Abstract
Functional recovery after cerebral ischemia is mediated by the regeneration of vascular networks and the restoration of synaptic architecture. Netrins have been implicated in neuronal pathfinding and angiogenesis. In this study, we investigated the expression of Netrin-4 and its putative receptors, deleted in colorectal cancer (DCC), Unc5A, and Unc5B after distal middle cerebral artery occlusion in mice. Netrin-4 protein was also administered intracerebroventricularly to examine its effect on angiogenesis and behavioral recovery. Netrin-4 protein was highly upregulated in the ischemic core as soon as 1 day after cerebral ischemia, with subsequent downregulation after 1 week. Its expression was limited to the area of blood—brain barrier damage and was seen on both blood vessels and astrocytic foot processes. Although there was not a significant upregulation of the putative Netrin-4 receptor Unc5A and Unc5B, there was a significant increase in expression of the DCC receptor on neuronal processes in the peri-infarct cortex. Intracerebroventricular administration of Netrin-4 into the ischemic brain increased blood vessel density, endothelial proliferation, and improved behavioral recovery at 1 week after stroke, but did not have an effect on blood—brain barrier permeability or infarct size. These findings suggest that Netrin-4 may improve poststroke functional recovery by enhancing blood vessel proliferation.
Introduction
Functional recovery after cerebral ischemia is governed by concerted mechanisms that promote the reorganization of neural and vascular circuits. Through a series of stages, connectivity among surviving neurons is redefined, which results in a form of cortical remapping that can compensate for lost function. This plastic process stems primarily from the regeneration of vascular networks, the induction of axonal outgrowth, and the restoration of synaptic architecture (Nudo, 2006). Several studies recently have provided strong evidence for an intertwined relationship between axon guidance and angiogenesis (Vogel, 2005; Weinstein, 2005). It has been demonstrated that several guidance molecules, which are secreted to provide attractive and repulsive signals during axonal outgrowth, play a similar role in the development of blood vessels (Vogel, 2005; Weinstein, 2005). The Netrin family of chemoattractants is one such example. The Netrins were first identified as extracellular factors secreted from the floor plate that attract spinal commissural axons toward the midline (Serafini et al, 1994). In mammals, the Netrin family includes Netrin-1, Netrin-3, Netrin-4, and the Netrin-related molecules, Netrin-G1 and Netrin-G2 (Wilson et al, 2006). Although Netrin-1 has been reported to increase after ischemic injury in rats subjected to transient middle cerebral artery occlusion (Tsuchiya et al, 2007), the role of Netrin-4 after ischemic injury is not known.
Mouse Netrin-4 is so named because its predicted size and secondary structure are highly similar to the other Netrins in mammals. During embryonic development, Netrin-4 is expressed widely in the brain, the spinal cord, especially the floor plate, and components of the peripheral nervous system, as well as other organs (Yin et al, 2000). In adult mice, Netrin-4 is expressed in both neural and nonneural tissue, with prominent expression seen in the kidney and the lung (Yin et al, 2000). In the adult brain, Netrin-4′s transcript is seen in granule cells of the cerebellum, pyramidal, and dentate neurons of the hippocampus, and cortical neurons (Yin et al, 2000). Although the function of Netrin-4 remains poorly understood, it has been shown to promote angiogenesis in animal models of diabetes and hindlimb ischemia (Wilson et al, 2006). Motivated by these findings, in the present study, we examined the expression pattern of Netrin-4, and its putative receptors, deleted in colorectal cancer (DCC), Unc5A, and Unc5B, and investigated whether intraventricular administration of Netrin-4 enhances angiogenesis and neurological outcome after cerebral ischemia. We report that Netrin-4 was upregulated in the ischemic cortex and its administration enhanced angiogenesis and behavioral outcome in a mouse model of cerebral ischemia.
Materials and methods
Focal Cerebral Ischemia
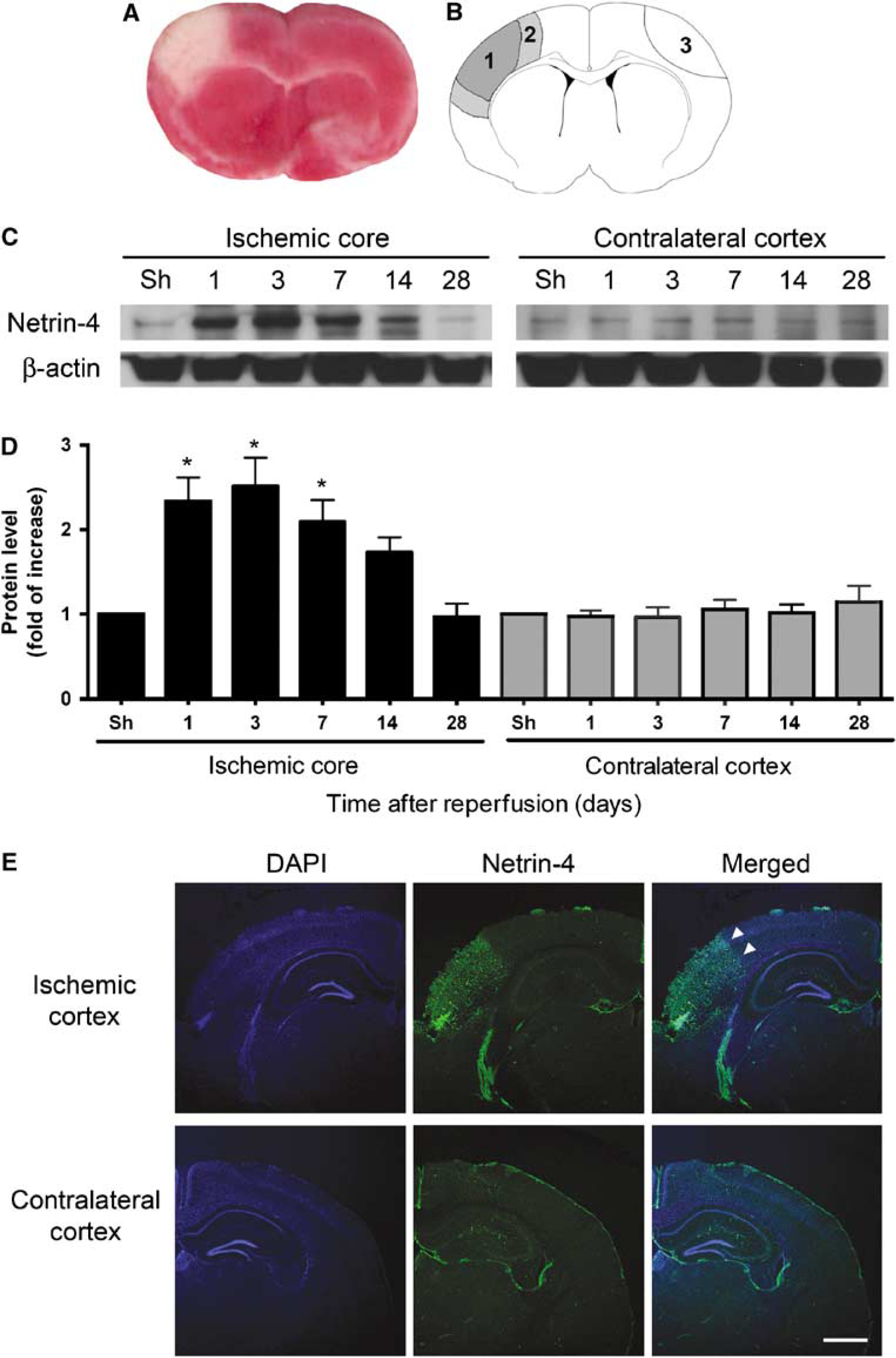
All experimental protocols were in accordance with the National Institutes of Health Guide for the Care and Use of Laboratory Animals and were approved by the Stanford University Administrative Panel on Laboratory Animal Care. Focal cerebral ischemia was induced in adult male C57BL/6J mice (30 to 35 g). Anesthesia was induced with 5% isoflurane and maintained with 1.5% to 2% isoflurane during surgery. Core body temperature was monitored with a rectal probe and maintained at 37°C with a heating pad. The left distal middle cerebral artery (dMCA) was exposed through a craniotomy and permanently cauterized above the rhinal fissure. The common carotid arteries were transiently occluded bilaterally for 20 mins and then released. This model of ischemia produces a focal infarct generally localized to the cortex (Figure 1A) (Carmichael, 2005). Sham-operated mice underwent identical surgery except that the dMCA and the common carotid arteries were not occluded.
