Abstract
Multidrug resistance-associated protein 1 (MRP1) acts as a defense mechanism by pumping xenobiotics and endogenous metabolites out of the brain. The currently available techniques for studying brain-to-blood efflux have significant limitations related to either their invasiveness or the qualitative assessment. Here, we describe an in vivo method, which overcomes these limitations for assessing MRP1 function, using positron emission tomography (PET) and a PET probe. 6-Bromo-7-[11C]methylpurine was designed to readily enter the brain after intravenous administration and to be efficiently converted to its glutathione conjugate (MRP1 substrate) in situ. Dynamic PET scan provided the brain time—activity curve after injection of 6-bromo-7-[11C]methylpurine into mice. The efflux rate of the substrate was kinetically estimated to be 1.4 h−1 with high precision. Moreover, knockout of Mrp1 gene caused approximately a 90% reduction of the efflux rate, compared with wild-type mice. In conclusion, our method allows noninvasive and quantitative assessment for MRP1 function in the living brain.
Keywords
Introduction
The blood—brain barrier (BBB) consists of a monolayer of tightly fused together brain capillary endothelial cells (BCEC) that restrict the transfer of hydrophilic substances from the systemic circulation to the brain (Bradbury, 1993; Pardridge, 1998; Reese and Karnovsky, 1967). In addition, metabolic enzymes and active efflux transporters serve as detoxification systems in the brain and accelerate the elimination of xenobiotics out of the brain (Loscher and Potschka, 2005b; Minn et al, 1991). Glutathione (GSH) conjugation is one of the detoxification systems in the brain and is typically catalyzed by GSH transferases (GSTs; Ghersi-Egea et al, 1994; Ghersi-Egea and Strazielle, 2001). This detoxification process is finally followed by the efflux process of GSH conjugates, which is often mediated by multidrug resistance-associated protein 1 (MRP1). MRP1 is a member of the ATP binding cassette superfamily of transporters and is expressed at the blood—brain and blood—cerebrospinal fluid interfaces (Dallas et al, 2006; Kruh and Belinsky, 2003; Lee et al, 2001). This protein is involved in protection from the toxic effects of oxidative stress as well as defense against xenobiotics and endogenous toxic metabolites. For instance, Mrp1 mediates the export of oxidized glutathione (GSSG) formed under oxidative stress from astrocytes (Minich et al, 2006). The release of GSSG might be an essential pathway for astrocytes to remove cellular GSSG to maintain reduced thiol reduction potential under oxidative stress. MRP1 has been also implicated in the protection against the toxicity of 4-hydroxynonenal, which is formed during oxidative stress, by extrusion of its GSH conjugate (Renes et al, 2000). MRP1 has an unusually broad substrate specificity and can transport not only hydrophilic compounds such as GSH conjugates but also numerous hydrophobic compounds including therapeutic drugs (Loscher and Potschka, 2005b). Additionally, it has been reported that a chemotherapeutic agent affects MRP1 expression in the mice brain (Joshi et al, 2005). Therefore, the in vivo quantitative assessment of brain MRP1 function is useful for examining the alteration of MRP1 related to brain diseases and for directly evaluating the effect on MRP1 caused by drugs including modulators used to improve brain drug delivery. However, little has been reported on a possible quantitative and noninvasive method to assess brain MRP1 function in vivo.
The assessment of brain MRP1 function can be achieved by determining the efflux kinetics of a radiolabeled GSH conjugate from the brain. The brain efflux index (BEI) is a useful tool to study the brain-to-blood transporters in vivo (Kakee et al, 1996); however, BEI requires the direct injection of a compound into the brain. The delivery of GSH conjugates into the brain cannot be successfully accomplished by intravenous administration, because the BBB prevents hydrophilic substances such as GSH conjugates from reaching the brain via simple diffusion. Recently, we have devised the metabolite extrusion method (MEM), a kind of prodrug/drug approach, for noninvasive assessment of GSH-conjugate efflux systems (Okamura et al, 2007). In MEM, a lipophilic compound labeled with a suitable radionuclide is used as a pro-probe, which readily crosses the BBB after intravenous injection and is immediately metabolized to a hydrophilic probe in the brain. This allows the noninvasive delivery of the hydrophilic probe into the brain, leading to the assessment of unidirectional efflux without the need for intracerebral injection (Figure 1).
