Abstract
Cerebral
INTRODUCTION
Mutations in a transmembrane receptor NOTCH3 cause cerebral
Vasoregulatory and circulatory disturbances are well established in CADASIL patients. Reduction of blood flow in cerebral white matter appears already around the age of 30 years and becomes more severe as the disease progresses. 10 Retinal arteries have been shown to appear similarly contracted as in hypertension and retinal blood flow is somewhat decreased.11,12 CADASIL patients have impaired endothelium-dependent vasodilation in peripheral resistant arteries13,14 while in cerebral resistance arteries the vasoreactivity could be enhanced by systemic application of L-arginine. 15 Moreover, alterations in vasoregulation have also been detected in caudal arteries of transgenic mice expressing mutated (p.Arg90Cys) human NOTCH316 and in cerebral arteries in transgenic mice expressing mutated (p.Arg169Cys) rat NOTCH3. 17
NOTCH3 protein is a large single pass transmembrane receptor expressed in adult human almost exclusively in VSMCs and pericytes
7
and is needed for structural integrity and generation of functional arteries.
18
Overexpression of the constitutively active NOTCH3 intracellular domain (N3ICD) in cultured VSMCs results in an increase of actin stress fibers and in steady-state levels of polymerized actin.
18
Analogously,
Upregulation of contractile proteins in our proteomic analysis of cultured human CADASIL VSMCs
20
prompted us to analyze in greater detail the consequences of expression of mutated NOTCH3 for the cytoskeleton of VSMCs. In the present work, we have analyzed the organization of actin cytoskeleton and adhesion complexes in cultured VSMCs derived from blood vessels of different organs from 10 CADASIL patients and from seven control individuals. All CADASIL cell lines are genetically genuine carrying either endogenous p.Arg133Cys or p.Gly528Cys mutation, and include a unique cell line homozygous for the p.Arg133Cys mutation. In addition, we investigated the changes in actin cytoskeleton in control VSMCs in which
MATERIALS AND METHODS
Generation of Patient Cell Lines and Cell Culture
Cell lines were established from blood vessels of genetically verified CADASIL patients and control subjects as previously described.
20
Nine CADASIL patients carried p.Arg133Cys mutation, eight were heterozygous and one was homozygous. In addition, one CADASIL patient carried p.Gly528Cys mutation. Vessels were obtained post partum from pregnancies, in which either of the parents was diagnosed with CADASIL (human umbilical cord arterial VSMC = HUmbVSMC and human placental VSMC = HPlaVSMC), from surgical operations (human systemic arterial VSMC=HArtVSMC) or
VSMC lines used in this study
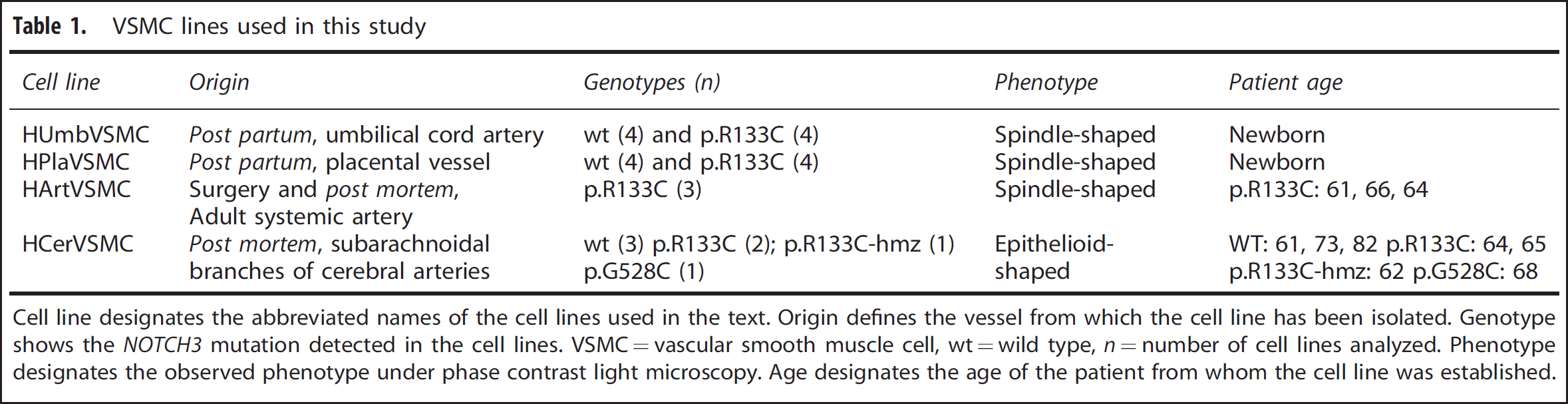
Cell line designates the abbreviated names of the cell lines used in the text. Origin defines the vessel from which the cell line has been isolated. Genotype shows the
The cells were infected with a human papilloma virus construct E6/E7 at early passage (p1 to p3) to partially immortalize the cell lines. The infection was verified by culturing the cells in the presence of G418 (Invitrogen, Auckland, NZ, USA) (400 μg/mL) for a 10-day period. Cells were identified to be VSMCs by α-smooth muscle actin (a-SMA) staining as primary cells (passage 1) and after the viral infection (passage 2 to 5) and all VSMC lines were screened negative for mycoplasma by VenorGeM mycoplasma detection kit (Minerva Biolabs GmbH, Berlin, Germany) or with DAPI staining. Passages of the cells were matched in each experiment and were maximally at passage 10, except for the NOTCH3-silenced cells, which were at passage 18.