Netrin-4 protein was upregulated in the ischemic core after cerebral ischemia. (
Western Blot
Animals were allowed to survive for 1 day, 3 days, 1 week, 2 weeks, and 4 weeks after reperfusion and killed with an overdose of isoflurane, followed by transcardial perfusion with phosphate-buffered saline (PBS). The brains were removed and the tissue corresponding to the ischemic core, peri-infarct cortex, and homologous contralateral cortex was dissected for Western blot. The ischemic core was defined as the central area of infarction, which was visibly discolored compared to the uninjured area. The periinfarct cortex was defined as tissue immediately adjacent to the infarct core in the ipsilesional cortex. Tissue from the homologous contralesional cortex was also isolated (Figure 1B). The corresponding regions from sham-operated mice were also dissected as controls. Brain tissue was homogenized in lysis buffer (9803; Cell Signaling Technology, Boston, MA, USA) and protease inhibitors (P8340; Sigma, St Louis, MO, USA) with pestle grinders (03-392-106; Fisher Scientific, Pittsburgh, PA, USA). The homogenate was centrifuged at 10,000g for 15 mins at 4°C and the supernatant was used for protein analysis. Protein concentration was determined with the BCA protein assay (23227; Pierce, Rockford, IL, USA).
Sodium dodecyl sulfate—polyacrylamide gel electrophoresis was performed as previously described (Zhao et al, 2005). Protein (20 µg) was run in each lane of a 4% to 20% Ready Gel (161 to 1105; Bio-Rad, Hercules, CA, USA) for 90 mins at 120V along with a size marker (161-0376; Bio-Rad). The protein on the gel was then transferred to a polyvinylidene fluoride membrane (RPN303F; Amersham, Piscataway, NJ, USA) and blocked in 5% milk (170 to 6404; Bio-Rad) to prevent nonspecific binding. Primary antibody was incubated overnight at 4°C: mouse monoclonal anti-human Netrin-4 antibody (1:200 dilution; 70 kDa; MAB1254; R&D Systems, Minneapolis, MN, USA); mouse anti-human DCC monoclonal antibody (1:200 dilution; 190 kDa; 554223; BD Pharmingen, San Jose, CA, USA); rabbit polyclonal anti-Unc5A (1:200 dilution; 98 kDa; 07-657; Upstate, Lake Placid, NY, USA); mouse monoclonal anti-rat Unc5B antibody (1:200 dilution; 90 kDa; MAB1006; R&D Systems). After washing, the appropriate horseradish peroxidase-conjugated secondary antibody was then incubated at room temperature for 1 h: horseradish peroxidase-goat anti-mouse IgG conjugate (1:200; 81 to 6,520; Invitrogen, Carlsbad, CA, USA), horseradish peroxidase-goat anti-rabbit IgG conjugate (1:200; 81 to 6120; Invitrogen). The signal was then detected with a chemiluminescence system (RPN2106; Amersham) and developed on Hyperfilm (RPN1677K; Amersham). To confirm even loading of proteins, membranes were stripped (21059; Pierce) and probed for β-actin (1:20,000 dilution; 42 kDa; A3854; Sigma). Films were scanned with a photoscanner and Image J (US National Institute of Health, Bethesda, MD, USA) was used for data analysis. The level of protein in each sample was normalized to the level of β-actin for that particular sample, and the normalized level at different time points was compared with the normalized level of sham-operated animals.
Tissue Preparation and Immunofluorescence Staining
At various time points after stroke, animals were killed and perfused transcardially with PBS (pH 7.4), followed by 4% paraformaldehyde in PBS (pH 7.4). The brains were removed and postfixed with 4% paraformaldehyde in PBS overnight at 4°C and then with 20% sucrose, 4% paraformaldehyde in PBS. Coronal brain sections of 40 µm in thickness were cut on a vibratome and stored in antifreeze solutions containing 30% ethylene glycol and 30% glycerol at −20°C.
Free-floating immunohistochemistry was used to localize the expression patterns of Netrin-4, DCC, Unc5A, and Unc5B. Slices were washed in PBS and blocked in 3% ormal goat serum (005-000-001; Jackson Immuno, West Grove, PA, USA) and 0.1% Triton X-100 (T8787; Sigma). Slices were incubated in primary antibody overnight at 4°C. The primary antibodies were as follows: goat anti-mouse Netrin-4 antibody (1:200 dilution; AF1132; R&D Systems), goat anti-mouse DCC antibody (1:200 dilution; AF844; R&D Systems), goat anti-rat Unc5A antibody (1:100 dilution; AF1405; R&D Systems), and goat anti-rat Unc5B antibody (1:100 dilution; AF1006; R&D Systems). To colocalize the expression of the above proteins with specific cellular makers, the following primary antibodies were used concurrently: biotinylated Lycopersicon esculentum lectin (1:200 dilution; B-1175; Vector Laboratories, Burlingame, CA, USA) for blood vessels, mouse antiβ- tubulin III (1:200 dilution; MMS-435P; Covance, Princeton, NJ, USA) or mouse anti-NeuN (1:200 dilution; MAB377; Chemicon, Temecula, CA, USA) for neurons, and guinea pig polyclonal antibody to human glial fibrillary acidic protein (GFAP, 1:500 dilution; 31223; Advanced Immunochemical, Long Beach, CA, USA) for astrocytes. After washing, appropriate secondary antibodies were then applied for 3 h at room temperature: Fluorescein (FITC) Donkey Anti-Goat IgG (1:500 dilution; 705-095-147; Jackson Immuno), Cy3 Donkey Anti-Mouse IgG (1:500 dilution; 715-165-150; Jackson Immuno), Cy3 Donkey Anti-Guinea Pig IgG (1:500 dilution; 706-165-148; Jackson Immuno), and Cy3 Streptavidin (1:500 dilution; 016-160-084; Jackson Immuno). Sections were also counterstained with 4′,6-diamidino-2-phenylindole dihydrochloride (DAPI) for 4 mins to detect cell nuclei (1:2,000; 268298; Calbiochem, San Diego, CA, USA).
Negative controls were routinely performed with each staining experiment by omitting the primary antibody. Sections were then mounted and examined under a confocal laser-scanning microscope (LSM510; Carl Zeiss, Oberkochen, Germany).
