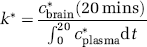Abstract
Nicotine exerts its central effects by activating pre- and postsynaptic nicotinic acetylcholine receptors (nAChRs). Presynaptic nAChRs modulate the release of many neurotransmitters that bind to postsynaptic receptors. These may be coupled to the activation of cytosolic phospholipase A2 (cPLA2), which hydrolyzes arachidonic acid (AA) from membrane phospholipids. We hypothesized that nicotine would modify brain signaling involving AA by binding to nAChRs. Nicotine (0.1 mg/kg, subcutaneously) or saline was injected 2 or 10 mins before infusing [1-14C]AA in unanesthetized rats. The AA incorporation coefficient k∗ (a marker of the AA signal) was measured in 80 brain regions by quantitative autoradiography. Nicotine, compared to saline, when administrated 2 mins before [1-14C]AA infusion, significantly decreased k∗ for AA in 26 regions, including cerebral cortex, thalamus, and habenula—interpeduncular regions, by 13% to 45%. These decreases could be entirely prevented by pretreatment with mecamylamine (1.0 mg/kg, subcutaneously). When administered 10 mins before [1-14C]AA infusion, nicotine did not alter any value of k∗. In summary, nicotine given to unanesthetized rats rapidly reduces signaling involving AA in brain regions containing nAChRs, likely by modulating the presynaptic release of neurotransmitters. The effect shows rapid desensitization and is produced at a nicotine dose equivalent to smoking one cigarette in humans.
Introduction
Nicotine, as the S(−)-isomer, is a major component in tobacco products and is an important cause of addiction (Henningfield and Fant, 1999). It binds to and activates nicotinic acetylcholine receptors (nAChRs) and can improve memory in animals and humans (Henningfield and Fant, 1999). Nicotinic acetylcholine receptors are thought to be involved in the pathophysiology of autism, schizophrenia, Parkinson's disease, Alzheimer's disease and depression, and to change with aging (Gotti et al, 2006). They are widely distributed in brain, particularly at presynaptic nerve terminals, where they modulate the release of acetylcholine, γ-aminobutyric acid (GABA), dopamine, serotonin, and glutamate in a concentration- and region-dependent manner. This presynaptic modulation is considered to be their predominant role in the brain (Gotti et al, 2006). Nicotinic acetylcholine receptors also are present on postsynaptic nerve terminals, neuronal cell bodies, and dendrites (Dajas-Bailador and Wonnacott, 2004; Gotti et al, 2006).
Nicotinic acetylcholine receptors are pentameric ligand-gated ion channels selective for Ca2+ and Na+. Combinations of different subunits produce different nAChR subtypes with distinct pharmacological properties. The homomeric α7 and heteromeric α4β2 receptor subtypes are highly expressed in the rat brain (Gotti et al, 2006). α4β2 nAChRs, which account for 90% of nAChRs, have a high affinity for nicotine, a moderate Ca2+ relative to Na+ permeability, and are reported to undergo desensitization within 10 to 15 mins in interneurons of the rat hippocampus and in whole-brain slices, as well as in other in vitro preparations (Alkondon et al, 2000; Dajas-Bailador and Wonnacott, 2004; Quick and Lester, 2002). α7 nAChRs have a low affinity for nicotine, a higher Ca2+ permeability than the other subtypes, and become desensitized within milliseconds in hippocampal neuron cultures (Dajas-Bailador and Wonnacott, 2004; Quick and Lester, 2002; Wang and Sun, 2005).
The released neurotransmitters (e.g., acetylcholine, dopamine, serotonin and/or glutamate), resulting from nicotine binding to presynaptic nAChRs, can bind to postsynaptic neuroreceptors (e.g., cholinergic muscarinic M1,3,5, dopaminergic D2-like, serotonergic 5-HT2A/2C and N-methyl-
Brain cPLA2 activation involving AA can be imaged in vivo by infusing radiolabeled AA intravenously after the administration of a drug acting at a cPLA2-coupled neuroreceptor, then measuring the tracer uptake into brain phospholipid in terms of an incorporation coefficient k∗ (brain radioactivity/integrated plasma radioactivity). k∗ for AA is independent of changes in cerebral blood flow, and thus is a specific marker of the brain AA signal (Basselin et al, 2006a, 2006b; Bhattacharjee et al, 2005; Robinson et al, 1992). Because AA is nutritionally essential and can neither be synthesized de novo in brain nor be converted in brain from its shorter chain linoleic acid (18:2n−6) precursor, k∗ for AA represents the plasma AA that has replaced the brain AA that was released by cPLA2 activation and then lost by metabolism to eicosanoids such as prostaglandin E2 (PGE2) (Basselin et al, 2006b).
As nicotine can increase synaptic concentrations of various neurotransmitters (see above) and thereby modulate AA release at postsynaptic sites (Vijayaraghavan et al, 1995), we hypothesized that acute administration of nicotine would alter k∗ for AA in brain regions having nAChRs. As nicotine readily enters the brain (Ghosheh et al, 1999) and nAChRs are known to be rapidly desensitized after exposure to nicotine (Alkondon et al, 2000; Quick and Lester, 2002), we also predicted that nicotine's signaling effects would be rapid and transient.
