Abstract
Cadherins are Ca2+-dependent cell adhesion molecules that are important in vertebrate nervous system development. We identified seven members of the cadherin superfamily (cadherin-4, cadherin-5, cadherin-6, cadherin-6, cadherin-11, protocadherin-1, and protocadherin-17) and an intracellular binding partner of δ-protocadherins, protein phosphatase 1α, as novel markers for developing blood vessels in the ferret brain. Some of the cadherin molecules are restricted to specific brain regions or a subset of blood vessels. The expression levels show a peak during perinatal vascular development. Our results suggest that multiple cadherins, which are also involved in neurogenesis, are regulators of angiogenesis in developing vertebrate brain.
Introduction
The development of a functional vascular system is a complex process and a primary requirement for embryogenesis. In the brain, concomitant vascular development meets the metabolic needs of growing neuronal populations. The vascularization of the brain takes place exclusively by means of angiogenesis (Plate, 1999). Angiogenesis is achieved by the pruning, remodelling, and extension of an existing primary vascular plexus in response to local cues, such as growth factors, adhesion molecules, and guidance factors. It is regulated by the capacity of endothelial cells to adhere to each other and to surrounding cells. Examples of adhesion molecules involved in angiogenesis are CD34, PECAM, VE-cadherin (cadherin-5, CDH5; Breier et al, 1996), N-cadherin, cadherin-10 (Williams et al, 2005), T-cadherin, and R-cadherin (Cavallaro et al, 2006) and protocadherin-1 (PCDH1; Redies et al, 2008). The presence of some cadherins in developing brain vasculature prompted us to ask whether other recently identified cadherins are also expressed during brain angiogenesis.
Cadherins are a large family of Ca2+-dependent cell adhesion glycoproteins, with more than 100 members in vertebrates. Cadherins are grouped into subfamilies that are designated as classic cadherins, desmosomal cadherins, flamingo cadherins, FAT cadherins, and protocadherins (Hirano et al, 2003). They play a role in a wide variety of developmental mechanisms, including cell proliferation, differentiation, recognition, migration and sorting, morphogenesis, signal transduction, and axon outgrowth (Gumbiner, 2005). The interaction of cadherins with the cytoskeleton through intracellular partners is crucial for their functioning. During vascular development, cadherins have been reported to regulate the formation of the primitive capillary plexus and its pruning, endothelial integrity, vascular permeability, formation of blood—brain barrier, pericyte stabilization, and tumor angiogenesis (Cavallaro et al, 2006). For example, gene silencing of VE-cadherin and N-cadherin in mice leads to early embryonic death with associated severe vascular anomalies (Cavallaro et al, 2006). With regard to its precise wiring in highly ordered and stereotyped networks, the formation of vasculature closely parallels neuronal development. Indeed, many of the molecules regulating vascular system development, like VEGFR, Eph, N-cadherin, Dlx1/2, Nkx2.1, Shh, and Pax6, are implicated also in neural development. It has therefore been proposed that common mechanisms are shared by vascular and neuronal development (Carmeliet, 2003). A large number of classic cadherins and δ-protocadherins (Vanhalst et al, 2005) are differentially expressed in brain structures and subtypes of neurons in the developing mammalian brain (Kim et al, 2007; Hertel et al, 2008; Krishna-K et al, 2008). In this study, we asked whether some of these cadherins and an intracellular binding partner of δ1-protocadherins, protein phosphatase 1α (PP1α; Vanhalst et al, 2005), are also expressed by developing blood vessels in the ferret, an animal model suitable for cerebrovascular research (Atkinson et al, 1989).
Materials and methods
Animals
The study was performed in 36 ferrets at 10 developmental stages (at 23 days after conception (E23), E30, E38, at postnatal day 2 (P2), P13, P25, P33, P46, P60, and at the adult stage). The series of sections analyzed was used previously to study visual cortical development (Krishna-K et al, 2008).
Reverse Transcriptase-PCR and Cloning
Probe synthesis and plasmids used were described previously (Krishna-K et al, 2008). In addition to the existing probes, we obtained cDNA fragments for CDH5, cadherin-19 (CDH19), and PP1α by reverse transcriptase-PCR and cloned them into pCR II-TOPO vector. Degenerate primers used and sequences obtained were submitted to GenBank (Table 1).
