Abstract
We present the case of a patient who developed myalgia as the primary symptom of envenomation by the eastern coral snake, Micrurus fulvius. The patient was evaluated and treated in the emergency department. Physical examination did not demonstrate any neuromuscular abnormalities. On consultation with the poison control center, the patient’s myalgia was determined to be an effect of envenomation, and 5 vials of North American coral snake antivenin were administered. The patient was admitted to the intensive care unit where his symptoms resolved. He was discharged the following day after remaining asymptomatic for 24 h.
Introduction
Coral snakebites are rare in the United States, accounting for only 25 to 50 of around 9000 (0.3–0.6%) annual total snakebites in the country. 1 The 3 coral snake species native to the United States and surrounding areas include Micrurus fulvius (eastern coral snake, ranging in the southeast United States), Micrurus tener (Texas coral snake, ranging in Texas, Louisiana, and northwestern Mexico), and Micruroides euryxanthus (Sonoran coral snake, ranging in Arizona and New Mexico as well as the Mexican state of Sonora) (Figure 1). 1 M fulvius is the only elapid snake found east of the Mississippi river, and unlike other venomous snakes in the eastern United States, its venom is neurotoxic by pre-synaptic and post-synaptic inhibition at the neuromuscular junction. The venom circulates via the lymphatic system and can lead to muscle weakness, with the most life-threatening complication being respiratory muscle weakness and pulmonary insufficiency that may be fatal if untreated.
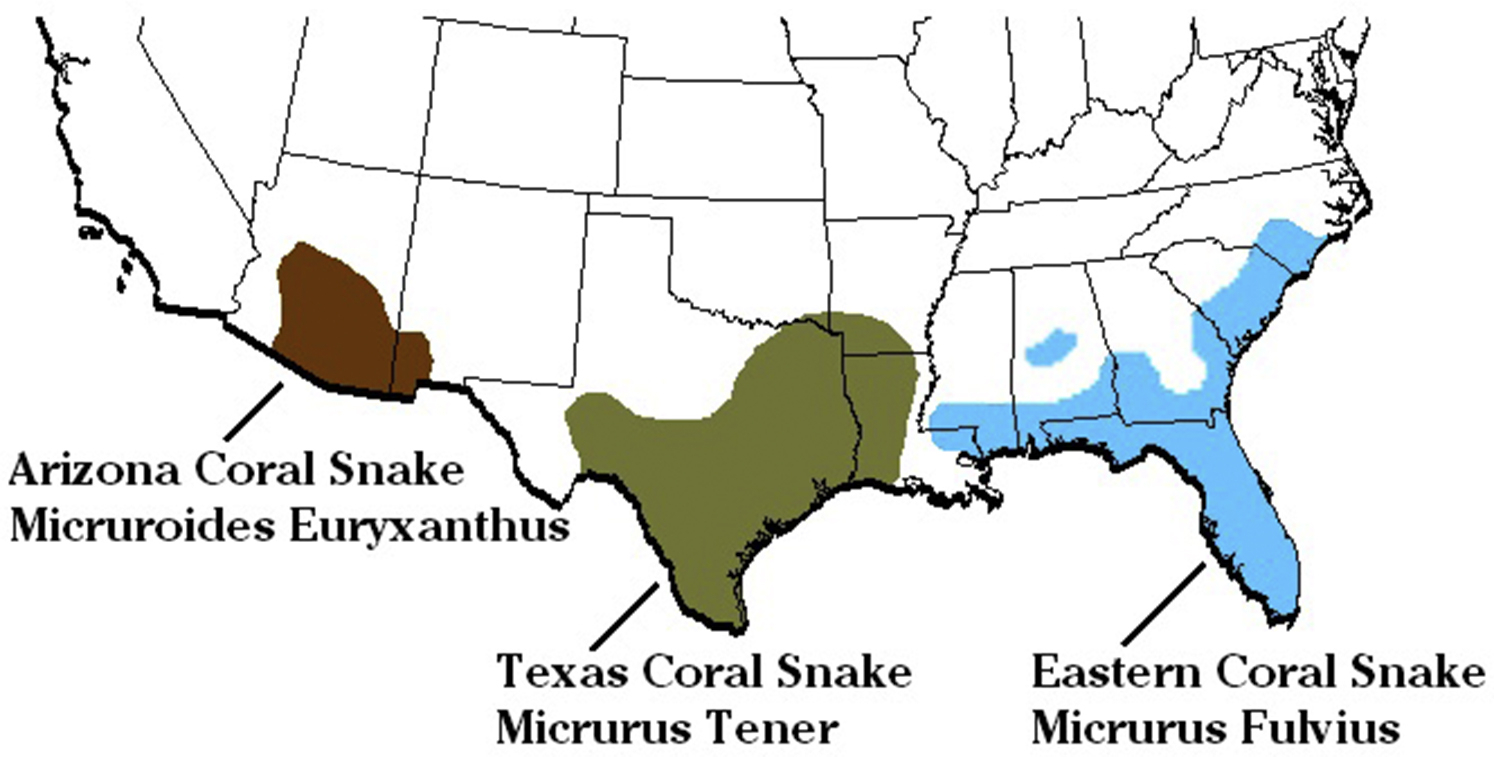
Range map of the 3 coral snake species found in the United States. Image by Howard Morland and released into the public domain.
Case Presentation