Netrin-4 Administration and BrdU Injection
To determine the effect of exogenous Netrin-4 on angiogenesis and behavioral outcome after stroke, Netrin-4 was infused into the lateral ventricle after induction of ischemia. At 1 day after reperfusion, mice were anesthetized with isoflurane and implanted with an osmotic minipump (1003D; 0000289; Durect, Cupertino, CA, USA). This pump model can hold a volume of 100 µL and can infuse solutions for 3 days at a rate of approximately 1 µL/h. The pump was filled with 100 µL of recombinant mouse Netrin-4 protein at a concentration of 25 µg/mL (1132-N4/CF; R&D Systems) in artificial cerebrospinal fluid (aCSF) that consists of 128 mmol/L NaCl, 2.5 mmol/L KCl, 0.95 mmol/L CaCl2, and 1.99 mmol/L MgCl2 (Sun et al, 2003). This formulation of the recombinant protein was free of carrier proteins. The cannula was implanted into the left lateral ventricle at 0.9 mm to the left of the midline, 0.1 mm posterior to the bregma, and 3 mm below the top of the skull (Kaya et al, 2005). Experimental animals (n = 5) were thus administered Netrin-4 (25 µg/mL) into the left lateral ventricle for 3 days, at 1 µL/h, beginning 1 day after reperfusion. This concentration of Netrin-4 was chosen because it had been shown that equivalent molar concentration of Netrin-4 has similar efficacy to vascular endothelial growth factor (VEGF) in inducing endothelial cell proliferation in vitro (Wilson et al, 2006). The equivalent molar concentration of Netrin-4 to VEGF in mass is approximately 4 to 1 (50 ng/mL Netrin-4 or 13 ng/mL VEGF). Because other groups have showed that 10 µg/mL of VEGF was sufficient to observe an angiogenic effect after ischemia by intracerebroventricular administration in vivo (Sun et al, 2003; Wang et al, 2006), we used 25 µg/mL of Netrin-4 in our experiments. This concentration is approximately half the molar concentration of that used with VEGF. Control animals (n = 5) were administered aCSF alone for the same length of time. At 1 day after reperfusion, 5-bromodeoxyuridine (BrdU, 50 mg/kg in 0.9% saline, i.p.; B9285; Sigma) was also administered to both groups once per day for 6 days. Animals were killed 7 days after induction of ischemia and used for immunohistochemical analysis.
Neurologic Assessment
Neurologic assessment of mice administered Netrin-4 or aCSF was performed 1 day before stroke and 7 days after stroke, which was immediately before they were killed for histologic evaluation, by an observer blind to the experimental conditions. A behavioral rating scale that evaluates the animals for motor function, sensory perception, and reflex was used (Li et al, 2000). The scale was graded from 0 to 14, with 0 being the normal score attained by animals tested before stroke and 14 indicating the possible maximum deficit. Animal tested at baseline did now show any deficit and received a score of 0.
Quantification of Blood Vessel Density and Endothelial Cell Proliferation
To compare blood vessel density between Netrin-4-treated and aCSF-treated control groups, free-floating immunohistochemistry was used. Sections were incubated with biotinylated L esculentum lectin (1:200 dilution; B-1175; Vector Laboratories) and followed with Cy3 streptavidin (1:500 dilution; 016-160-084; Jackson Immuno) and counterstained with DAPI (1:1,000; 268298; Calbiochem). After treatment with 2mol/L HCl for 30 mins at 37°C, sections were then incubated with rat anti-BrdU (1:500; OBT0030CX; Accurate Chemical, Westbury, NY, USA) and followed with Fluorescein (FITC) Donkey Anti-Rat IgG (1:500; 712-095-153; Jackson Immuno). Sections were then mounted for analysis.
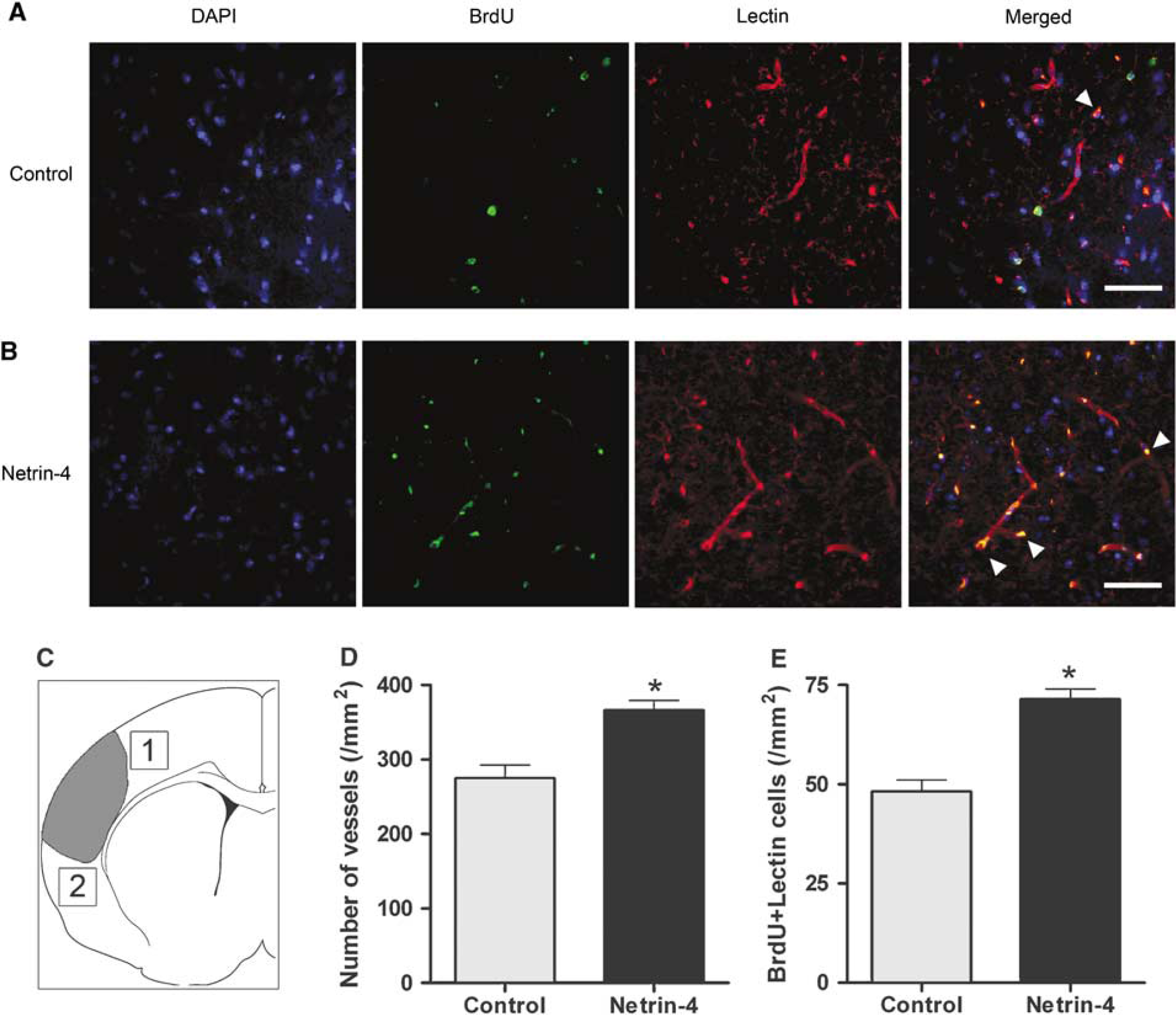
A total of 8 coronal sections (40 µm thick) spaced 160µm apart from each animal (n = 5 per group) were used for analysis. Each section was examined under a confocal microscope, and 3 nonoverlapping high-power fields at ×40 magnification were imaged from each dorsal and ventral cortical peri-infarct cortex area by an investigator blind to the experimental condition (Figure 4C). A total of six high-power fields were thus imaged from each section. An investigator blind to the experimental conditions then counted the number of lectin-positive blood vessels and the number of lectin-positive blood vessels that also colocalized with BrdU in each imaged high-power field. In the case that a blood vessel gives off multiple branches, each branch was counted as one additional vessel. The number of lectin-positive and lectin/BrdU-positive vessels was divided by the total area sampled to give an estimate of density.
