Abstract
We report a case of prolonged motivational deficit as a sequela of high altitude cerebral edema (HACE), the most severe form of neuropsychiatric dysfunction arising from traveling to high altitude. Magnetic resonance imaging of the brain showed hyperintense lesions in the globi pallidi bilaterally on T2-weighted images. Single-photon emission computed tomography showed hypoperfusion in dorsolateral and orbital prefrontal cortices bilaterally and in the anterior cingulate cortex. This case suggests that a prolonged motivational deficit can occur in patients with HACE. The case may also suggest that HACE can cause network disturbances between the prefrontal cortex and the globi pallidi.
Introduction
High altitude cerebral edema (HACE) is the most severe form of high altitude neurologic illness. Altitude illness includes several forms of neuropsychiatric dysfunction, including high altitude headache, acute mountain sickness, 1 and HACE. 2 HACE presents with the most severe manifestations, including disturbance of consciousness and ataxia. 3 Although many cases of nonfatal HACE have been reported, 4 -8 the neuropsychiatric course and prognosis are still unclear. One report 9 described 2 cases with subcortical dementia after HACE and drew attention to persistent neuropsychiatric dysfunction. The mechanism of dysfunction remains unclear. We report a patient who presented with apathy, a motivational deficit that affects emotional, cognitive, and behavioral functioning, after the symptoms of HACE resolved.
Case Report
A 36-y-old, right-handed Japanese male with no history of neurologic, psychiatric, or physical disorders traveled from Tokyo, Japan (40 m above sea level) to La Paz, Bolivia (3640 m above sea level), for the purpose of climbing. After 1 wk in La Paz, he was found comatose in his hotel. There was no history of alcohol or drug use.
He was transported to a local hospital in La Paz where he was diagnosed with HACE and high altitude pulmonary edema. He recovered consciousness after 3 d of treatment. He was discharged from the hospital 6 d after regaining consciousness. He then returned to Japan.
After returning to Japan, he had lost motivation, which caused difficulty in working. Three months after this episode, he presented to our hospital with marked motivational deficit on the Japanese version 10 of Starkstein’s apathy scale, 11 with a score of 28 points (range: 0–42 points, cutoff score of ≥16 indicates apathy). In particular, he showed a lack of interest in learning new things or in new experiences and a lack of concern about his personal problems, and he had a flat affect. He had no depression-related symptoms other than motivational deficit. Clinical examination by a neurologist (MI) found no hard neurological signs, including motor abnormalities. His mini-mental state examination 12 score was 29 of 30; he lost a point only on orientation in place, which is normal. His scores were normal on the third-edition Wechsler adult intelligence scale, 13 Benton visual retention test, 14 trail making test, 15 and Wisconsin card sorting test. 16