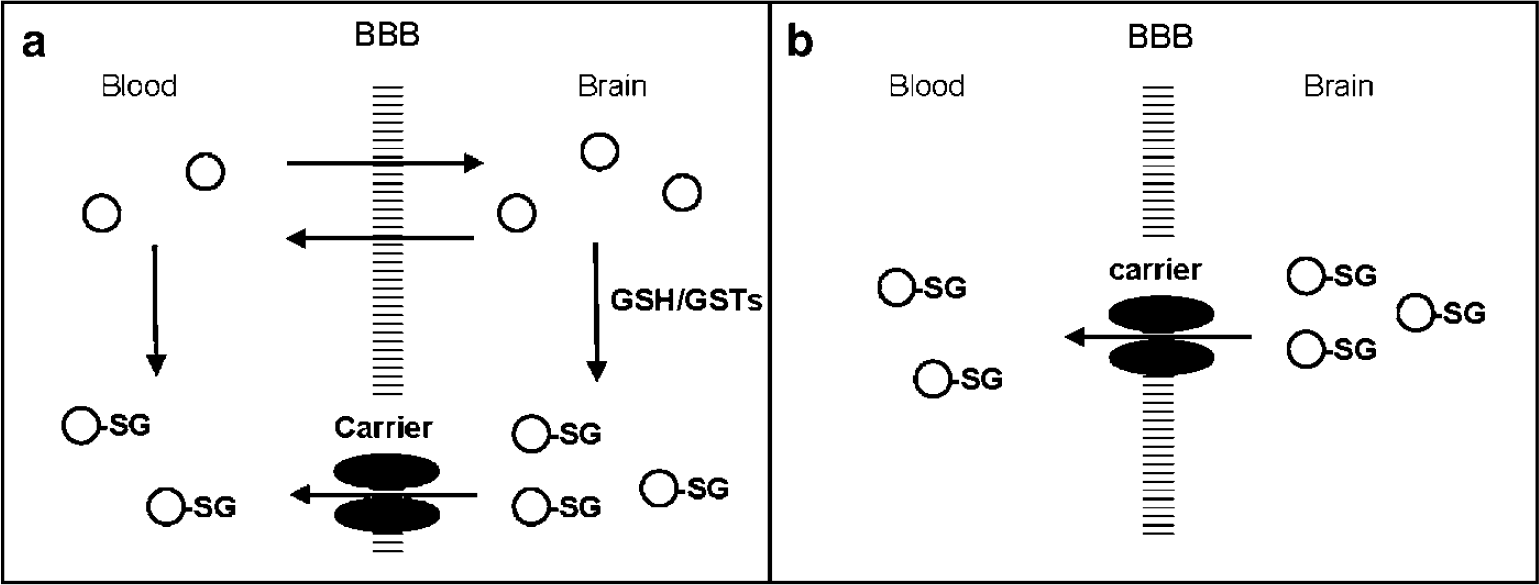
Metabolite Extrusion Method. When a pro-probe (open circles) is intravenously administered, it readily enters the brain via simple diffusion and effectively transforms to a probe of GSH conjugate (circle-SG) in situ. The probe is extruded by carrier-mediated efflux systems from the brain to the blood. The brain kinetics depends on both pro-probe and probe immediately after the intravenous injection (
Our previous study showed that a purine derivative labeled with 14C (half-life: 5,730 years), a specifically designed pro-probe, could enter the brain and rapidly transform to its GSH conjugate in situ after intravenous injection into rats (Okamura et al, 2007). Furthermore, this GSH conjugate, S-(6-(7-methylpurinyl))glutathione (PSG), was found to be a good substrate for MRP1 using inside-out membrane vesicles. In this study, we confirmed whether 6-bromo-7-[14C]methylpurine ([14C]7m6BP) as a pro-probe can transform to [14C]PSG in Mrp1 knockout (KO) and wild-type (WT) mice. Next, 7m6BP was labeled with 11C (half-life: 20.4 min), a positron emitting nuclide. The brain kinetics of [11C]7m6BP in Mrp1 KO and WT mice were investigated using positron emission tomography (PET) as an external detector, which allows the noninvasive measurement of the in vivo tissue concentration of a radiolabeled compound as a function of time and also the assessment of physiologic and biochemical functions in the living body (Namba et al, 1999; Reivich et al, 1979).
Materials and methods
General
All commercially available starting materials and solvents were of reagent grade or better and were used without further purification. [14C]Methyl iodide (specific activity: 2.15 GBq/mmol) was purchased from Amersham Biosciences Corp. (Piscataway, NJ, USA). Thin layer chromatography (TLC) plates (silica gel 60 F254) were purchased from Merck Ltd (Tokyo, Japan). Proton nuclear magnetic resonance (1H-NMR) spectroscopy was performed using a JEOL JNM AL-300 (300 MHz) spectrometer (JEOL Ltd, Tokyo, Japan) with chemical shifts reported in units of parts per million (p.p.m.). High-resolution mass spectrometry (HRMS) was performed on a JEOL JMS-SX102 spectrometer (JEOL Ltd). The relative radioactivity of compounds on TLC was quantified using an imaging phosphor plate system (BAS 1800 system; Fuji Photo Film Co., Tokyo, Japan).
Synthesis
S-(6-(7-methylpurinyl))glutathione: This compound was prepared from 6-chloro-7-methylpurine with GSH in the same manner as described previously (Okamura et al, 2007). 1H-NMR (δ, D2O): 1.92 (2H, br), 2.27 (2H, br), 3.50 (1H, br ov), 3.58 (1H, br ov), 3.72 (2H, s), 3.84 (1H, dd, J=3.31, 14.3), 3.93 (3H, s), 4.70 (1H, br), 8.17 (1H, s), 8.53 (1H, s); HRMS (m/z): [M+H]+ calcd (found) for C16H22O6N7S, 440.1352 (440.1352).