A 60-y-old male with a history of hypertension, alcohol dependence, splenectomy after a traumatic splenic laceration, and envenomation by a cottonmouth snake, Agkistrodon piscivorus, requiring antivenin administration over a decade prior presented to the emergency department (ED) via ambulance after receiving a bite from a coral snake approximately 1 h before arrival. He noticed the snake in a coop outside and sustained a bite on his left index finger while attempting to remove the snake. He captured the snake and placed it in a jar. The paramedics arrived and obtained a photograph of the snake (Figure 2). The patient then released the snake. He only sustained the single bite. On arrival, he complained of swelling to the left index finger and pain gradually radiating up his left arm with associated left arm stiffness.

Photograph of the snake that bit the patient, confirming its identity as the eastern coral snake Micrurus fulvius. Important features include color bands fully encircling the body, and every other body ring is yellow.
Presenting vital signs in the ED were temperature of 36.8° C, heart rate of 79 beats·min-1, blood pressure of 173/100 mm Hg, respiratory rate of 16 breaths·min-1, and oxygen saturation of 98% on room air. On physical examination, he had superficial wounds and edema on the flexor surface of the left second finger distal interphalangeal joint with a small amount of dried blood (Figure 3). He maintained full range of motion and 5/5 strength in the fingers, wrists, elbows, and shoulders bilaterally. He was awake and alert with no focal neurologic deficits, intact cranial nerves II-XII, normal sensation, normal reflexes, normal coordination, normal muscle strength in his lower extremities, and normal tone. The left second finger had a capillary refill of less than 2 s. His left hand did not have any cyanosis, and his fingers did not demonstrate clubbing. He had intact radial pulses bilaterally. His cardiac exam demonstrated a regular rate and rhythm, normal S1 and S2 sounds, no murmurs, no rubs, no gallops, and no clicks. His lungs were clear to auscultation bilaterally with normal respiratory effort. His abdomen was soft, nontender, nondistended, and did not demonstrate rebound or guarding.