Magnetic resonance imaging (MRI) of the brain showed hyperintense lesions in the globi pallidi bilaterally on T2-weighted images (Figure 1A). Additionally, on T2∗-weighted gradient-echo MRI, punctate intensities were seen in white matter areas, especially in the corpus callosum (Figure 1B). Single-photon emission computed tomography (SPECT) showed hypoperfusion in dorsolateral, orbital prefrontal cortices and in the anterior cingulate cortex (ACC) bilaterally (Figure 2). His motivational deficit was partially improved by treatment with amantadine, an agent used for the modulation of dopaminergic systems, and nicergoline, an agent used for the management of cognitive function disorders. The Japanese version of Starkstein’s apathy scale score recovered to 20 points after 16 mo of treatment.
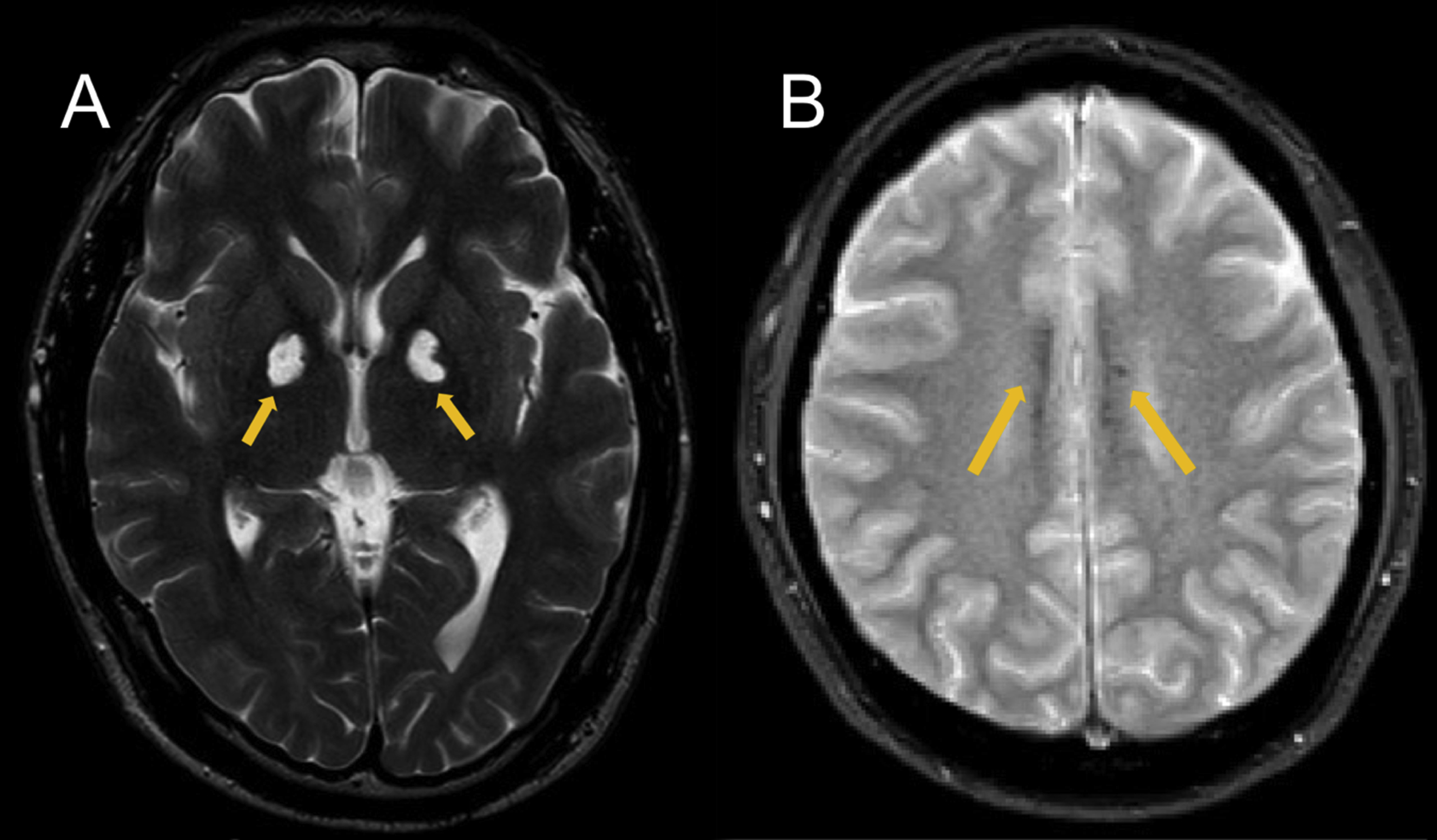
Brain magnetic resonance images of the patient. A, T2-weighted image. Yellow arrows indicate hyperintense lesions in the globus pallidi bilaterally. B, T2∗-weighted image. Yellow arrows indicate punctate intensities in the corpus callosum.
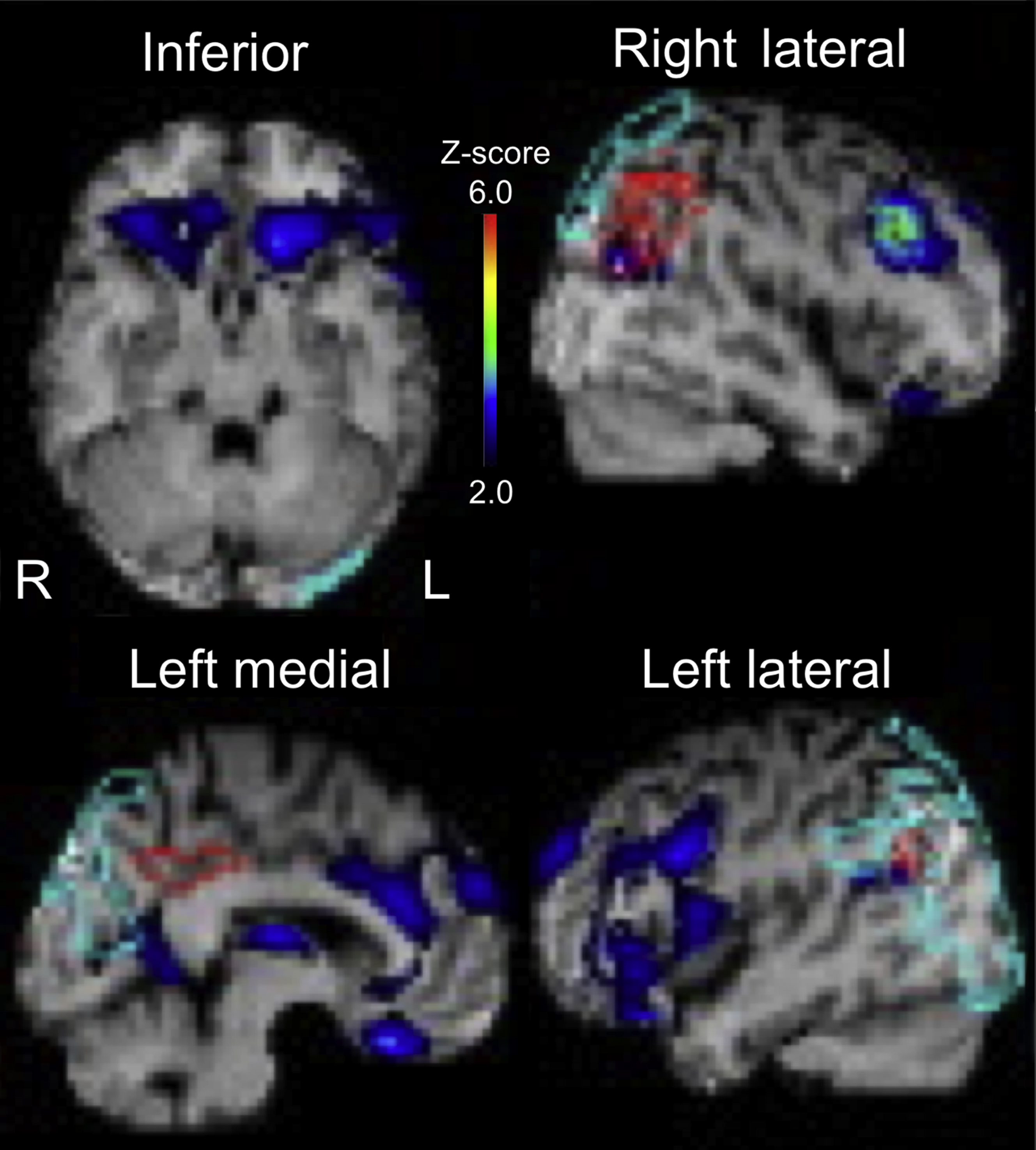
Result of easy Z-score imaging system analysis of brain single-photon emission computed tomography images of the patient. A higher Z-score indicates lower regional cerebral brood flow. Hypoperfusion included the orbital prefrontal cortices (inferior view), anterior cingulate cortex (left medial view), and dorsolateral prefrontal cortices (right and left lateral view).
Discussion
Apathy is a syndrome of primary motivational loss (ie, not attributable to emotional distress, intellectual impairment, or diminished level of consciousness). 17 The MRI and SPECT examinations showed bilateral lesions in the globi pallidi, subcortical structures that are part of the basal ganglia, and hypoperfusion in the prefrontal cortical areas and ACC. The globi pallidi are known to have abundant connections with the prefrontal cortex. 18 Previously, patients with bilateral lesions in the globi pallidi have been reported to manifest frontal lobe symptoms including apathy,19,20 suggesting possible impaired communications between the 2 areas. 21 Pathology and neuroimaging studies have provided evidence supporting this notion. 22 The observations in the present case suggest that frontal lobe symptoms after HACE may arise from disturbances of circuits between the prefrontal cortex and the globi pallidi because of impairment of the pallidi. Research techniques such as diffusion tensor imaging would be necessary to directly examine circuit disturbances between these regions. The present case had different sequelae than those in 2 other reported cases of subcortical dementia. 