6-Bromo-7-methylpurine and 6-bromo-7-[14C]methylpurine: Using 6-bromopurine as the starting material in place of 6-chloropurine, these compounds were synthesized in the same manner as described previously (Okamura et al, 2007). 6-Bromo-7-methylpurine yield: 6%; 1H-NMR (δ, CDCl3): 4.17 (3H, s, NCH3), 8.21 (1H, s, H-8), 8.81 (1H, s, H-2); HRMS (m/z): [M+H]+ calcd (found) for C6H6N4Br, 212.9776 (212.9814) and 214.9756 (214.9795). Radiochemical yield for 6-bromo-7-[14C]methylpurine was 10% to 20%. Radiochemical purity (greater than 97%) was determined by TLC using chloroform/ethanol (9:1; v/v) and ethyl acetate/ethanol (9:1; v/v).
6-Bromo-7-[11C]methylpurine: Carbon-11 was generated by the 14N(p, α)11C nuclear reaction using a CYPRIS HM-18 cyclotron (Sumitomo Heavy Industries Ltd). Preparation of [11C]CH3I and subsequent 11C-methylations were performed automatically by using a synthetic apparatus for 11C-labeled compounds developed by Suzuki et al (1985). High-pressure liquid chromatography (HPLC) was performed using a JASCO HPLC system for radioactive runs. Effluent radioactivity from the HPLC was determined using a NaI (Tl) scintillation detector system. [11C]CH3I was trapped in anhydrous acetone (300 μL) containing 6-bromopurine (3 mg) and K2CO3 (1 mg) at −10°C. The reaction vessel was heated at 100°C for 5 mins, and then the solvent was removed in a stream of nitrogen. After an addition of ethyl acetate (1 mL), the radioactive mixture was transferred onto an HPLC column (COSMOSIL 5SL-II, 10 I.D. × 250 mm; Nacalai Tesque Inc., Kyoto, Japan) and eluted with AcOEt/EtOH (85/15) at a flow rate of 5 mL/min. A minor radioactive fraction having a retention time of 9.5 mins was collected in a flask. After evaporation of the solvents from the flask under reduced pressure, [11C]7m6BP was reconstituted in sterile saline (3 to 5 mL). By this procedure, 0.30 to 0.58 GBq of [11C]7m6BP was obtained as a saline solution in a total synthesis time of 30 mins after a 12 to 15 mins proton bombardment at a beam current of 15 μA. The radiochemical yield was 4% to 9% (decay corrected to end of bombardment). The identification and radiochemical purity of [11C]7m6BP obtained after the formulation were determined by HPLC and TLC using an authentic sample of 7m6BP. HPLC was performed on a COSMOSIL 5C18-AR-II (Nacalai Tesque Inc.) column (4.6 I.D. × 250 mm) with a mobile phase of H2O/MeOH (75:25; v/v) at a flow rate of 0.9 mL/min. The retention time of the authentic sample was 10.1 mins. TLC was developed with ethyl acetate/ethanol (9:1; v/v), and then the air-dried TLC plate was covered with a thin plastic film (lumilar film) and placed in a cassette in contact with an imaging phosphor plate for several hours. The radiochemical purities determined by HPLC and TLC were both greater than 95%. The specific activity of [11C]7m6BP was determined by comparison of UV absorbance with that of known standards to be 40 to 84 GBq/μmol at the end of synthesis.
Animals
Male FVB mice and Mrp1 KO mice were purchased from Clea Japan Inc. (Tokyo, Japan) and Taconic Farms Inc. (Germantown, NY, USA), respectively. Mice weighing 26 to 36 g (10 to 16 weeks) were used throughout this study and were allowed free access to food and water. In this study, animals were treated and handled according to the ‘Recommendations for Handling of Laboratory Animals for Biomedical Research’, compiled by the Committee on Safety and Ethical Handling Regulations for Laboratory Animal Experiment, NIRS.
Analysis of the Chemical form in the Brain of Wild-Type and Mrp1 Knockout Mice
Mice were decapitated at 15 and 60 mins after intravenous injection of [14C]7m6BP (333 kBq in 200 μL saline). The head was quickly dipped in liquid nitrogen and the brain was removed. The brain was homogenized in 67% ethanol in water and then centrifuged at 4°C. The supernatant was concentrated in a stream of nitrogen, followed by centrifugation and filtration through a 0.45 μm filter. The concentrated sample was analyzed using an HPLC system consisting of a model L-2130 pump (Hitachi High-Technologies Corporation, Tokyo, Japan), a model L-2420 UV-VIS absorbance detector set at 288 nm (Hitachi High-Technologies Corporation), and a COSMOSIL 5C18-AR-II column (4.6 I.D. × 250 mm; Nacalai Tesque Inc.). The column was eluted with a mobile phase of acetonitrile/water containing 0.1% trifluoroacetic acid (10:90; v/v) at a flow rate of 1.0 mL/min. The eluate from the column was collected in 0.5 mL fractions, and radioactivity in the fractions was measured by a liquid scintillation counter (LS 6000; Beckman Instruments, Fullerton, CA, USA). Both the recovery of radioactivity in HPLC elutions and the extraction efficiency of that to the supernatant were more than 95%.