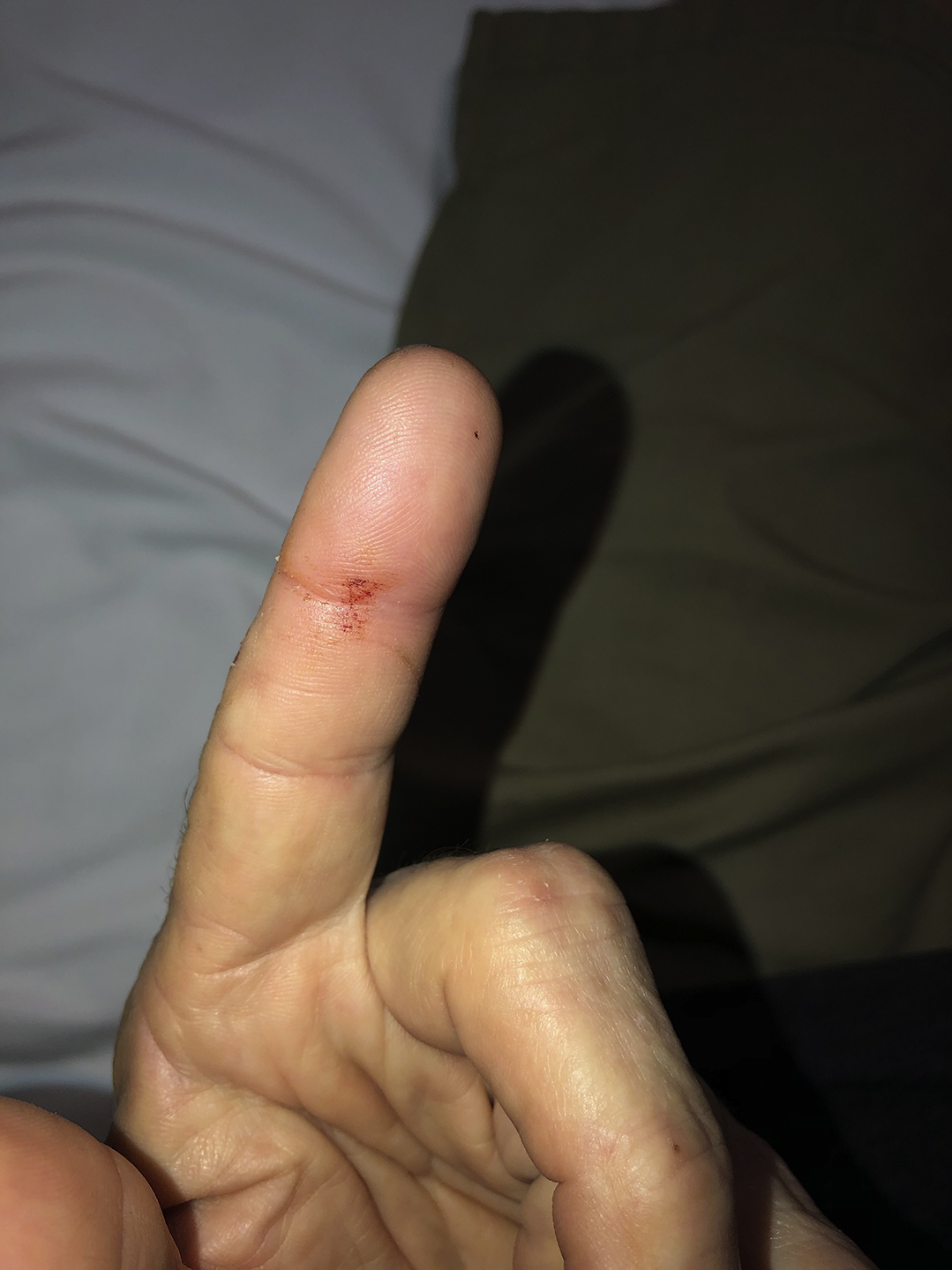
Photograph of the bite mark on the patient’s left index finger.
The patient was placed on continuous cardiac monitoring. An x-ray of the left hand was obtained that did not demonstrate any retained teeth in the soft tissue. His tetanus vaccine was updated. He was given 4 mg of morphine for his pain. Laboratory evaluation included complete blood cell count, complete metabolic panel, creatine phosphokinase, prothrombin time, and international normalized ratio and did not show any abnormalities. An electrocardiogram was not obtained.
University of Florida (UF) Health Jacksonville: Florida poison information center was consulted, and the toxicologist on call recommended the patient elevate the left upper extremity at or above the level of the heart. The toxicologist also requested 5 vials of North American coral snake antivenin (NACSA) (produced by Wyeth, a subsidiary of Pfizer) due to the patient having the neurologic symptoms of tingling and myalgia in his left upper extremity. The antivenin was brought via ambulance from Lake County fire rescue’s venom 2 unit.