To test these hypotheses, nicotine (0.1 mg/kg, subcutaneously) was injected 2 and 10 mins before infusing [1-14C]AA intravenously in unanesthetized rats. Arachidonic acid incorporation coefficients k∗ were measured 20 mins after beginning tracer infusion with quantitative autoradiography in 80 identified brain regions. In addition, the nonspecific nAChR antagonist, mecamylamine (1.0 mg/kg, subcutaneously), was given alone or before nicotine administration. The doses and time points of measurement were based on studies of brain glucose metabolism in unanesthetized rats (London et al, 1988a, 1988b).
Materials and methods
Animals
Experiments were conducted following the Guide for the Care and Use of Laboratory Animals (National Institutes of Health Publication No. 86-23) and were approved by the Animal Care and Use Committee of the Eunice Kennedy Shriver National Institute of Child Health and Human Development. Three-month-old male Fischer CDF (F-344)/CrlBR rats (Charles River Laboratories, Wilmington, MA, USA) were housed in an animal facility in which temperature, humidity, and light cycle were regulated, with free access to water and food (Rodent NIH-31 auto 18-4 diet, Zeigler Bros, Gardners, PA, USA). The diet contained (as percentage of total fatty acids) 20.1% saturated, 22.5% monounsaturated, 47.9% linoleic, 5.1% α-linolenic, 0.02% AA, 2.0% eicosapentaenoic, and 2.3% docosahexaenoic acid.
Chemicals
[1-14C]AA in ethanol (50 mCi/mmol, 99% pure) was purchased from Moravek Biochemicals (Brea, CA, USA). (−)-Nicotine hydrogen tartrate, mecamylamine hydrochloride, HEPES (4-(2-hydroxyethyl)-1-piperazineethanesulfonic acid), and fatty acid-free bovine serum albumin were purchased from Sigma Chemical Co. (St Louis, MO, USA). Sodium pentobarbital was purchased from Abbott Laboratories (North Chicago, IL, USA). Nicotine was dissolved in saline (0.9% NaCl), and solution pH was adjusted to 7.0 with 1
Arterial and Venous Catheter Placement
A rat was anesthetized with 2% to 3% halothane in O2, and PE-50 polyethylene catheters were inserted into the right femoral artery and vein, as described previously (Basselin et al, 2006a). The wound was closed with surgical clips, and the rat was wrapped loosely, with its upper body remaining free, in a fast-setting plaster cast, which was then taped to a wooden block. Surgery lasted 20 to 25 mins. The rat was allowed to recover from anesthesia for 3 to 4 h in a quiet environment maintained at 23 °C. Arterial systolic and diastolic pressures and heart rate were measured with a blood pressure recorder (Model 2400, Gould, Cleveland, OH, USA). Arterial blood pH, partial pressure of oxygen (pO2), and partial pressure of carbon dioxide (pCO2) were measured with a blood gas analyzer (Model 248, Bayer Health Care, Norwood, MA, USA). All physiologic parameters were measured before drug injection and 10 mins after [1-14C]AA infusion.
Treatment Schedules
Rats were injected with 0.1 mg/kg nicotine, followed at 2 or 10 mins by an intravenous infusion of [1-14C]AA. Control animals were given saline followed at 2 mins by [1-14C]AA infusion. Additional groups were administered mecamylamine (1.0 mg/kg) at 17 mins before [1-14C]AA infusion, or mecamylamine (1.0 mg/kg) at 15 mins followed with 0.1 mg/kg nicotine at 2 mins before [1-14C]AA infusion. Drugs as well as saline were injected subcutaneously in a volume of 1 mL/kg.
Radiolabeled Arachidonic Acid Infusion
A pump (Model 22, Harvard Apparatus, Natick, MA, USA) was used to infuse a rat intravenously at a constant rate of 0.4 mL/min for 5 mins, with [1-14C]AA (170 μCi/kg) in 2 mL of 5 mmol/L HEPES buffer, pH 7.4, containing 50 mg/mL fatty acid-free bovine serum albumin. A total of 13 timed arterial aliquots (80 to 120 μL) were collected before, during, and after tracer infusion to time of death 20 mins later. At 20 mins after starting the infusion, the rat was killed with an overdose of sodium pentobarbital (80 mg/kg, intraperitoneally) and then decapitated. The brain was removed (<30 secs), frozen in 2-methylbutane at −40 °C, and stored at −80 °C for later quantitative autoradiography.
Radiolabeled Unesterified Arachidonic Acid in Plasma
The arterial blood samples were centrifuged immediately (30 secs at 18,000 g). Total lipids were extracted from 30 μL of plasma, with 3 mL chloroform/methanol (2:1, by volume) and 1.5 mL 0.1
Quantitative Autoradiography
Frozen brains were cut in serial 20-μm-thick coronal sections in a cryostat at −20 °C. Sections were exposed together with calibrated [14C]methylmethacrylate standards to Ektascan C/RA film (Eastman Kodak Company, Health Imaging Group, Rochester, NY, USA) for 5 weeks. Brain regions from autoradiographs, identified from a stereotaxic rat brain atlas (Paxinos and Watson, 1987), were sampled in both hemispheres. The average of bilateral measurements for each region from three consecutive brain sections was used to calculate regional radioactivity (nCi/g of brain) by digital quantitative densitometry, using the public domain NIH Image program 1.62 (developed at the US National Institutes of Health and available on the Internet at http://rsb.info.nih.gov/nih-image/). Regional AA incorporation coefficients k∗ (mL plasma per sec per g brain) were calculated as (Robinson et al, 1992),
where c∗plasma (nCi/mL) is the plasma radioactivity determined by scintillation counting, c∗brain (20 mins) (nCi/g) is brain radioactivity determined by densitometry at 20 mins, and t (mins) is the time after beginning of the [1-14C]AA infusion.