Positron Emission Tomography Scanning Procedures
Thirteen mice (seven WT mice and six Mrp1 KO mice) were used for scanning. Body temperature was monitored using a rectal thermometer throughout PET scanning, and was maintained within the normal range using a heat lamp. Scans were performed with an Inveon Dedicated PET system (Siemens Medical Solutions, Knoxville, TN, USA), which has a transaxial field of view of 10 cm and an axial field of view of 12.7 cm. [11C]7m6BP (12 to 16 MBq) was administered as a 0.1 to 0.3 mL intravenous bolus injection to each mouse, which was maintained under isoflurane anesthesia during the scanning periods. Data were acquired by the animal tomograph for 60 mins after injection in 20 frames divided as follows: 4 × 1 mins, 8 × 2 mins, and 8 × 5 mins frames. Without attenuation correction, the data were reconstructed using Fourier re-binning and filtered back projection with a ramp filter cutoff at the Nyquist frequency into images with a 128 × 128 matrix size and 3 times zoom, to give a voxel size of 0.26 × 0.26 × 0.8 mm3. After image reconstruction, ellipsoidal regions of interest covering 14 slices were manually placed on a summed PET image (sagittal view of the brain) and transferred to all of the frames of images to generate time—radioactivity concentration (Bq/mL) curves for whole brain.
Efflux Rate Constant
Brain radioactivity (% injected dose/mL) during the period of 15 to 60 mins after injection versus time was fitted to the monoexponential function by linear least-squares regression to obtain the efflux rate constant (keff). The slope of the best fit was expressed as the efflux rate constant (h−1) ±s.e.
Effect of Substrate Dose on the First-Order Efflux Rate Constant
A single mouse (WT) was repeatedly used at weekly intervals. The mouse received the intravenous injection of [11C]7m6BP containing 0.36 (no carrier added), 3.6, 47, 230, 480, and 4,700 nmol of unlabeled 7m6BP dissolved in saline in the randomized order. PET scanning followed by the estimation of the efflux rates was performed as described above.
Statistical Analysis
Statistical differences in efflux rates between WT and Mrp1 KO mice (n=6 for each group) were analyzed using two-tailed Wilcoxon rank sum test, a type of nonparametric test. The level of significance was set at 0.01.
Results
Analysis of the Chemical form in the Brain of Wild-Type and Mrp1 Knockout Mice
The quantitative assessment of MRP1 function requires the delivery of a hydrophilic radiolabeled compound (MRP1 substrate) into the brain. To confirm that [14C]7m6BP was converted to [14C]PSG (GSH conjugate), the chemical form in the brain tissue was analyzed by HPLC at 15 and 60 mins after intravenous injection of [14C]7m6BP into WT and Mrp1 KO mice. The retention times for the authentic PSG and 7m6BP were 5.6 and 10.4 mins, respectively. The result clearly confirmed the complete conversion of [14C]7m6BP to [14C]PSG in the brain tissue of WT and Mrp1 KO mice (Figure 2).
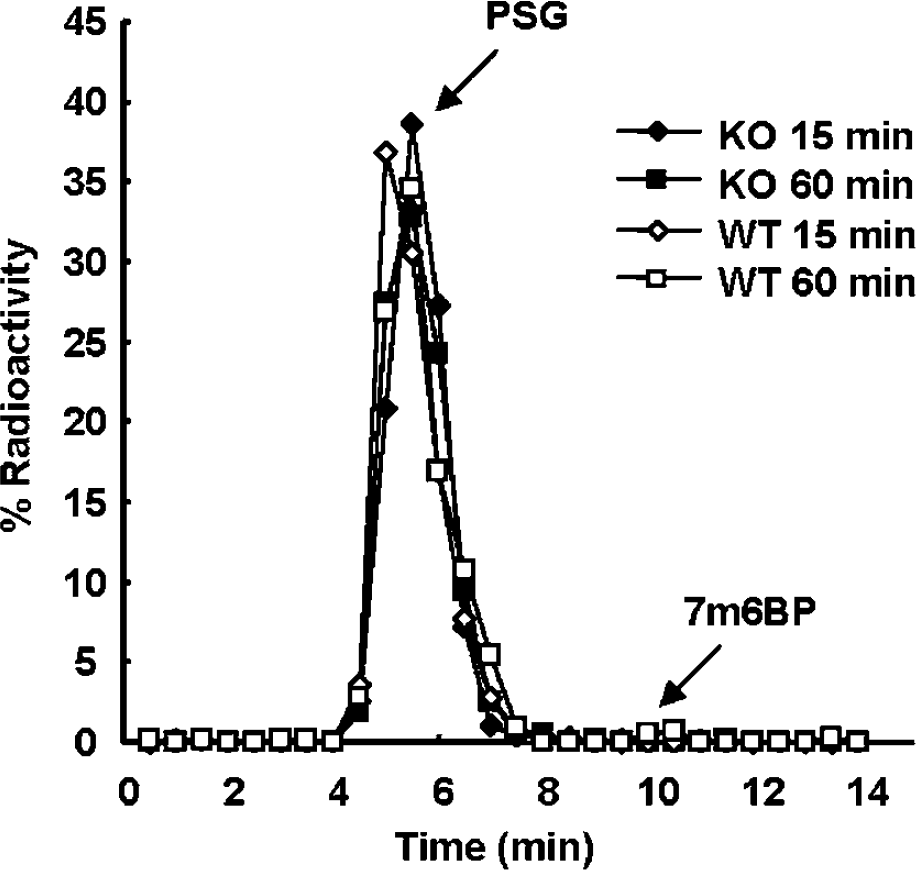
Analysis of chemical form. HPLC analysis of brain tissue extract at 15 (diamond) and 60 (square) mins after the intravenous injection of [14C]7m6BP into Mrp1 KO (solid) and WT (open) mice. Data are shown from a single mouse for each time point and each group.