Expression profile of 18 cadherins and PP1α in blood vessels of the developing ferret braina
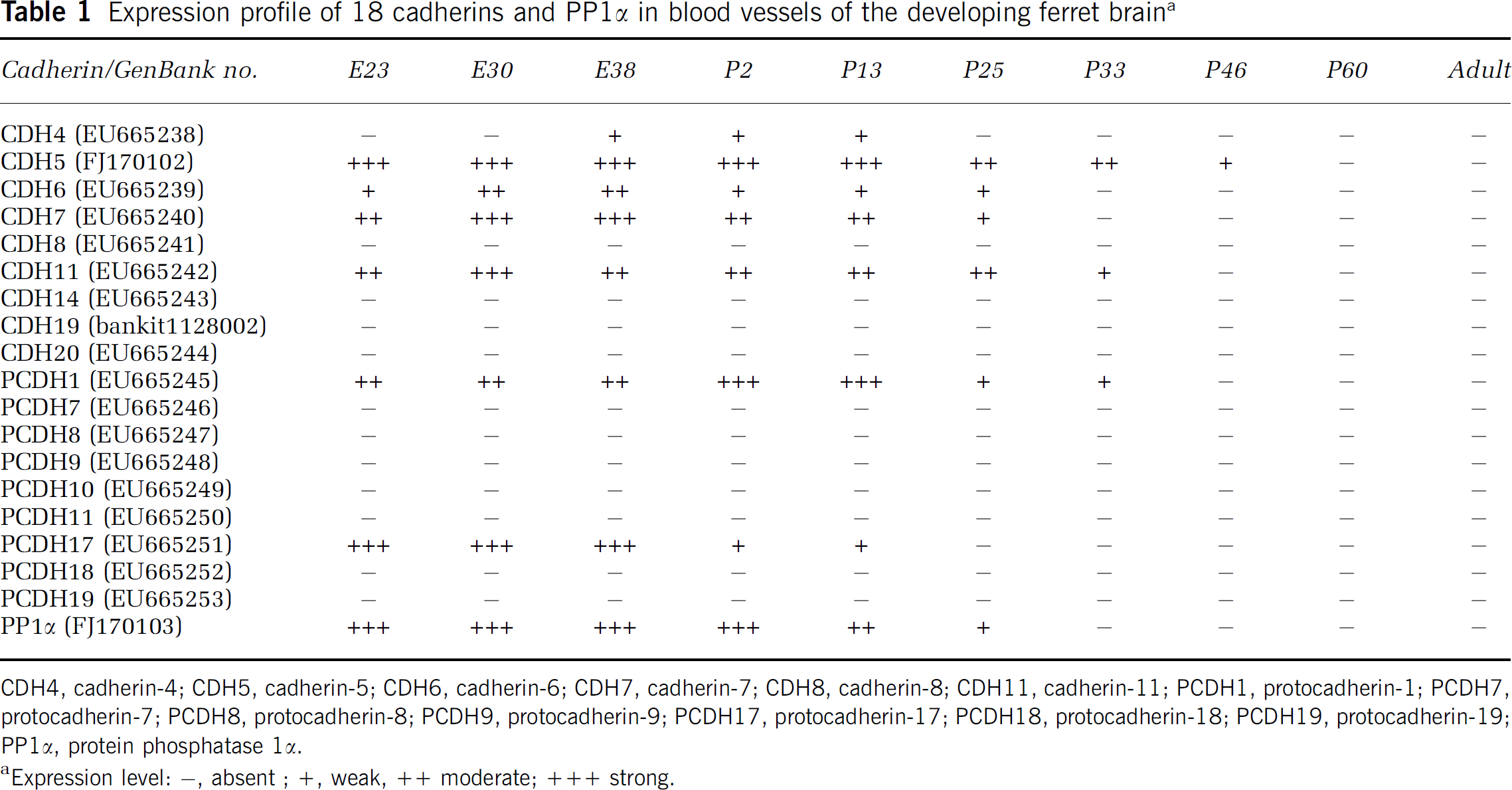
CDH4, cadherin-4; CDH5, cadherin-5; CDH6, cadherin-6; CDH7, cadherin-7; CDH8, cadherin-8; CDH11, cadherin-11; PCDH1, protocadherin-1; PCDH7, protocadherin-7; PCDH8, protocadherin-8; PCDH9, protocadherin-9; PCDH17, protocadherin-17; PCDH18, protocadherin-18; PCDH19, protocadherin-19; PP1α, protein phosphatase 1α.