Brain Prostaglandin E2
In separate experiments, rats were given nicotine (0.1 mg/kg, subcutaneously) or saline for 7 mins. Later, they were anesthetized with Nembutal® (50 mg/kg, intraperitoneally) and subjected to head-focused microwave irradiation (5.5 kW, 3.6 secs; Cober Electronics, Stamford, CT, USA) to stop brain metabolism. Frozen half-brains were weighed, homogenized with 18 volume of hexane/isopropanol (3:2 by volume) using a glass Tenbroeck homogenizer and the homogenate was centrifuged for 5 mins at 800 g. Tissue residues were then rinsed with 3 × 2 volume of the same solvent. The resultant lipid extract was concentrated to dryness under nitrogen and resuspended in an enzyme immunoassay buffer provided with a polyclonal PGE2 assay kit (Oxford Biochemical Research, Oxford, MI, USA).
Statistical Analysis
Data are expressed as means±s.d. Physiologic parameters were analyzed by paired t-tests in the same animal before and after drug injection (GraphPad Prism Software, San Diego, CA, USA; http://www.graphpad.com). The significance of drug treatments on arterial plasma radioactivity and k∗ values for each brain region was determined by one-way analysis of variance. For each of the 80 regions, we used one-way analysis of variance to test for significant differences among group means. We set our α at 0.05 and all tests were two-tailed. We then applied a false discovery rate (FDR) procedure (Benjamini and Hochberg, 1995) to control the FDR at 0.05. Each significant analysis of variance result that survived the FDR procedure was then followed up with Bonferroni's post hoc test for multiple mean comparisons. Our post hoc comparisons corresponded to our hypotheses and included all groups versus control, and mecamylamine+nicotine (2 mins) versus nicotine (2 mins).
Results
Physiology and Arterial Plasma Radioactivity
As illustrated in Table 1, nicotine (0.1 mg/kg, subcutaneously) at 2 mins significantly decreased heart rate by 24%, compared with saline. Nicotine at 2 mins increased pO2 by 13%, whereas it reduced pCO2 by 12%. Mecamylamine alone decreased heart rate by 14% and blood pressure by 22%. The combination of mecamylamine and nicotine blocked the nicotine-induced heart rate response but decreased blood pressure by 20%.
Effects of nicotine and mecamylamine on physiological parameters
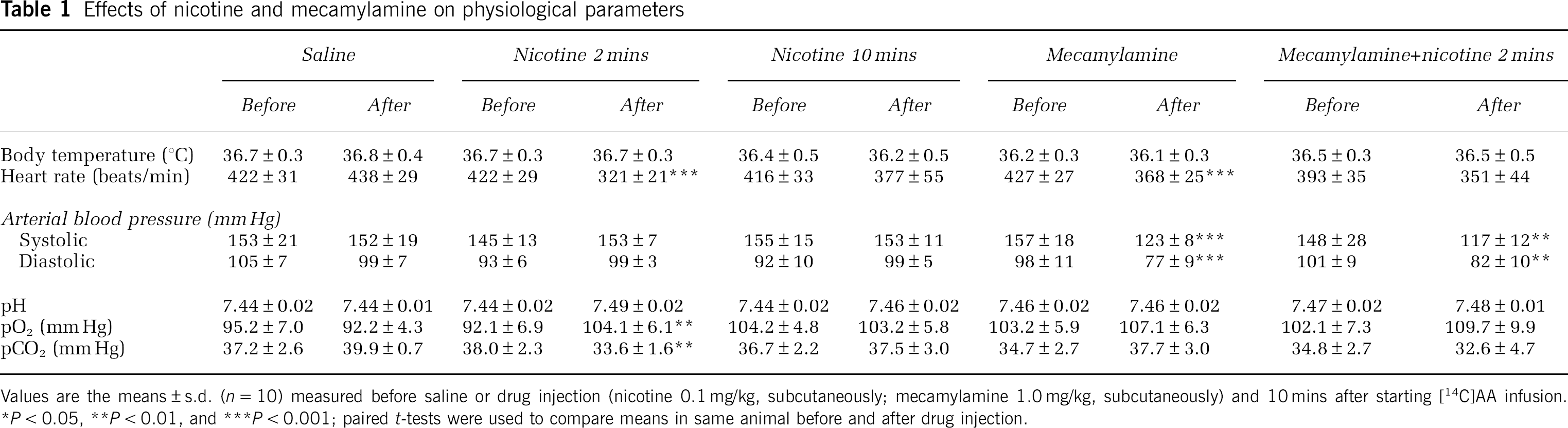
Values are the means±s.d. (n=10) measured before saline or drug injection (nicotine 0.1 mg/kg, subcutaneously; mecamylamine 1.0 mg/kg, subcutaneously) and 10 mins after starting [14C]AA infusion. ∗P<0.05, ∗∗P<0.01, and ∗∗∗P<0.001; paired t-tests were used to compare means in same animal before and after drug injection.