9 The cause of differences among patients is unclear. It may be that there are variations in the areas of the globi pallidi that are affected by HACE because the globi pallidi have subregions that connect to different areas in the prefrontal cortex.18,21 Another possible cause is the difference in age. The patient in the present case was 36 y old, whereas the patients in the 2 other reported cases with subcortical dementia were both in their 60s. 9 Study of additional cases is necessary to understand how symptoms that occur after HACE are associated with damaged areas in the globi pallidi and with aging.
On T2∗-weighted gradient-echo MRI, our patient showed punctate intensities in white matter areas including the corpus callosum, indicating microhemorrhages (MHs) in these regions. MHs in the corpus callosum is an MRI finding specific for HACE rather than acute mountain sickness, and is associated with greater clinical severity of HACE.7,23,24 Although the mechanism by which HACE induces MHs in the corpus callosum is unclear, it has been suggested that disruption of the blood–brain barrier is due to hypoxic overperfusion predominantly in the corpus callosum, which is supplied by small, short, and perforating arteries. 3 Therefore, MRI with T2∗-weighted gradient-echo and especially susceptibility-weighted images may be useful to diagnose HACE in patients with neurological issues after travel to high altitude. 7
In this patient with prolonged apathy after the resolution of HACE, brain MRI showed bilateral lesions in the globi pallidi and MHs in white matter areas including the corpus callosum. Using SPECT, hypoperfusion in the prefrontal cortical areas and ACC were observed. This suggests that frontal lobe symptoms can occur after HACE owing to network disturbances between the prefrontal cortex and the globi pallidi. This observation may lead to a better understanding of the pathophysiological mechanisms underlying the development of apathy in patients with HACE.
Footnotes
Acknowledgements
Acknowledgments: We thank the technical staff at Nihon University Itabashi Hospital for the acquisition of psychological and image data.
Author Contributions: Study concept and design (YK, MS, MK, MU); acquisition of data (MS, MI, SB, HN); interpretation of data (YK, MS, TS, KY, HK, MU); drafting of the manuscript (YK, MI, MK); critical revision of the manuscript (MS, SB, TS, KY, HK, MU); and approval of final manuscript (all authors).
Financial/Material Support: None.
Disclosures: YK reports grants and personal fees from Idorsia Pharmaceuticals Japan, outside the submitted work. MS reports personal fees from Otsuka Pharmaceutical, Eisai, Meiji Seika Pharma, MSD, Pfeiser, Takeda Pharmaceutical, and grants from Novartis, outside the submitted work.