In Situ Hybridization
cRNA probes were produced with T7 or SP6 RNA polymerase followed by labeling with digoxigenin or fluorescein to generate antisense probes. In situ hybridization was performed as described previously with antisense and sense probes for all cadherins (Krishna-K et al, 2008).
Double Tyramide-FISH and Fluorescent Immunohistochemistry
The in situ hybridization protocol was modified by introducing tyramide-coupled Alexa fluorophore A488 or A568 as reaction products by CARD (Krishna-K et al, 2008). Subsequently, fluorescent immunostaining was performed on the same slices by using anti-mouse fibronectin rabbit antiserum (kind gift of Richard Hynes, MIT, USA). The sections were viewed and photographed under a confocal laser scanning microscope (SP5; Leica Microsystems, Germany).
Results
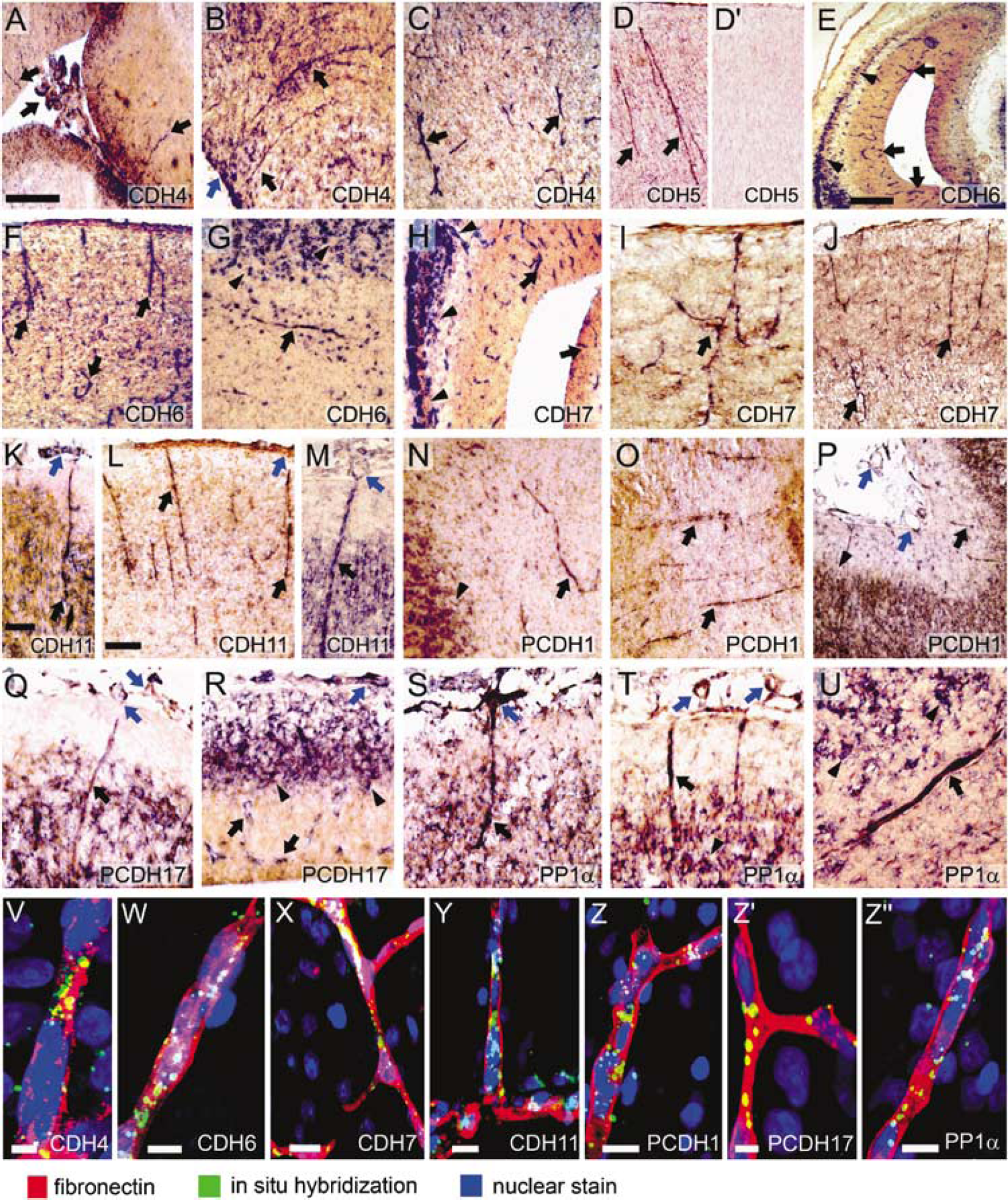
The expression of eight classic cadherins, nine δ-protocadherins and PP1α was mapped by in situ hybridization from early embryonic stages to the adult stage in the ferret brain (Table 1). We found that 7 of the 18 cadherins (cadherin-4 (CDH4), CDH5, cadherin-6 (CDH6), cadherin-7 (CDH7), cadherin-11 (CDH11), protocadherin-1 (PCDH1), and protocadherin-17 (PCDH17); Figure 1A–1R) and PP1α (Figure 1S–1U) are expressed by blood vessels in the developing brain (black arrows in Figure 1; Table 1) and meninges (blue arrows in Figure 1). No specific labeling was seen with sense probes (Figure 1D′). Embryonic blood vessels showed strong and ubiquitous expression of CDH11, PCDH17, and PP1α, whereas expression of CDH4, CDH6, and PCDH1 was restricted to blood vessels in specific brain regions. Double labeling with antibodies against fibronectin, a marker for embryonic brain blood vessels, confirmed the vascular expression of the seven cadherins and PP1α (Figure 1V–1Z″). However, particularly at postnatal stages, only a subset of fibronectin-positive blood vessels expressed the cadherins. Note that many of the cadherins are also expressed by neurons in the same brain regions (arrowheads in Figure 1).
(
Cadherin-4 (R-Cadherin)
The expression of CDH4 (R-cadherin) starts to appear in brain blood vessels at E38 (Figure 1A). The meninges are also positive. The expression is weak and restricted to cerebral cortex. The signal becomes stronger at P2 (Figure 1B), decreases until P13 (Figure 1C) and disappears by P25.
Cadherin-5 (VE-Cadherin)
Strong staining was observed from the earliest stage studied (E23) and persists until P13 in most brain regions (Figure 1D). Thereafter, expression decreases and is not seen after P46.
