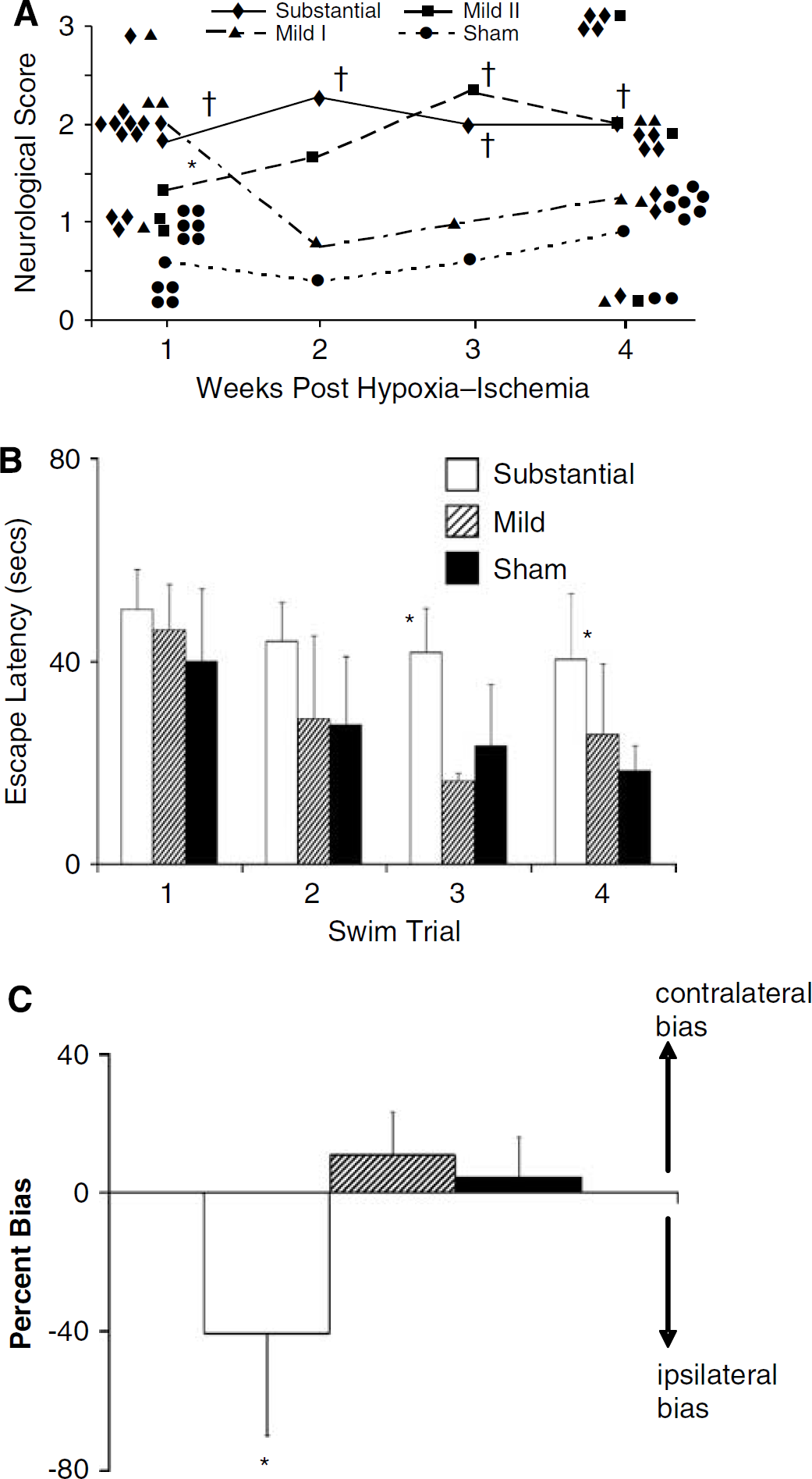
Effect of severity of the cerebral hypoxic—ischemic insult on behavioral outcomes in subgroups of animals imaged with MR. Neurological scores (
For the mild group of rat pups randomized to behavioral testing, only those allocated to neurological score testing had a portion of the group (3/7) with gross permanent brain damage. Thus, for the neurological score test, the mild group was subdivided into those with and without gross brain damage and analyzed separately. For all three behavioral tests, the groups of animals with a mild insult without gross damage had behavioral responses similar to sham controls. The mild subgroup with gross permanent cerebral damage had a significant deficit on neurological score testing. Thus, when gross brain damage was apparent, MTR and T2 imaging changes were still obvious in both white and gray matter at 2 days after hypoxia—ischemia without full recovery being observed after 1 week. Such MR imaging changes corresponded to animals with a significant neurological deficit. If gross damage was not apparent, MR imaging measures recovered by 1 week and there were no significant behavioral deficits.
Discussion
This is an original study in immature brain that provides novel information concerning the effects of a hypoxic—ischemic insult on MTR decreases and their evolution in immature brain, on their regional variation in white compared with gray matter, and on their correspondence with severity of insult. We show, to our knowledge for the first time, that (1) early MTR changes are indicative of tissue that is at high risk of permanent injury. (2) Magnetization transfer ratio imaging is sensitive for detecting mild ischemic injury in immature white matter, and (3) after mild hypoxia—ischemia in immature brain, there can be acute MTR changes and no significant T2 changes, whereas both are evident with more severe injury.