Genotyping
The genotype of the cell lines [presence or absence of the c.475C > T (p.Arg133Cys) or c.1582G > T (p.Gly528Cys) mutation] was verified by amplifying exon 4 (for analysis of mutation c.475C > T) or exons 9 and 10 (for analysis of mutation c.1582G > T) of
Silencing of NOTCH3 in Vascular Smooth Muscle Cells with Lentiviral Vectors
To generate lentiviral transduction particles, a set of five pLKO.1 vectors encoding shRNA targeting
Validation of NOTCH3 silencing in the transduced cells was carried out by Western blot analysis 3 days after puromycin selection at passage 18. To assess the level of silencing, ImageJ analysis software (NIMH, Bethesda, MD, USA) was used to calculate the relative density of each band (N3 full lenght
Immunocytochemistry
Vascular smooth muscle cells were fixed with paraformaldehyde for analysis of cell surface NOTCH3 and with methanol for analysis of intracellular NOTCH3 and other intracellular proteins. Fixed cells were blocked with 3% bovine serum albumin in phosphate-buffered saline for 30 minutes and incubated with primary antibodies, followed by appropriate Alexa-fluor labeled secondary antibody (Molecular Probes, Eugene, OR, USA).
All available cell lines were analyzed for immunofluorescence for NOTCH3 and actin: HUmbVSMC: CADASIL
Western Blot Analysis
Cultured cells at passage 5 or at 18 (shRNA-silenced cells) were washed three times with phosphate-buffered saline and lysed in RIPA-buffer with complete mini protease inhibitor cocktail (Roche, Meylan Cedex, France) on ice for 15 minutes. Cells were scraped from the culture plates and sonicated 3 × 30 seconds on water bath sonicator. Cell suspension was centrifuged at 10,000 g for 15 minutes at 4°C. Insoluble cell debris was discarded. Protein concentrations were measured by Quant kit (GE Healthcare, Amersham, UK). Western blot was performed according to the manufacturer's instructions. Antigens were detected with the primary antibodies followed by HRP-labeled secondary anti-mouse or anti-rabbit antibodies and ECL-plus substrate (GE Healthcare). Blots were scanned and visualized with a fluorescence scanner (Typhoon 9400, GE Healthcare).
Primary Antibodies
Primary antibodies used in Western blot and immunocytochemistry were NOTCH3 clone 1E4, NOTCH3 clone 5E1, both directed against N3ECD (both kind gifts from Dr A Joutel), NOTCH3 ICD (D11B8, Cell Signaling, Beverly, MA, USA), α-SMA (Sigma), HES5 (M-104, Santa Cruz Biotechnology, Heidelberg, Germany) β-Actin (Sigma), vinculin (Biohit, Helsinki, Finland), integrin β1, 21 tensin (BD Transduction Laboratories, Mississauga, Canada).
Statistical Analyses
Significance of the differences of average area of adhesion complexes and the intensities in Western blots was analyzed with Student's
RESULTS AND DISCUSSION
We have previously shown that cultured patient CADASIL HUmbVSMCs have altered expression of several proteins involved in actin polymerization and VSMC contraction, and that CADASIL VSMCs have impaired spontaneous contractility.
20
In this study, we have analyzed the association of CADASIL mutations and shRNA silencing of
Growth Characteristics of the Vascular Smooth Muscle Cells
Vascular smooth muscle cell lines derived from blood vessels of different organs shared common features, but were morphologically different from each other (Table 1). HUmbVSMCs, HPlaVSMCs, and HArtVSMCs appeared as typical VSMCs. 22 They were large, elongated and had prominent nucleus. In culture, they oriented themselves into parallel bundles, became spindle-shaped and have a tendency to grow in multilayered aggregates. CADASIL and control HCerVSMCs were more irregular in shape than HArtVSMCs and their appearance resembled the epithelioid-like phenotype of VSMCs isolated from human thoracic artery. 23 All CADASIL VSMCs were larger in size and had slower proliferation rate than the corresponding control VSMCs, consistent with a previous study performed with HEK293 cells expressing mutant NOTCH3 (p.R133C or p.C185R). 24 The proliferation rates of the VSMCs varied according to the origin of the cell line: HPlaVSMC > HArtVSMC ≥ HUmbVSMCs > HCerVSMC.