The mean integral of radioactivity in the plasma organic fraction (nCi × sec)/mL, the input function in Eq. 1, did not differ significantly between groups: 200,957±33,731 after saline; 214,412±12,364 after nicotine (2 mins), 173,297±17,958 after nicotine (10 mins), 218,876±16,926 after mecamylamine, and 211,397±38,314 after mecamylamine and nicotine (2 mins).
Regional Brain Arachidonic Acid Incorporation Coefficients, k∗
Figure 1 illustrates coronal autoradiographs from rats injected with saline, nicotine 2 mins, nicotine 10 mins, and mecamylamine and nicotine 2 mins before [1-14C]AA infusion. Compared with saline, reductions in k∗ for AA are evident when [1-14C]AA was injected at 2 but not 10 mins after nicotine. Pretreatment with mecamylamine blocked these 2-min reductions.
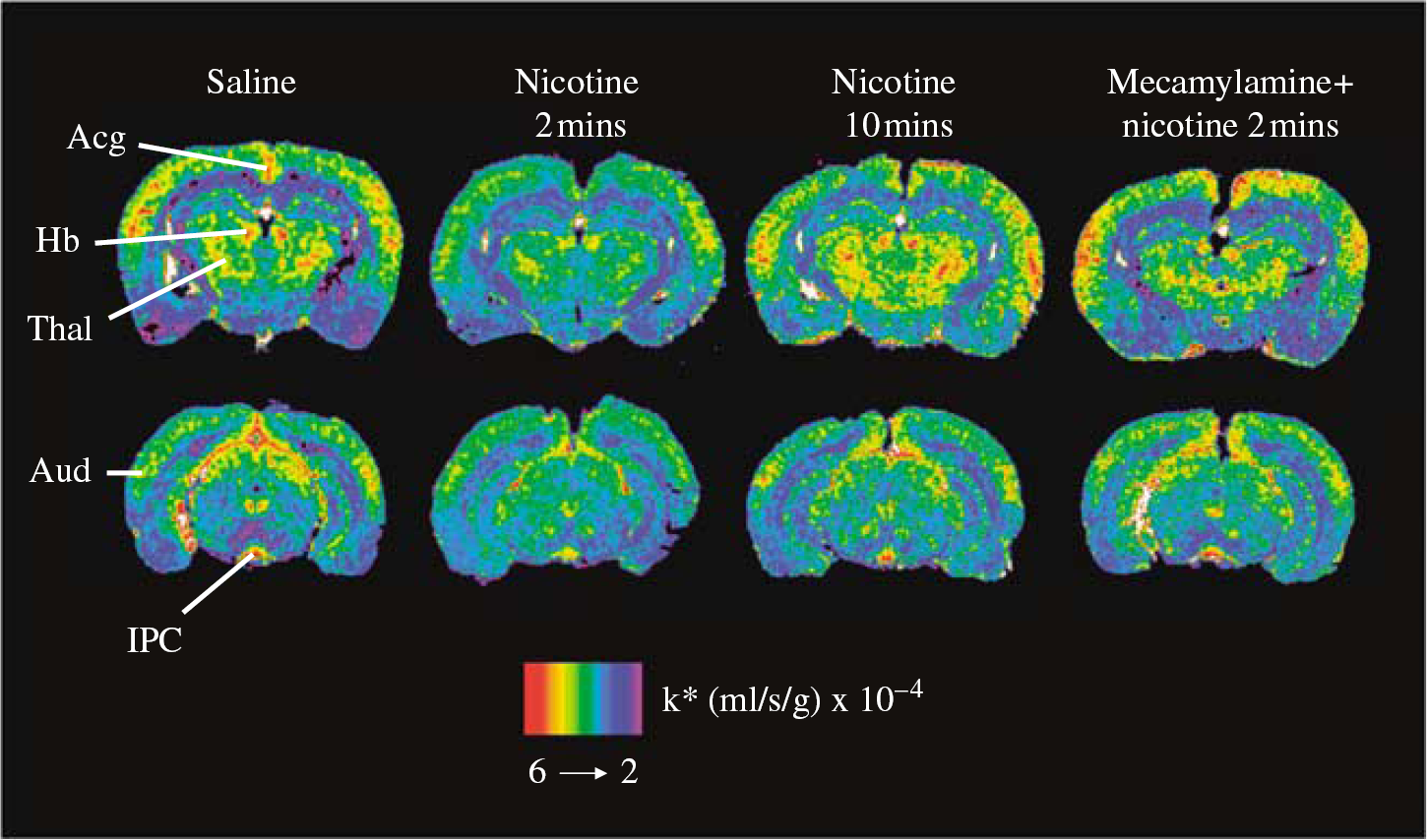
Coronal autoradiographs showing effects of nicotine on AA incorporation coefficients k∗ in rats. Studies involved injection of saline or nicotine and measuring k∗ for AA after 2 or 10 mins, and injection of nicotine after pretreatment with mecamylamine. Values of k∗ (mL/s per g brain × 10−4) are given on a color scale. Abbreviations: Acg, anterior cingulate cortex; Aud, auditory cortex; Hb, habenular nucleus; IPC, interpeduncular nucleus; Thal, thalamus.