Before the administration of antivenin, the patient was informed of the risks of allergic reaction and possibility of serum sickness, a type III hypersensitivity reaction that may develop days to weeks after administration and includes symptoms such as fever, chills, joint aches, and muscle cramps. The patient verbalized understanding and agreed to proceed with antivenin administration. The patient also reported tolerating a different antivenin for his A piscivorus bite over 10 y prior without anaphylaxis, serum sickness, or any other adverse event. The patient received 5 vials of NACSA over 1 h by the ED registered nurse. The antivenin was administered approximately 4 h after envenomation.
The patient was subsequently admitted to the intensive care unit (ICU) for frequent monitoring of his neurologic and pulmonary status. Prior to transfer to the ICU, the patient’s vital signs were temperature of 36.8° C, heart rate of 66 beats·min-1, blood pressure of 179/97 mm Hg, respiratory rate of 16 breaths·min-1, and oxygen saturation measured by pulse oximetry (SpO2) of 97% on room air. His symptoms resolved later in the afternoon, and he never developed respiratory distress. Per recommendations of the poison control center, the patient was monitored for 24 h once asymptomatic and subsequently discharged the day after presentation to the ED. At discharge, the patient’s vital signs were temperature of 36.9° C, heart rate of 75 beats·min-1, blood pressure of 125/74 mm Hg, respiratory rate of 22 breaths·min-1, and SpO2 of 99% on room air. The patient was instructed to follow up with his primary care physician in 3 d. He was advised to return to the emergency department immediately if he developed increased swelling or erythema at the bite site or difficulty breathing. Although it is unknown whether he followed up as instructed, he did return to the ED 6 mo later for an unrelated complaint.
Discussion
Coral snakes are the only terrestrial snakes native to the United States belonging to the Elapidae family. This family is diverse and widespread, including other types of snake such as cobras, sea snakes, and sea kraits. Other species of coral snake are found in Central and South America as well as Asia. The venom of most Elapidae is neurotoxic, unlike the hemotoxic venom found in the US Crotalinae, or pit vipers. The patient in this case had previously been bitten by a pit viper, A piscivorus, over 10 y prior to presenting with the M fulvius bite.
In the United States, coral snakebites are uncommon compared to Crotalinae bites. The infrequency of coral snakebites is attributable to their living primarily underground and with peak periods of activity around dawn and dusk. 2 Deaths from coral snakes are rare in the United States, with only 1 fatality documented since 1967. 3 Coral snake-related fatalities are also rare in other regions. A review of coral snake envenomations in Argentina from 1979 to 2003 documented no deaths, and a review of coral snakebites in Brazil documented 6 reported fatalities in the period of 1867 through 2015, with no deaths reported since 1972.4,5 Asian coral snake envenomations remain understudied, and neurotoxicity from these species has rarely been documented. 6
A key initial step in treating patients presenting with snakebites is identification of the snake. This may be straightforward when patients are bitten by pet snakes, but identification is more difficult when patients are bitten by wild snakes. Clear pictures capturing the entire body of the snake are optimal, taken from a distance of the length of the snake to be outside of the snake’s striking range. 7 Although people may be tempted to kill the snake, this will make the snake engage in defensive behavior and may result in a second bite.
All 3 coral snake species native to the United States typically have bands of black, yellow/white, and red coloration. They usually have black noses and every other midbody ring is yellow/white. The bands completely encircle the body, going across the belly as well. These features differentiate coral snakes from several nonvenomous mimic species in their US range. 7 A common rhyme for coral snake identification, “red on yellow, kill a fellow; red on black, venom lack” is generally true only of coral snake species native to the United States. However, regional and anomalous variations of M fulvius and M tener have been documented, and an expert should be consulted if the identification of the snake is in doubt. 7
Outside the United States, coral snakes demonstrate an array of colorations and patterns. Some species in Central and South America have red bands touching black bands, so the rhyme popular in the United States is not applicable elsewhere. Other South American species lack any banding. Many Asian coral snake species appear unlike their American counterparts, with some species lacking bands and others possessing longitudinal bands. Some Asian coral snakes incorporate additional colors such as gray, brown, and blue.