Expression and Cellular Localization of NOTCH3
We followed the expression of NOTCH3 by immunostaining with the 1E4 antibody, which is directed against N3ECD, and detects both N3ECD and full-length NOTCH3. Cells were fixed with paraformaldehyde for analysis of cell surface and with methanol for analysis of intracellular NOTCH3.
Subcellular localization of NOTCH3 in CADASIL vascular smooth muscle cells (VSMCs). For detection of cell surface NOTCH3, VSMCs were fixed with 3.5% phosphate-buffered paraformaldehyde. NOTCH3 forms more aggregates on the cell surface in CADASIL than in control VSMCs (
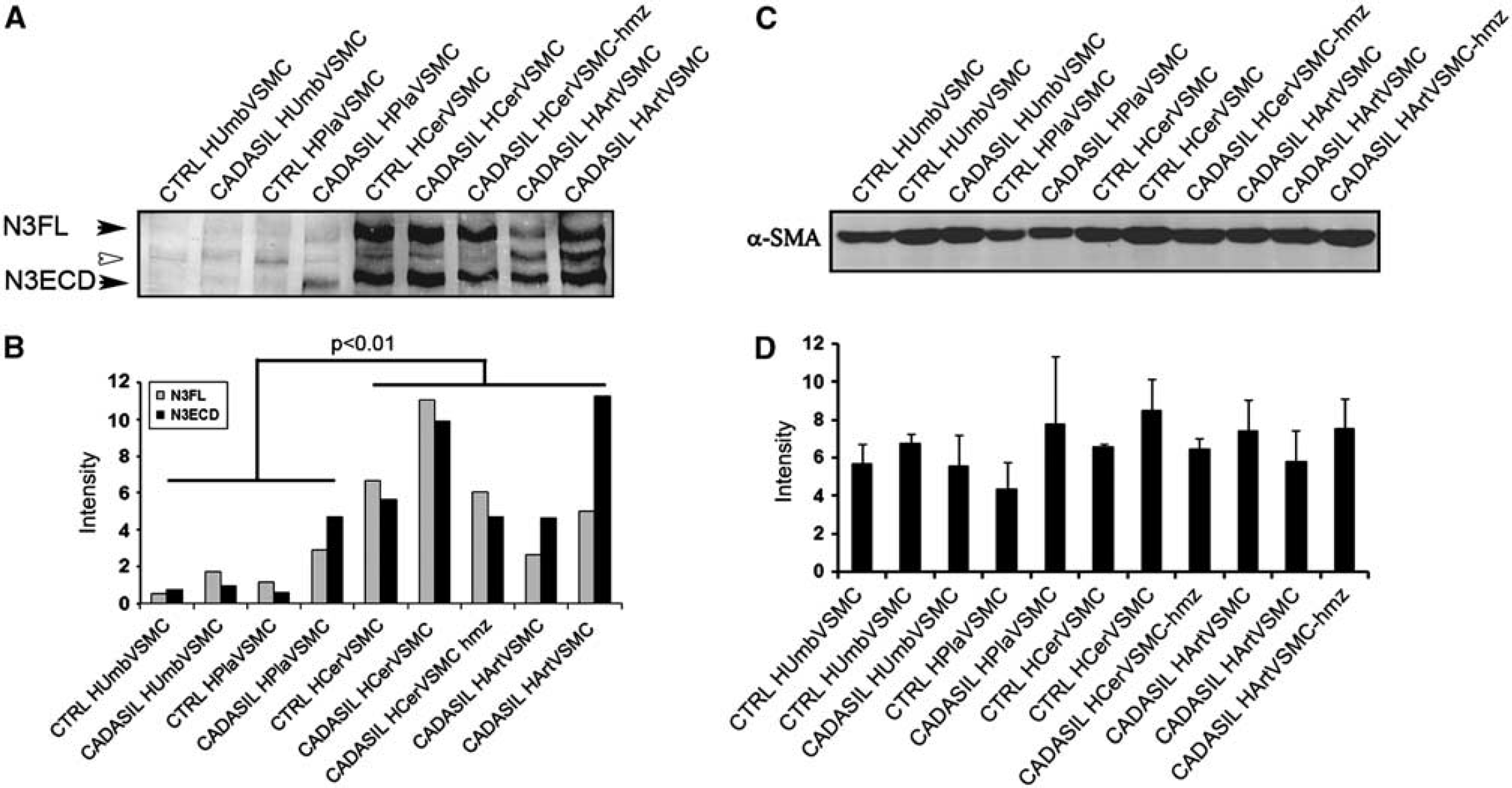
Total NOTCH3 and α-SMA expression in CADASIL and control vascular smooth muscle cells (VSMCs). (
Several different experimental models using transfected cells have been applied to analyze the processing of mutated NOTCH3. These have shown impaired or protracted S1 cleavage27,29 or have indicated that mutated NOTCH3 is correctly processed.28,30 Because in our Western blot analysis, there was no statistically significant difference in the full-length NOTCH3/N3ECD-ratio between CADASIL versus control VSMC lines, the S1 cleavage of mutated NOTCH3 appears to occur normally in CADASIL VSMCs. The data suggest that in human VSMCs with endogenous NOTCH3 expression, the microenvironment in Golgi and endoplasmic reticulum is able to prevent intracellular interreceptor crosslinking and aggregation of the mutated receptor. Thus, the aggregation does not occur until the receptor reaches the cell surface and therefore the accumulation of N3ECD on VSMCs in CADASIL is most likely due to its local aggregation and/or its impaired clearance.