Netrin-4 colocalized with blood vessels in the ischemic core. (
Netrin-4 colocalized with astrocytic endfeet in the peri-infarct cortex border. (
Intracerebroventricular administration of Netrin-4 enhanced angiogenesis. (
Quantification of Infarct Size
To visualize the area of infarction, triphenyltetrazolium chloride staining was performed at 24 h (Figure 1A). To compare infarct size between control and Netrin-4-treated mice, cresyl violet staining was performed at 1 week after dMCA occlusion (dMCAO). Eight coronal sections (40 µm thick) spaced 160 µm apart from each animal (n = 5 per group) were used for analysis. The StereoInvestigator software (Stereo Investigator, Williston, ME, USA) was used to draw contours around the intact ipsilesional cortex and the contralesional cortex under −10 magnification to determine the area of each region by an investigator blind to the experimental conditions. The infarct core can be distinguished as a dystrophic, hypercellular area. To correct for edema, the area of infarction was expressed as the percentage of the intact contralesional cortex and was determined according to the following formula: (1–(area of intact ipsilesional cortex)/(area of the contralesional cortex)). Infarct volume was calculated by multiplying the area of infarction with the section thickness and the total number of sections for each animal.
Quantification of Blood—Brain Barrier Leakage
To visualize the area of blood—brain barrier (BBB) leakage, 4% Evans blue (E2129; Sigma) in 0.9% saline was administered via an Osmotic minimump (1003D; 0000289; Durect) that was implanted intraperitoneally for 3 days after dMCAO. The Evans blue azo dye binds tightly to albumin and appears red under rhodamine optics (Li et al, 2003). After tissue processing and fixing as described above, sections were stained with Netrin-4 antibody (1:200 dilution; AF1132; R&D Systems), followed by Fluorescein (FITC) Donkey Anti-Goat IgG (1:500 dilution; 705-095-147; Jackson Immuno) and DAPI (1:2,000; 268298; Calbiochem). Colocalization of Netrin-4 and Evans blue was examined under a confocal microscope (Figure 3C).
To compare the volume of blood—brain barrier leakage between control and Netrin-4-treated mice, albumin staining was performed at 1 week after dMCAO. Eight coronal sections (40-µm thick) spaced 160 µm apart from each animal (n = 5 per group) were used for analysis. After tissue preparation and fixation as described above, sections were incubated with the albumin primary antibody (1:200; MAB1455; R&D Systems) overnight at 4°C, followed by biotinylated secondary antibody (1:500; 715-065-140; Jackson Immuno) for 3h at room temperature. After incubation in ABC solution (PK-6100; Vector Laboratories) for 1h, the sections were exposed to DAB (SK-4100; Vector Laboratories). The StereoInvestigator software (Stereo Investigator) was used to draw contours under ×10 magnification around the area in the ipsilesional cortex not stained with albumin and the contralesional cortex to determine the area of each region by an investigator blind to the experimental conditions. To correct for edema, the area of blood—brain barrier leakage was expressed as the percentage of the intact contralesional cortex and was determined according to the following formula: (1–(area of unstained ipsilesional cortex)/(area of the contralesional cortex)). Volume of BBB leakage was calculated by multiplying the area with the section thickness and the total number of sections for each animal.
Statistical Analysis
The statistic software package PRISM (GraphPad, San Diego, CA, USA) was used for data analysis. All means are presented±s.e.m. Statistical analyses for the Western blot experiments were performed with the use of analysis of variance followed by post hoc t-tests with the Bonferroni correction. Comparison of blood vessel density, blood vessel proliferation, neurologic score, volume of blood—brain barrier leakage, and infarct volume between control and Netrin-4-treated group was made with an unpaired t-test. A value of P < 0.05 was considered significant.
Results
Netrin-4 Protein Increased in the Ischemic Core After Ischemia
Because level of expression after ischemia may reflect potential function, we determined whether Netrin-4 was upregulated after ischemia by examining its protein level at various time points with Western blots. Compared with the expression level of sham-operated animals (n = 4), Netrin-4 expression increased in the ischemic core significantly at day 1 by 2.3 ± 0.28-fold, at day 3 by 2.5 ± 0.34-fold, at day 7 by 2.1 ± 0.26-fold (P < 0.05, n = 4 per time point), and decreased to baseline level at day 14 and day 28 (Figures 1C and 1D). No significant changes compared with sham-operated animals were observed in the contralesional cortex (Figures 1C and 1D). Immunohistochemical analysis with an antibody specific to Netrin-4 showed that at day 1, Netrin-4 was mostly expressed in the infarct core of the ischemic cortex, with very little expression in the contralesional cortex (Figure 1E). Similar staining was obtained at day 3 and day 7 after ischemia (data not shown). Netrin-4 was also seen in areas of the hippocampus bilaterally, which most likely reflects basal expression, as previously reported (Yin et al, 2000). This evidence suggests that Netrin-4 is upregulated after cerebral ischemia and may be important in subsequent molecular processes.