Nicotine, injected 2 mins before [1-14C]AA infusion, decreased k∗ significantly in 26 of 80 brain regions, by 13% to 45% (Table 2). Affected regions included the prefrontal cortex layer IV (−25%), frontal cortex 10 layers I (−30%) and IV (−30%), frontal cortex 8 layer IV (−20%), anterior cingulate cortex (−32%), motor cortex layer V (−25%), somatosensory cortex layers II/III, IV, and V (−23% to −30%), auditory cortex layer I (−31%), olfactory tubercle (−26%), lateral and medial caudate putamen (−24% to 25%), medial septal nucleus (−29%), lateral and medial habenular nuclei (−35% to 39%), dorsal lateral geniculate nucleus (−34%), anteroventral and anteromedial thalamic nuclei (−24% to 40%), reticular thalamic nucleus (−27%), parafascicular thalamic nucleus (−39%), supraoptic nucleus (−13%), posterior hypothalamic nucleus (−26%), interpeduncular nucleus (−45%), substantia nigra (−27%) and gray matter of the cerebellar (−30%). The effects on k∗ dissipated by 10 mins after nicotine injection.
Mean values of arachidonic acid incorporation coefficients k∗ in rats at the baseline (in response to saline) and in response to nicotine and mecamylamine
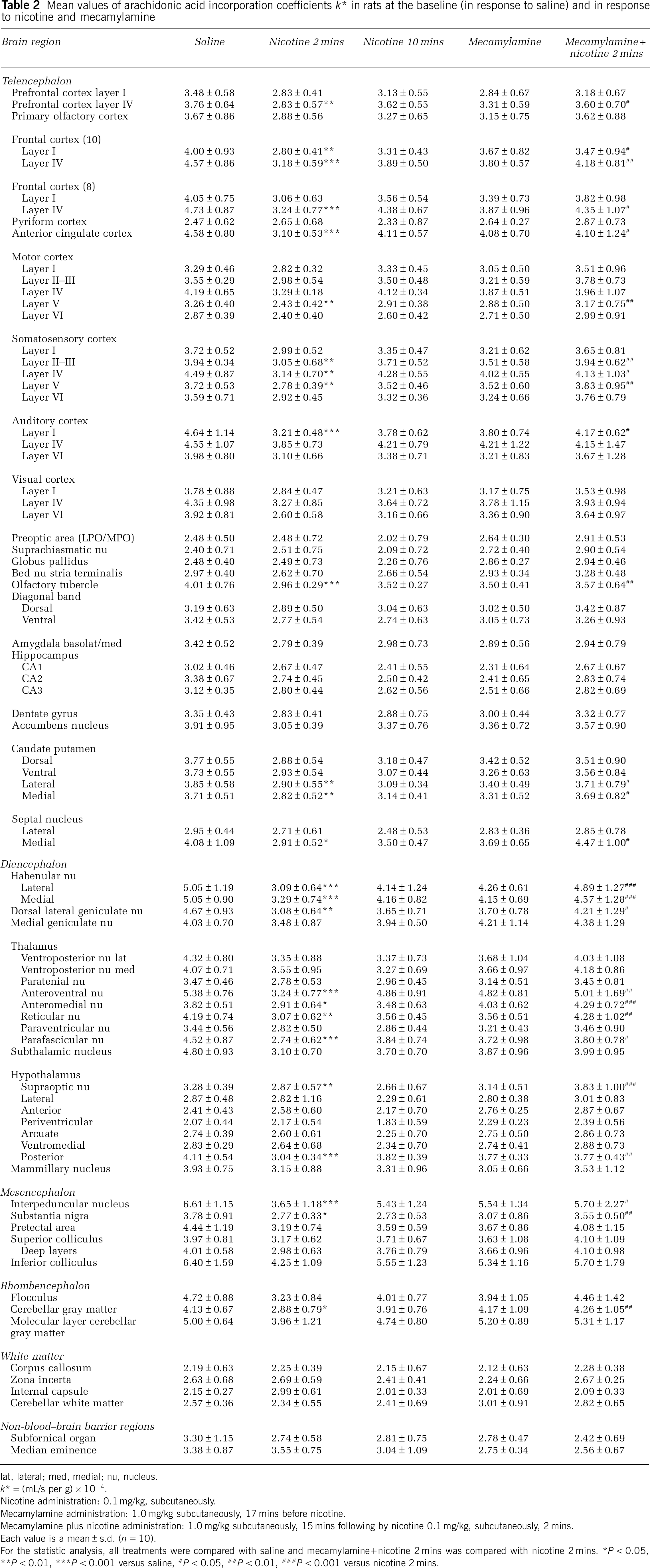
lat, lateral; med, medial; nu, nucleus.
k∗=(mL/s per g) × 10−4.
Nicotine administration: 0.1 mg/kg, subcutaneously.
Mecamylamine administration: 1.0 mg/kg subcutaneously, 17 mins before nicotine.
Mecamylamine plus nicotine administration: 1.0 mg/kg subcutaneously, 15 mins following by nicotine 0.1 mg/kg, subcutaneously, 2 mins.
Each value is a mean±s.d. (n=10).