Clinical Presentation
Coral snake venom is neurotoxic. In the most concerning presentation, envenomated patients develop a descending paralysis with ptosis and bulbar palsies appearing first. Early symptoms may include slurred speech and double vision. If untreated, fatalities from M fulvius envenomation are usually due to respiratory failure secondary to paralysis of the diaphragm muscle. 8 As this case demonstrates, however, initial symptoms may involve myalgia in the absence of abnormal neurologic physical exam findings. A review of 387 M fulvius envenomations in Florida between 1998 and 2010 found pain (41%) and paresthesia (28%) to be the most common clinical findings of M fulvius envenomation, with paralysis (3%) and respiratory depression (3%) being comparatively rare findings. 9 Symptoms of neurotoxicity were noted to take up to 13 h to manifest. 9 This review does not report myalgia as a symptom displayed by any of the 387 cases investigated. An older case series of 20 patients treated for M fulvius envenomations at UF Gainesville noted muscle tenderness in 10% of cases. 10
Because respiratory failure is the most feared complication of coral snake envenomation, it is important to assess the airway and respiratory functions. Respiratory function should be monitored using negative inspiratory force and forced vital capacity. Unlike Crotalinae envenomation, M fulvius envenomation does not typically result in laboratory abnormalities, and patients presenting after M fulvius bites do not require trending of laboratory values during treatment and observation.
Mechanism of Envenomation and Neurotoxicity
The mechanism of North American coral snake envenomation is unlike that of Crotalinae. Crotalinae have hinged fangs that can deliver venom very quickly while striking. Coral snakes have smaller fixed fangs in the front of their mouths. 11 A common misconception is that coral snakes must latch onto and “chew” to deliver venom to their target; in fact, coral snakes are capable of rapidly injecting venom with a brief bite. Unlike Crotalinae bites that often leave distinct puncture wounds, bites from coral snakes often leave no mark, and when marks are left they may be difficult to see on a patient’s skin. 12
M fulvius venom contains a mixture of proteins that result in neurotoxicity, with alpha neurotoxins leading to post-synaptic nicotinic acetylcholine receptor inhibition. 13 Another component, phospholipase A2, produces neurotoxicity by inhibiting pre-synaptic nicotinic acetylcholine receptors. 8 The venom travels via the lymphatic system and can ultimately lead to descending paralysis. Phospholipase A2 has also been shown to cause intravascular hemolysis, myotoxicity, and rhabdomyolysis in mouse models; however, there is no histologic evidence to suggest that similar effects occur in humans envenomated by M fulvius.14,15 Mild elevations of creatine phosphokinase, rhabdomyolysis, and coagulopathy have been reported in envenomations by the South American coral snake Micrurus lemniscatus helleri and the Asian banded coral snake Calliophis intestinalis, and patients bitten by these species will benefit from a more extensive laboratory workup than patients bitten by M fulvius.4,16,17
Snake venom lethality is described using the term median lethal dose, or LD50. The LD50 indicates the amount of venom needed to kill 50% of the test population if untreated, with a lower LD50 indicating a more lethal venom. The LD50 of M fulvius venom, determined using mice models, has been reported at 0.32±0.12 mg·kg-1 if administered intravenously (IV).8,18 In comparison, the LD50 of M tener venom has been reported at 0.78±0.14 mg·kg-1 IV, indicating that M fulvius venom is more deadly. 18 -20 The LD50 of M euryxanthus has not been determined, but no human fatalities have been attributed to bites from this species. 21
Some Central and South American Micrurus species also use phospholipase A2 as the primary neurotoxic component. Such species include M nigrocintus, with an LD50 of 0.3 to 0.5 mg·kg-1 IV, and M mosquitensis, with an LD50 of 0.20-0.61 m·kg-1 IV.22,23 Other Micrurus species in Central and South America utilize 3-finger toxins that can lead to postsynaptic neurotoxicity, cytotoxicity, cardiotoxicity, and anticoagulant and antiplatelet activity.24,25 Such species include M alleni, with an LD50 of 0.74±0.16 mg·kg-1 IV, and M clarki, with an LD50 of 0.42 to 1.38 mg·kg-1 IV.22,26 Species of Asian coral snake, including Calliophis intestinalis, have been documented to contain both phospholipase A2 and 3-finger toxins in their venom. 17