Brain Kinetics After [11C]7m6BP Injection
Figure 3A shows typical time—radioactivity curves determined by animal PET after the intravenous injection of [11C]7m6BP into WT and Mrp1 KO mice. Brain radioactivity in both mice reached a maximum level at 1.5 mins after injection. However, quite different kinetics between WT and Mrp1 KO mice were observed thereafter; brain radioactivity was rapidly cleared in WT mice whereas it was almost constant up to 60 mins in Mrp1 KO mice. Brain radioactivity during the period of 15 to 60 mins was plotted in a semilog scale (Figure 3B inset). As a result, the semilog plot exhibited linearity (each correlation coefficient: 0.99).
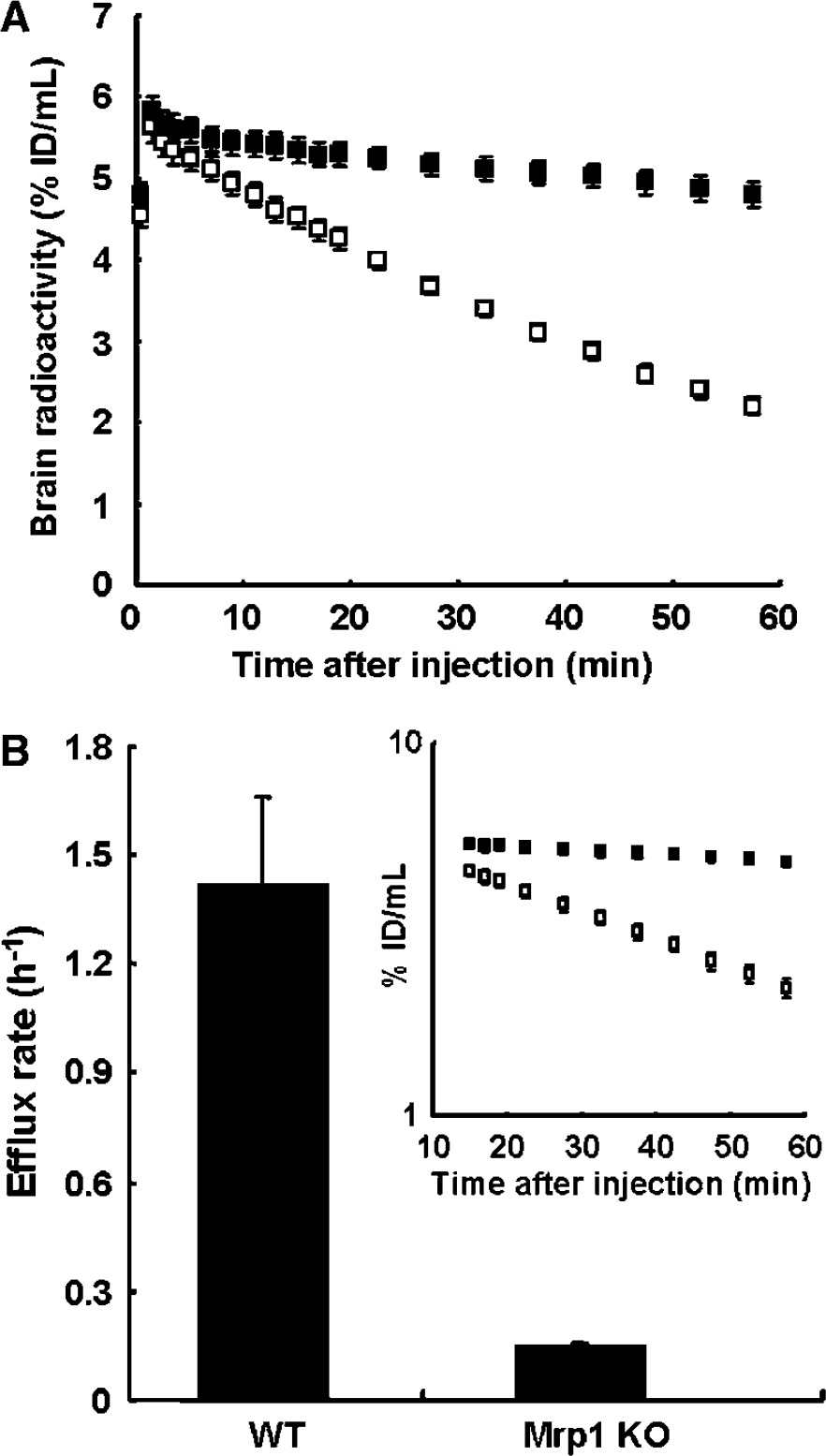
Time—radioactivity curves in the brain obtained by animal PET after intravenous injection of [11C]7m6BP into Mrp1 KO (solid symbols) and WT mice (open symbols) (
Efflux Rate Estimated from the Brain Kinetics of Wild-Type and Mrp1 Knockout Mice
The precise efflux rates of [11C]PSG could be estimated from the slopes. The coefficients of variation (s.e. divided by mean) of individual efflux rates in WT and Mrp1 KO mice were calculated to be less than 1.0% and 3.2% to 5.7%, respectively. The efflux rates (h−1) in WT and Mrp1 KO mice were 1.4±0.24 and 0.15±0.010 (mean±s.d.), respectively. Knockout of the Mrp1 gene caused a significant reduction in the efflux rate of PSG (Wilcoxon rank sum test, W=21, α=0.01).