For the statistic analysis, all treatments were compared with saline and mecamylamine+nicotine 2 mins was compared with nicotine 2 mins. ∗P<0.05, ∗∗P<0.01, ∗∗∗P<0.001 versus saline, #P<0.05, ##P<0.01, ###P<0.001 versus nicotine 2 mins.
Mecamylamine administered alone had no effect on k∗. When administered 15 mins before nicotine, it completely blocked the effects of nicotine at 2 mins (Table 2). Patterns of changes in k∗ due to nicotine and mecamylamine are illustrated in Figure 2.
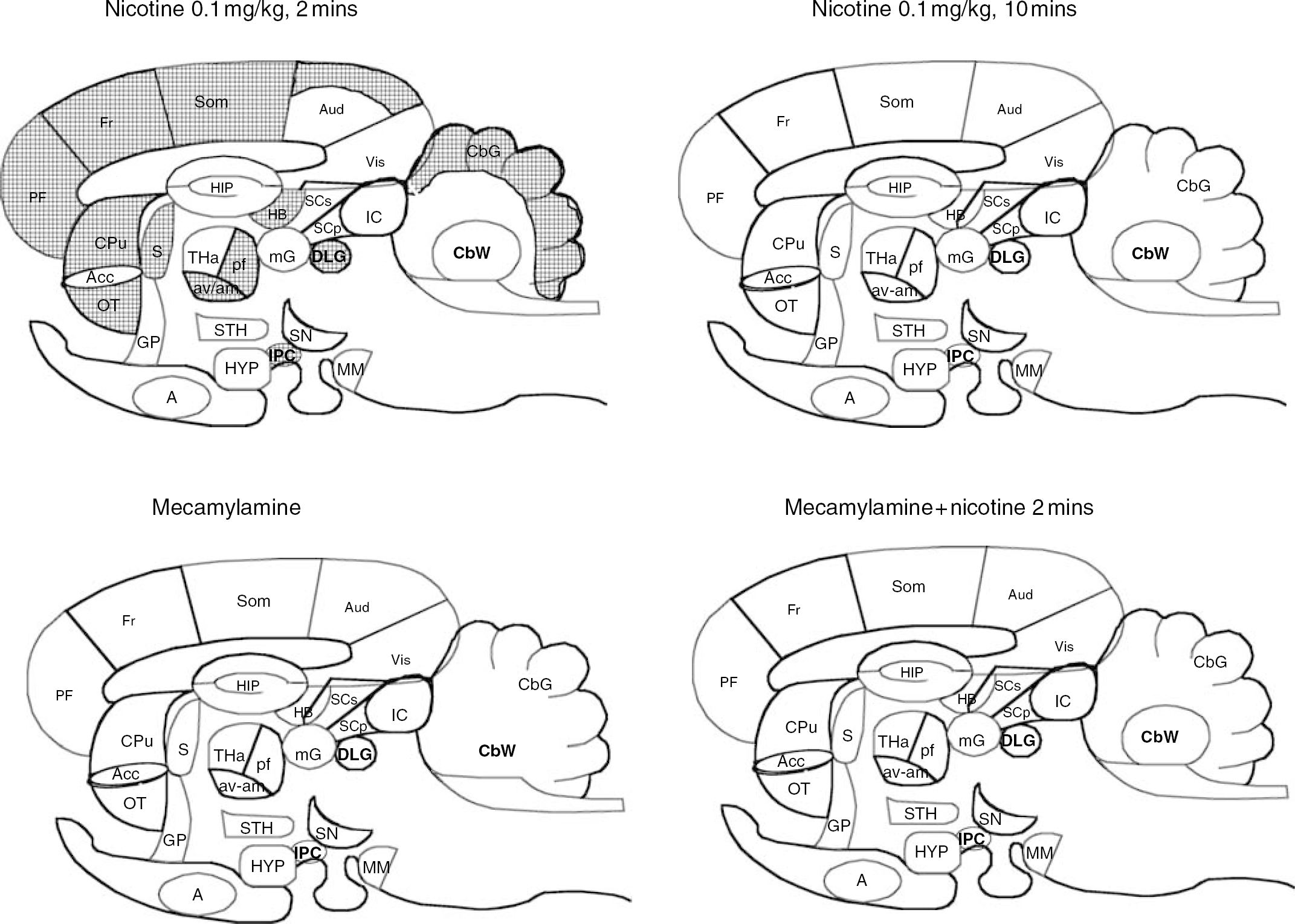
Difference patterns of k∗ responses to nicotine and mecamylamine in sagittal representation of the rat brain. Regions in which k∗ was decreased significantly are hatched. List of regions: A, amygdala; Acc, nucleus accumbens; Aud, auditory cortex; am, anteromedial thalamic nucleus; av, anteroventral thalamic nucleus; CbG, cerebellar gray matter; CBW, cerebellar white matter; CPu, caudate putamen; DLG, dorsal lateral geniculate nucleus; Fr, frontal cortex; GP, globus pallidus; HB, habenular complex; HIP, hippocampus; HYP, hypothalamus; IC, inferior colliculus; IPC, interpeduncular nucleus; MM, mammillary nucleus; mG, medial geniculate nucleus; OT, olfactory tubercle; PF, prefrontal cortex; pf, parafascicular thalamic nucleus; SN, substantia nigra; S, septum; Som, somatosensory cortex; SCp, deep layer of superior colliculus; SCs, superficial layer of superior colliculus; STH, subthalamic nucleus; THa, thalamus; Vis, visual cortex.