Actin Filaments
Experimental studies have provided evidence of the role of NOTCH3 in actin filament polymerization
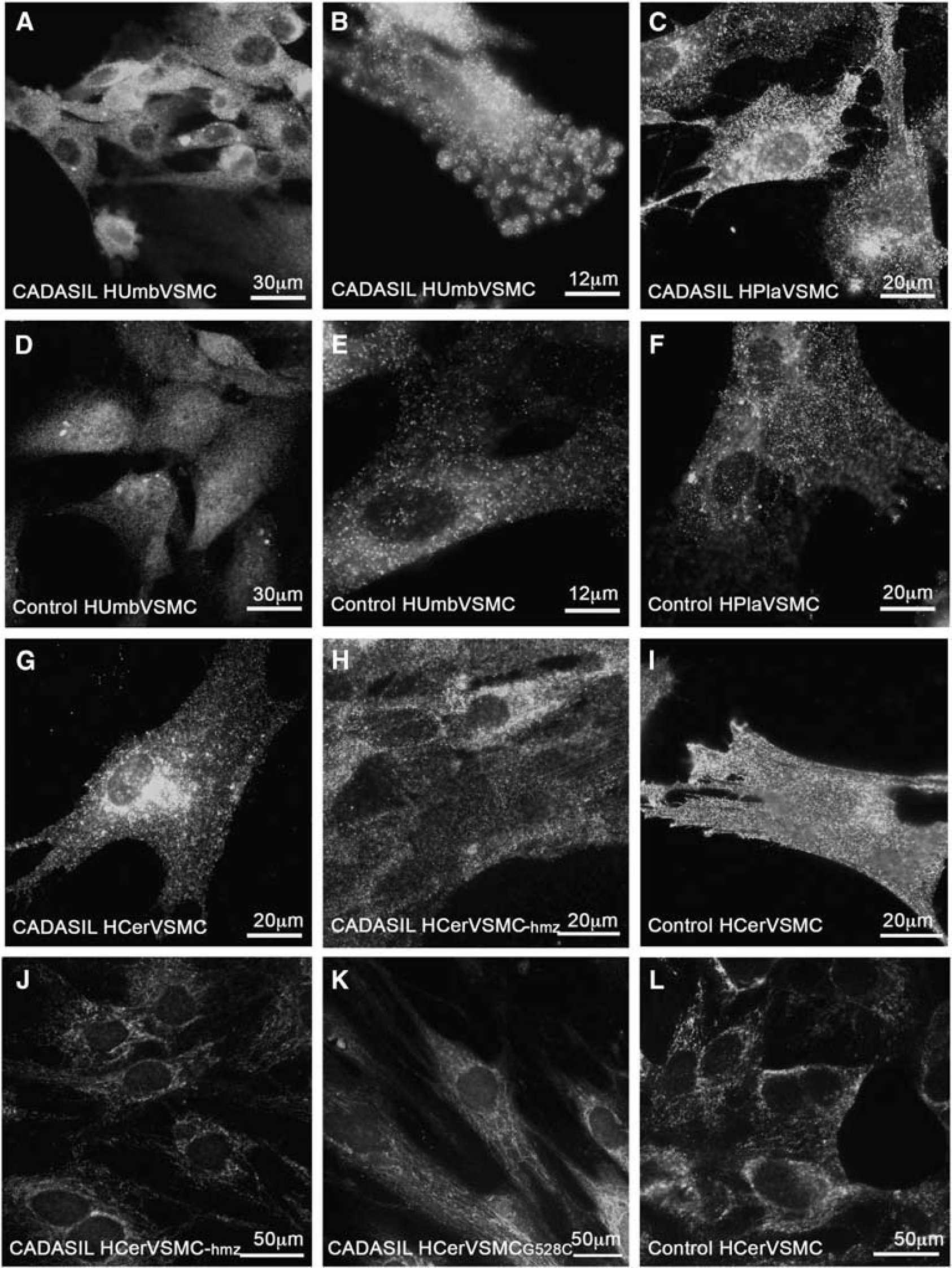
The staining revealed altered organization of actin filaments in CADASIL VSMCs as compared with control VSMCs (Figure 3). In all CADASIL VSMC lines isolated from the newborn (HUmbVSMCs and HPlaVSMCs; Figure 3A: CADASIL HUmbVSMC, D control HumbVSMC), the abnormalities in actin filaments were only minor: many of the cells appeared as control cells and about 39% had minor abnormalities in the organization of the actin cytoskeleton (representative images shown). Abnormalities in the organization of actin filaments were most severe in CADASIL HCerVSMCs: on average 72% of the HCerVSMCs had major abnormalities in the organization of the actin cytoskeleton (representative images shown) (Figures 3B, C, and F, inset I, control E, inset H). In 64% of HCerVSMCs from a heterozygous p.Arg133Cys patient (Figure 3B), actin filaments circled the cell and there were few nodes. In 69% of HCerVSMCs derived from a CADASIL patient with heterozygous p.Gly528Cys mutation showed similar nodes and circular orientation of filaments (Figure 3C). In 82% of HCerVSMCs-hmz (Figure 3F, inset I), actin filaments were in robust bundles and they formed several nodes with clusters of haphazardly oriented filaments (representative image shown). Such substantial deviations were not detected in any of the heterozygous CADASIL VSMC lines. In all, 60% of CADASIL HArtVSMCs also exhibited prominent abnormalities: fewer and shorter actin filaments, some with abnormal nodes and increased branching (Figure 3G). Similar correlation of altered actin filament organization was observed between HCerVSMCs and HArtVSMCs cell lines established from same patient (p.R133C; age 64).