Effect of Substrate dose on the First-Order Efflux Rate Constant
The efflux rate constant should not be affected by the injection dose as long as the first-order condition is satisfied. To confirm this, we investigated the effect of the substrate dose on the efflux rate (Figure 4). The efflux rates remained nearly constant (approximately 1.4 h−1) in the range of 0.36 nmol (no carrier added) to 47 nmol, and decreased at high doses (230 to 4,700 nmol).
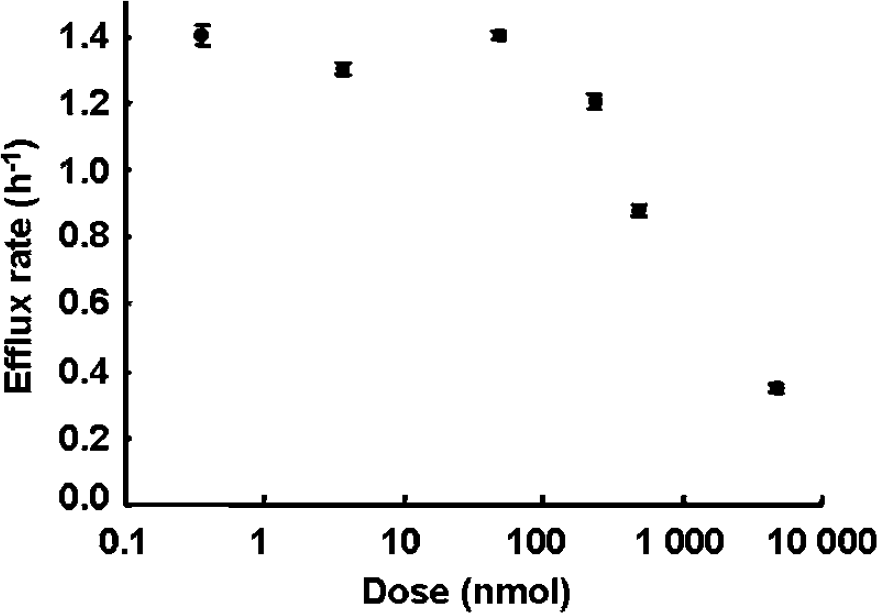
Effect of substrate dose on the first-order efflux rate constant. Each efflux rate are shown from the same single mouse that received the intravenous injection of [11C]7m6BP containing 0.36, 3.6, 47, 230, 480, and 4,700 nmol of unlabeled 7m6BP. Values are given as the mean±s.e.
Discussion
The concept of MEM includes three major requirements of the pro-probe: (1) it readily enters the brain, (2) efficiently transforms to the hydrophilic probe in situ, and (3) the generated probe specifically undergoes the export by a target transporter. Our previous study showed that 6-chloro-7-[14C]methylpurine satisfied the two requirements of the pro-probe and that MRP1 mediated the transport of PSG (probe) in vitro (Okamura et al, 2007). In this study, 7m6BP was applied as a new pro-probe; the reaction rate with GSH is higher than that of 6-chloro-7-methylpurine in the brain homogenate of WT mice (data not shown). This is a desirable property for MEM, and 7m6BP was expected to transform into PSG in the brain of Mrp1 KO and WT mice. Indeed, it was shown that [14C]7m6BP was rapidly converted to [14C]PSG in the mouse brain. Brain radioactive compounds were nearly completely composed of PSG from 15 mins after the intravenous injection. For noninvasive and external measurement, 7m6BP was radiolabeled with 11C-nuclide by methylation with 11CH3I of 6-bromopurine. This provided a sufficient amount of [11C]7m6BP for animal PET studies, although the radiochemical yield was not high because the methylation mainly produced an isomer with 11C-methyl at the C-9 position of the purine ring.
We have shown for the first time that PET using the pro-probe of [11C]7m6BP can noninvasively assess the efflux transport of the GSH conjugate out of the brain of living mice. According to the MEM concept, the brain radioactivity comprises the mixture of [11C]7m6BP and [11C]PSG in the early phase after injection of [11C]7m6BP into mice and gradually changes to [11C]PSG alone. Analysis of the chemical form confirmed that the time—radioactivity curve reflects the kinetics of [11C]PSG alone from 15 mins after injection. Moreover, linearity was obtained by the semilog plot of brain radioactivity during the period of 15 to 60 mins, showing that [11C]PSG was monoexponentially extruded from the brain. Theoretically, the first-order rate constant is independent of the substrate concentration. The coadministration of unlabeled 7m6BP provided no change of the efflux rate constants up to approximately 100-fold greater than the amount of no carrier added (Figure 4). The [11C]PSG efflux follows the first-order kinetics under the condition of trace amount (less than 47 nmol). Therefore, we can exclude the effect on the efflux rate in our experiment caused by dose variability because of differences in a specific activity. The decreased rate of efflux at higher doses might be caused by the repeated administration of 7m6BP and its measurement in a single mouse; however, the drug dose was randomly injected into the mouse. In addition, we confirmed the reproducibility in another mouse at the highest dose alone (data not shown). Therefore, the decrease in the efflux rate at higher doses shows the alteration from the first-order kinetics to the zero-order kinetics, but not a cumulative toxic effect. In Mrp1 KO mice, the efflux rate markedly decreased as compared with that of WT as shown in Figure 3B. This provides evidence that Mrp1 specifically pumped [11C]PSG out of the brain in vivo. From these results, brain Mrp1 function can be noninvasively assessed by estimating the washout rate of brain radioactivity, i.e., the efflux rate of the GSH conjugate, using PET and [11C]7m6BP.