Prostaglandin E2 Concentration
Nicotine compared with saline did not significantly modify the net brain PGE2 concentration (20.0±3.1 versus 18.7±3.6 ng/g, n=4, P>0.05).
Discussion
Nicotine (0.1 mg/kg, subcutaneously) significantly decreased k∗ for AA at 2 mins postinjection in 26 of 80 brain regions, by −45% to −13%, but not at 10 mins postinjection. The decreases at 2 mins were mediated by nAChRs, as pretreatment with the nonspecific nAChR antagonist, mecamylamine, prevented them. Nicotine decreased heart rate, whereas mecamylamine given before nicotine blocked the change. The negative effect of nicotine on the heart rate, reported previously (London et al, 1988a), has been ascribed to nicotine-induced catecholamine release from central and peripheral sources (Gozzi et al, 2006). The mecamylamine effects on blood pressure and heart rate are consistent with previous reports (Gozzi et al, 2006).
These data support our hypotheses that nicotine would alter k∗ for AA and that its effect would be transient, consistent with reported receptor desensitization in in vitro studies. The decrements in k∗ caused by nicotine at 2 mins generally occurred at [3H]nicotine-binding sites (London et al, 1985). The habenula nuclei, interpeduncular nucleus, thalamic nuclei, and dorsal lateral geniculate nucleus, which had significantly reduced values of k∗ (by −35% to −39%), have high binding capacities for [125I]epibatidine (3.2 to 5.7 fmol/mg). This agonist has a high affinity for α4β2 nAChRs and for nAChRs containing the α3 but not the α7 subtype (Perry et al, 2002). Reductions in k∗ (by −20% to −30%) were also observed in the frontal, somatosensory, and anterior cingulate cortex, substantia nigra, caudate putamen, and cerebellum having lower [125I]epibatidine-binding capacities (1.1 to 2.0 fmol/mg) (Perry et al, 2002). In contrast, k∗ was not significantly affected by nicotine in the hippocampus, which has almost no [3H]nicotine-binding sites (London et al, 1985), very low [125I]epibatidine-binding capacity (0.5 to 0.8 fmol/mg) (Perry et al, 2002), and contains mostly the α7 receptor subtype (Gotti et al, 2006). Thus, these data and evidence that α4β2 nAChRs are reported to undergo desensitization within 10 to 15 mins in vitro (Alkondon et al, 2000; Dajas-Bailador and Wonnacott, 2004; Quick and Lester, 2002) and that α7 nAChRs become desensitized within milliseconds in hippocampal neuron cultures (Dajas-Bailador and Wonnacott, 2004; Quick and Lester, 2002) suggest that nicotine's 2-min effect on the AA signal was largely mediated by α4β2 and α3 subtypes. Significant reductions in k∗ for AA at 2 mins after nicotine were also evident in auditory (medial habenula nucleus and auditory cortex layer I) and visual (lateral habenula nucleus and dorsal lateral geniculate nucleus) circuits. The parafascicular thalamic nucleus, where a reduction also occurred, can participate in visual oculomotor circuits (Van der Werf et al, 2002). All these regions belong to the cholinergic system (Woolf, 1991). Indeed, the cortex, interpeduncular, thalamus, and caudate putamen are innervated by the pontomesencephalic and/or basal forebrain cholinergic cells.
Nicotine concentrations peak in the rat plasma and brain 10 mins after subcutaneous nicotine, and then decrease with half-lives of 20 and 50 mins, respectively (Ghosheh et al, 1999). Thus, although brain nicotine concentrations were higher at 10 than at 2 mins, significant reductions in k∗ for AA were only observed at 2 mins, consistent with rapid nAChR desensitization (Alkondon et al, 2000; London et al, 1988a, 1988b; Quick and Lester, 2002). Metabolites of nicotine (cotinine, nornicotine, and norcotinine) likely did not play a role at 2 mins as they appear in the rat brain only 30 mins after subcutaneous nicotine (Ghosheh et al, 1999).
Thus, the data are consistent with the responses in k∗ for AA to nicotine at 2 mins being mediated by the activation of α4β2 and α3β4 receptor subtypes, as α7 receptors are very rapidly desensitized (Quick and Lester, 2002). Desensitization of nAChRs has been shown in hippocampal and whole-brain slices in vitro, making this report the first demonstration on neuroimaging of receptor desensitization in brain in vivo. Pretreatment with selective high-affinity agonists might help to identify the exact subtype of nAChRs involved in desensitization (Gozzi et al, 2006). The consequences of nAChR desensitization are multiple, and include homologous upregulation of nAChRs, hypersensitization of muscarinic receptors, modulation of gene expression for ion channels, receptors and signaling systems, learning and memory, and neuroprotection (Wang and Sun, 2005).