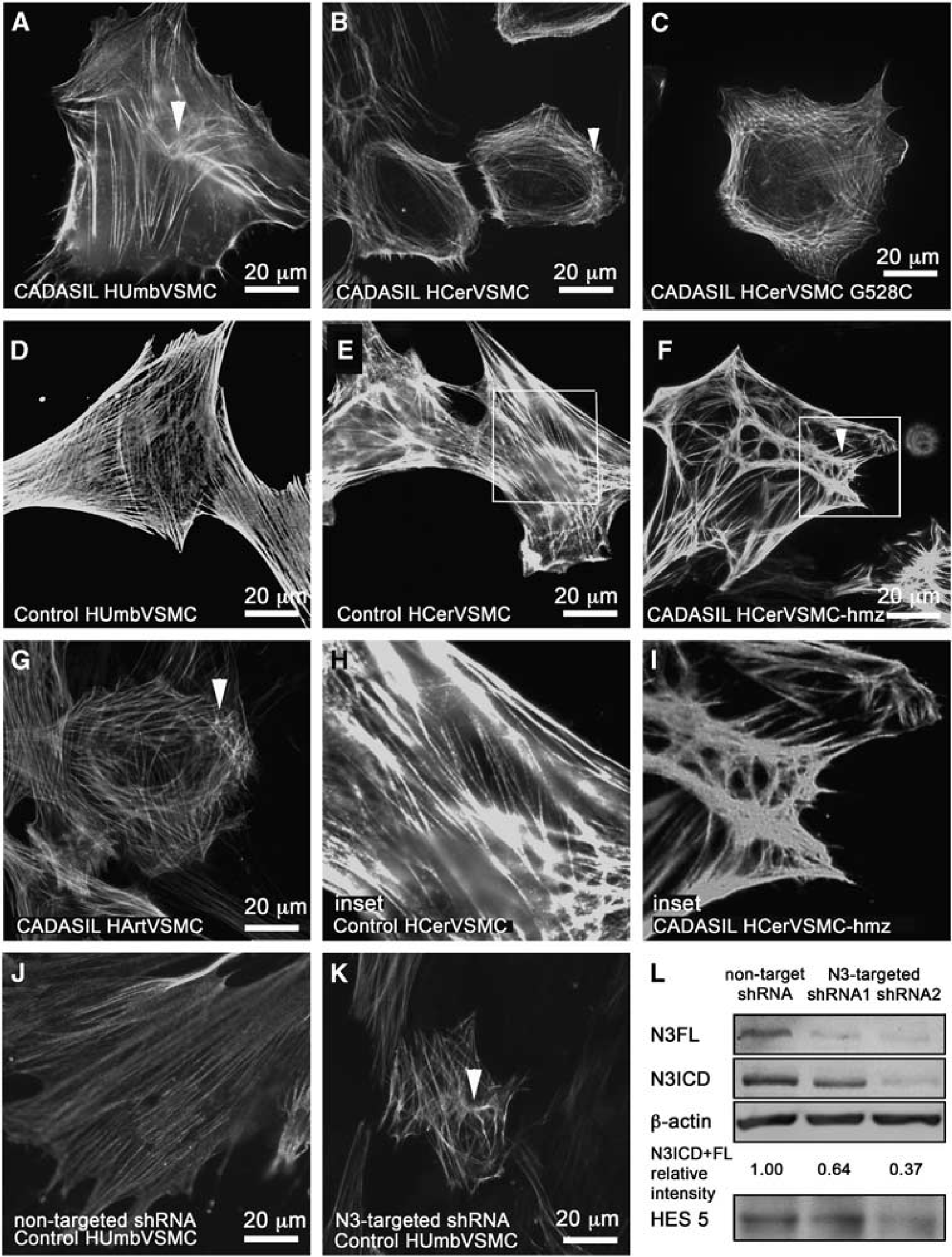
Actin cytoskeleton in CADASIL vascular smooth muscle cells (VSMCs). VSMCs fixed with methanol were immunostained for smooth muscle cell α-actin (α-SMA). α-SMA filament have altered organization in CADASIL VSMCs. In all CADASIL VSMCs the α-SMA positive actin filaments are short, poorly organized and form nodes (clusters of short haphazardly oriented filaments, selected five marked with arrowheads) (
Vascular smooth muscle cell lines from adult arteries (HCerVSMCs and HArtVSMCs) produced greater amount of NOTCH3 than the VSMCs from newborn blood vessels (HUmbVSMCs and HPlaVSMCs) (Figure 2). Therefore, the degree of abnormalities in actin filaments appeared to correspond to the NOTCH3 expression level. In addition, the actin abnormalities where more prominent in homozygous cell line compared with heterozygous cell lines (82% versus 69% and 64%) (Figure 3), supporting the role of NOTCH3 in organization of actin cytoskeleton network. To see if the NOTCH3 mutation affects the whole actin cytoskeleton and not just the α-SMA organization, we performed total actin filament staining with phalloidin (actin α, β, and γ). Phalloidin staining showed similar irregular filaments as α-SMA immunostaining (not shown), which suggests that the whole actin filament cytoskeleton is disorganized in CADASIL cells. Our results suggest that homozygousity gives rise to more severe phenotype in VSMCs, although patient studies have not shown major difference between homozygous and heterozygous patients. 33 This may reflect the effects of genetic modifiers, life choice factors, and complexity of intact tissue compared with cell culture.
We have demonstrated that