There seems to be a difference in the efflux pathway from the brain to the blood across the BBB between BEI and MEM, because BEI deposits a radiolabeled probe in the extracellular space whereas MEM appears to generate it in the intracellular space. Considering that the volume of BCEC is very small (approximately 0.8 μL/g brain; Gjedde and Christensen, 1984), [11C]7m6BP rapidly equilibrates between BCEC and blood after intravenous injection, and then it can diffuse into the large compartment of the brain tissue according to a concentration gradient. In addition, Ghersi-Egea et al (1994) reported that GST activity was 0.36 nmol/min/mg of protein in the cerebral cortex whereas it was not detectable (<0.05 nmol/min/mg) in the microvessel preparation. Cytosolic GST activity in cultured glial cells is much higher than that in granule cells or in the brain (Huang and Philbert, 1995; Sagara and Sugita, 2001). Given these findings, it seems likely that [11C]PSG formation may occur within BCEC, and neurons, and primarily in glial cells. Hence, [11C]PSG should cross an additional membrane to be eliminated from the brain tissue compared with BEI. RT-PCR analysis revealed the expression of Mrp1 and other Mrp family members in WT FVB mouse astroglia-rich cultures (Minich et al, 2006). Mrp1 mRNA transcript was also detected in the primary cultures of embryonic rat brain neurons (Hirrlinger et al, 2002). MRP1 on parenchyma cells as well as BCEC may significantly contribute to [11C]PSG efflux out of the brain. As shown in Figure 5, PSG generated within brain parenchyma cells must cross at least three membranes to be eliminated from brain to blood across the BBB: parenchyma cell membrane (the first step), abluminal and luminal membranes of BCEC (the second and third steps). Mrp1 may take part in the first and third step, the efflux process. In this study, the carrier proteins involved in the second step of the uptake process remain unclarified; however, these might include organic anion transporters and organic anion transporting polypeptides (Kusuhara and Sugiyama, 2005).
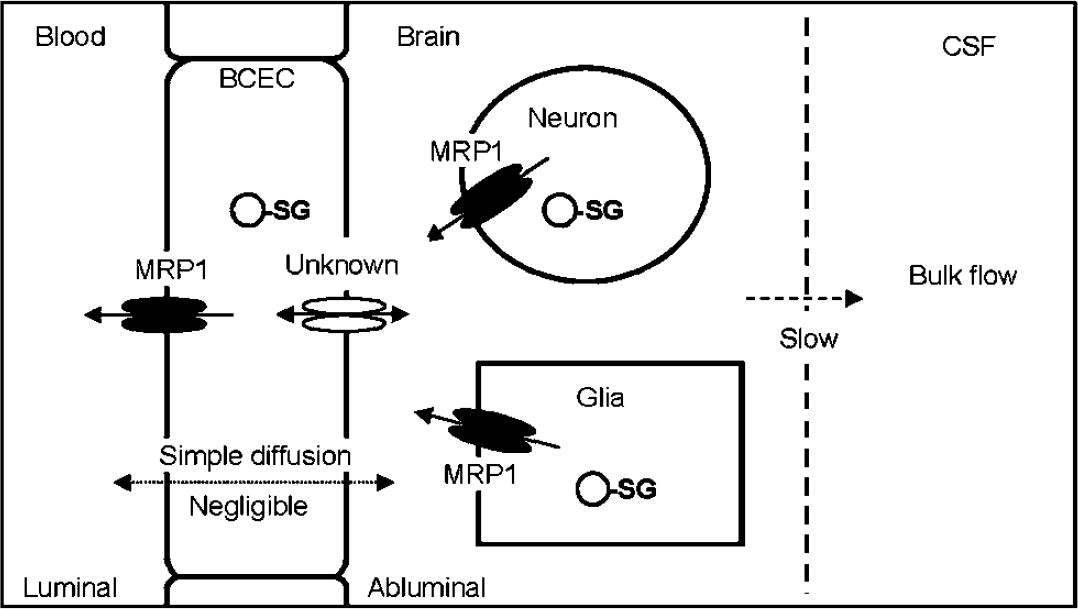
Putative pathway and mechanism for the efflux transport of PSG. PSG (circle-SG) will be generated within glial cells, neuron, or BCEC. Based on our previous and present studies, the efflux pathway of PSG via simple diffusion is negligible, and the elimination by bulk flow is relatively small. PSG formed within parenchyma cells is first transported to the extracellular space by MRP1, and then taken up into BCEC by as yet unknown transporter. Finally, PSG is extruded into the blood by MRP1 at the luminal side of BCEC. This diagram does not represent other transporters although these are possibly involved in PSG efflux out of the brain.