Netrin-4 Colocalized with Blood Vessels in the Ischemic Core
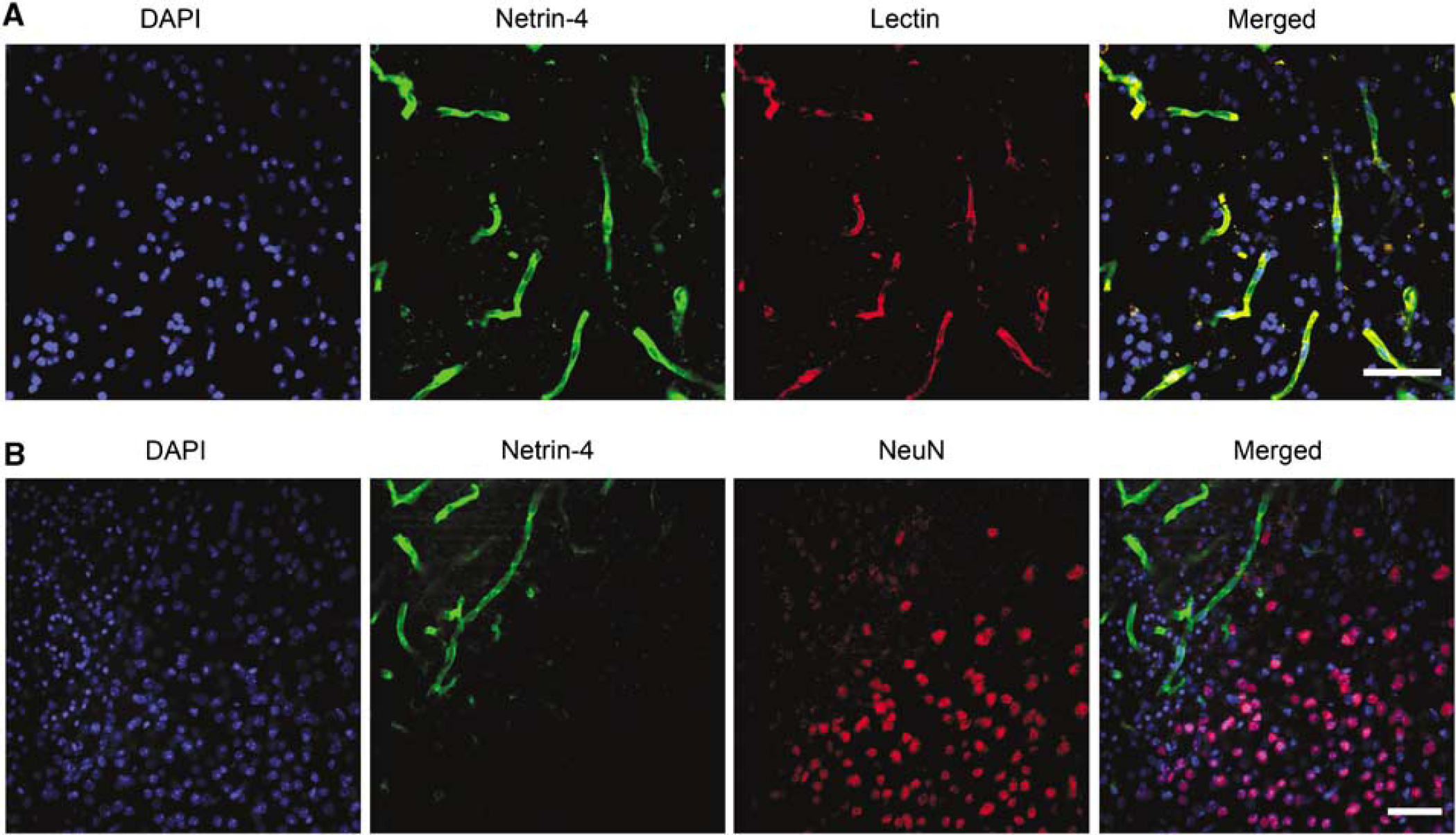
Because a previous study has implicated a role of Netrin-4 in angiogenesis (Wilson et al, 2006), we attempted to determine whether Netrin-4 colocalized with marker for blood vessels. We found that Netrin-4 colocalized with blood vessels as soon as day 1 (Figure 2A) and continued to be seen at day 3 and week 1 after dMCAO (data not shown). Moreover, to determine the location of Netrin-4 expression relative to the infarct region, we performed double immunohistochemistry for Netrin-4 and the neuronal marker NeuN. In our model of ischemia, the ischemic core is surrounded by normal appearing NeuN-positive neurons and GFAP-positive reactive astrocytes, which delineate the peri-infarct cortex. inside the ischemic core, NeuN-positive neurons die and thus no longer express NeuN. At day 1 after ischemia, Netrin-4 was significantly expressed in the ischemic core and the ischemic border, but Netrin-4 expression did not extend deeply into the periinfarct cortex where the majority of NeuN-positive cells were located (Figure 2B). This pattern continued to be seen at day 3 and week 1 (data not shown), during which Netrin-4 was located mainly in the ischemic core and border of the peri-infarct cortex.
Netrin-4 Delineated Astrocytic Foot Processes in the Peri-Infarct Cortex
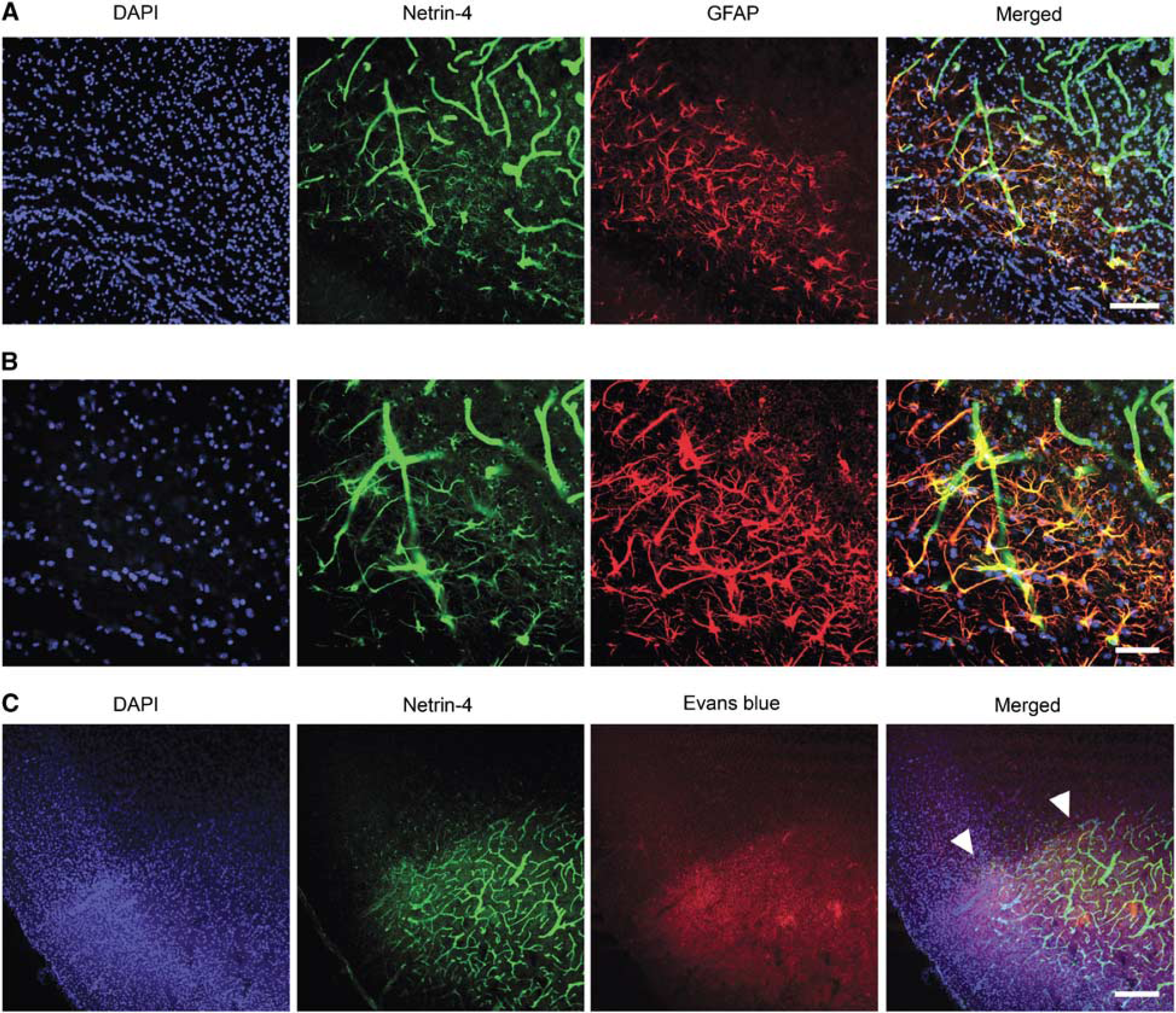
After injury to the central nervous system, astrocytes proliferate and help repair the blood—brain barrier to limit the extent of cellular injury (Silver and Miller, 2004). These activated astrocytes upregulate the expression of GFAP and become hypertrophic. Interestingly, immunohistochemical analysis showed that GFAP-positive astrocytes also expressed Netrin-4 at day 1 (Figure 3A), and later time points at day 3 and day 7 (data not shown). The GFAP-positive astrocytes formed a distinct border in the peri-infarct cortex and surrounded the Netrin-4-positive blood vessels in the ischemic core. Higher magnification of this region showed that Netrin-4-positive astrocytes wrapped their foot processes around the Netrin-4-positive blood vessels, reminiscent of the blood—brain barrier that is formed by tight association between astrocytes and endothelial cells (Figure 3B). Moreover, Netrin-4 expression also localized to the area of blood—brain barrier leakage, as showed by the presence of extravasated Evans blue (Figure 3C). These findings implicate a potential function of Netrin-4 in blood—brain barrier repair.