Cadherin-6
At the earliest stage examined (E23), weak staining was observed in some blood vessels of the brain stem. At E30, expression is prominent in the telencephalon, where positive blood vessels are seen in the mantle zone and ventricular zone (Figure 1E). In the hindbrain, signal becomes weak from E30 to E38. Staining in the telencephalon gradually decreases until P13 (Figure 1F). Some blood vessels in the white matter are also weakly to moderately positive, for example at P25 (Figure 1G). At later stages, most of the blood vessels do not show any expression.
Cadherin-7
Moderate staining for CDH7 was seen in hindbrain blood vessels at E23. At E30 (Figure 1H), E38 (Figure 1I), and P2 (Figure 1J), moderate to strong expression was observed in blood vessels throughout the brain, both in the ventricular zone and mantle zone. At P2 and P13, blood vessels in frontal cortical regions, including white matter, are stained more strongly than in caudal telencephalic regions. Signal decreases at P25 and disappears at P33.
Cadherin-11
Ubiquitously strong expression by blood vessels in all brain regions and in the meninges is seen from E23 to E38, both in the ventricular layer and mantle layer (Figure 1K). At P2, signal has become moderate in most areas, but remains strong in some blood vessels in the cortical mantle (Figure 1L). At P13 and P25, only blood vessels in the cortex remain weakly to moderately positive (Figure 1M). No signal is observed after P33.
Protocadherin-1
From E23 and E38, blood vessels in the telencephalon are moderately positive, and a weak to moderate signal is seen in meninges. The expression is moderately strong between P2 (Figure 1N) and P25 (Figure 1O), particularly in the cerebral cortex and diencephalon; it is no longer observed after P33 (Figure 1P).
Protocadherin-17
PCDH17 expression is very strong and ubiquitous in blood vessels of all brain regions and in the meninges at E23, both in the ventricular and mantle layer (Figure 1Q). This expression profile persists at E30 and E38 (Figure 1R). The staining becomes weak at P2 and is no longer seen after P13.
Protein Phosphatase 1α
PP1α shows strong ubiquitous staining in the blood vessels and meninges at E23 (Figure 1S). Staining persists at E30 and E38 (Figure 1T). At P2, vessels exhibit only a weak signal, except for superficial blood vessels in the neocortex that maintain a strong expression. At P13, a subset of blood vessels retains moderate staining, also in the white matter (Figure 1U). The expression becomes weak at P25 and disappears thereafter.