Treatment
The poison control center or in-house toxicology service should be consulted immediately for all coral snakebites. In the pre-hospital setting, a pressure dressing may be applied around and proximal to the bite area to slow lymphatic flow only if doing so will not delay transport to a healthcare facility. At the healthcare facility, the wound should be cleaned, and the patient should receive a tetanus vaccine if not up to date on vaccinations. An x-ray may be obtained to assess for retained teeth from the snake embedded in soft tissue. Snakebites rarely result in bacterial infections, so prophylactic antibiotics should not be administered unless the wound is contaminated or the patient has evidence of a bacterial infection. 27
The only Food and Drug Administration (FDA)-approved antivenin for M fulvius and M tener is NACSA. NACSA is a horse-derived IgG antibody that directly neutralizes the venom. Before the introduction of NACSA in 1967, the mortality rate of coral snake envenomations was around 10%. 28 The recommended dose is 3 to 5 vials immediately, but patients may require up to 15 vials with further vials administered for persistent or worsening symptoms at the recommendation of a medical toxicologist. 29 Neurologic sequelae may progress despite administration of antivenin. Therefore, antivenin should be administered at the first sign of possible neurotoxicity to reduce the chances of diaphragm paralysis. 28 NACSA is effective against the venoms of the Micrurus coral snakes native to the United States. The efficacy of NACSA against M euryxanthus venom is unproven, but supportive care is the mainstay of M euryxanthus envenomations as bites have only been recorded to result in minor effects. 3 The FDA-approved antivenin for bites from the Crotalinae native to the United States is crotalidae polyvalent immune fab (ovine) (produced by BTG, a subsidiary of Boston Scientific).
Horse-derived antivenin can cause both acute and delayed hypersensitivity reactions. 9 Acute hypersensitivity reactions are common and managed by stopping the infusion, laying the patient supine, and administering fluids and epinephrine. Refractory cases can be treated with epinephrine infusions for persistent hypotension and nebulized albuterol for persistent bronchospasm.
Serum sickness is a delayed hypersensitivity reaction that may occur 5 to 14 d after initial administration of antivenin. There is no current consensus on serum sickness symptomatology. Rash, fever, myalgia, pruritus, epigastric pressure, thrombocytopenia, anorexia, and arthralgia have been described. Treatment for serum sickness consists of steroids, antihistamines, and supportive care. 30
As coral snakebites are rare in the United States, NACSA is intermittently produced. NACSA is currently produced by Wyeth, a subsidiary of Pfizer, and is in adequate supply. In the past, NACSA has undergone several cycles of ceased production and extension of the expiration date until the existing supply has been depleted, requiring the production of more antivenin. This last occurred with a lot of NACSA initially marked with an expiration date of January 31, 2017 that was extended several times until January 31, 2020.
Although NACSA shortages are not an issue at this time, several alternative non-FDA approved treatments have been investigated. Anecdotally, expired antivenin may be fully effective if the product appears clear in the vial, but opaque product seems to have decreased efficacy. 31 For situations where neither in-date nor expired NACSA are available, animal studies have been performed suggesting that alternative antivenins such as Australia/New Guinea polyvalent antivenom (produced by Instituto Clodomiro Picado in Costa Rica), tiger snake antivenom (produced by Seqirus in Australia), and Mexican coral snake antivenom (Coralmyn) may be efficacious in M fulvius envenomations. 32 -35 Whenever a non-FDA approved treatment such as expired NACSA or an exotic antivenin is given for M fulvius envenomation, the physician should notify the US FDA Vaccines, Blood, and Biologics section (1-301-827-3524) and consult local hospital policy on the administration of experimental drugs.