Furthermore, we must consider other efflux routes for PSG out of the brain in addition to the BBB. The efflux of a solute from the brain (keff) generally includes a number of pathways and mechanisms. Based on previous reports (Cserr et al, 1981; Groothuis et al, 2007), keff can be considered to be the sum of three parallel conductances; kp for the diffusional efflux across the BBB, kcsf for the convective efflux into cerebrospinal fluid, and kx for the carrier-mediated efflux: keff=kp+kcsf+kx. Brain radioactivity during the period of 1 to 60 mins was extremely low after intravenous injection of 35S-labeled PSG into rats (Okamura et al, 2007). Furthermore, PSG was almost completely retained in the brain of Mrp1 KO mice (Figure 3A). These results indicate that PSG hardly crosses membranes in the brain via simple diffusion; therefore, the total efflux rate of PSG can be approximately equal to be the sum of convective efflux and carrier-mediated efflux (keff≈kcsf+kx). For the value of kcsf, Groothuis et al (2007) assumed a value of 0.3 h−1, which is the average of the efflux rate for nontransported, nonmetabolized, water-soluble compounds. The total efflux of PSG (0.15 h−1) in Mrp1 KO mice was lower than the convective efflux (0.3 h−1), although whether kcsf of mice is equivalent to that of rats remains unknown. From this finding, it may be inferred that [11C]PSG is almost completely trapped within brain parenchyma cells because of knockout of Mrp1 gene; subsequently, the convective flow only modestly contributes to the value of keff.
There are currently two recognized methods for assessing in vivo active efflux transporters in the brain. One is the BEI method that requires the intracerebral injection of a radioactive substrate for target efflux transporters (Kakee et al, 1996). Brain efflux index allows us to simply estimate the carrier-mediated transport rate constant for the substrate out of the brain, which is a quantitative index of efflux transporter function. However, BEI is limited to animal studies because of invasiveness and also the need for a large number of animals. The other is a noninvasive method based on intravenous injection of a lipophilic radioactive substrate for efflux transporters, a diffusible probe. The diffusible probe enters the brain across the BBB by simple diffusion, and subsequently migrates from the brain to the blood via two routes of back-diffusion and carrier-mediated transport. Usually, this method enables us to determine the ratio of influx rate to efflux rate defined as the distribution volume, which is useful for the qualitative assessment of efflux transporter function. It has been reported that the distribution volume of [11C]verapamil reflects the in vivo modulation of P-glycoprotein, an active transporter (Bart et al, 2003). In the case of a diffusible probe, however, the efflux rate depends on not only carrier-mediated transport but also back-diffusion rate. Even if the arterial probe concentration is monitored, kinetic analyses cannot separate these rates, thereby compromising the quantitative assessment. Meanwhile, MEM is based on the intravenous injection of a lipophilic ‘pro-probe’ that can be converted to a hydrophilic probe in the brain. MEM possesses advantages of both BEI and the noninvasive method described above. Furthermore, MEM requires no blood sampling (input function) because the efflux rate can be estimated from brain kinetics after the complete conversion of the pro-probe to the probe.
Several applications are possible because [11C]7m6BP allows the repeated assessment of brain MRP1 function in a single individual. The inhibition of efflux pumps such as P-glycoprotein and MRP1 by inhibitors has been recognized to be a useful strategy for the delivery of a sufficient amount of a therapeutic drug into the brain (Loscher and Potschka, 2005a, 2005b). Because MRP1 accepts anticancer and anti-HIV drugs as a substrate, this pump might be involved in limiting the entry of these therapeutic drugs into the brain (Loscher and Potschka, 2005a). [11C]7m6BP and PET will be appropriate for the estimation of the dose required to modulate MRP1 in vivo, thereby avoiding side effects and enhancing the therapeutic efficacy. Interestingly, a slight increase in MRP1 expression was observed in frozen hippocampal samples from patients with Alzheimer's disease (Sultana and Butterfield, 2004). The samples also showed a concomitant increase in adducts of MRP1 bound to 4-hydroxy-2-trans-nonenal. [11C]7m6BP, or a similar such probe that could be used in humans, could perhaps detect an alteration of MRP1 function in neurodegenerative disorders because it enables the precise estimation of MRP1 function. MRP1 is involved in protecting not only the brain but also peripheral tissues from xenobiotics and endogenous metabolites (Leslie et al, 2005). Given the nature and properties of [11C]7m6BP, it might become possible to apply [11C]7m6BP to assessment of MRP1 function in peripheral tissues, for instances, tumor tissue in which overexpression of MRP1 is well known for one of the factors that cause drug resistance (Deeley et al, 2006).
In conclusion, this study has shown that MEM with [11C]7m6BP is a powerful tool to noninvasively and quantitatively assess brain efflux systems, especially Mrp1 function. As PSG probably traces the efflux route similar to that of endogenous and exogenous GSH conjugates generated in the brain, [11C]7m6BP may facilitate evaluation of the true effect of a therapeutic drug on MRP1 and may also provide new insight into the physiologic role of the efflux systems for GSH conjugates out of the brain.
Footnotes
Acknowledgements
The authors are grateful to cyclotron crew of the National Institute Radiological Sciences for technical support in radioisotope production.
The authors state no conflict of interest.