Although the organization of actin filaments showed variable degrees of abnormalities, Western blot analyses disclosed no statistically significant quantitative alteration of α-SMA levels between CADASIL and control VSMC cell lines (Figures 2C and 2D).
Recently, a weak hypomorphic
Our results showed that CADASIL patient's VSMCs derived from blood vessels of different vascular beds are differentially affected by the disease. Furthermore, it has been demonstrated in
Adhesion Sites in Cerebral Autosomal Dominant Arteriopathy with Subcortical Infarcts and Leukoencephalopathy Cells
Cell adhesion is regulated by dynamic assemblies of structural and signaling proteins that link the filamentous actin cytoskeleton to the ECM. To assess the major CADASIL-related differences in VSMC adhesion sites, we analyzed the distribution of adhesion molecules, which are associated with the organization of actin filaments, in two CADASIL (HUmbVSMC and HArtVSMC) and two control (HUmbVSMC and HCerVSMC) VSMC lines.
Arterial abnormalities in CADASIL include widening of the subendothelial space, thickening of the basement membrane and detachment of VSMCs from each other (Figure 4H).
38
Vinculin is a key molecule at adhesion sites (focal complexes and focal adhesions) anchoring actin filaments to the membrane bound integrins. Immunostaining of VSMCs for vinculin showed a markedly greater number of adhesion sites in CADASIL than in control VSMCs derived from newborn tissue (HUmbVSMC) (Figures 4A and 4C). These adhesion sites were spread widely over the basal surface of the CADASIL VSMCs, whereas in the control VSMCs they were mostly at cellular extensions. The sizes of individual adhesion sites were smaller in CADASIL than in control HUmbVSMCs (E): in CADASIL HUmbVSMCs the average size of individual adhesion sites was 40.7% (±3.5%) of that in control HUmbVSMCs (
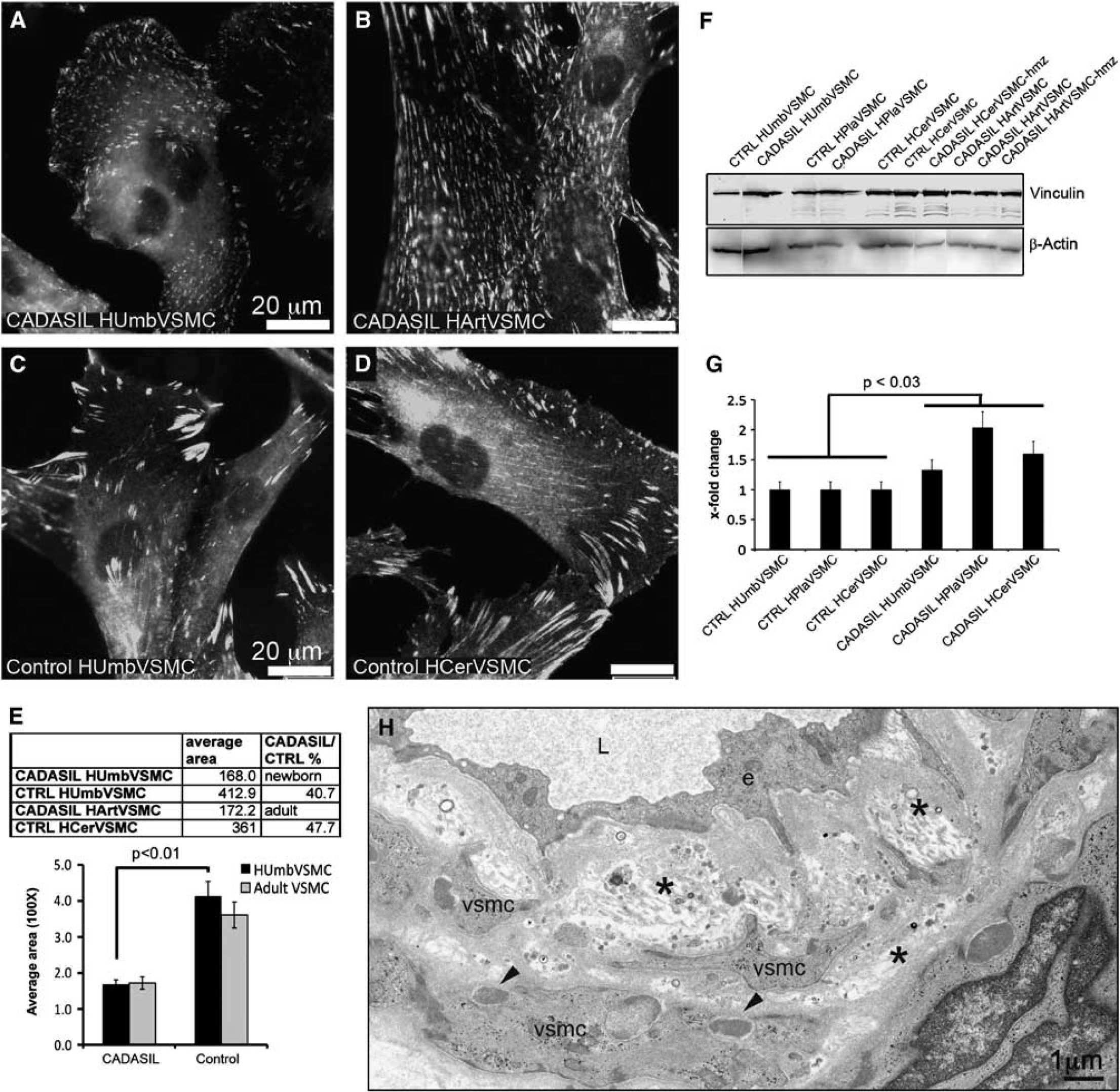