Exotic antivenins may be stocked by US zoos, but there may be a delay of several hours if antivenin needs to be transported from a zoo to a hospital. When such a delay is anticipated, clinicians may consider administering an anticholinesterase medication such as neostigmine, but this decision should only be made with the consultation of a medical toxicologist. If neostigmine is given, an anticholinergic agent such as atropine or glycopyrrolate should also be administered to counteract excessive effects of neostigmine.
Alternative antivenins are also being investigated. A novel equine F(ab’)2 antivenom for M fulvius has been administered to 26 patients in phase 3 clinical trials.36,37 This antivenin is currently used only for experimental protocols, and the full results of the clinical trials have not been released. Efforts to develop a combined antivenin to Micrurus species are underway in South America, where Micrurus species diversity is greater.38,39 As NACSA is prone to periods of shortages lasting several years, a combined Micrurus antivenin might be investigated for use in M fulvius envenomations in the future. Research is also underway to develop a more efficient method of producing antivenin for the South American species Micrurus corallinus using genetic immunization with a synthetic multiepitope gene followed by booster doses with recombinant protein. 40 Application of a similar method to M fulvius might result in a less labor-intensive, more widely available antivenin.
Intubation may be required even if a patient receives antivenin. Criteria for intubation include a forced vital capacity less than 50% of the predicted value, a negative inspiratory force closer to 0 than -30 cm H2O, or a maximal expiratory pressure less than 40 cm H2O. Patients requiring intubation following M fulvius envenomation often make a complete recovery, but those who are intubated without receiving antivenin may require several weeks of ventilator support.9,41
Disposition
All patients presenting to the ED after a bite by a US coral snake species should be admitted to the ICU for hourly neurologic checks and pulmonary function tests. 9 Patients should not be discharged until they have been asymptomatic with normal neurologic examinations and pulmonary function tests for a period of 24 h beginning from the initial time the patient is asymptomatic. Patients who require intubation tend to do well but may require ventilator support for over 1 wk. All patients discharged after receiving antivenin should be instructed to return to the ED immediately if they exhibit any signs or symptoms of anaphylaxis or serum sickness. Patients should be advised to follow up with their primary care physician in the week after discharge. Routine laboratory monitoring is not required.
Conclusions
Although rare, bites by coral snakes in the United States require prompt recognition of signs and symptoms of envenomation. Early administration of NACSA reduces but does not eliminate the risk of respiratory failure requiring intubation. In-date NACSA is currently available but has had periods of low supply lasting several years in the past, and research into other agents is ongoing. Alternative methods of coral snake antivenin production and a single antivenin for all Micrurus species are being investigated in South America, and these advances may contribute to a more regularly available antivenin for envenomations by coral snakes in the United States. The aim of this case report is to highlight a unique presentation of M fulvius envenomation where the patient’s primary symptom was myalgia. The most worrisome presentation of patients envenomated by M fulvius is a descending paralysis worrisome for subsequent respiratory failure, but pain and paresthesia are more common presenting symptoms. Another aim of this case is to discuss management of this rare condition and potential future directions for its treatment.
Footnotes
Acknowledgements
Acknowledgments: The authors acknowledge Dr Brett Lorenzetti, MD for his contribution in patient management and Dr Latha Ganti, MD, MS, MBA, FACEP for her contribution to fostering a culture of research at their institution.
Author Contributions: Study concept and design (NT); acquisition of the data (NT); drafting of the manuscript (NT, MH); critical revision of the manuscript (NT, JLW); approval of the final manuscript (JLW, NT, MH).
Financial/Material Support: This research was supported (in whole or in part) by HCA Healthcare and/or an HCA Healthcare affiliated entity.
The views expressed in this publication represent those of the authors and do not necessarily represent the official views of HCA Healthcare or any of its affiliated entities.
Disclosures: None.