Intracerebroventricular Administration of Netrin-4-Enhanced Angiogenesis
Because a previous study has implicated a function of Netrin-4 in angiogenesis (Wilson et al, 2006), we attempted to determine whether administration of Netrin-4 would enhance angiogenesis in the periinfarct cortex. Compared with the control group (n = 5), the group that received Netrin-4 (n = 5) had a significantly higher blood vessel density and more blood vessel proliferation at 1 week after stroke (P < 0.05) (Figures 4A and 4B). In the peri-infarct cortex, the number of lectin-positive blood vessel was 275 ± 17.68 per mm2 in the control group and 366 ± 12.99 per mm2 in the Netrin-4-treated group (Figure 4D). To determine blood vessel proliferation, the number of blood vessels positive for both BrdU and lectin was also quantified. The number of lectin and BrdU-positive vessels was 48 ± 2.93 per mm2 for the control group and 71.5 ± 2.59 per mm2 for the Netrin-4-treated group (Figure 4E). Similar analysis for the contralesional cortex showed no significant differences in angiogenesis between the control and Netrin-4 treated groups (Supplementary Figure 1). These findings thus suggest Netrin-4 that administration can enhance angiogenesis in the peri-infarct cortex. No effect on angiogenesis in the contralateral cortex was observed (Supplementary Figure 1).
Intracerebroventricular Administration of Netrin-4 Improved Behavioral Outcome But Did Not Have a Significant Effect on Infarct Size or Blood—Brain Barrier Leakage
Improvement in angiogenesis has been correlated with an improvement in behavioral outcome (Slevin et al, 2006). As a result, we used a behavioral rating scale that evaluates the animals for motor function, sensory perception, and reflex (Li et al, 2000) to compare behavioral recovery between the control and the Netrin-4-treated group. Animals treated with Netrin-4 showed a significant improvement compared with control animals (P< 0.05) (Figure 5A). The control group had a score of 1.6 ± 0.24 whereas the Netrin-4-treated group had a score of 0.08 ± 0.2 (n = 5 per group). To determine whether the improvement in behavioral recovery may be due to differences in infarct size or blood—brain barrier leakage, we also compared these two variables. However, no significant differences were found between the 2 groups. The volume of blood—brain barrier leakage, expressed as a percentage of the contralesional hemisphere, in the control group was 21.9 ± 2.58% and that for the Netrin-4-treated group was 20.96 ± 1.72% (Figure 5B). Likewise, the infarct volume, expressed as a percentage of the contralesional hemisphere, in the control group was 17.00 ± 0.76% and that for the Netrin-4-treated group was 17.19 ± 1.84% (Figure 5C). These findings thus suggest that Netrin-4 induction of angiogenesis played an important role in promoting neurologic outcome.
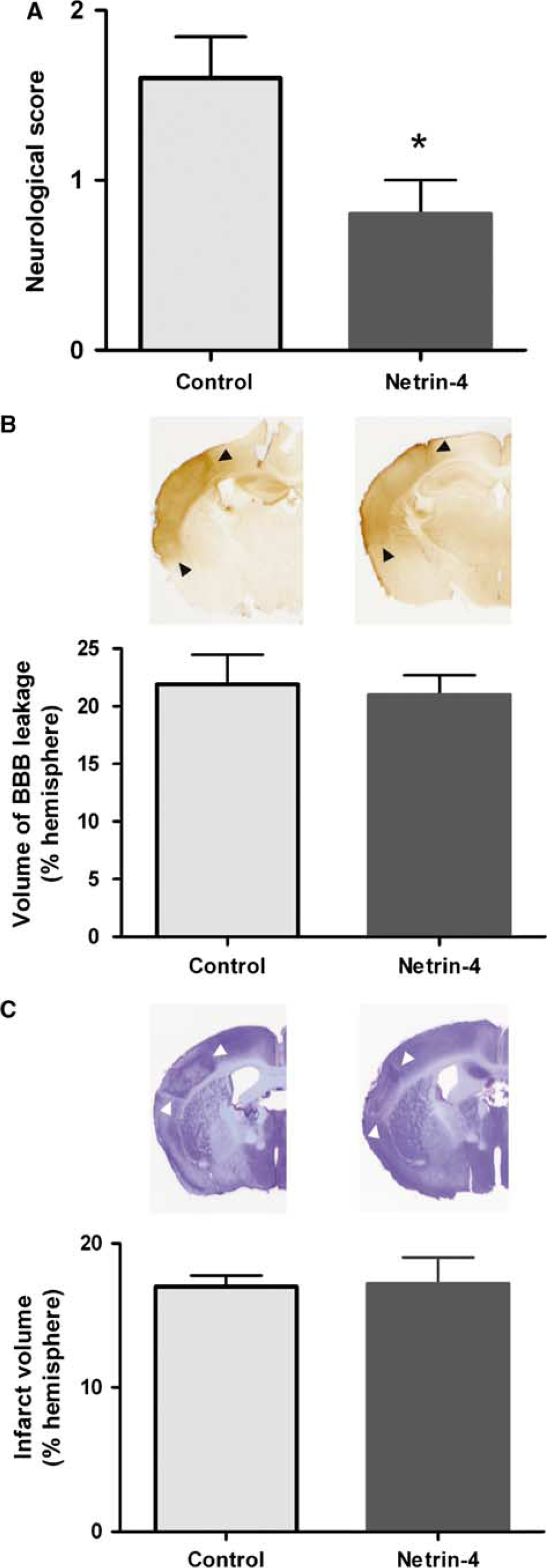
Netrin-4 administration improved behavioral outcome but did not have a significant effect on infarct size or blood—brain barrier permeability. (
Expression of the Putative Netrin-4 Receptor DCC, but not Unc5A or Unc5B, was Upregulated in the Peri-Infarct Cortex after Stroke
To better understand the mechanisms by which Netrin-4 may exert its effect, we also examined the distribution of its putative receptors, DCC and Unc5A, which have been shown to bind to Netrin-4 in vitro (Qin et al, 2007). We also examined the distribution of Unc5B, as it has been shown to be important for angiogenesis (Navankasattusas et al, 2008). However, Western blot analysis of the expression level of Unc5A and Unc5B did not detect any significant changes in the peri-infarct cortex as compared with sham-operated controls (Figures 6A and 6B). In the peri-infarct cortex, Unc5A was found on NeuN-positive neurons (Figure 6C), whereas Unc5B was found on β-tubulin III-positive neuronal processes at day 1 after dMCAO (Figure 6D). In the ischemic core, there was a significant loss of expression of both Unc5A and Unc5B that corresponded with the loss of neurons (data not shown). However, the putative Netrin-4 receptor, DCC, was significantly upregulated in the peri-infarct cortex after dMCAO. Compared with the expression level of sham-operated animals (n = 4) in the ischemic core, DCC expression increased significantly at day 1 by 3.5 ± 0.36-fold, at day 3 by 3.8 ± 0.34-fold, at week 1 by 2.7 ± 0.18-fold, at week 2 by 2.63 ± 0.32 (P<0.05, n = 5 per time point), and decreased to baseline level at day 28 (Figure 7A). No significant changes compared with sham-operated animals were observed in the contralesional cortex. Immunohistochemical analysis with an antibody specific to DCC showed DCC to be in neuronal processes, by its colocalization with β-tubulin III, at day 1 (Figure 7B) and continued to be seen at day 3, week 1, and week 2 (data not shown). There was also a significant loss of DCC in the ischemic core that corresponded with the loss of neurons (data not shown). This evidence suggests that DCC is upregulated after cerebral ischemia and may be important in neuronal plasticity.