Vinculin positive adhesion sites in CADASIL vascular smooth muscle cells (VSMCs). Methanol fixed cells immunostained for vinculin show markedly greater number of small-sized adhesion sites in (
Adhesion sites are large multimolecular complexes that interconnect cells to neighboring cells or to the surrounding ECM. Adhesion is mediated by transmembrane integrins and associated cytosolic adapter proteins, which provide a direct link to the actin filaments. In VSMCs integrins (mainly αvβ3 and α5β1) are considered to be involved for example in the regulation of contractile function and myogenic behavior.
39
Adhesion sites are dynamic structures, which are transformed by continuous association and dissociation of proteins according to received signals and thus their molecular composition is heterogeneous. In culture, the adhesion sites are transformed (matured) from (1) early state
To assess the maturation of the adhesion sites, we analyzed the distribution of β1 and β3 integrins and the adapter protein tensin. Integrin β1 appeared as ribbon-like structures, which were more frequent in CADASIL than in control VSMCs. In both CADASIL and control VSMCs (Figures 5A–5F), integrin β1 positive ribbon-like structures were either positive or negative for vinculin, the former representing focal complexes or focal adhesions and the latter mature fibrillar adhesions. 40 Integrin β3 localized to the same structures as vinculin and did not show substantial difference between CADASIL and control VSMCs (data not shown). Remarkably, a greater number of adhesion sites were positive for tensin in CADASIL than in control VSMCs. (Figures 5G–5L) and the average volume of tensin immunoreactivity was significantly higher in CADASIL cells compared with control cells (P < 0.01) (Figure 5M).