Magnetization Transfer Ratio is Reduced in Gray Matter after a Substantial Ischemic Insult
Much of the evidence regarding MT imaging of ischemic brain is available from studies in adults, and these support the observation that a substantial cerebral ischemic insult results in reductions in MTR within gray matter at subacute or prolonged time points after the insult (Hanyu et al, 1998; Knight et al, 2005; Tourdias et al, 2007). At acute time points after an ischemic insult, MTR changes have been reported to be variable with either a lack of major changes within the first few hours (Hanyu et al, 1998; Makela et al, 2002) or a significant decrease (Ewing et al, 1999; Knight et al, 2005). Variability can be related to differences in MT imaging methods and/or the types and severity of ischemic injury investigated.
Magnetization transfer ratio imaging after cerebral ischemia in immature brain has been infrequent. One clinical study of children with cerebral infarcts (Pui and Wang, 2005) showed some trend for reductions in MTR after a stroke, but there was a wide age range (newborn to 17 years), infarct types, and recovery times investigated. The present results show directly that after a substantial hypoxic-ischemic insult in immature brain there are early (i.e., 1 h) reductions in MTR and increases in T2, which evolve progressively in both white and gray matter as the infarct matures.
Magnetization Transfer Ratio is Sensitive for Detecting Hypoxic—Ischemic White Matter Injury
Previous studies on MTR imaging in adults have observed reductions in MTR in chronic white matter lesions of potential ischemic origin (Fazekas et al, 2005; Kado et al, 2001). Magnetization transfer ratio imaging in several white matter regions in infants with periventricular leucomalacia reported a relatively reduced MTR or an arrested myelination within the white matter of the splenium (Xydis et al, 2006). The present study shows that MTR changes are readily detected in white matter at acute in addition to prolonged times in immature brain after a hypoxic—ischemic insult. Indeed, after a mild insult that has been shown to be associated with relatively selective white matter injury (Meng et al, 2006), MTR imaging but not standard T2 imaging detected significant early white matter changes. This lack of sensitivity of standard imaging for detecting ischemic injury soon after reperfusion is consistent with our previous report that noted no significant white matter changes in T2 and apparent diffusion coefficient (ADC) ipsilaterally early (1 h) after hypoxia—ischemia (Meng et al, 2006). The sensitivity for MTR imaging to detect white matter ischemic injury is supported by a report of an enhanced ability of MT imaging compared with T2 imaging to detect early beneficial effects of therapy with an intracellular adhesion molecule-1 antibody for focal cerebral ischemia in rats (Jiang et al, 2001). Considering that often changes in MTR are a ssociated with changes in T2, the reduced MTR with hypoxia—ischemia is at least, in part, likely because of tissue edema, particularly at acute time points. In addition, ischemic and developmental alterations in macromolecules and cellular structures, such as myelin, also need to be considered as potential contributors to the MTR changes.
Changes in MTR and T2 and Their Relationship to Permanent Damage
In addition to detecting early reductions in MTR in white matter after a mild insult, the present study showed that there was a progression of imaging changes that differed according to final damage observed. Others have previously reported that a relatively mild hypoxic—ischemic insult can result in a subgroup of animals that ultimately progress to significant tissue damage at 8 weeks after insult compared with another group assessed at 2 weeks after insult (Geddes et al, 2001). We found that animals either did or did not develop gross damage chronically after a mild hypoxic—ischemic insult and these two groups could be distinguished by differences in their imaging changes at 2 days after insult. After a mild hypoxic—ischemic insult, animals without gross damage, but subtle histological changes, had modest white matter decreases in MTR and increases in T2, and these resolved by 1 week after insult. In contrast, for animals in the mild group with gross damage, pronounced imaging changes occurred in white matter and to a lesser extent in cortex. At 1 week, there was a transient recovery of MR changes; however, only in cortical gray matter. Chronically, there were imaging changes remaining in white and gray matter along with evidence of sizeable tissue damage. In these animals, there was significant gray matter involvement even though less than that in the substantial injury group. Thus, this mild insult is considered to model the type of periventricular white matter injury observed in premature or term infants that is often accompanied by cortical or basal ganglia involvement (Back, 2006; Kinney, 2006). Our behavioral results suggest that there is functional recovery in the mild group with subtle histological changes, whereas obvious deficits occur if there is gross damage with cortical involvement. Thus, similar to neonatal infants, neurological outcomes appear worse if both gray matter and white matter are affected. Note that although the mild insult produced only transient MR imaging changes and subtle histological changes, deficits in function may be present but would need a finer or more sensitive behavioral test to be detectable.
In contrast, in the substantial ischemic insult group, significant MTR and T2 changes were present at all times with changes in gray matter often being more pronounced than those in white matter. Over time after insult, the imaging changes enhanced until the detection of very large increases in T2 and decreases in MTR at 4 weeks after insult, reflecting the presence of cavitation. This group also exhibited significant functional deficits. Thus, rather marked early decreases in MTR and increases in T2 were predictive of major gross damage and functional deficits.
To conclude, MTR detects well ischemic injury in immature brain. At acute times, MTR imaging appears more sensitive for detecting mild ischemic injury than T2 imaging, particularly in white matter. Although acute MTR changes may not be able to predict definitively which animals will have poor outcomes, MTR imaging does help identify a group at increased risk for obvious neurological deficit. Pronounced MTR and T2 changes occurred after a substantial insult and were indicative of severe ischemic injury that evolved to cystic infarct or cavitation. Whether there is a threshold for magnitudes of early MTR changes that can help predict final outcome will require further study with experiments designed to address this issue. These results encourage further study of MTR imaging for its potential for early diagnosis of white matter ischemic injury in infants affected by perinatal hypoxia—ischemia.
Footnotes
Acknowledgements
R Kokotailo assisted with the MTR optimization studies. T Foniok performed the optimization and MR imaging acquisition. D Kirk provided technical assistance during MR imaging, and S Shivji and S Lama assisted with histology and image analysis.