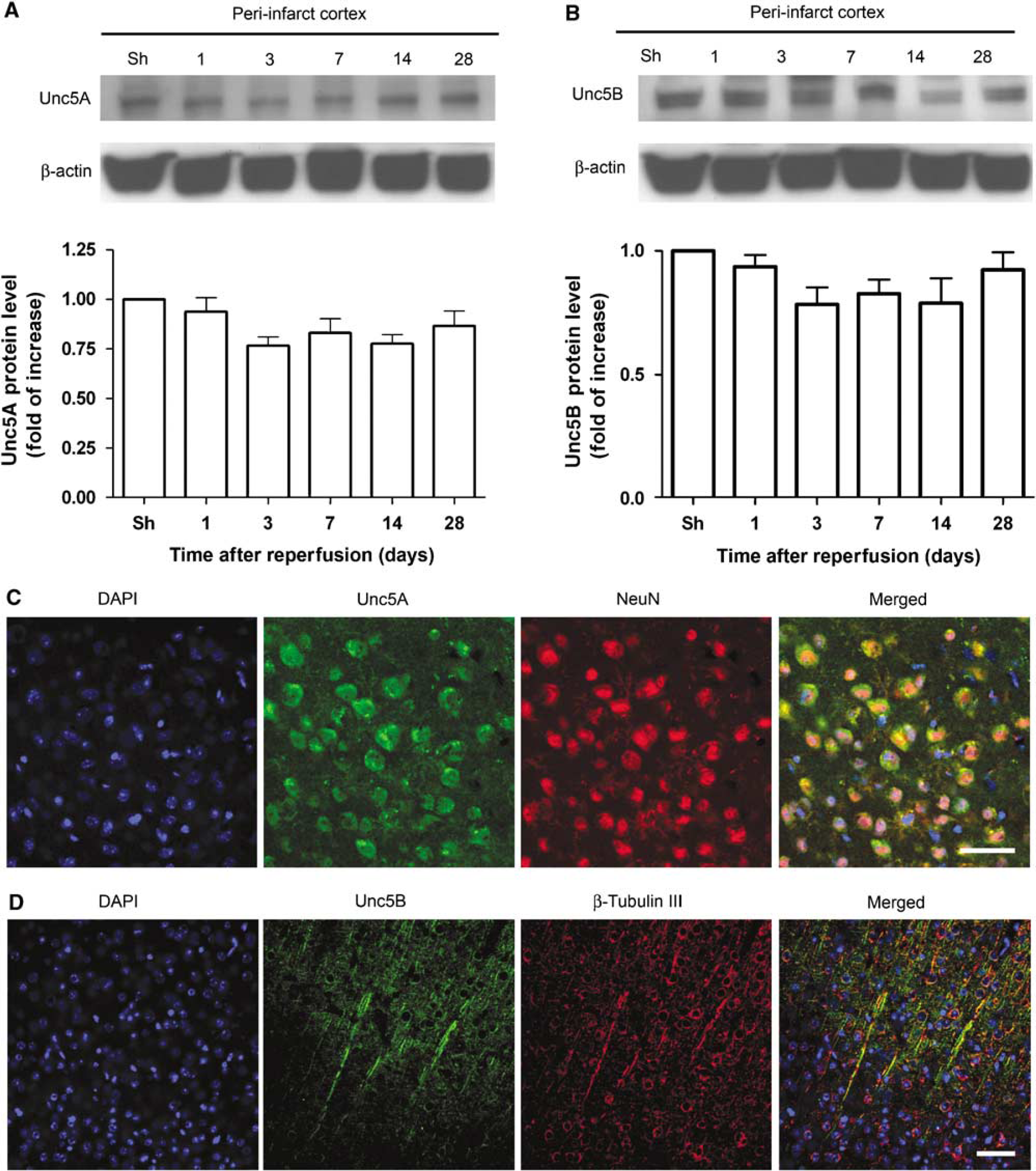
Expression level of Unc5A and Unc5B did not change significantly after cerebral ischemia. (
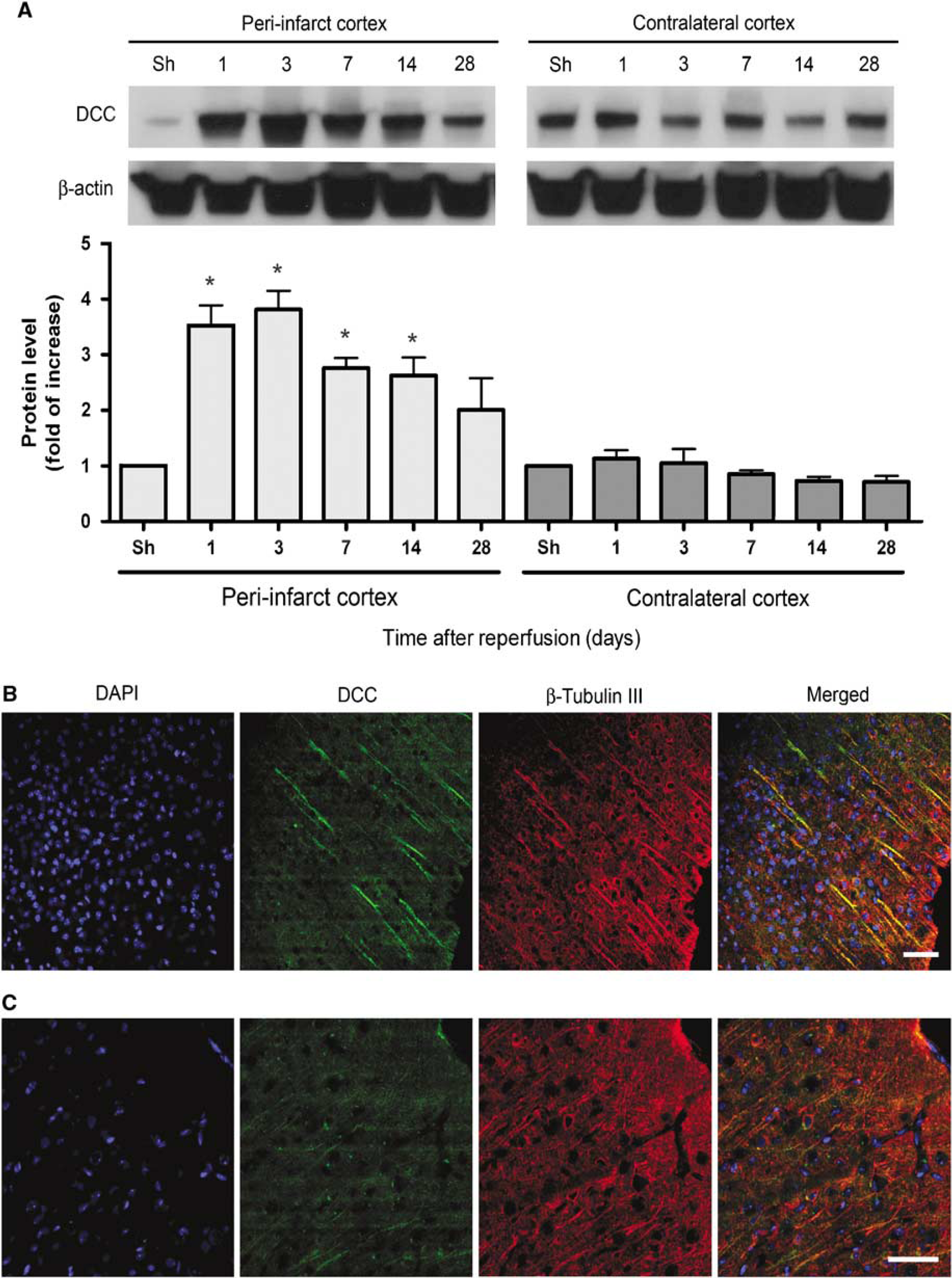
DCC protein was upregulated in the peri-infarct cortex after cerebral ischemia. (
Discussion
In this study, we showed that Netrin-4 was significantly upregulated in blood vessels and astrocytes after stroke, most specifically in the ischemic core where the BBB was severely disrupted. Intracerebroventricular administration of Netrin-4 led to improvements in angiogenesis in the peri-infarct cortex and behavioral outcome, but did not have a significant effect on synaptogenesis (Supplementary Figure 2), infarct size, or blood—brain barrier leakage. The putative Netrin-4 receptor, DCC, was also found to be upregulated in the peri-infarct cortex. Because Netrin-4 has been shown to promote angiogenesis in vitro and its administration after hindlimb ischemia promoted neovascularization and enhanced blood flow (Wilson et al, 2006), our findings, in the context of cerebral ischemia, support the hypothesis that Netrin-4 can act as an angiogenic factor for improving behavioral outcome after stroke.