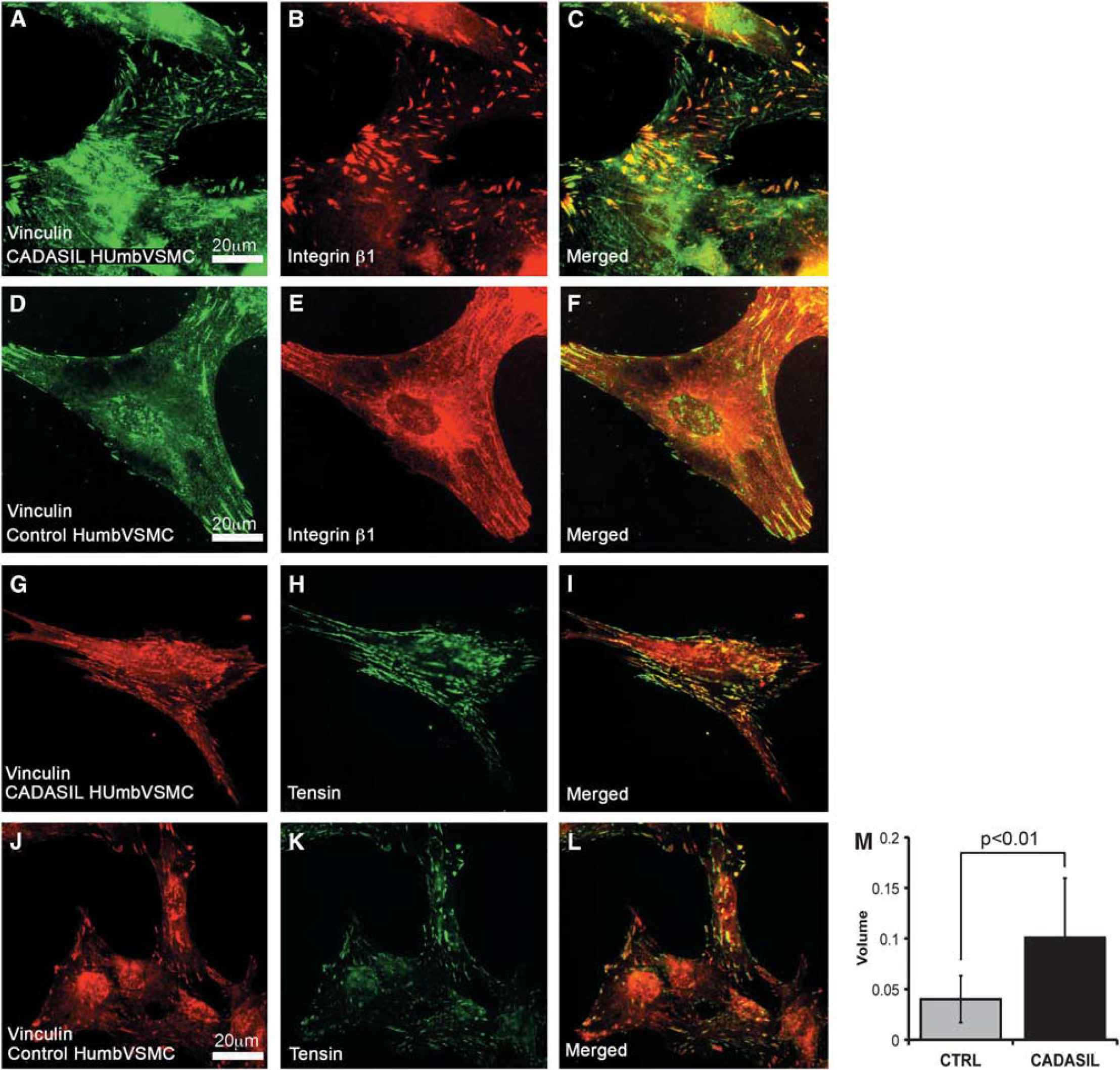
Integrin β1 and adapter protein tensin in adhesion sites. Methanol fixed vascular smooth muscle cells (VSMCs) were immunostained with integrin β1 or tensin antibody. (
Both talin and tensin are adapter proteins that bind to the same region in the tail of β integrins. 41 Talin is usually found in early adhesion (focal complexes and focal adhesions) and it functions mainly as a structural adaptor, whereas tensin is usually found in more mature adhesion sites (late focal adhesions and fibrillar adhesions) and it appears to assemble a signaling complex. 40 Thus, the switch of talin to tensin in binding to integrins determines the change from a structural adhesion to an adhesion that interacts with signaling.
Structure, subcellular distribution, and molecular composition of adhesion sites in CADASIL VSMCs suggests a switch in the balance of adhesion sites from focal complexes towards more mature and stable focal and fibrillar adhesions in CADASIL VSMCs. This change in adhesion site maturation together with altered actin cytoskeleton might affect the ability of the cells to respond to different stimuli, for example to narrowing or dilating the arterial lumen. Interestingly, also VSMCs of transgenic mice expressing human NOTCH3 with a mutation p.Arg90Cys exhibit cytoskeletal changes with the presence of more numerous and larger cytoplasmic dense bodies as well as more frequent and thicker dense plaques (tissue equivalent to adhesion sites in cultivated cells). 42 The observed enlargement of subendothelial space of the arteries and the loss of intercellular connections of VSMCs in CADASIL patients (Figure 4H)4,5 and in mouse expressing mutated NOTCH342 might be caused by alterations in actin filament structures, deviant formation of the adhesion sites or changed integrin affinity/avidity, which all could be initiated by abnormal NOTCH3 function.
CONCLUSIONS
Cultured genuine patient derived CADASIL VSMCs expressing endogenous mutated
CONFLICT OF INTEREST
The authors declare no conflict of interest.