The expression of the above cadherins and PP1α by blood vessels was confirmed by double labeling with a marker for brain blood vessels (fibronectin; red in Figure 1V–1Z″).
Discussion
For the first time, expression of 18 members of two cadherin subfamilies, classic cadherins and δ-protocadherins, were studied in blood vessels at different stages of vertebrate brain development. Using in situ hybridization and immunohistochemistry, we show that 7 out of the 18 cadherins and PP1α (Table 1) are expressed under a tight spatiotemporal control by cerebral blood vessels during development.
Cadherin Expression by Blood Vessels During Development
The cadherins and PP1α are expressed from the earliest stage studied (E23) until about P25. In higher vertebrates, brain angiogenesis starts at the beginning of neurogenesis and proceeds up to the last wave of neuronal migration, when the basic scheme of vascular network is completed (Plate, 1999). As the brain grows in thickness, unbranched vessel, which migrate radially into the nervous wall, express the cadherins (Figure 1J–1M, 1P, 1Q, 1S and 1T). During this migration, cadherins may contribute to the adhesion between endothelial cells or between endothelial cells and surrounding pericytes, as shown for VE-cadherin and N-cadherin, respectively (Breier et al, 1996; Cavallaro et al, 2006). Cadherin expression reaches highest levels at perinatal stages (Table 1). At corresponding stages in the rat, maximal endothelial cell proliferation and gliogenesis takes place. Similarly, expression of Flk-1, Flt-1, PECAM-1, and VEGF peaks in vessels of the premature brain and then declines (Ogunshola et al, 2000; Yang et al, 2003). The peak during this critical developmental period suggests that cadherins are proangiogenic factors that regulate the growth, migration, and controlled pruning of the newly forming vascular network. Because the cadherins are not expressed by astrocytes (Krishna-K and Redies C, unpublished data), they are unlikely to play a role in the interaction between endothelial cells and astrocytes (Zerlin and Goldman, 1997). The identification of PP1α in developing blood vessels leads to the question of which molecules are regulated downstream by this phosphatase.
Region- and Subtype-Specific Expression of Cadherins by Blood Vessels
Expression of CDH4, CDH6, and PCDH1 is restricted to particular areas of developing brain, in particular to cerebral cortex. Another cadherin that shows a regionally restricted expression pattern is cadherin-10; it is found at the surface of the mouse cortex but not of the cerebellum (Williams et al, 2005). Interestingly, some cadherins (CDH7 and CDH11) are expressed by specific blood vessels although the neighboring ones do not show expression. Several studies have shown that arteries and veins are likely to differ in their expression of molecular markers. For example, ephrinB2 expression is confined to the arterial endothelium whereas EphB4 expression is higher in veins (Wang et al, 1998). Whether the cadherins are expressed also by a specific blood vessel type or during a specific stage of angiogenesis remains to be investigated.
A Common Cadherin-Based Mechanism Behind Angiogenesis and Neurogenesis?
The seven cadherins expressed by blood vessels are also known for their spatiotemporally regulated expression by neurons in restricted brain regions (arrowheads in Figure 1; Kim et al, 2007; Hertel et al, 2008; Krishna-K et al, 2008) and for their roles in brain regionalization, cell-specific expression and guided migration. Other genes, like ephrins, netrins, slits, and semaphorins, are also concomitantly expressed and reported to play dual roles in both neural and vascular development (Carmeliet, 2003). This similarity led to the suggestion that there might be an intrinsic or operative relationship or both between neurogenesis and angiogenesis (Carmeliet, 2003; Vasudevan et al, 2008). The identification of a novel panel of cadherin markers for developing brain blood vessels provides a basis for the design of functional assays to study the role of these genes in neuroangiogenesis, also in brain tumors.
Footnotes
Acknowledgements
Ferrets were kindly provided by Dr Dieter Wolff and his colleagues at the Federal Institute of Risk Assessment in Berlin-Marienfelde, Germany. We thank Dr Richard Hynes for his kind gift of the fibronectin antibody, Ms Monique Nuernberger for help in the initial part of the study, and Ms Nicole Mergel and Ms Jessica Heyder for technical assistance. This work was supported by the Interdisciplinary Clinical Research Center of the University of Jena (IZKF Jena, TP 1-16).
Disclosure/conflict of interest
The authors declare that they have no conflict of interest.