In the model of ischemia used here, we have shown that the upregulation of Netrin-4 was limited to areas of blood—brain barrier leakage, as evident by the presence of Evans blue from the systemic circulation. The localization of Netrin-4 to the ischemic core is similar to that seen with VEGF, which also showed an upregulation in the ischemic core as soon as the first day after stroke (Zhang et al, 2002). Near the border of the peri-infarct cortex, Netrin-4 was upregulated mostly in those astrocytes whose endfeet wrapped tightly around Netrin-4-positive blood vessels. This morphologic evidence thus posits a potential role of Netrin-4 in blood—brain—barrier function. Although we were not able to show a beneficial effect of Netrin-4 administration on blood—brain barrier function or infarct size, it is possible that loss-of-function experiments with transgenic animals may prove more informative. It is also possible that a more acute time course of administration or different dosage may need to be tried to show a neuroprotective effect of Netrin-4. Lastly, albumin is a relatively small molecule, so although we were not able to show a positive result with albumin, it remains to be determined whether administration of Netrin-4 reduces permeability to larger molecules.
After an ischemic stroke, there exists a periinfarct cortical region immediately surrounding the ischemic core, where partially affected neurons receiving intermediate perfusion have a greater capacity for survival and structural reorganization if blood flow is maintained through the formation of new blood vessels (Slevin et al, 2006). Angiogenesis has been shown to take place in the peri-infarct cortex after focal ischemia in rodents (Hayashi et al, 2006), and the extent of angiogenesis is correlated with survival in patients with stroke (Krupinski et al, 1994). Thus, identifying the factors that can enhance angiogenesis in the peri-infarct cortex may lead to the development of new therapies with the potential to improve repair of the vascular network after stroke. Although Netrin-4 expression after stroke did not extend too deeply into the area of unaffected neurons, it was found in the ischemic core, where there was a loss of neurons, and in the ischemic border, where partially affected neurons resided. We also found that administration of Netrin-4 after stroke resulted in higher blood vessel density and more blood vessel proliferation in the peri-infarct cortex. This suggests that one potential function for the endogenous upregulation of Netrin-4 near the ischemic border may be to induce new blood vessels formation and thus improve collateral blood flow and survival for those neurons partially affected by ischemia.
Although neurons in the ischemic core eventually die, the potential angiogenic role of Netrin-4 in this area may function to enhance macrophage infiltration and removal of necrotic tissue (Manoonkitiwongsa et al, 2001). It was shown that in an area roughly corresponding to the ischemic core in our model of ischemia, there was an increase in angiogenesis that correlated with an increase in the number of macrophages after stroke. It was thus suggested that the increased number of collateral vessels in the ischemic infarct facilitated entry of inflammatory cells to clear away tissue debris in this area. Nevertheless, although the precise functions of angiogenesis after cerebral ischemia still remain to be elucidated, the angiogenic effect of Netrin-4 supports previous findings. It was shown that Netrin-4 had similar potency to VEGF in stimulating endothelial cell migration, proliferation, and tube formation in vitro and in enhancing angiogenesis and blood flow after hindlimb ischemia in vivo (Wilson et al, 2006). For these reasons, understanding the mechanisms behind the angiogenic effect of Netrin-4 would be very useful.
Biochemical evidence has showed that Netrin-4 can bind to the DCC and Unc5A receptors in vitro (Qin et al, 2007). As a result, we attempted to characterize their expression pattern after stroke, along with Unc5B. We were interested in Unc5B because it has been shown to be important for guidance events in vascular morphogenesis (Lu et al, 2004) and its deletion in the embryonic endothelium of mice resulted in a deficiency of arterioles in the placental labyrinth (Navankasattusas et al, 2008). However, after dMCAO, there was not a significant increase in the expression of Unc5A and Unc5B and their expression was mostly seen in neurons in the peri-infarct cortex, where there was little upregulation of Netrin-4. In contrast, there was a significant increase in the expression of DCC in neuronal processes in the peri-infarct cortex, starting at day 1 and continued until 2 weeks after dMCAO. In a model of ischemia in rat, DCC has been shown to increase by immunohistochemistry (Tsuchiya et al, 2007), but we also confirmed its increase in our experiments with Western blot data. The increase in DCC was mostly seen in neurons and did not overlap with the location of Netrin-4 upregulation, which was found mostly in nonneuronal structures in the ischemic core. We tested the possibility that Netrin-4 may be mediating behavioral recovery by signaling to neurons and affecting synaptic density, but we detected no changes in synapses in the ipsilesional or contralesional hemispheres due to exogenous Netrin-4 (Supplementary Figure 2). Interestingly, in a different study using coimmunoprecipitation, Netrin-4 was not found to bind to any known Netrin-1 receptor including DCC, Neogenin, Unc5A, and Unc5B (Wilson et al, 2006). As a result, it is likely that a currently unidentified receptor mediates the endogenous effect of Netrin-4 after stroke.
Several agents that enhance angiogenesis have been shown to improve behavioral recovery after stroke. Chief among them is VEGF, which through its angiogenic effect, improved functional outcome after cerebral ischemia (Sun et al, 2003; Zhang et al, 2000). Some other agents that enhance angiogenesis after ischemia include Niaspan (Chen et al, 2007), statins (Chen et al, 2003), and Erythropoietin (Li et al, 2007). Our findings that Netrin-4 administration improved angiogenesis and behavioral outcome after ischemia provides another example of blood vessel remodeling that is correlated with functional recovery after ischemic injury.
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