The basis of the reductions in k∗ for AA at 2 mins after nicotine is not immediately evident insofar as agonist stimulation of cPLA2-coupled postsynaptic D2-like, M1,3,5, 5-HT2A/2C, and NMDA receptors increased k∗ for AA in unanesthetized rats (Basselin et al, 2006a, 2006b; Bhattacharjee et al, 2005; Qu et al, 2003). Nicotine can provoke Ca2+ entry into the cell through nAChRs and voltage-gated Ca2+ channels, and also can trigger Ca2+ release from intracellular stores (Dajas-Bailador and Wonnacott, 2004). However, as nAChRs are largely presynaptic (see the Introduction section), it is unlikely that this release activated Ca2+-dependent AA-selective cPLA2, which is localized mainly at postsynaptic excitatory sites in rat brain (Ong et al, 1999). The reductions in k∗ at 2 mins were not likely due to cPLA2 inhibition by nicotine, although nicotine (10 μmol/L to 1 mmol/L) has been reported to inhibit neuronal, cytosolic, and membrane-bound PLA2 activities in vitro (Marin et al, 1997), because the brain nicotine concentration increased between 2 and 10 mins (Ghosheh et al, 1999), when significant k∗ changes were absent.
Although nicotine decreased regional k∗ values in 26 of 80 brain regions at 2 mins, it did not reduce the net brain concentration of PGE2. This may have reflected its transient effect on k∗ for AA, as studies in which AA turnover was chronically reduced showed reductions in cPLA2 activity as well as in PGE2 concentrations (Lee et al, 2007).
There are several possible causes for the reductions in k∗ for AA at 2 mins after nicotine, and these might be explored with appropriate antagonist pretreatment in future studies. One possible cause is that acute nicotine-stimulated GABAergic inhibition of neuronal activity (Contreras et al, 1999). Indeed, submicromolar concentrations of nicotine, in addition to activating nAChRs, enhanced the frequency and amplitude of GABAergic postsynaptic currents in interneurons of rat hippocampal and striatal slices (Alkondon et al, 2000). Another is that acute nicotine reduced glutamatergic signaling involving AA. Nicotine has been reported to reduce presynaptic glutamate release (Levy et al, 2006; Stapleton et al, 2003), which would be expected to reduce baseline values of k∗ for AA by decreasing postsynaptic NMDA receptor-mediated Ca2+ entry into cells and thereby reduce cPLA2 activation (Basselin et al, 2006a).
Although most nAChRs are presynaptic, some are found at postsynaptic sites in the brain (Dajas-Bailador and Wonnacott, 2004; Gotti et al, 2006). However, postsynaptic α4β2 nAChRs are unlikely to have been involved in nicotine's acute effect on k∗, as they have a low Ca2+ permeability compared with NMDA receptors (Rogers and Dani, 1995) and do not increase intracellular Ca2+ levels sufficiently to activate cPLA2, which requires 300 nmol/L to 1 μmol/L (Clark et al, 1995; Dajas-Bailador and Wonnacott, 2004).
The estimated brain nicotine concentration after 0.1 mg/kg subcutaneous nicotine injection in rats is ∼150 ng/g (Ghosheh et al, 1999). For comparison, in humans who smoke a single cigarette, the brain nicotine concentration reaches 20 to 70 ng/mL and results in 90% occupancy of α4β2 receptors for several hours (Brody et al, 2006). Thus, we would expect that k∗ for AA in the brain would be transiently reduced in humans who smoke just one cigarette. This prediction could be tested directly with positron emission tomography (PET) and [1-11C]AA, following the two-measurement paradigm (2 and 10 mins) employed in the rat study (Esposito et al, 2007).
The acute reduction in the AA signal by nicotine may have a neuroprotective action. N-methyl-
In vivo imaging after the intravenous injection of radiolabeled 2-deoxy-
A PET study of six drug-naive human volunteers reported that intravenous 1.5 mg nicotine significantly reduced brain glucose metabolism in 9 of 30 measured bilateral regions, and caused a 9.5% reduction in global glucose metabolism (Stapleton et al, 2003). In addition, PET studies using intravenous [15O]water showed that nicotine decreased normalized regional cerebral blood flow in the amygdala in healthy cigarette smokers (Rose et al, 2003), and in the left anterior temporal cortex and right amygdala after 12 h of smoking deprivation (Zubieta et al, 2001). Our data and these latter studies are consistent in indicating a negative effect of acute nicotine on different aspects of brain activity and metabolism. To our knowledge, the mechanisms for the nicotine-induced reduction of cerebral glucose metabolism and blood flow are not agreed upon.
In summary, nicotine, at a dose that produced a brain nicotine concentration in rats equivalent to that produced by smoking one cigarette in humans, reduced k∗ for AA in 26 of 80 brain regions examined at 2 mins after administration, but did not significantly affect k∗ for AA at 10 mins. As brain nicotine concentrations are reported to be higher at 10 than at 2 mins, this difference is consistent with nicotine receptor desensitization, which to date has been reported only in in vitro preparations. The reductions in k∗ for AA at 2 mins could be prevented by pretreatment with the reversible receptor antagonist mecamylamine, and likely reflected nicotine's modulation of presynaptic neurotransmitter release by nAChRs. Selective antagonists might be used in the future to distinguish which neurotransmitters and which neuroreceptors are involved in the process. In addition, PET might be used to see whether acute nicotine or smoking has a similar transient effect on AA signaling in the human brain.
Footnotes
Acknowledgements
We thank Dr Angelo O Rosa for his helpful comments on this paper.
The authors declare no conflict of interest.